Abstract
This study was conducted to assess the prevalence of depressive symptoms, sleep disturbances, and subjective cognitive complaints in patients with HIV receiving highly active antiretroviral therapy. Participants completed the “Center for Epidemiological Studies Depression Scale” (CES-D) and a questionnaire on sleep disturbances and subjective cognitive complaints. Mean age of the 799 participants was 43.7 years and 67% were men. Adjusted prevalence of CES-D was 35.4% (95% confidence interval [CI]: 32.0-38.7), with no significant differences between gender and age groups. Sleep disturbances were more prevalent in older versus younger participants (74.0% [95% CI: 70.4-77.7] versus 63.3% [95% CI: 56.8-69.8]). Cognitive complaints were more prevalent in women (52.3% [95% CI: 46.4-58.2]) when compared with men (48.2% [95% CI: 44.7-51.6]). Hepatitis C virus coinfection was a strong predictor of depressive symptoms. Male gender and detectable viral load were independent risk factors for sleep disturbance. A higher CES-D score was an independent risk factor for sleep disturbance and cognitive complaints.
Introduction
In the pre-highly active antiretroviral therapy (HAART) era, depressive symptoms and mood disorders were known to be substantial problems for patients infected by HIV with or without AIDS. 1,2 Although there is some evidence indicating that with the introduction of HAART, the rates of mood disorders may have decreased, findings have been inconsistent, and mood disorders continue to be a substantial problem in both women and men with HIV. 3 –6 Major depression is one of the most common mood disorders in HIV-seropositive individuals, with rates of depression ranging from 8% to 67%. 7 The wide variation in rates across studies may depend on the patient populations studied and the method of assessment. Furthermore, several studies have shown that up to 85% of patients living with HIV/AIDS report some depressive symptoms that may not reach diagnostic criteria for a major depression diagnosis. 8 –13
Attention to HIV-associated depression is important since its presence may be associated with HIV disease progression and mortality, after controlling for CD4 counts and viral load. 14 Depression has also been shown to influence the quality of life 15 and medication adherence, 9,15,16 being an independent risk factor for nonadherence to HAART regimens. 17 –19 Conversely, depressive symptoms and lack of treatment adherence are considered independent predictors of HIV clinical progression and mortality. 16
Despite its high prevalence, HIV-associated depression is frequently unrecognized and undertreated. 20 As in other medical illnesses, physicians may fail to diagnose depression as they might consider it an expected reaction to illness or a declining functional status related to a major medical disease. 21
There are some other symptoms that deserve attention in HIV-positive individuals, due to the elevated rates reported and the possible relationship with depressive symptoms and/or the progression of the infection. Sleep disturbance is an important issue for HIV-infected patients, with prevalence reports of 30% to 40%. 22 Psychological morbidity seems to be a major determinant of insomnia, although other variables may play a role including antiretroviral medication, drug and alcohol use, and potentially a direct role of HIV in the brain. Sleep disturbance has been described in all stages of HIV-related illness and may lead to chronic fatigue, reduced physical and social functioning, and an overall reduction in quality of life. 23
HIV can cause a wide range of neurocognitive complications such as mental slowing, memory loss, and difficulties in complex tasks requiring executive functions. 24 After HAART, the prevalence of milder forms of HIV-associated neurocognitive disorders seems to have increased with reported prevalence between 20% and 50%. 25 –27 Many HIV patients report cognitive complaints in clinical interviews, and little data exist regarding the positive predictive value of cognitive complaints for the presence of minor cognitive deficits in HIV-positive patients. Subjective neurocognitive complaints are often correlated with depression or other psychological symptoms and do not seem to correlate with objective cognitive impairment as has been found in some medical conditions, such as multiple sclerosis and chronic fatigue syndrome. 28 ,29 Rourke et al 30 shows that depression in HIV-infected patients is associated with cognitive complaints, whereas those with more severe objective impairment tend to underreport cognitive difficulties.
The main objective of this study was to evaluate the prevalence of depressive symptoms in HIV-positive patients treated with HAART in Spain. Secondary objectives were to determine the prevalence of sleep disturbances and subjective cognitive complaints in this population. Additionally, independent risk factors associated with depressive and other central nervous system (CNS) symptoms were also analyzed.
