Abstract
Background. Even optimal nerve reconstruction after facial nerve damage leads to defective reinnervation because of misdirected axonal sprouting and polyinnervation of the end plates of the facial muscles. Objective. The authors studied whether temporary chemical denervation of the contralateral nonlesioned hemiface with botulinum toxin (BTX) would increase regeneration of the lesioned buccal branch of the facial nerve and improve functional recovery of the whisker pad. Methods. The experiments were performed in 65 adult rats distributed in 4 interventions: (1) buccal–buccal nerve anastomosis (BBA), (2) BBA plus ipsilateral injection of BTX into the whisker pad, (3) BBA plus contralateral BTX injection, or (4) BTX injection without any surgery. Sequential preoperative and postoperative retrograde fluorescence tracing at 4 weeks after surgery quantified the accuracy of reinnervation. Functional recovery was measured by biometrical image analysis of whisking behavior at 12 weeks after surgery. Results. After BTX injection without any surgery, muscle paralysis was transient, and the animals restored normal nerve terminals and normal vibrissal function at 8 weeks after treatment. After BBA and ipsilateral or contralateral BTX injection, the degree of correct reinnervation increased significantly to 61% in comparison to 27% after BBA without any other intervention. Enhanced correct reinnervation was accompanied by a significant improvement of whisking after contralateral but not after ipsilateral injection of BTX. Conclusions. These results provide evidence that transient contralateral muscle paralysis helps improve the morphological and functional regeneration after facial nerve repair.
Keywords
Introduction
Even with the use of optimal modern microsurgical techniques, functional repair of the facial nerve or of other peripheral nerves is often insufficient. The recovery of resting tone, voluntary movements, and emotional expression remain poor. 1 Further functionally very important clinical signs are abnormal movements (synkinesia) and altered reflex activity, for example, abnormal blink reflex. 2 Neurobiologically, these phenomena are based on misdirected reinnervation caused by collateral sprouting, terminal sprouting, and transaxonal exchange of nerve impulses between the regenerating axons. 3 Another key finding is the hyperexcitability of the axotomized facial motoneurons. 4 However, it has also been reported that the lesioned facial motoneurons are less responsive to afferent stimulation, 5 perhaps because of decreased synthesis of transmitter-related compounds 6 or impaired connectivity caused by the stripping of axosomatic afferents. 7
Recently, we have shown that regeneration of axotomized facial motoneurons can be improved by modifying the trigeminal sensory input. Deafferentation of the whisker pad by resection of the infraorbital nerve ipsilateral to the facial nerve lesion deteriorated facial nerve regeneration. In contrast, contralateral deafferentation reduces postoperative axonal sprouting of the lesioned facial motoneurons and leads to a significant functional improvement of whisking behavior in animals with facial nerve transection and immediate repair. 8,9 We hypothesized that the contralateral deafferentation could be interpreted as a form of constraint-induced therapy forcing the animals to use the ipsilateral lesioned and reinnervated hemiface and whisker pad. To prove this hypothesis and to modify the approach in a manner that would be applicable for clinical use, we performed experiments in which unilateral facial nerve repair was combined with a simultaneous but transient chemical denervation of the contralateral hemiface with botulinum toxin (BTX) type A. Here, we show that this approach results in a pronounced improvement of functional recovery in rats.
Methods
Animal Model
The experiments were performed in 65 female Wistar rats (175-200 g; strain HsdCpb:WU, Harlan-Winkelmann, Borchen, Germany). Four different types of surgical interventions were performed: (1) ipsilateral buccal–buccal nerve anastomosis (BBA), (2) BBA plus ipsilateral injection of BTX into the whisker pad, (3) BBA plus contralateral BTX injection, or (4) BTX injection without any surgery. Methodologically, the animals were divided into 3 groups (A-C): Group A was used to optimize the BTX type A dose; group B was used for the retrograde tracing experiments; and group C was used for vibrissal movement analyses. More details about the experimental groups are given in Table 1. All examinations, that is, cell counting after tracing, analysis of the nerve terminals, and whisker movement analyses, were performed under blinded conditions by 2 independent examiners. The rats were kept on standard laboratory food (Sniff, Soest, Germany) and tap water ad libitum with an artificial light/dark cycle of 12 hours light on and 12 hours light off. The experiments were conducted according to the German Law for Animal Protection and were approved by the local animal care committee (Bezirksregierung Köln, Az. 23.203.2-K35, 16/04). The guidelines of the National Institutes of Health for animal care were closely observed.
Overview of the Different Experimental Animal Groups (N = 65 Rats)

Abbreviations: BTX, botulinum toxin; R, right side; BBA, buccal–buccal nerve anastomosis; L, left side.
