Abstract
Background:
Persons living with HIV (PLWH) need practical tools to self-manage their condition.
Methods:
We conducted a proof-of-concept study among PLWH to assess whether patients could learn to use a personal health record (PHR) on a hand-held device (iPod Touch) to manage their condition. We began individual trainings and later adapted this to group training. We assessed usability, acceptability and also effects on self-efficacy for treatment adherence using the HIV Treatment Adherence Self-Efficacy Scale (HIV-ASES).
Results:
Nine PLWH participated in the individual training and 29 participated in the group sessions. The participants were largely middle aged, low-income and of racial/ethnic minorities. The sessions were well attended and participants fully engaged in tasks and shared learning. Most participants stated they intended to use the PHR and reported improved self-efficacy in treatment adherence (P = .05) particularly on the integration of treatment adherence into one's routine (P < .02).
Conclusions:
Training PLWH in use of a handheld PHR shows promise.
Introduction
Among countries where combination antiretroviral therapy (cART) is widely available, life expectancy for persons living with HIV (PLWH) has dramatically increased over the past 15 years. 1,2 High levels of patient adherence to treatment are required for patients to maximize their quality of life (through improved immune function), to achieve near normal life expectancy, 3,4 and to reduce transmission of HIV to others. 5 This level of adherence requires effective self-management. 4 Self-management means that patients assume an active and informed role in key aspects of their condition in collaboration with their personal clinician. 6 Personal health records (PHRs) whereby patients assume ownership of their own health information including tracking their test results, cancer prevention, medications, and health care appointments represents a potentially important tool for enabling effective self-management. 7 Beyond this, the self-reliance and motivation engendered by successful self-management can also facilitate the adoption of healthier lifestyle practices such as optimizing diet, exercise, and avoidance of harmful behaviors (smoking cessation, etc). 6,8
Many PLWH confront various barriers to self-management, such as establishing their own goals for management of their chronic condition including factors that impede daily adherence to cART. Examples of barriers include anxiety and depression, forgetfulness, fear of disclosure of one’s HIV status, substance abuse, complicated medication regimens, side effects from cART, and work/family responsibilities. 9 Common facilitators to self-management and medication adherence for PLWH within developed countries include positive attitude and experience toward cART, presence of high self-worth as a person, acceptance of HIV status, recognition of the need for strict medication adherence, use of reminder tools, and having a simple treatment regimen. 9
Studies have demonstrated that patient involvement in their own care leads to better outcomes, 10,11 and empowering patients with information so that they can play a key role in developing and managing their own health care is one of the driving forces behind providing patients access to their own medical information. However, access to private computers where patients can securely review their medical information is not available to all. Handheld devices can provide secure access to the patients’ PHR, but it is unknown whether this approach is acceptable and feasible to patients with low literacy level, who often lack experience using computers.
The objective of this study was to conduct a pilot study to establish feasibility and acceptability of a technology-enabled self-management program for PLWH. We chose to use the iPod Touch (an iPhone without standard telephone capability) for this pilot because of its low cost, ease of use, and its direct applicability to iPhone/iPad devices that may be preferred by some users. Following, we describe the development of this program and report our preliminary findings.
Methods
Context
The study was conducted using a community-based participatory research approach (CBPR). The CBPR involves active engagement of community members in all aspects of the research process. 12,13 Our partnership membership in Rochester, New York, included PLWH, HIV physician leaders, case managers, outreach workers, and researchers from the University of Rochester Developmental Center for AIDS Research (D-CFAR). Partnership members identified the need and assisted in the design of the intervention, the recruitment of participants, the trainings, and in the interpretation of the findings.
Selection of Handheld Device
The iPod Touch was selected for ease of use and because it presented the lowest cost option at the time among smart devices. In addition, at the time our project began, user-friendly PHR applications were only available for iPods/iPhones as opposed to other handheld devices. Moreover, most PLWH in our community use standard cell telephones that cannot operate PHR applications, even if such applications were available. In addition, the iPod Touch is Internet-enabled, allowing free Internet access to health information through “hot spots.” It is not encumbered by the eligibility and cost requirements for a “smart” telephone contract. Finally, we needed to choose a single device and a single PHR to facilitate training. Handheld devices were purchased through grant support and provided to participants to keep when they completed the training.
Selection of PHR Application
Partnership members helped identify low-priced, easy to use PHR applications for the iPod. The PHRs allow persons to enter, store, and retrieve medical information that is password protected. Typically, PHRs include fields to store information regarding medications, tests and procedures, allergies, preventive care, and documents. They also offer options for appointment and medication reminders. We piloted 2 PHR applications, Capzule PHR and My Medical. Both were available for less than $5 on iTunes. At the time our program began, these were the only PHR applications we identified. We selected MyMedical based on user feedback from one-on-one trainings; most participants found it somewhat easier to learn to use.