Patients and Methods
Study Design
This was a probabilistic stratified multistage survey developed in Spain to determine the prevalence of depression (main objective) and other CNS symptoms (secondary objective) in HIV patients treated with HAART. The survey sampling frame was a national population of 60 000 HIV patients under HAART treatment, attending Spanish hospitals. In order to establish the prevalence of depressive symptoms in a representative probabilistic sample, we considered in advance a prevalence of depressive symptoms of 40%. A total of 573 surveys should be collected in order to determine the true prevalence with a confidence level, 1 – α, 0.95, and a relative error of 10%. Considering multistage design effect and nonresponse, sample size was increased up to 808 patients and the participants were recruited by 101 hospital and health centers in Spain. Prior to the sampling stages, the participating centers were stratified among the 17 Spanish Autonomous Communities (SAC). The number of centers enrolled per SAC was fixed proportionally to the number of HIV-infected patients censed in each SAC. In the first stage of sampling, centers in each SAC were randomly selected with probability proportional to the number of HIV-infected patient visits the last year. In the second stage, physicians were selected by simple random sampling among those attending HIV-infected patients in each selected center. In the third stage, each participating physician selected 8 HIV patients under treatment with HAART by systematic sampling among visits scheduled during the 1-month planned survey period per center. To maintain the probabilistic structure, physicians or patients who refused to participate were not reposited, and the survey was finally administered to 799 HIV-infected patients, from September to December 2008. The sampling fraction was 0.013 (0.011-0.021 depending on SAC considered).
Approval for the study was given by the Ethics Committee of Hospital Vall d’Hebrón (Barcelona, Spain), and written informed consent was obtained from each patient before survey administration.
Study Procedures
Demographic and medical data obtained from medical records included age, gender, time since estimated HIV infection, time since HIV diagnosis and/or AIDS diagnosis, current and nadir CD4 count, current HIV viral load, and time since hepatitis B virus (HBV), and hepatitis C virus (HCV) infection, if relevant. The use of concomitant drugs (antidepressants and HCV treatment) was also recorded.
The survey was self-administered and it was composed of 2 questionnaires. To measure depressive symptoms, we used the Spanish version of the 20-item Center for Epidemiological Studies Depression Scale (CES-D) developed by Radloff 31 and translated and validated by Gonzalez et al 32 in arthritis population and Vazquez et al 33 in the general population. Participants indicated how often, over the past week, they experienced each of the 20 symptoms described in the CES-D scale, ranging from 0 (never) to 3 (all the time). Scores of 16 or above on the total score are suggestive of clinical depression. 31 The second questionnaire was designed specifically for this study and collected patients’ subjective reports pertaining to sleep disturbance and cognitive complaints. Sleep disturbance was considered if the patient answered “yes” to any of the 3 questions about difficulties in falling asleep, broken sleep, and bad dreams or nightmares during the night. Cognitive complaints were considered if the patient answered “yes” to the questions about difficulties in concentration at work, studies, or homework, and/or reported to forget things very often. Finally, sexual desire was explored with a single question, “Do you have loss of or diminished sexual desire?”
Statistical Analysis
Summary data are shown as mean ± standard deviation or median and first to third quartiles (Q1-Q3) for quantitative data, and as frequency and percentage for qualitative data. Raw and adjusted prevalence with its confidence interval of 95% (95% CI) was calculated for CNS disturbances in the total sample, as well as by sex. In order to study the prevalence in age groups, the sample was segmented by age tertiles to conform 3 groups, <40 years, 40 to 46 years, and >46 years, of approximately the same size. Student t test was used to compare mean groups and chi-square to compare proportions. Logistic regression analysis was performed in a subset of 411 patients, in which information was available on all variables involved, in order to study the predictors of CES-D score ≥16, sleep disturbance, and cognitive complaints. The set of independent variables included gender, age (years), time from HIV diagnosis (years), HCV coinfection (yes/no), viral load (undetectable/detectable), CD4 counts (cells/mm3), nadir CD4 count (cells/mm 3 ), and CES-D score (the last one only in models with sleep disturbance and cognitive complaints as dependent variable). Bivariate odds ratio (OR) were calculated considering each independent variable and then a multivariate model was built, exploring with the forced entry method and then by conditional forward stepwise method, with probabilities of .05 and .10 for entry and exit variables, respectively. Statistical significance was set at P < .05. Statistical analyses were carried out using SPSS v15.0 software.
Results
Description of Participants
A total of 799 patients participated across 101 health centers. Baseline characteristics are shown in Table 1. Mean age of patients was 43.7 ± 8.7 years and 67.1% were men. In all, 152 patients (19.0%) had detectable HIV viral load (>50 copies/mL), 254 patients (31.8%) had HCV coinfection, and 40 patients (5.0%) were coinfected with HBV. Information about antidepressant treatment and CES-D score was collected on 696 patients. The use of antidepressant drugs was more prevalent in patients with CES-D score ≥16 (20%) compared to those with CES-D score <16 (2.6%; P < .0005).