Preparation and Injection of BTX Type A
The BTX type A formulation Dysport (Ipsen Pharma, Ettlingen, Germany) was used. The content of a vial of BTX (500 MU) was dissolved in 2.5 mL saline, resulting in a solution of 200 MU/mL. All injections were applied subcutaneously in the center between row A and B of the vibrissal hairs using a 1-mL syringe and a 26 G canula.
Dose Finding: Temporary Facial Palsy by BTX Injection
The aim of the dose-finding study was to detect the dosage of BTX sufficient to paralyze completely the ipsilateral (right) vibrissal musculature without paralysis of the contralateral (left) vibrissal musculature or other neighboring musculature. No facial nerve surgery was performed in these animals. Six different dosages of BTX—5, 10, 20, 40, 60, and 80 MU per whisker pad—were injected unilaterally in 6 animals of group A. The vibrissal movements of the animals were observed for 154 days. To objectively analyze the observations, the movements of the vibrissal hairs were recorded digitally and analyzed (see below) 2, 7, 14, 21, 28, 35, 84, and 154 days after injection. To confirm the effects, the chosen optimal dose of 20 MU was injected in 1 additional animal and the movement analysis was repeated.
For fluorescence histochemistry of target-muscle end plates, 2 animals per time point were killed at 2, 7, 28, 56, and 84 days after injection of 20 MU BTX. As in previous studies, we selected the levator labii superioris muscle as a representative of the external vibrissal muscles. 10 The animals were fixed by perfusion (see below). Under a surgical microscope, the levator labii superioris muscle on the injection side and contralateral side of the face was dissected free and cryoprotected by immersion in 30% sucrose. Longitudinal sections (50-µm thick) were cut on a cryostat and mounted on SuperFrost/Plus slides (Carl Roth, Karlsruhe, Germany). To visualize intramuscular axons and motor end plates, every third section through the muscle was double stained with an antibody against synaptophysin (staining the regenerating nerve fibers) and α-bungarotoxin (staining the postsynaptic nicotinic acetylcholine receptors) through the following steps: (1) 0.2% Triton X-100 (Fluka Chemie, Buchs, Switzerland) in Tris-buffered saline (TBS), pH 7.4, for 10 minutes at room temperature (RT); (2) incubation with Cy2-conjugated α-bungarotoxin (MoBiTec, Göttingen, Germany; dilution 1:200 for 20 minutes at RT); (3) incubation with the antisynaptophysin SY38 antibody (Boehringer, Ingelheim, Germany) raised in rabbit (dilution 1:8000 in TBS plus 0.8% (w/v) bovine serum albumine BSA) for 24 hours at RT; (4) 5.0% (v/v) normal sheep serum (Sigma, No. S-3772) in TBS for 30 minutes at RT; and (5) incubation with sheep antirabbit IgG Cy3 conjugate (1:400; Sigma, St Louis; in TBS plus 0.8% normal sheep serum for 1 hour). For specificity controls (1) omission of the primary antibody yielded blank sections, and (2) omission of the secondary biotinylated antibody also yielded blank sections.
Sections were observed with a Zeiss Axioskop 50 epifluorescence microscope through a Cy2-specific filter (no. F41-054; HQ 470/40x Q505 LPc HQ727/30m; AHF Analysentechnik, Tübingen, Germany) and through a Cy3-specific filter (no. F41-057; D548/10x Q565 LPc HQ 670/75m; AHF Analysentechnik). Individual end plates were identified in every third section of a muscle except for end plates at the sections’ surfaces that appeared to be cut. For image analysis, separate color images of Cy2- and Cy3-labeled end plates and nerve fibers were created through the 2 different filter sets using a CCD video camera system (Optronics Engineering Model DEI-470, Goleta, CA) combined with the image analyzing software Optimas 6.5. (Optimas Corporation, Bothell, WA). The generated masks of each Cy2-labeled end plate was superimposed over the Cy3 image. With this approach, all end plates stained by Cy2 or Cy3 alone and double labeled with Cy2 and Cy3 could be identified. The quality of end plate innervation was evaluated by a semiquantitative approach in each section: 0 points if the end plate was labeled only by Cy2 (only bungarotoxin labeling of the end plate), 1 point if a Cy3-labeled regenerating nerve fiber was seen in the surrounding of the Cy2-labeled end plate without contact, and 2 points if the Cy3-labeled nerve fiber had direct contact to the Cy2-labeled end plate. The mean score of all sections per animal was calculated.
