Abstract
Background. Ankle impairments are closely associated with functional limitations in children with cerebral palsy (CP). Passive stretching is often used to increase the range of motion (ROM) of the impaired ankle. Improving motor control is also a focus of physical therapy. However, convenient and effective ways to control passive stretching and motivate active movement training with quantitative outcomes are lacking. Objective. To investigate the efficacy of combined passive stretching and active movement training with motivating games using a portable rehabilitation robot. Methods. Twelve children with mild to moderate spastic CP participated in robotic rehabilitation 3 times per week for 6 weeks. Each session consisted of 20 minutes of passive stretching followed by 30 minutes of active movement training and ended with 10 minutes of passive stretching. Passive ROM (PROM), active ROM (AROM), dorsiflexor and plantarflexor muscle strength, Selective Control Assessment of the Lower Extremity, and functional outcome measures (Pediatric Balance Scale, 6-minute walk, and Timed Up-and-Go) were evaluated before and after the 6-week intervention. Results. Significant increases were observed in dorsiflexion PROM (P = .002), AROM (P = .02), and dorsiflexor muscle strength (P = .001). Spasticity of the ankle musculature was significantly reduced (P = .01). Selective motor control improved significantly (P = .005). Functionally, participants showed significantly improved balance (P = .0025) and increased walking distance within 6 minutes (P = .025). Conclusions. Passive stretching combined with engaging in active movement training was of benefit in this pilot study for children with CP. They demonstrated improvements in joint biomechanical properties, motor control performance, and functional capability in balance and mobility.
Introduction
Cerebral palsy (CP), the most common physical developmental disability in childhood, represents a group of permanent postural, motor control, and movement disorders because of nonprogressive lesions or anomalies of the brain. The disorder affects developmental activity and function of the child. Not only the motor functions are impaired, children with CP often face challenges such as poor coordination, sensory disturbances, communication difficulties, and musculoskeletal and cognitive deficits. 1 Ambulation requires neurological control (ie, voluntary and reflexive control over multiple joints and agonist/antagonist muscle groups simultaneously) and musculoskeletal integrity, so gait is often disrupted significantly in CP. 2-4
Children with CP continue to be at risk for deformity during growth years. There is a risk of losing functional mobility during adolescence because of ongoing secondary impairments such as muscle fascicle shortening, weakness, and bony deformity. 5-7 Increased muscle tone and poor selective motor control affect children with CP and these impairments may contribute to decreased frequency and variety of voluntary movement. 8 Children with these secondary impairments may also experience a loss of independence. 9
Cerebral palsy is a persisting, but not unchanging, disorder of movement and posture. 1 Prevention of secondary impairment and promotion of functional independence aims, in part, to reduce spasticity and increase range of motion (ROM). 8,10-12 Training-induced induction of neuroplasticity has been demonstrated by functional imaging with rehabilitation. 13 Improvements in motor control may also occur about a single joint in an isolated movement, but whether improvements carry over to more complex tasks still needs to be investigated. 4 Despite interventions throughout childhood, many individuals with CP who achieve ambulation in childhood lose this ability in early to middle adulthood. 9,14
Impaired ankle function is a frequent cause of limitation in ambulation. 15 Contractures and spasticity affect alignment and create an imbalance in the involved limb. In the ankle and foot this will lead to decreased foot contact during stance and impaired prepositioning of the foot in the swing phase, 2 attributes of gait that are important for efficient ambulation. 16 Weakness of ankle plantarflexors, and the resulting limitation of the plantarflexor joint moment, can lead to crouch gait in children with CP, which may deteriorate further without intervention. 16 Such gait abnormalities increase submaximal walking energy expenditure almost 3-fold compared with typically developing children. 17
Common methods for reducing muscle overactivity or imbalance include neurolysis (botulinum toxin injections, phenol injections), selective dorsal rhizotomy, partial immobilization (ankle foot orthosis), and lengthening or releasing tendons through surgery. These approaches do improve lower extremity biomechanical alignment during gait in the short term, but they perhaps do so at a cost that may only be realized in the longer term. 18
Active and passive ROM exercises are incorporated into the management of children with CP. 19-21 Stretching serves an important component of physical therapy intervention to treat spasticity and contracture. 22-24 Exercise incorporates active movement to maintain or increase joint ROM and/or muscle strength. 20 Children with CP are significantly weaker than their peers 25 in most or all muscles around a joint. 26
In pediatric rehabilitation, eliciting cooperation in children with CP and motivating them to exercise the impaired joints enthusiastically can be challenging. The level of difficulty of therapy should be considered so as not to frustrate children while still meeting the required treatment intensity. However, very few available devices and protocols conveniently address these issues. Furthermore, there is insufficient study regarding how treatment of limb impairment can be translated into daily functions. Hence, our aim was to investigate the efficacy of the combination of passive stretch and active movement training delivered by a rehabilitative robot for children with CP.