Sample Characteristics

Abbreviations: HBV, hepatitis B virus; HCV, hepatitis C virus; Q1 to Q3, 1st to 3rd quartile.
Prevalence of Depressive and Other CNS Symptoms
Raw and adjusted prevalence and its 95% CI of depressive and other CNS symptoms are shown in Table 2. The CES-D score ≥16 was considered as the cutoff and was reached or exceeded by 280 patients (35.0%). The adjusted prevalence was 35.4% (95% CI: 32.0-38.7). No significant differences were found related to gender or age group.
Prevalence of Depressive Symptoms, Sleep Disturbance, and Cognitive Complaints in HIV-Positive Patients under HAART Treatment
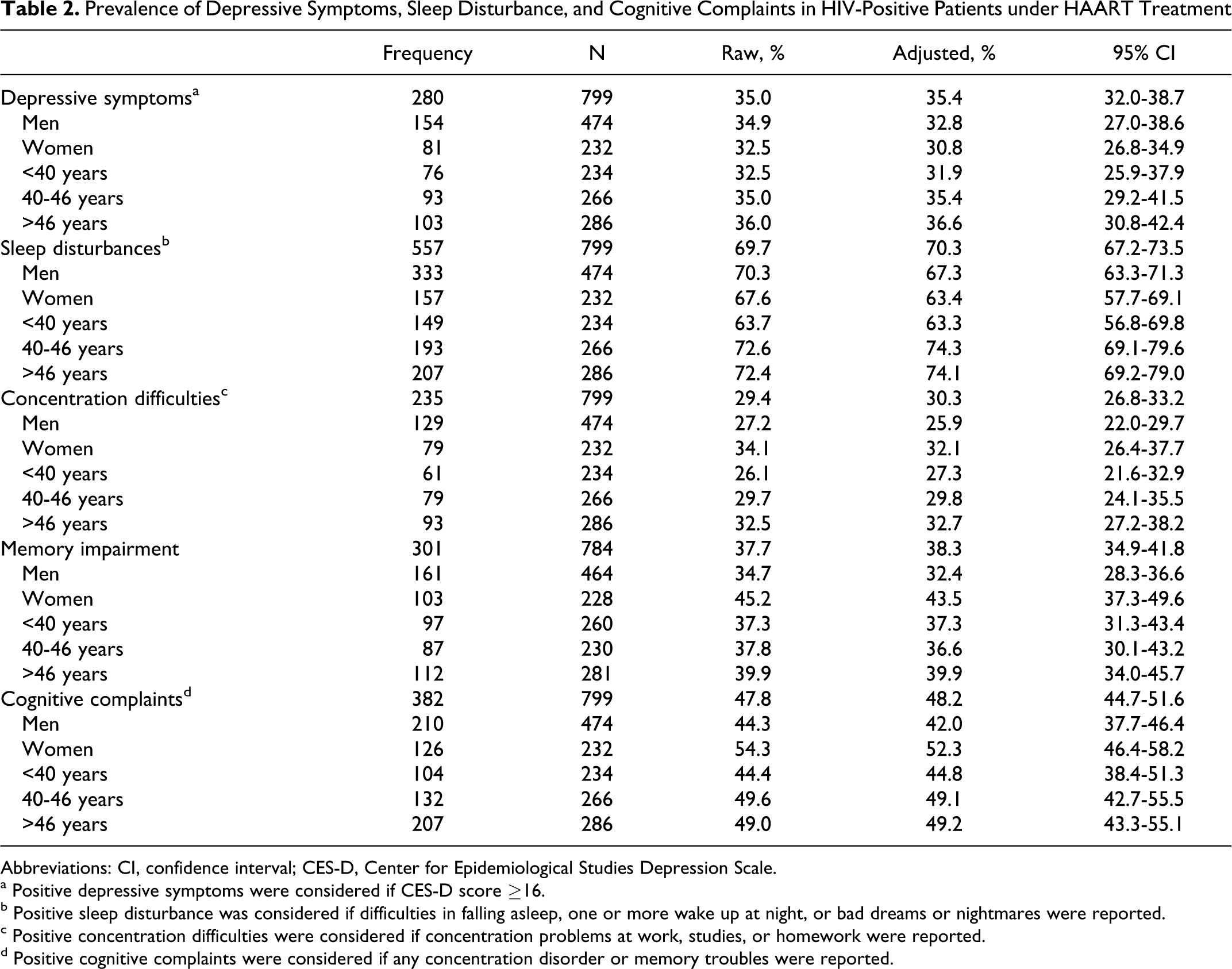
Abbreviations: CI, confidence interval; CES-D, Center for Epidemiological Studies Depression Scale.
a Positive depressive symptoms were considered if CES-D score ≥16.
bPositive sleep disturbance was considered if difficulties in falling asleep, one or more wake up at night, or bad dreams or nightmares were reported.
cPositive concentration difficulties were considered if concentration problems at work, studies, or homework were reported.
dPositive cognitive complaints were considered if any concentration disorder or memory troubles were reported.