Development of Training
Training methods were developed and refined by conducting one-on-one training sessions with 9 PLWH referred by clinicians in our partnership. These trainings focused on learning how to use the handheld device and the PHRs. We used experience from these individual training sessions to guide development of the group training, both in terms of content deemed useful by trainees, format (hands on with guided instruction), and pace (ensuring competence of 1 step before proceeding to the next). Once the one-on-one training was completed and the PHR was selected, we began recruitment for our pilot study.
Sample and Recruitment
Participants were recruited through partnership member referrals and flyers posted at the partnership medical offices. We applied minimal inclusion and exclusion categories for participation. Participants were required to be adult (18 years or older) PLWH and receiving care at 1 of the 5 participating clinical sites. Participants were excluded if they could not read English or lacked capacity to provide informed consent. We aimed to recruit 10 PLWH per group and conducted 3 groups in addition to the individual training sessions. One hundred and thirty patients called to express their interest. From these, 40 were randomly selected and 80% consented to participate.
Training Curriculum
The 6-week training curriculum was developed collaboratively with partnership members. The project coordinator served as the primary trainer. Case managers, outreach workers, and participants from previous sessions served as assistant trainers. Space for classes was provided by organizations participating in the partnership. Training locations were conveniently located and offered free wireless Internet access.
Classes were for 90 minutes. They included an overview of the topic of HIV self-management including cancer reduction. This first class addressed (1) basic HIV pathogenesis; (2) cancer risk among PLWH and risk reduction strategies; and (3) the role of medication adherence in viral suppression. This was followed by 5 training classes in the use of the handheld device and the PHR. Instruction was step-by-step using a projected image from an iPad (essentially a larger version of an iPod with projection capabilities). Participants performed specific device tasks (creating a password and accessing the Internet) and PHR tasks (creating a password, entering personal health data, etc), during each class. Training sessions also included how to identify free hot spots (where wireless signals were located), how to find reliable health-related Internet sites, and where to find free applications for smoking cessation and physical fitness.
Participants rehearsed tasks (completed homework assignments) between the weekly classes. The trainers reviewed each participant’s task competency at each session. Participants needing extra help to perform tasks were offered up to 3 one-on-one sessions with a trainer.
A participant from the first training group was selected to assist as a peer trainer. The peer trainer completed required training for human subjects protection before assisting other participants. Participants received an iPod Touch at the end of the training in order to use their PHR applications.
Measures
Baseline measures were assessed (by the project coordinator) 2 to 3 weeks before the first training session. Follow-up measures were assessed approximately 1 month after the last training session. Baseline assessment included participant sociodemographic characteristics (age, sex, race, marital status, ethnicity, education, and income), health literacy (using the REALM, health word test), 14 and health (self-rated using a 5-point scale). 15 We also assessed attitudes toward technology using the Attitudes Towards Computers scale, 16 a 6-item scale with a score ranging from 6 to 42; the higher score indicating a more positive attitude toward computers than a lower score. We assessed participants’ perceived self-efficacy for HIV treatment using the HIV Treatment Adherence Self-Efficacy scale pre- and posttraining. It is a 12-item scale with scores ranging from 10 to 120 that includes 2 subscales. 17 The first is a 9-item treatment integration subscale that assesses patients’ integration of medication adherence into their daily lives, for example, “In the past month, how confident have you been that you can integrate treatment into your daily routine?” The second is a 3-item perseverance subscale that assesses patient perseverance to adhere to the medications in the face of various challenges. Finally, we conducted assessments of usability posttraining. Participants rated various aspects of usability for the device and the PHR based on a series of 7-point Likert-type scale.
Statistical Analysis
The mean and standard deviation for age and percentages for other categorical variables were provided to describe the sample. We assessed pre- and postintervention scores for self-efficacy for adherence to treatment using paired t tests.
Results
A summary of the participant characteristics is shown in Table 1. Most participants were middle aged, single, male, minority, and had low levels of education and income. Nearly 30% had very low health literacy based on the REALM assessment. 14 Thirty percent reported their health as fair or poor. One participant in 6 reported no computer experience. One in 5 participants never used a computer and 1 in 3 never used e-mail. Nonetheless, computer anxiety was fairly low.
Characteristics of the Sample.