Methods
Participants
Twelve children (6 girls, 6 boys) who ranged in age from 5 to 15 years (mean age of 8 years 6 months) with spastic CP participated in this study; 6 had diplegia, and 6 showed signs of hemiplegia. All had impaired ankle function that consisted of reduced ROM and reduced selective motor control of his/her lower extremity. Table 1 lists the characteristics of the participants.
Participant Characteristics
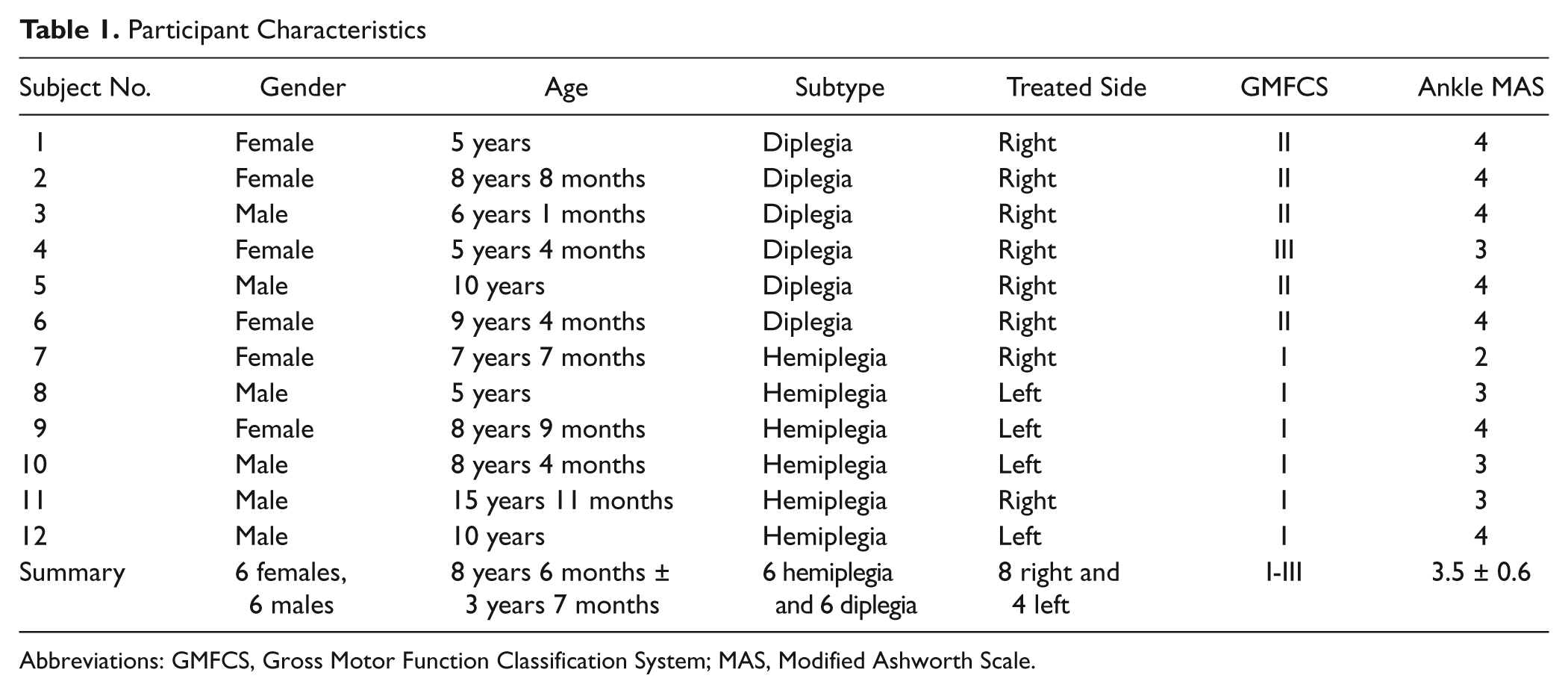
Abbreviations: GMFCS, Gross Motor Function Classification System; MAS, Modified Ashworth Scale.