Surgical Procedures: BBA
The animals were anesthetized with ketamine hydrochloride (100 mg/kg body weight) and xylazine (5 mg/kg) intraperitoneally. All surgical procedures were performed under an operation microscope. To perform the BBA, the buccal branch of the right facial nerve (innervating the whisker pad musculature) was exposed, transected, and immediately resutured end-to-end with 1 epineural atraumatic 11-0 suture (Ethicon EH 7438G, Braunschweig, Germany. To prevent regrowth of the marginal mandibular branch into the distal buccal nerve stump, the marginal mandibular branch was ligated. 9 Finally, the wound was closed by three 4-0 skin sutures. A total of 6 and 10 animals of group B and C, respectively, received a BBA, a BBA plus BTX injection in the ipsilateral whisker pad, a BBA plus BTX into the contralateral whisker pad, or only a BTX injection without BBA (for injection method, see above). For BTX injection, the optimal dose of 20 MU was used.
Sequential Preoperative and Postoperative Retrograde Fluorescence Tracing
For the sequential preoperative and postoperative retrograde fluorescence labeling of the facial motoneurons, Fluoro-Gold (FG) and Fast Blue (FB), respectively, were used. FG was purchased from Fluorochrome Inc, Denver, CO, and FB from EMS-Chemie, Groß-Umstadt, Germany. Under diethylether anesthesia, 10 days before surgery, 1 mg FG dissolved in 100 µL distilled water containing 2% dimethyl sulfoxide (DMSO) was injected in the whisker pad muscles at the midpoint between the 2 dorsal vibrissal rows. 11 Also, 28 days after facial nerve surgery, all rats of group B received injections of 1% FB in 100 µL distilled water with 2% DMSO into the whisker pad musculature, exactly at the same site of earlier FG injection.
Then, 10 days after the postoperative labeling, the rats were transcardially perfused with 0.9% NaCl in distilled water for 60 s followed by a fixation with 4% formaldehyde in 0.1 M phosphate buffer, pH 7.4, for 20 minutes under deep ether narcosis. After removal of the whole brains, the brainstems were dissected and cut coronally in 50 µm sections on a vibratom. The sections were observed on a Zeiss Axioskop 50 epifluorescence microscope through a custom-made bandpass filter set for FB (no. F31-000; excitation D 436/10; beam splitter 450 DCLP; barrier filter D470/40; AHF Analysentechnik), which allows recognition of FB-labeled motoneurons only. Observations through a custom-made HQ-Schmalband-filter set (no. F36-050; excitation D 369/40; beam splitter 400DCLP; barrier filter HQ 635/30; AHF Analysentechnik) visualized all motoneurons containing FG. This filter combination restricts the fluorescence cross-talk between FG and FB to a minimum and allows an optimal quantitative analysis of FG + FB double-labeled neurons. 11 For the image analysis, separate color images of FG and FB retrogradely labeled facial motoneurons were created through the different filter sets using a CCD video camera system (Optronics Engineering Model DEI-470, Goleta, CA) combined with the image analyzing software Optimas 6.5 (Optimas Corporation, Bothell, WA). The generated masks of FG-labeled cells were superimposed over the FB image of the lesioned facial nucleus. With this approach, all cells stained by FG, FB, and double-labeled by FG + FB could be readily identified. The labeled cells were counted on the computer screen. 12 Using the fractionator principle in the 50-µm thick sections, all labeled motoneurons with visible cell nuclei were counted in every third section through the facial nucleus on the operated and unoperated sides. 13
Functional Analysis of Facial Nerve Regeneration
The mystacial vibrissae of the rat are innervated by the buccal branch of the facial nerve. 14 The method of video-based motion analysis of vibrissae motor performance has been thoroughly established and tested in a series of recent experiments. 10,15,16 Briefly, only 2 large vibrissae of the caudal C-row on each side of the face were used for biometric analysis. Recently, we have shown that such a cutback is sufficient to obtain an objective reflection of whisking behavior. 16 Under light ether anesthesia, all other vibrissae of animals in group C were clipped with small fine scissors. Videotaping of the whisking of C-row vibrissae followed. Using a digital camcorder (Panasonic NV DX-110 EG), animals were videotaped for 3 to 5 minutes during active exploration. After calibration, video images of whisking behavior were sampled at 50 Hz (50 fields/s, video camera shutter opened for 4 ms). For analysis of whisking biometrics, 1.5-s sequence fragments from each animal were selected and captured by a 2D/Manual Advanced Video System (Motus 2000, PEAK Performance Technologies, Inc, Englewood, CO). The geometrical model consisted of 3 reference points (for details see Tomov et al 16 ): (1) a point in the medial sagittal line (perpendicular to a line connecting both orbits) close to the end of the nose, (2) a point corresponding to the medial angle of the left orbita, and (3) a point corresponding to the medial angle of the right orbita. Each vibrissa was represented in the spatial model by 2 points: its base and a point on the shaft 0.5 cm away from the base. Applying this model, we were able to collect and evaluate data on the following parameters: (1) protraction (ie, the forward movement of the vibrissae) measured by the rostrally open angle (in degrees) between the midsagittal plane and the hair shaft (maximal protractions are represented by minimal angle values); (2) whisking frequency, that is, cycles of protraction and retraction (backward movement in seconds); (3) amplitude (the difference between maximal retraction and maximal protraction in degrees; (4) angular velocity during protraction in degrees/s; and (5) angular acceleration during protraction in degrees/s2 (results not shown).
