Abstract
Background. Spontaneous visual improvement in people with an optic glioma (OG) of the anterior or retrochiasmatic optic pathways is rare. Objective. To evaluate the effects on visual function of nerve growth factor (NGF) eye drop administration in a patient with severe visual impairment due to a low-grade OG. Methods. A 45-year-old woman with OG and long-standing optic nerve atrophy was assessed before and after 2 NGF treatment courses. The drug used was 2.5S murine NGF. One milligram of NGF, diluted in saline solution, was administered onto the conjunctiva of both eyes for 10 consecutive days 3 times a day for each treatment. The follow-up was performed by clinical, neuroradiologic, and electrophysiological tests (electroretinogram and visual evoked potentials [VEPs]) at the end of each treatment and 30 and 60 days later. Results. A repeated subjective and objective improvement of visual function (>3 lines visual acuity; >40° visual field; >50% VEP amplitude increase, Wilcoxon test P < .01) was recorded after NGF treatment. These measures tended to deteriorate toward baseline values 60 days from the end of each NGF treatment. No ocular or systemic side effects were observed throughout treatment. Conclusions. Conjunctival NGF may be a beneficial adjunct therapy for visual loss in patients with OG, possibly exerting its effects on residual viable optic pathways.
Introduction
Several neoplastic diseases, such as craniopharyngioma and optic glioma (OG), can compromise visual function involving both anterior and retrochiasmatic optic pathways. No specific therapy is currently available for OG-induced visual loss. Nerve growth factor (NGF) is the first discovered neurotrophin involved in the development and survival of sympathetic, sensory, and forebrain cholinergic neurons. 1 In experimental animal models, NGF promotes nerve terminal outgrowth and neuron recovery after inflammatory, ischemic, and toxic injuries. 2 Given its actions favoring neuronal survival, NGF has been proposed for the treatment of some traumatic, ischemic, and neurodegenerative brain diseases. 3,4 Intraventricular NGF administration may ameliorate symptoms in adults with Parkinson’s and Alzheimer’s disease and improves cerebral blood flow in infants with hypoxic–ischemic brain injury. 5-8 Exogenous NGF showed neuroprotective effects also on the visual system 9 because of the presence of NGF receptors on the conjunctiva, cornea, and retinal ganglion cells (RGCs). 10 The effects of NGF and other neurotrophins, such as brain- derived neurotrophic factor (BDNF), are mediated via uptake by RGCs, anterograde transport along the optic nerve, and release to the postsynaptic geniculate neurons. 11 In experimental animal models, intraocular NGF administration keeps RGCs from degeneration after optic nerve transection and ocular ischemia and leads to the rescue of axotomized forebrain cholinergic neurons, 12,13 whereas conjunctivally applied NGF proved to be effective in patients with corneal ulcers and severe glaucoma. 14,15 Indeed, it has been reported that NGF administered to the conjunctiva rapidly reaches the retina and the optic nerve where this neurotrophin exerts its biological activities through the uptake by RGCs. 16 Based on these evidences, we report the first known case of NGF effect on visual loss reversal in a patient with OG.
Case Report
A 45-year-old woman, affected since childhood by genetically confirmed neurofibromatosis type I, had a diagnosis of anterior prechiasmatic OG at age 33 years with bilateral and steadily progressive deterioration of both central and peripheral vision. At admission neuro-ophthalmologic examination showed bilateral proptosis of 3 mm, normal extrinsic and intrinsic ocular motility. Best corrected visual acuity was as follows: right 0.2 (−6 sph), left 0.1 (−8 sph); near distance: 5th size (Jaeger reading chart) bilaterally. Ishihara plate testing showed bilateral red-green axis dyschromatopsia. Goldmann visual field testing showed bilateral concentric, severe constriction with both V/4e and II/4e isopters (main diameter = 15° and 10°, respectively). Visual evoked potentials (VEPs) pattern reversal and flicker (8 Hz) stimulation showed severe bilateral amplitude attenuation with an increased time-to-peak. Electroretinograms (ERGs) showed attenuated amplitudes bilaterally of the inner retina–related flicker 1st and 2nd harmonic components.
Based on the role of NGF in important biochemical and molecular mechanisms of the optic pathways, we decided to treat this patient by NGF eye drop administration when marked and progressive visual loss was no longer responsive to conventional treatment with steroids and mannitol. The second NGF course was carried out after 60 days in the same fashion, according to an institutional review board/ethical committee–approved protocol.