Participation criteria required that the participants (a) were able to walk with an assistive device independently (Gross Motor Function Classification System [GMFCS] levels I, II, III), (b) had no surgical intervention within the preceding year and no serial casting within 6 months, and (c) had enough cognitive ability to actively participate in the training protocol. This study was approved by the institutional review board of Northwestern University, Chicago. All parents gave informed parental consent, and all participants gave informed verbal assent before the experiment.
Experimental Setup
A portable rehabilitation robot with computer game interface was used to deliver the designed protocol and to measure the biomechanical data, that is, specify what biomechanical data the robot measured. The robotic device was equipped with a torque sensor, a servomotor, and a digital controller. It was connected to a personal computer for display and user interface. The interface allowed adjustment of the applied torque value, motion velocity, and difficulty levels of the exercise games, such as assistance level and resistance level, according to each child’s ability.
Procedures
The participants came to our laboratory 3 days a week for 6 weeks. The same training protocol was used for each training session for 20 minutes of warm-up passive stretching, 15 minutes of assisted-active movement, 15 minutes of resisted-active movement, and 10 minutes of cooldown passive stretching.
Training Protocol
Each participant was seated on a comfortable chair fixed to the floor with the robotic device in front of the participant at the appropriate distance to keep the participant’s knee extended (Figure 1). This arrangement was kept the same at each training session, as well as during pre- and postevaluations. We aligned the participant’s ankle dorsiflexion axis with the rotation axis of the device and strapped the foot onto the footplate of the robot (see Figure 1). The ROM limits and applied torque of the rehabilitation robot were determined for passive stretching and controlled by the intelligent algorithm that allows strenuous and safe passive stretching. 27
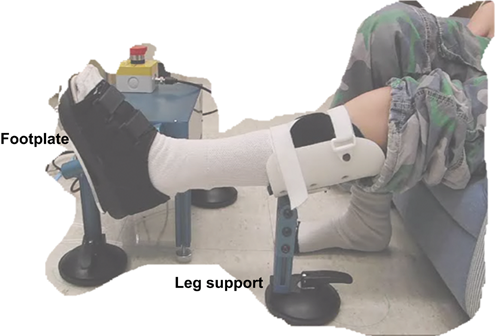
Training setup. Participant was seated on the chair with knee extended. The leg was supported by the leg support and the foot was strapped onto the foot plate with the ankle joint aligned to the rotation axis of the rehabilitation robot.
Two types of active movement training were completed by the participants in which they voluntarily dorsiflexed and plantarflexed their ankle to play computer games. The first type of active movement training was assisted active movement training. In this condition, movement assistance came from the robotic device, while the participant dorsiflexed and plantarflexed his/her ankle within the initially determined passive range of motion (PROM). The range of the movement target in the computer games was scaled to the individual participant’s PROM, which allowed assisted movement training within the full PROM. The second type of active movement training was resisted active movement training. The participant in this condition had to overcome the resistance added by the robotic device and dorsiflex and plantarflex his/her ankle voluntarily within his/her active range of motion (AROM). The range of occurring targets in this training mode was reduced from the PROM according to each participant’s AROM. Each data collection session ended with 10 minutes of passive stretching of the ankle using the same parameters employed in the warm-up stretching program.
Outcome Evaluations
Clinical, functional, and laboratory evaluations were performed before and after the 6-week training period. A pediatric physiatrist performed the Modified Ashworth Scale (MAS, 0-5) 28 and Modified Tardieu Scale (MTS) for spasticity, the Pediatric Balance Scale (PBS), and the Selective Control Assessment of the Lower Extremity (SCALE). 41 Functional evaluations included 6-minute walk and Timed Up-and-Go (TUG). Biomechanical measures were PROM, AROM, and muscle strength.