Statistical Analysis
Data are presented as mean values ± standard deviation. For all parameters, the data from all 4 experimental groups were tested in a 1-way analysis of variance (1-way ANOVA) procedure for overall experimental effects. If significant effects were detected (P < .05), comparisons of all groups were performed using the Bonferroni post hoc test at a significance level of P < .05. For analysis, SPSS software, version 17.0 for Windows (SPSS Inc, Chicago, IL) was used.
Results
Optimal Dose of BTX for Temporary Facial Palsy
The paralysis of the whisker pad was complete 3 days after BTX injection in all animals, independent of the chosen dosage—that is, even a dosage of 5 MU BTX revealed this effect. Using image movement analysis, a significant decrease in whisker movements was seen just 2 days after injection (detailed data not shown). The contralateral side was also paralyzed after injection of 40, 60, or 80 MU. The animal died 21 days after injection of 80 MU. Using 5 to 20 MU, the paralysis was completely reversible clinically after 35 days, and image analysis revealed a recovery to normal values for all parameters (frequency, amplitude, angular velocity, and angular acceleration) 56 days after BTX injection. The effect of 20 MU was confirmed in a second animal; 20 MU were chosen as dosage for an optimal effect without side effects.
Chemical denervation of the levator labii superioris muscle was already complete 2 days after BTX injection. The first reinnervated end plates were seen after 7 days (mean score = 0.25), and the reinnervation of all end plates was complete after 28 days (mean score = 1.75; Figure 1).
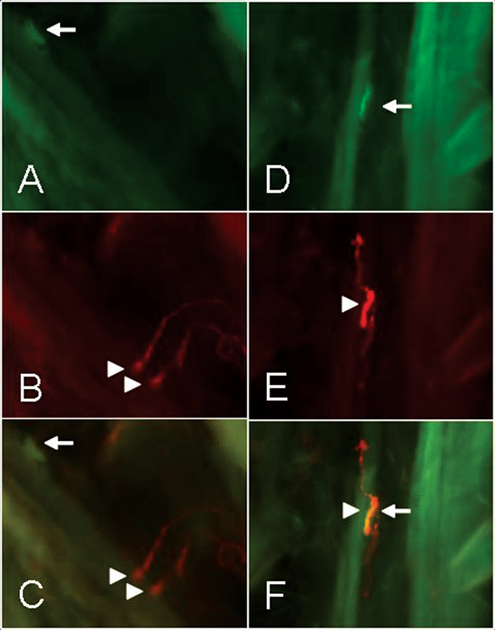
Neuromuscular junctions of the rat whiskerpad muscles 10 days after (A, B, C) and 84 days after botulinum toxin injection into the whiskerpad (D, E, F). Fluorescence labeling of the nerve terminals with alpha-bungarotoxin of the nicotinergic acetylcholine receptors (A, D) and of the nerve axons with antineuronal SY38 antibody (B, E). Superposition of the pictures in C and F. Longitudinal 50 µm cryostat sections 250x.
Analysis of Quality of Axonal Reinnervation
Neuronal labeling 28 days after BBA showed that all FG-labeled neurons were located in the lateral facial subnucleus (Figure 2). The distribution pattern of the FB-labeled neurons innervating the whisker pad musculature after BBA was changed. We observed neurons projecting to the whisker pad musculature, which were located outside the lateral facial subnucleus in other subnuclei. The quantitative estimates revealed no postoperative loss of neurons but a hyperinnervation of the whisker pad—that is, on average, about one third more motoneurons innervated the whisker pad after surgery in comparison to the situation prior to surgery (Table 2, Figure 3A). However, only 27% of these motoneurons were double labeled; that is, only 27% of them belonged to the original motoneuron pool and reinnervated the correct target; the rest were ectopic nerve cells from other subnuclei. The number of double-labeled motoneurons was significantly lower than after BBA plus ipsilateral or contralateral injection of BTX or after BTX injection without surgery (ANOVA, Bonferroni P < .001).