On the basis of our questionnaire, the adjusted prevalence of sleep disturbance was 70.3% (557 of 706; 95% CI: 67.2-73.5) and the adjusted prevalence of cognitive complaints was 48.2% (382 of 706; 95% CI: 44.7-51.6). Concentration difficulties were reported by 235 patients, adjusted prevalence 30.3% (95% CI: 26.8-33.2), and memory difficulties were reported by 301 patients, adjusted prevalence 38.3% (34.9-41.8). With respect to sleep disturbances, there were no differences in prevalence by gender but were more frequent in patients older than 40 years compared with younger patients. Cognitive complaints (mainly memory difficulties) were more prevalent in women. Differences by age group were not found.
The relation of loss of or diminished sexual desire with other variables was studied. Loss of or decrease in sexual desire was independent of gender (P = .250) or HCV coinfection (P = .499). Patients with decreased sexual desire compared to patients with conserved sexual desire were older (45.6 ± 9.4 versus 41.9 ± 7.7; P < .0005) and showed higher mean CES-D scores (19.6 versus 11.2; P < .0005). Differences in time from HIV diagnosis were not found (P = .930).
Logistic regression analysis was done to study the prognostic risk factors for depressive symptoms, sleep disturbance, and cognitive complaints. Results of bivariate and multivariate models for each dependent variable are shown in Table 3. The HCV coinfection was an independent risk factor for CES-D score ≥16, OR 1.599 (95% CI: 1.025-2.429). Male sex (OR 1.853 [95% CI: 1.134-3.044]), detectable viral load (OR 2.069 [95% CI: 1.069-4.004]), and higher CES-D score (OR 1.091 [95% CI: 1.062-1.120]) were independent risk factors for sleep disorders. Higher CES-D score (OR 1.107 [95% CI: 1.081-1.113]) was an independent risk factor for cognitive complaints.
Bivariate and Multivariate Models of Association between Independent Variables and CES-D Score ≥16, Sleep Disturbance, and Cognitive Complaints

Abbreviations: CI, confidence interval; OR, odds ratio.
aMultivariate forced entry P values.
bMultivariate last step of forward stepwise entry P values and OR.
Discussion
We have designed a survey focusing on 3 of the most studied areas in HIV neuropsychiatry: depressive symptoms, sleep disturbance, and subjective cognitive complaints. We have found that in the HAART era, significant depressive symptoms based on the items of the CES-D scale are still very common in both HIV men and women in Spain (35.4% of the sample). Although there have been several studies in Europe, United States, and Africa regarding the prevalence of these symptoms, there is no current study in the Spanish HIV population. The prevalence of depression in patients with HIV/AIDS is higher than that in the general population, which has been established as 10.5% by the Global Burden of Disease study from the World Health Organization, 34 although this percentage may vary depending on age, gender, socioeconomic status, and/or comorbidity. 35 Ciesla’s meta-analysis 36 indicates that major depression rates for individuals with HIV infection are almost twice as high as in the general population.
Current studies in HIV-positive participants, most of them done in African countries, have found different rates of depressive symptoms ranging from 14% to 54%, probably as a result of different methodologies and the use of different instruments to assess depressive symptoms and different cutoff on the CES-D. 37 –39 The high prevalence of depressive symptoms in our study underscores the importance of screening for depression in outpatient clinics in Spain. Evidence shows that the prevalence of depression in the general population is higher among women than among men across all ages and ethnic groups. 40 –42 However, we have not found any difference between the prevalence of depressive symptoms and gender in our study, after controlling for the use of antidepressant treatment which was higher in women.