Passive ROM and joint stiffness measurement
The participant sat on the chair with the foot strapped onto the footplate. Various levels of terminal torque were used to move the ankle joint at a consistent velocity of 10°/s for each participant. For consistency between pre- and postevaluations, the levels of torque used in the 2 evaluations were the same for each participant. However, different peak torques may have been used for different participants, considering differences in body size, relative strength, and so on. The PROM measurement was only performed in dorsiflexion since the proposed protocol in this study was aimed at stretching the stiff calf muscle, which was believed to lead to an increase in the participant’s dorsiflexion and a reduction in the joint stiffness, with the joint stiffness defined as the slope of the ascending limb of the torque–angle relationship (hysteresis loop) at 10° of ankle dorsiflexion. 29
Active ROM and strength measurement
Each participant was asked to actively dorsiflex the ankle joint from the end of plantarflexion while a visual feedback bar was displayed on the screen, so that with more dorsiflexion, the length of the bar became longer. Three trials with a 1-minute rest interval were completed. The values of the 3 trials were averaged as the measured AROM. For strength testing, the footplate of the device was locked at approximately 10° of plantarflexion. The participant was then asked to maximally dorsiflex the ankle to make the displayed bar as long as possible. Finally, participants were instructed to plantarflex as much as possible to measure plantarflexor muscle strength.
Statistical Analysis
The nonparametric Wilcoxon signed-rank test was used to examine intervention-induced changes of the ordinal data (SCALE, MAS, and PBS), with significance levels set at P < .05. To compare differences before and after intervention in the biomechanical measures (AROM, PROM, and strength) and in the functional outcome data (6-minute walk and TUG), paired t tests were conducted, with significance levels of P < .05.
Results
All twelve participants completed the 18 sessions of training in addition to the pre- and postevaluations. After the 6-week training period, participants showed significant improvements in their ankle ROM and function with additional improvements in adjacent joints as well.
Biomechanical Measures
Both passive and active ankle dorsiflexion increased significantly as a result of the 6-week training program (see Figure 2A and B). The passive dorsiflexion was 20.7 ± 4.6° and 26.6 ± 4.9° before training and after training, respectively (P = .002), whereas the active dorsiflexion was 3 ± 6.4° prior to training and 10.1 ± 8.7° after training (P = .02).
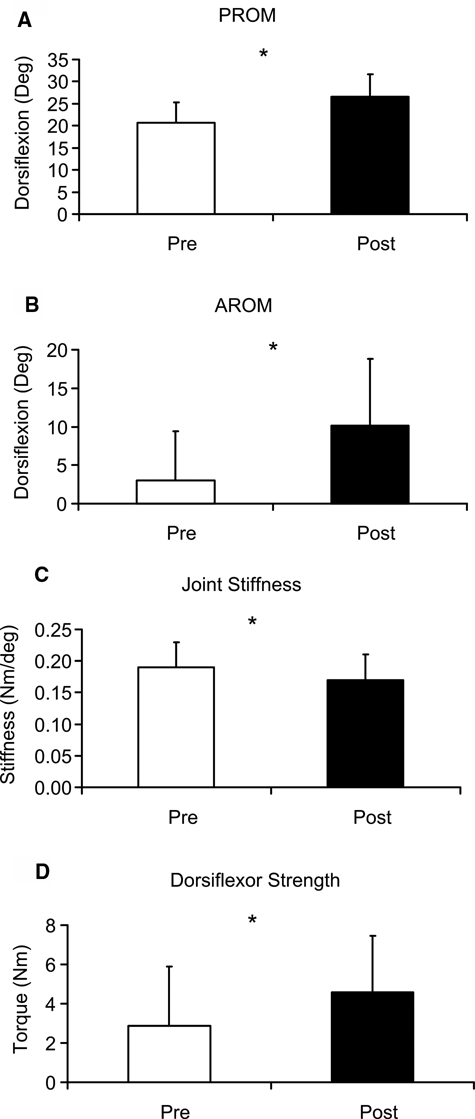
Biomechanical measures (A, passive range of motion [PROM] of dorsiflexion; B, active range of motion [AROM] of dorsiflexion; C, joint stiffness; D, Strength of dorsiflexor). *Indicates significant difference (P < .05).
Joint stiffness was also reduced with training from 0.19 ± 0.04 N m/° before training to 0.17 ± 0.04 N m/° after training; P = .03 (Figure 2C). The strength of dorsiflexors also increased from 2.9 ± 3.0 N m (mean ± SD) before training to 4.6 ± 2.9 N m after training; P = .001 (Figure 2D). No change in plantarflexor strength was found (P = .12).