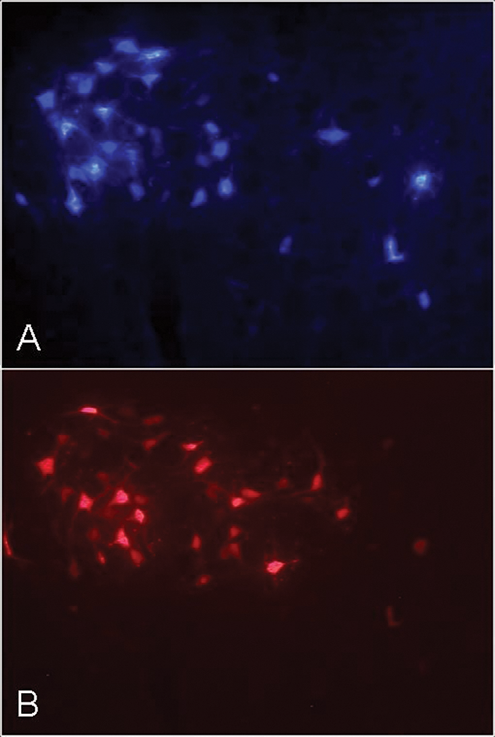
Facial motoneurons in the brainstem of the rat 38 days after buccal-buccal nerve repair and sequential retrograde fluorescence tracing with Fluoro-Gold (A) 10 days before surgery and Fast Blue (B) 28 days after surgery. Coronal 50 µm vibratom sections; 50x.
Recovery of Selective Reinnervation of the Whisker Muscles Revealed by Sequential Preoperative and Postoperative Retrograde Fluorescence Tracing
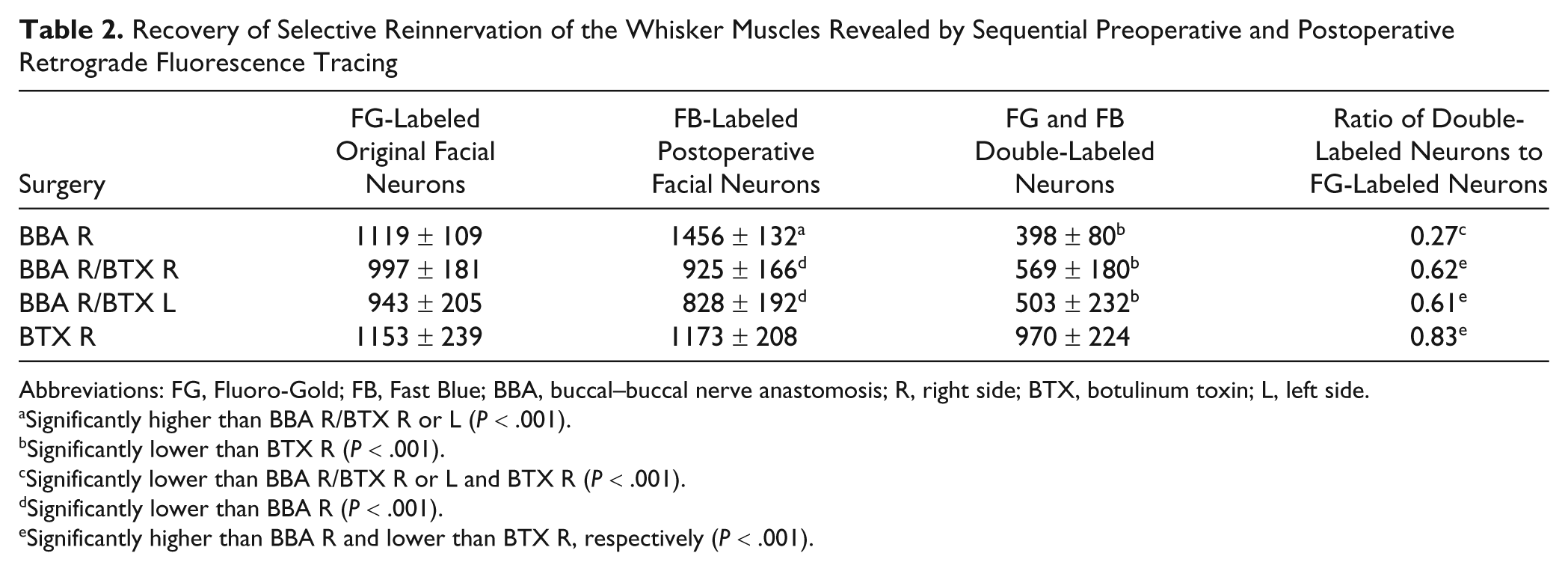
Abbreviations: FG, Fluoro-Gold; FB, Fast Blue; BBA, buccal–buccal nerve anastomosis; R, right side; BTX, botulinum toxin; L, left side.