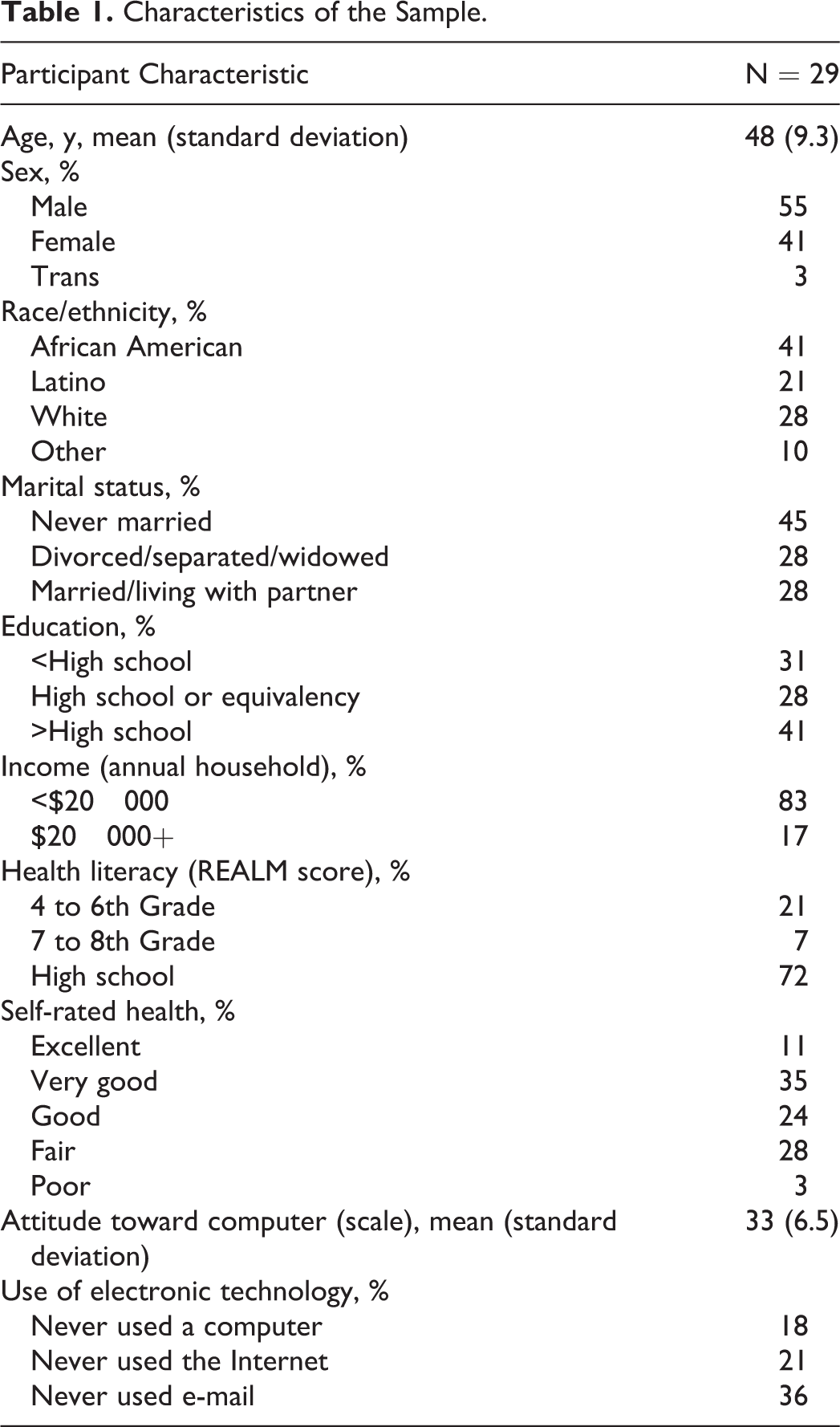
Of the original 30 participants, 28 completed all the training sessions. Participants were fully engaged in training sessions. Participants with greater proficiency readily assisted less experienced ones. Of all, 7 participants received 2 extra one-on-one trainings. Of all, 3 participants (10%) reported their device was lost or stolen at 30-day follow-up.
Participants reported improved self-efficacy for treatment adherence (Table 2). Most of these changes were driven by the subscale that measures self-efficacy for integration of medication taking into one’s routine. Participant’s written comments in posttraining surveys indicated their preference for the device calendar to track appointments and the PHR for medication reminders.
Pre- and Postintervention Scores for the HIV Treatment Adherence Self-Efficacy Scale (HIV-ASES).