Clinical Evaluations and Functional Outcomes
The catch angle in dorsiflexion (Tardieu R1) was reduced (P = .02), but no change was seen in other joints. A decrease in MAS score was also seen at the ankle (P = .01). Table 2 shows the changes obtained in selective motor control of the lower extremity joints for each participant after the 6-week training program.
Selective Control Assessment of the Lower Extremity (SCALE) a

Each row corresponds to a subject. Asterisk * denotes no change, − denotes decrease of 1 gradation, + denotes increase of 1 gradation, and ++ denotes increase of 2 gradations.
SCALE scores increased significantly after the 6-week training period (P = .003). Significant improvements were seen at the ankle (P = .002). In addition, significant improvement in selective motor control of the hip and foot were also observed (P = .04 and P = .02, respectively).
Figure 3A-C highlights the functional changes before and after 6 weeks of training. The participants showed functional improvements in terms of balance and walking distance. All participants improved in balance (P = .003). Within 6 minutes, the participants also walked further than before the 6-week intervention (mean of 314.39 ± 90.57 m before training compared with 365.63 ± 110.39 m after training; P = .02). TUG scores did not change after the 6-week training program (P = .21).
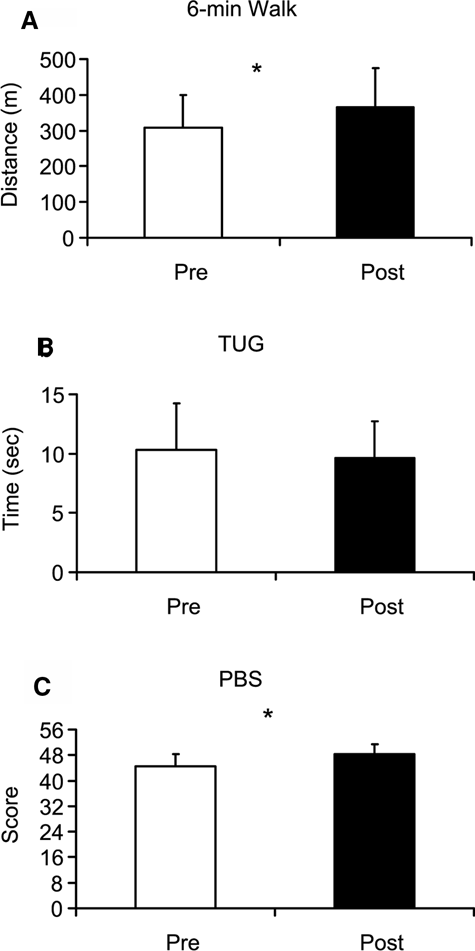
Functional outcome measures (A, 6-minute walk; B, Timed Up-and-Go [TUG]; C, Pediatric Balance Scale [PBS]) before and after the 6-week intervention. *Indicates significant difference (P < .05).
Discussion
This study investigated the efficacy of combined passive stretching with active movement training delivered by a portable rehabilitation robot. The results demonstrated that passive stretching combined with biofeedback games can benefit children with CP who have impaired ankle function. We used ankle position as biofeedback; the participants could see the controlled object of the game moving proportionally to the actual ankle position. Passive stretching lengthened the spastic muscles, as indicated by the increased PROM as well as decreased joint stiffness, which may facilitate muscle force generation and ankle function. 27 The subsequent biofeedback game playing further improved ankle control and functional mobility, as shown in the improved AROM, muscle strength, 6-minute walk tests, and balance. In addition, spasticity reduction at the ankle was found, possibly related to the passive stretching and active movement training.
Active and passive movement training is incorporated into therapeutic programs 20,21,30 to improve the functional ROM of the ankle joint. Passive stretching is a critical component of maintaining muscle flexibility; however, stretching alone may not be enough. For children with CP, the possible reasons for insufficient effect may be due to immobility and/or undesired muscle activations associated with spasticity or dystonia. 19 Hence, spasticity reduction and improved motor control are needed. From this perspective, the focus of the robotic on encouraging movement immediately after flexibility increased enabled children with CP to experience a repertoire of demanding movement control activities, which helped improve their motor control ability.