Significantly higher than BBA R/BTX R or L (P < .001).
Significantly lower than BTX R (P < .001).
Significantly lower than BBA R/BTX R or L and BTX R (P < .001).
Significantly lower than BBA R (P < .001).
Significantly higher than BBA R and lower than BTX R, respectively (P < .001).
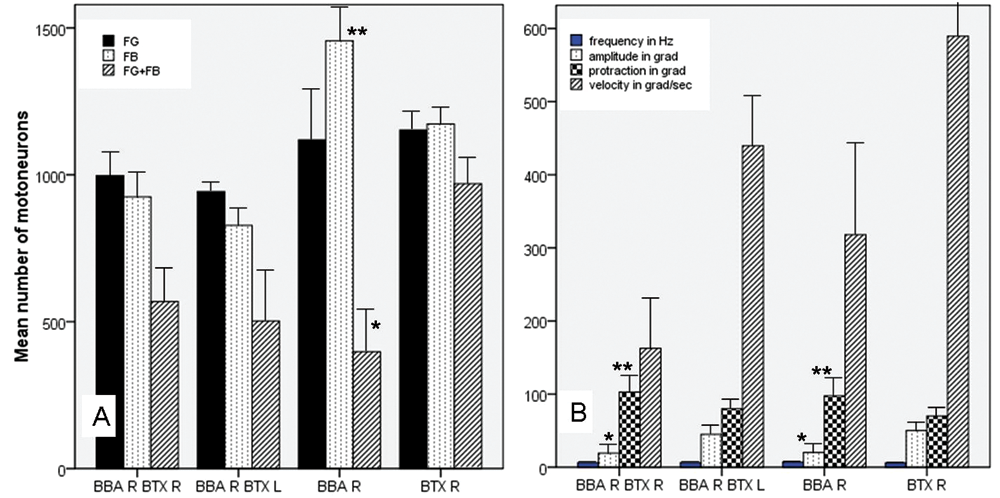
Comparison of the results of retrograde tracing (A) and whisking behavior (B) between the different experimental groups (mean values and standard deviation). To avoid confusion, only the statistically significant differences (P < 0.001) of the comparisons to the BTX R group are indicated; all values are shown in Table 2 and 3; * = significantly lower than BTX R; ** = significantly higher than BTX R.
Before BBA plus ipsilateral or contralateral injection of BTX, the FG-labeled motoneuron pool was correctly localized in the lateral facial subnucleus. Also after BBA plus ipsilateral or contralateral injection of BTX in the whisker pad, ectopic innervation of the whisker pad was seen, but the (hyper-)innervation was significantly reduced (see Table 2; ANOVA, Bonferroni P < .001). Furthermore, the rate of correct reinnervation increased significantly in comparison to the BBA group without any other manipulation (ANOVA, Bonferroni P < .001).
After injection of BTX into the right whisker pad without any other surgery, all retrogradely labeled neurons (FG, FB, and FG + FB) were localized exclusively in the lateral facial subnucleus. A misdirected reinnervation was not seen, which means that the BTX injection did not change the whisker pad innervation (Table 2).
Functional Analysis of Facial Nerve Reinnervation
In normal intact Wistar rats, the mystacial vibrissae are erect, with anterior orientation. During exploration, they move forward and backward with a frequency of about 6 Hz. The angle of maximal protraction of the vibrissae is about 70°. The amplitude, that is, the difference between maximal protraction and maximal retraction, of whisking is about 50°, and the movements have a sagittal angular velocity of about 530°/s. 16 After any surgery, the whisking behavior stopped immediately as well as within 2 days after BTX injection without any further surgery. In all experimental groups, whisking behavior improved until 56 days and plateaued until 84 days after surgery (data not shown). The whisking behavior 84 days after different interventions is summarized in Table 3 and Figure 3B.