A single cause for the high prevalence of depression in the HIV population has not been identified, but a combination of genetic, biochemical, and environmental factors are hypothesized to contribute. 43 In addition, research suggests that HAART may affect mood as an adverse effect. 44 However, much has also been written about the role of HAART as a protective factor for the occurrence of depressive symptomatology. 45,46 In our study, we have not found any relation between viral load or markers of immune status (CD4 counts) and depressive symptoms and the only clinical variable that influences the risk of depression is HCV coinfection. Nevertheless, this effect cannot be attributed to the HCV medication because only 10.6% of the coinfected patients were taking peg-interferon plus rivabirine at the time of the study. This is in accordance with the results of other authors. 47 As social and psychological factors potentially play a greater role in the genesis of depression compared with the neuropathological effects of the HIV and/or HCV infection, these factors deserve further investigation.
Because depression may affect the global functioning of the participants and influence the progression of HIV infection, both detection and treatment of depression should be a major priority. However, one of our results is that at least 80% of patients with symptoms suggestive of clinical depression did not receive pharmacological treatment. This finding is consistent with the one found by Israelski et al 20 who showed that only 43% of the patients in their study were receiving concurrent psychiatric treatment.
We have found a higher prevalence of sleep disturbances (70.3%) compared with the prevalence of less than 20% in the general population. 48,49 The high prevalence could be explained by several specific factors of this population group such as HIV infection, HAART, and depressive symptoms. The signs and symptoms of depression are similar in HIV-infected and noninfected patients, although patients with HIV infection may more frequently have sleep and appetite disturbance. 50 In our study, a higher viral load and higher depressive symptomatology were independent risk factors for the presence of sleep disturbance, suggesting that there could be a double influence related to the virus and psychological morbidity. With respect to subjective cognitive complaints, 48.2% of the participants reported lack of concentration or memory difficulties, the last being predominantly in women. As in other studies, the only variable that was associated with the emergence of cognitive complaints is having more depressive symptoms. 51 Other clinical variables such as HCV coinfection, higher viral load, disease duration, and nadir and current CD4 count did not seem to be the risk factors for the cognitive complaints in our study. There is a need to include in future studies an objective neuropsychological assessment and to explore the relationship between subjective cognitive complaints, depressive symptoms, and objective cognitive functioning.
This study has several limitations. First, although CES-D is a useful screening tool, it frequently yields false positives independently of the population in which it is applied. 52 –54 Second, questions concerning sleep disturbance and cognitive complaints have been extracted from other studies 5,55,56 and applied in the absence of specific psychometric tests. Third, lack of information affects our study in 3 ways, (a) we did not collect information concerning the number and type of HAART and treatment duration that could have affected depressive and other CNS symptoms, (b) we have not used structured or semistructured diagnostic interviews to diagnose depression that would offer superior reliability and validity and would have increased the quality of our study, and (c) because the survey was completed in the waiting room, the missing data could not be collected later. Finally, interpretations of the predictive associations between risk factors and outcomes remain difficult and prospective studies are needed to demonstrate causality.
We consider this study as one of special relevance, first, because of the probabilistic nature of the survey and a relatively large sample size with a fraction sampling higher than 0.01; second, due to the current interest in the mental health of patients with HIV infection, which includes the need for increased identification of mental illness as well as provision of psychiatric services in an accessible setting.
In summary, we have shown that 35.4% of HIV-positive patients receiving HAART treatment in Spain report depressive symptoms suggestive of depressive disorders. Since the prevalence of depression in patients with HIV/AIDS is much higher than in the general population, it would be important to evaluate carefully in future prospective studies whether this high prevalence could be related to the virus, HAART, or other social factors. In addition, it is necessary to assess how depression affects medical outcome, compliance, quality of life, and high-risk behaviors. Given that HAART therapies have greatly increased life expectancy and transformed HIV infection into a potentially chronic disease, physicians face a major long-term challenge to anticipate and manage distress and psychiatric disorders, all of which could result from a chronic infectious disease that requires lifelong treatment. Successful treatment for depressive symptoms could influence the reported rates of sleep difficulties and cognitive complaints and may significantly improve the quality of life. HIV caregivers should be sensitive to the possibility of depressive symptoms and other CNS symptoms in order to detect them early and to provide individualized care to patients with HIV.
Footnotes
Acknowledgments
The authors acknowledge Dr Jesús Garrido for the statistical analysis of the study and Dr Fernando Sánchez-Barbero (HealthCo, Spain) for assistance in the preparation of this manuscript.
Declaration of Conflicting Interests
Esteban Ribera has received honoraria, speaker’s fees, consultant fees, and funds for research from Abbott Laboratories, Bristol-Myers Squibb, Boehringer Ingelheim, Gilead, Janssen-Cilag, MSD, Pfizer, Roche Farma, Schering Plough, and Glaxo Smith Kline.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