The computer games engaged the children in the exercises. This approach may help improve functions along the neural pathway between signal reception (sensory cues) and movement execution (actuator), 31 as the movement training through computer games allows participants to receive biofeedback of the ankle position during the game playing. Biofeedback treatment can help individuals develop greater awareness of and an increase in voluntary control. 32 Our results showing improvements in SCALE, 6-minute walk, and PBS indicate not only changes in passive biomechanical properties but also improvements in selective motor control, gait, and balance. The closed-chain training design was employed to enhance sensory input by stimulating the proprioceptors in stretched or tensioned tissues, which may facilitate motor control. Sensory input may also reduce body image deficiency, thereby improving voluntary movement control. 33
Changes of SCALE scores showed improvement in the target training joint (ankle), as well as adjacent (foot) and proximal (hip) joints of the lower limb. Intervention-induced enhancement of ipsilateral descending motor pathway activity might have contributed to the improved performance observed at the hip joint and foot. Maintenance of ipsilateral tracts seems to play a certain role for higher compensatory potential in the motor system. 34 Upregulation of ipsilateral cortical activity and increased excitability of ipsilateral corticofugal and segmental cord recrossing fibers may help preserve some degree of motor function. The improvement in foot motor control might be due to expression of synergistic motion resulting from upregulation of ipsilateral projection. 35 The ipsilateral hemisphere of hemiparetic patients contributes to the control of normal voluntary movements. 35,36 Of note is that, in our study, not only children with hemiplegic CP but also children with diplegic CP showed improvement in joints other than the ankle. Further study with larger samples and different types of CP, with the addition of brain imaging studies, are needed to investigate these phenomena.
Muscle strengthening in children with CP may raise concerns about increasing spasticity. 37,38 On one hand, some investigators believe excessive effort should be avoided since it may increase spasticity and impair motor control. 37 On the other hand, it was reported that strength training was not associated with an increase in muscle tone, 38 so physical therapy intervention strategies can be aimed at enhancing, rather than restricting, muscle function in CP. 30,39 In our study, 15 minutes of resistance training was employed. Consistent torque was added to both dorsiflexion and plantarflexion during active movement according to the participants’ capabilities. No increase in spasticity was found after the 6-week training. Rather, ankle spasticity had decreased after training. Furthermore, dorsiflexor muscle strength increased with the training, although there was no significant increase in plantarflexor strength. During game playing, dorsiflexors were forced to actively contract against resistance compared with their relatively ineffective contraction during daily activities including gait. Also, the plantarflexors are often overactive during gait. 40 The intensity of active plantarflexion during game play might not have been high enough to increase muscle strength; the level of resistance was set to the child’s comfort level. Since our muscle strength measurement was done in an antigravity position, further investigations into providing training and evaluating muscle strength in a functional position against gravity should be pursued. The active movement training focused more on the motivating repetitive movement training than on muscle strengthening. Therefore, the improvement in movement control was probably more due to robot-assisted repetitive movement training and motor learning than due to strengthening.
The effectiveness of deliberately training an underused limb through repetitive practice has become a widespread rehabilitation principle, developed in part through studies of constraint-induced movement therapy. 19 This assumption remains to be proven clinically, 13 especially in relation to the optimal dosage of intensive training. Intensive training may not only be tiring and stressful for children and their families, with a consequent low compliance, but also rather expensive. 13 The effective intensity also depends on the severity of CP. In our study, the intensity of an hour per day, 3 days per week over 6 weeks was well tolerated by the participants with mild to moderate CP (GMFCS levels I-III). None of the participants withdrew from the study, and parents were satisfied with the training.
We did not require that children stop possible school-based, traditional rehabilitation during the robotic therapy, which might have influenced results. We did not quantitatively evaluate the lasting effect of the protocol.
The positive outcome of this study suggests that future studies include a larger sample of children with CP including GMFCS levels I to IV in clinical settings and perhaps at nonlaboratory settings to explore the impact of combined passive and active training of a single joint on adjacent joints. Further studies must include control subjects and seek the optimal dose of therapy and then examine for the translation of gains to daily functional movements and participation in daily life.
Footnotes
Acknowledgements
We acknowledge gratefully the support from the participants and their families.
The author(s) declared a potential conflict of interest (e.g. a financial relationship with the commercial organizations or products discussed in this article) as follows: Li-Qun Zhang and Yupeng Ren have equity positions in Rehabtek LLC, which is involved in developing and commercializing the ankle rehabilitation robot used in this study.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The authors acknowledge the funding supports from the National Institutes of Health (R42HD043664), National Institute on Disability and Rehabilitation Research (H133F090042, H133S080074, H133E10007, and H133G010066), and National Science Foundation (IIP-0750515).