Functional Recovery of Whisking Revealed by Digital Motion Analysis 84 Days After Surgery
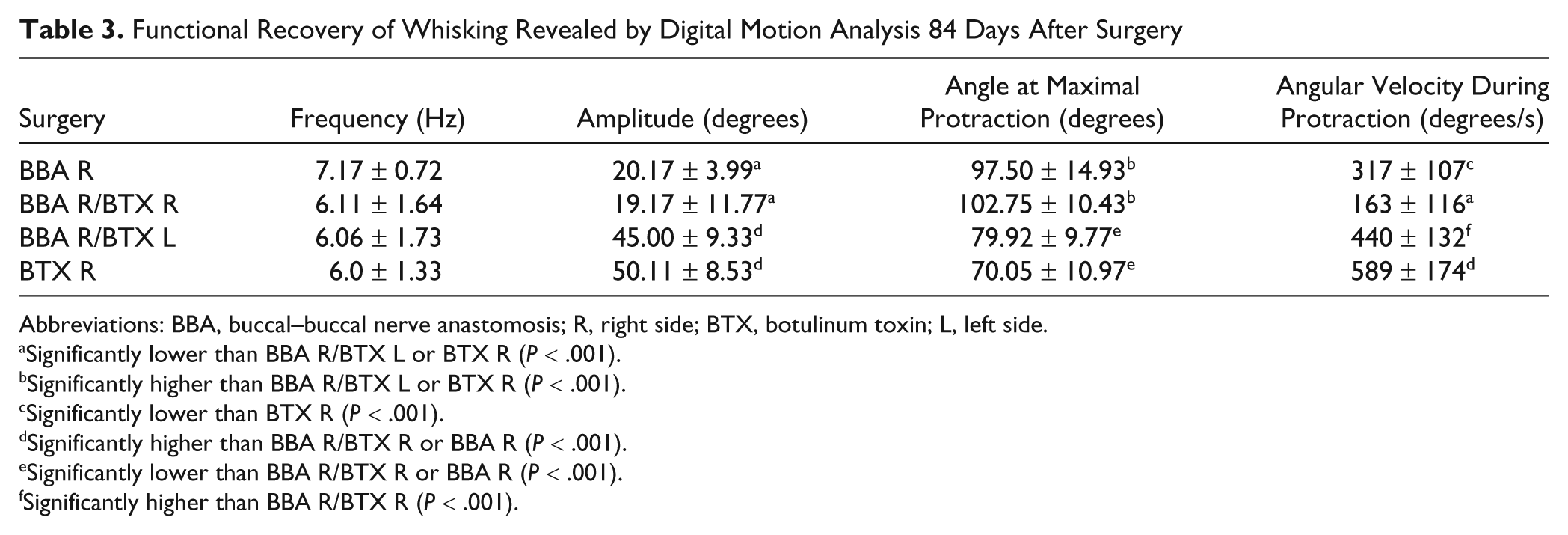
Abbreviations: BBA, buccal–buccal nerve anastomosis; R, right side; BTX, botulinum toxin; L, left side.
Significantly lower than BBA R/BTX L or BTX R (P < .001).
Significantly higher than BBA R/BTX L or BTX R (P < .001).
Significantly lower than BTX R (P < .001).
Significantly higher than BBA R/BTX R or BBA R (P < .001).
Significantly lower than BBA R/BTX R or BBA R (P < .001).
Significantly higher than BBA R/BTX R (P < .001).
After BBA, the average whisking frequency was slightly enhanced to 7.17 Hz, but there was no significant difference from the other animal groups (Table 3). The amplitude was significantly reduced after BBA in comparison with BBA plus contralateral BTX injection or with BTX injection without surgery (ANOVA, Bonferroni P < .001). Concerning the whisking amplitude, no difference was seen between the BBA and the BBA plus ipsilateral BTX injection group. The angle at maximal protraction after BBA was significantly higher than after BBA plus contralateral BTX injection or with BTX injection without surgery (ANOVA, Bonferroni P < .001). Also, for this parameter, no difference was seen between the BBA and the BBA plus ipsilateral BTX injection group. The angular velocity during protraction of the vibrissae was decreased after BBA in comparison with BTX injection without surgery (ANOVA, Bonferroni P < .001). There was no significant difference as compared with the other animal groups.
BBA plus ipsilateral BTX injection had different effects compared with BBA plus contralateral BTX injection (Table 3). After ipsilateral injection, the amplitude remained significantly low, the angle at maximal protraction high, and the angular velocity low in comparison with BTX injection without surgery (ANOVA, all Bonferroni P < .001). In contrast, after BBA plus contralateral BTX injection (Figure 4), all parameters reached the values after BTX injection without surgery and were significantly better than after BBA and ipsilateral BTX injection or BBA without additional injection (ANOVA, all Bonferroni P < .001, except for angular velocity).

Projection of the functional analysis of the vibrissae movement recovery on photography of a rat with measurement of angles, angular velocity, and angular acceleration of the intact and operated side during protraction.
In summary, after BTX injection without surgery, normal whisking was observed 84 days after the injection (Table 3). This functional recovery was significantly better than that observed after BBA alone or BBA plus ipsilateral BTX injection (ANOVA, all P < .001), but not in comparison with BBA plus contralateral BTX injection.