The drug used was 2.5S NGF, purified and lyophilized from male mouse submandibular glands and prepared according to the method of Bocchini and Angeletti. 17 The submandibular glands of adult male mice were explanted under sterile conditions and the tissues were homogenized, centrifuged, and dialyzed. This aqueous gland extract was then passed through subsequent cellulose columns, thereby separating NGF by adsorption. The first step was gel filtration at pH 7.5, wherein most of the active NGF was eluted in the 80 000 to 90 000 molecular weight range. The samples obtained were analyzed by spectrophotometry at a wavelength of 280 nm to identify NGF-containing fractions. Specificity of fractions was determined by Western blot analysis. NGF purity (>95%) was estimated by high-performance liquid chromatography column equipped with a guard column calibrated with 40 mg of purified and bioactive murine 2.5S NGF standard. The NGF obtained was then dialyzed and lyophilized under sterile conditions and stored at −20° until used. Biological activity of purified NGF was evaluated by in vitro stimulation of neurite outgrowth in rat pheochromocytoma PC12 cells over a period of 7 to 14 days. NGF was dissolved in 0.9% sterile saline solution in concentrations of 200 µg/mL. The concentration of NGF in this solution was stable for the entire treatment time. One milligram of NGF, diluted in 5 mL of saline solution (0.2 mg/mL), was administered onto the conjunctiva of both eyes for 10 consecutive days 3 times a day for each treatment. This dose is considered sufficient to reach and stimulate NGF receptors in most cerebral cholinergic areas of the brain and optic pathways, as previously reported in the literature. 5
No ocular or systemic side effects occurred. Brain magnetic resonance imaging (MRI) evaluation, performed at baseline and after 120 days, did not show any significant change in tumor volume after NGF treatment (Figure 1 A-D). The follow-up was performed by clinical and electrophysiological tests at the end of each treatment and after further 30 and 60 days. A subjective and objective improvement of visual acuity and visual field (>3 lines visual acuity; >40° visual field; Wilcoxon test, P < .01; magnitude of change >99 percentile of the test–retest variability for both visual acuity and visual field 18,19 ; Figure 2A and B) allowing her to walk confidently, watch TV, and recover the customary activities was recorded after 48 hours from the beginning of each treatment. Also, VEPs and ERGs showed an improvement after NGF administration (>50% increase in VEP amplitude; changes >99 percentile of the test–retest variability 20 ; Figure 2C and D). Visual acuity, visual field, and VEPs tended, following the improvement, to deteriorate toward baseline values after 60 days from the end of each NGF treatment, suggesting the need to continue NGF administration every 2 months.
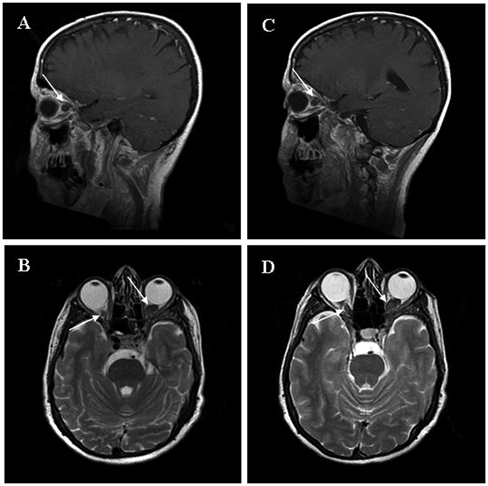
Sagittal T1-weighted magnetic resonance imaging scan (MRI) of the brain and orbits showing the glioma affecting the left optic nerve (white arrow) at baseline (A) and 4 months after nerve growth factor (NGF) administration (C). Axial T2-weighted MRI showing both right and left optic nerves involved by bilateral optic gliomas (white arrow) at baseline (B) and 4 months after NGF administration (D). No significant change in tumor volume after NGF treatment was found
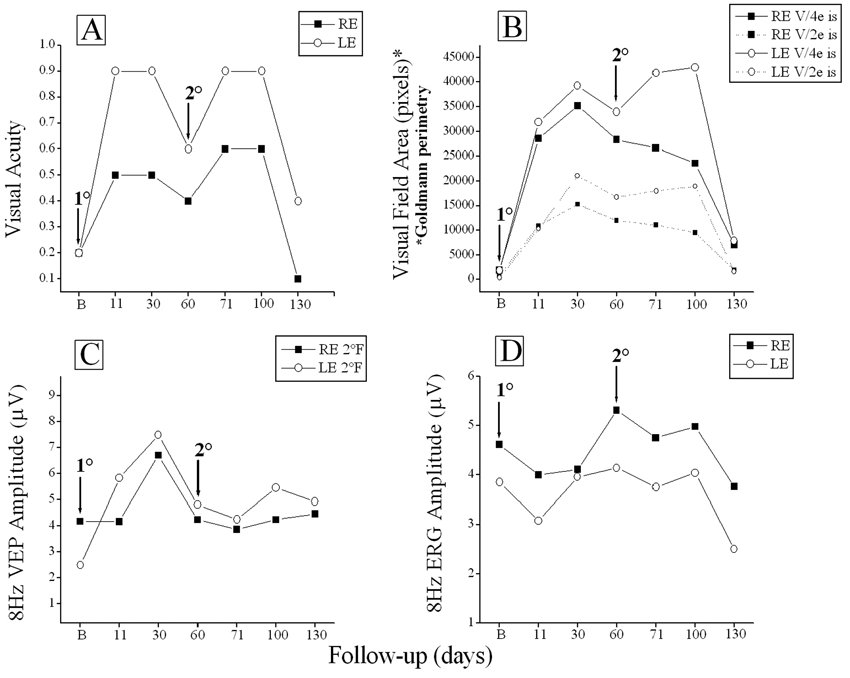