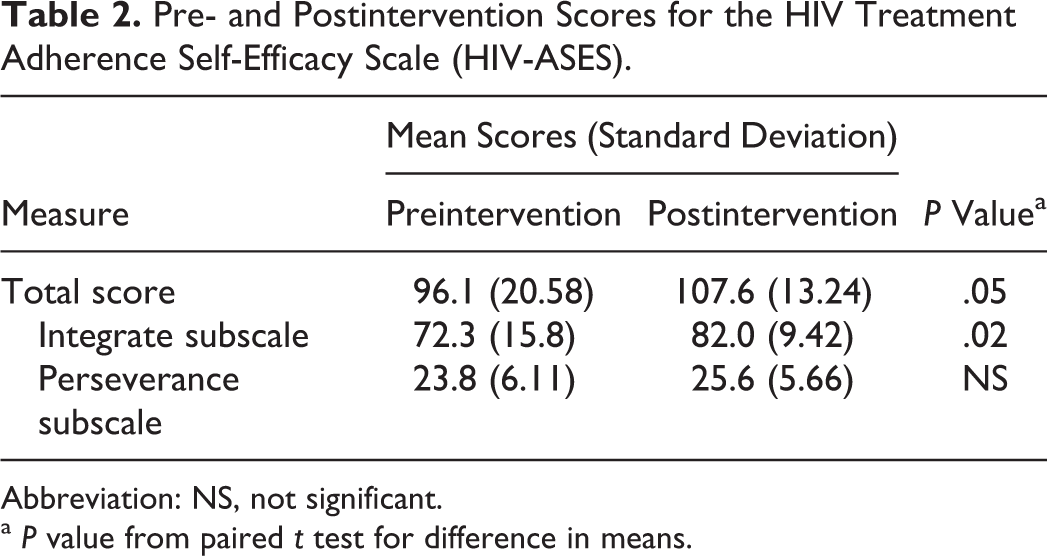
Abbreviation: NS, not significant.
a P value from paired t test for difference in means.
Participants reported high levels of satisfaction with the handheld device (Table 3) and with the PHR (Table 4). More than 90% reported they were satisfied with the ease of use of the device and the PHR. Most importantly, 100% of participants planned to use the PHR during or after their doctor visits.
Postusability Questionnaire for the iPod.

Postusability Questionnaire for the PHR.

Discussion
These findings show that it is feasible to train PLWH with fairly low levels of education (31% <high school) and low computer literacy in use of a PHR application for a handheld device. Specifically, we observed high levels of interest in the program, high rates of consent, and high levels of attendance and active engagement. Most participants were able to learn how to use the device and PHR. Only a small minority of participants required additional one-on-one help sessions. Participants rated the ease of use of the PHR device as high and all intended to use the PHR.
Our findings are consistent with the previous studies that focus on improving attitudes, knowledge, and skills relevant to health information technology for poor and minority persons. 18 –20 Kalichman et al developed an intervention based on a model of Information-Motivation-Behavioral Skills of behavior change that targeted PLWH with no prior Internet experience. 21 Using a randomized controlled study design, they found their training program improved the use of Internet for health, information coping, and social support. 20 Robinson and Graham reported that a 50-minute training session boosted self-assessed Internet skills among 18 PLWH. 22 Consistent with these findings, we found that group-based training was both feasible and acceptable.
Increasingly PLWH will have access to their health information, whether through printouts of their health records by their clinicians or through access to Internet-based portals that allows access to their PHRs. 7,23 However, the mere provision of patient health information data online alone is not sufficient. 24 Patients need the means to store and view these data in ways that are meaningful to them. Use of a CBPR approach that engaged PLWH, clinicians, and case managers in the design and development process of the training program and in the selection of the PHR helped optimize the use of the PHR. Use of handheld applications for this purpose will become more feasible as the price of handheld devices (including smart telephones) declines. Moreover, the finding that the training was associated with improvements in perceived efficacy for adherence, particularly for integration into one's daily routine, is consistent with a recent systematic review of the literature that concluded that tailored, multifunction technologies improve cART adherence. 25
Our findings are largely confined to feasibility and acceptability based on pre-/postmeasures. Further research using more rigorous study designs and objective outcomes is needed to determine whether this intervention is efficacious in promoting adherence.
In conclusion, our study suggests that training PLWH in use of PHRs on handheld devices is feasible and acceptable. Future research is needed to assess the impact on outcomes.
Footnotes
Acknowledgments
We thank URMC Cancer Center, AIDS Care, and Unity Health System for facilitating our group training sessions for PLWH, and Vi Luong for designing our brochure. The following individuals and organizations participated in the partnership and contributed to the success of this project: Julian Brown Jr,1 Catherine Bunce, RN, MS,2 Nayef El-Daher, MD, PhD,3 Steven Fine, MD, PhD, HIV C,4,5 Steve Marcus,6 Lizz Ortolani-Walls, MSW,1 and Renee Rivoli.3 The organizations are (1) AIDS Care, (2) University of Rochester Medical Center, Developmental Center for AIDS Research (D-CFAR), (3) Unity McCree McCuller Wellness Center, (4) University of Rochester Medical Center, Department of Medicine/Infectious Disease Division, (5) Anthony Jordan Health Center, and (6) Highland Hospital, Department of Family Medicine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Center to Reduce Cancer Health Disparities of the National Cancer Institute (3U01CA116924-05S1,S4) and in part by the University of Rochester Developmental Center for AIDS Research Grant P30 AI078498 (NIH/NIAID) and the University of Rochester School of Medicine and Dentistry.