Discussion
The methodological approach used here has several advantages and a few disadvantages. The combination of morphological and quantitative evaluation of selective reinnervation of the target musculature by retrograde fluorescence tracing and functional assessment by digital motion analysis of the vibrissae allows a precise estimation of the quality of facial nerve regeneration. 3,16-18 Previously, we have shown that terminal sprouting at the end plates of the target musculature is also an important factor influencing the degree of defective healing after nerve regeneration. 3 This and the role of the supraspinal centers controlling locomotion of the whiskers 19-23 was not analyzed in the present study. Hence, the effects of the BTX treatment on these localizations have to be studied in future investigations. Nevertheless, the application of BTX had a strong effect on collateral sprouting, enhancing the correct reinnervation of the whisker pad from 27% to 61% after facial nerve reconstruction. This effect was much more pronounced than the one previously observed after contralateral deafferentation by resection of the infraorbital nerve when only 41% of the motoneurons innervating the target belonged to the original neuron pool. 9
The enforcement of correct reinnervation of the original target may explain the functional improvement of postlesional whisking only in part. First, ipsilateral injection of BTX also improved the degree of proper motoneuronal reinnervation, but the functional results were significantly worse compared with contralateral BTX injection. Second, it has been shown that functional reinnervation after facial nerve reconstruction can be good even if collateral sprouting remains abnormally high as, for example, in blind rats in which neocortical plasticity appears to be augmented. 21 Also, the effect of mechanical stimulation of the whiskers during the time course of regeneration was mainly related to the decreased pathological terminal sprouting at the end plates. 19 We will elucidate the effect of BTX during facial nerve regeneration both on terminal sprouting and on the ipsilateral and contralateral sensorimotor cortex in future studies. From intracortical microstimulation analysis, we see that the motor cortex reorganization starts within 1 day after BTX injection in the whisker pad, is effective for 6 days, and is mainly reversible after 12 days. 24 After facial nerve axotomy and repair, reorganization is seen for about 4 weeks. 24 We speculate that the BTX-induced temporary inactivation of the contralateral motor cortex representation areas causes an increased activation of the ipsilateral hemisphere leading to better functional results. In our model, ipsilateral motor cortex means the (contralateral) motor cortex to the facial nerve lesion site. The increased activation might simply reflect the removal of transcallosal inhibition from the contralateral hemisphere, as has been shown in stroke patients, 25 or active compensatory activity especially in the dorsal premotor cortex responsible on the non-BTX side supporting the functional motor reorganization and regeneration, as has been shown by cortical after transcranial magnetic stimulation. 26
The approach of using BTX to influence the contralateral side simultaneous to the facial nerve regeneration on the ipsilateral side has 2 important differences in comparison to previous studies where we used a deafferentation of the contralateral side by resection of the infraorbital nerve. 8,9 Both manipulations hinder normal facial motor nerve function on the nonlesioned side. But first, deafferentation causes a permanent blockage of the sensory input, whereas BTX injection leads to transient block of the motor output. The present results confirm that the effects of the neurotoxin are completely reversible (for a review see Meunier et al 27 ). The transient paralysis of the contralateral side was sufficient to affect the ipsilateral side, as shown in this study. Furthermore, it is unlikely that a permanent paralysis would yield better results because facial nerve regeneration in the rat is completed within 28 days after BBA, and the results are stable for at least 1 year after surgery. The transient paralysis approach facilitates the translation of these experimental results from the bench to the bedside because BTX is widely used in clinical routine for applications involving the face, such as in the treatment of blepharospasm or hemifacial spasm, with transient effect and low morbidity. 28
The second important difference is the effect of the peripheral manipulation on the sensorimotor cortex. Whereas BTX injection (transiently) copies the effects of facial nerve axotomy on short-term motor cortex reorganization, input deprivation by infraorbital nerve resection does not influence the cortical reorganization. 29 This would imply no or a much lower deregulation of the interhemispheric inhibition and no or a much lower activation of the contralateral hemisphere that is reorganized to influence facial nerve regeneration. Furthermore, BTX does not hinder the sensory input to the whiskers. We have shown in a series of experiments that the sensory input, especially the enforcement of the sensory input by mechanical stimulation of the whiskers, is very important for good functional facial nerve regeneration. 19,30 For other targets, such as forearm and hand muscles, it has been shown that sensory input modulates excitability in both motor cortices simultaneously. 31 Again, it becomes obvious that in future studies, we need to analyze in more detail the cortical effects of our approach.
Footnotes
Acknowledgements
This study was supported by the Köln-Fortune Forschungsprogramm (O.G.-L.). The skilful technical assistance of N. Lange is greatly appreciated.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: the Köln-Fortune Forschungsprogramm (O.G.-L.).