Right after the end of each treatment, visual acuity (A) recovered in both eyes by 3 to 7 lines (significant [P < .05] increase or decrease is ≥2 lines). (B) Lateral visual fields reported in relative units following planimetric analysis, recovered by several log units of area indicating a main increase in lateral perception of 40° in RE and 35° in LE. (C) VEPs showed an increase in amplitude in parallel with visual acuity improvement. (D) ERGs showed amplitude enhancement 60 days after the first NGF course and then tended to decline
Discussion
This report shows that NGF eye drop administration may ameliorate visual acuity, visual field, VEPs, and ERGs amplitudes in this patient with OG, as indicators of visual loss reversal after NGF treatment. The improvement of VEPs and ERGs suggests molecular events within surviving RGCs leading to a recovery of their physiological responses. It is known that RGCs express NGF receptor (TrkA) and that NGF, binding to TrkA, upregulates Bcl-2 protein, which protects cells from apoptosis by preventing caspase activation. 21 Furthermore, intravitreal NGF delivery to the retina and optic nerve is crucial to the survival of RGCs and for functional recovery of the retina following ocular ischemia and hypertension in experimental animal models. 13,22 Last, conjunctivally applied NGF has been shown to reach sharply the retina and optic nerve where this neurotrophin exerts its biological activities. 16 In our patient, the electrofunctional changes after NGF administration were immediately followed by clinical improvement. These effects confirm the key role of neurotrophins in modulating RGC function and visual cortical neuronal activity, as reflected by receptive field size and response latency. 23,24 Amelioration of both clinical and electrophysiological parameters lasted for about 60 days after NGF treatment, indicating that NGF effect on optic pathways had a prolonged duration. This prolonged and stable NGF effect may be related not only to a protective activity against neural apoptosis but also to the formation of new neural pathways, since it is known that NGF promotes neuronal repair and axonal regeneration. 25,26 NGF acts on different levels to promote neuronal recovery following ischemic, inflammatory, and traumatic injuries: through a neosynaptogenetic mechanism, by directly affecting precursor cells, and/or by induction of other growth factors, such as BDNF, whose neuroprotective effects on visually evoked RGC responses after optic nerve section have been reported. 11,27
Caution is necessary. NGF promotes proliferative activity of glioblastoma cells through direct interaction with activation of RAS and MAPK Erk1/2 pathway. 28 The level of NGF increases in the malignant glioma tissue, 29 although the opposite has been found for low-grade astrocytomas, 30 whereas growth control of glioma by NGF has been shown in vivo. 31 Currently, drugs that block RAS and mTOR are under investigation for treatment of NF1-associated malignancies (see www.clinicaltrials.gov). Potential complications of long-term therapy using topical NGF treatment on NF1-associated OG include an increase in tumor size. A recent 6-month, phase I trial of topical NGF in children with OG from our group did not show any growth by MRI.
Prospective and controlled clinical trials should be conducted to explore the efficacy of conjunctivally applied NGF on optic nerve function. In parallel, animal studies could identify NGF-induced mechanisms in genetically engineered mice with knockout of the NF-1 gene that develop OG. 32 These animal models could evaluate the changes and correlations between NGF and other neurotrophic factors, such as BDNF, basic fibroblast growth factor, ciliary neurotrophic factor, and synaptic vesicle modifications.
Footnotes
Antonio Chiaretti and Benedetto Falsini contributed equally to this study.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
