Abstract
Background
Limited data are available on patterns of resistance mutations in pediatric patients in southern Africa, where HIV-1 subtype C (HIV-1C) predominates.
Methods
Retrospective chart review of pediatric patients. Nucleoside reverse transcriptase inhibitor (NRTI)- and nonnucleoside reverse transcriptase inhibitor (NNRTI)-associated resistance mutations quantified from population-based sequencing genotypic resistance assay results taken at time of first-line antiretroviral therapy (ART) failure (first-line ART = stavudine [d4T] or zidovudine [ZDV] + lamivudine [3TC] + nevirapine [NVP] or efavirenz [EFV]).
Results
Total number of patients with resistance assays analyzed is 45. Nucleoside reverse transcriptase inhibitor-associated mutation frequencies noted were M184V (n = 41; 91.1%); thymidine analogue mutations (TAMs; n = 20; 44.4%); >1 TAM (n = 9; 20%); TAM-2 pathway (n = 10; 22.2%); TAM-1 pathway (n = 7; 15.6%); TAM-1 and TAM-2 pathways (n = 3; 6.7%); K65R (n = 2; 4.4%); Q151M (n = 1; 2.2%); and L74V (n = 0; 0%). Nonnucleoside reverse transcriptase inhibitor-associated mutation frequencies noted were associated with notable resistance to either/both NVP and EFV (n = 40; 88.9%); K103N (n = 15; 33.3%); ≥1 mutations associated with etravirine (ETR) failure (K101E, Y181C, and G190A; n =20; 44.4%); and ≥2 notable NNRTI mutations (n = 12; 26.7%).
Conclusions
In this cohort, low-genetic barrier mutations were common, as were TAMs, including more than 1 TAM. Mutations compromising nonthymidine analogue backbones were rare, suggesting that it is likely that children who fail first-line NRTI backbones containing d4T or ZDV/3TC would still respond to abacavir (ABC), didanosine (ddI), and, for adolescents, tenofovir (TDF). Our data support the empiric continuation of 3TC in second-line regimens.
Keywords
Introduction
The extent of the HIV/AIDS epidemic in southern Africa is well appreciated, including its impact on children. Botswana has one of the world’s highest HIV prevalence figures, estimated at 25.0% among adults aged 15 to 49, including a large cohort of HIV-infected children (as many as 19 000 children aged 0-14). 1 Botswana’s national antiretroviral therapy (ART) program has been in place since November 2001. National pediatric treatment guidelines include standardized indications for ART initiation, first- and second-line drug regimens, and empiric management of first-line virologic failure. These, along with a strong commitment from the Government of Botswana and its partners, have facilitated a thorough response to pediatric HIV/AIDS in Botswana, with almost nearly all the 9000 children in need of ART receiving it. 1
Great strides in the prevention of mother-to-child transmission (PMTCT) have also been made in Botswana. The national PMTCT program was launched in 1999 and achieved nationwide coverage in late 2001. The single-dose NVP (sd-NVP) component of PMTCT for mothers and infants was introduced in November 2003. Currently, the national 2 mother-to-child transmission rate is <5%, and the future of pediatric HIV management in the country is increasingly recognized to be one of the challenges associated with the long-term administration of ART. 3 Implicit among these is the specter of HIV antiretroviral (ARV) drug resistance, a growing concern as pediatric ART programs realize scale-up in similar settings. Despite the threat of ARV drug resistance to treatment success in resource-limited settings, there is little pediatric data from the southern African region.
Most pediatric data on HIV-1 ARV drug resistance come from the western world, where HIV-1 subtype B (HIV-1B) predominates. 4 –8 Less is known about resistance patterns among HIV-1 subtype C (HIV-1C) in children, including in southern Africa, where HIV-1C is very common (approximately 99% of the subtypes in Botswana). 9,10
Existing data on patterns of resistance mutations in HIV-1C vary. Thymidine analogue mutations (TAMs) typically occur sequentially in 1 of 2 distinct pathways (TAM-1 [M41L, L210W, and T215Y] and TAM-2 [67N, 70R, 215F, and 219Q/E/N/R]), with TAMs accumulating in a given pathway overtime if thymidine analogue antiretrovirals (stavudine [d4T] or zidovudine [ZDV]) are continued in patients experiencing viral replication. Some data show unique patterns of TAMs (hybrid pathways) in HIV-1C in adults in Botswana, 11 whereas data from adults and children in South Africa and other HIV-1C predominant countries show mutations comparable with those found among HIV-1B viruses. 12,13 Most studies of HIV-1C mutations have shown high prevalence of common, low genetic barrier mutations such as M184V and nonnucleoside reverse transcriptase inhibitor (NNRTI). 14 –18 However, some have shown considerable prevalence of TAMs and other mutations associated with nucleoside reverse transcriptase inhibitors ([NRTIs] such as K65R, L74V, and Q151M) that can compromise the NRTI backbones used in second-line therapies. 19 –21
Generally, first-line ART for children in southern Africa has traditionally followed World Health Organization (WHO) recommendations of 2 NRTIs + 1 NNRTI, that is ZDV or d4T + lamivudine (3TC) + NVP or efavirenz (EFV); and 2 NRTIs + lopinavir/ritonavir (LPV/r) is currently recommended by WHO as first-line ART for infants and children less than 24 months who have been exposed to NNRTIs. 22 Abacavir (ABC) has been recently recommended by WHO for pediatric first-line regimens and has replaced ZDV or d4T in many regional programs, including Lesotho and South Africa 22 –24 ; however, it has yet to be included in first-line ART in Botswana, where standard pediatric first-line ART (when there is no history of NNRTI exposure through PMTCT or maternal treatment) is ZDV/3TC/NVP or EFV and pediatric second-line ART is d4T/ABC/LPV/r. 25 Given the limited availability of genotypic resistance assays in the region, most ART programs have relied on empiric first-to-second-line ART switches when children are failing ART and when ARV drug resistance is suspected; according to Botswana’s current HIV treatment guidelines (2008), resistance assay is recommended only after second-line ART failures. 25
Located in Gaborone and opened in 2003, the Botswana-Baylor Children’s Clinical Centre of Excellence (BCOE) is Botswana’s largest pediatric HIV treatment center, with more than 1800 children receiving ART. 26 During 2005 and 2006, prior to publication of Botswana’s current guidelines recommending empiric switch, children being treated at the BCOE had resistance assay performed at the time of first-line virologic failure when resistance rather than nonresistance causes (nonadherence, pharmacokinetics, drug interactions, etc) of ART failure were suspected. This study was performed with the purpose of reviewing these results and exploring their implications to practice in Botswana and similar settings.
Materials and Methods
A retrospective chart review was performed to identify and quantify NRTI- and NNRTI-associated mutations among pediatric and adolescent patients at the BCOE with resistance assay profiles available at the time of virologic failure on first-line ART. Using the Botswana HIV treatment guidelines, 25 virologic failure was defined as viral load (VL) >400 copies/mL on 2 consecutive occasions. Genotypic resistance testing was performed by HIV-1 ViroSeq Genotypic System (Abbott Molecular–Abbott Park, Illinois) at the Botswana–Harvard HIV Reference Laboratory. Drug resistance mutations were defined using the Stanford University HIV Drug Resistance Database (http://www.hivdb.stanford.edu) as substitutions at the following positions: for NRTI—M41L, E44D, A62V, K65R, D67N, S68G, 69i, K70R, L74V, V75T, F77L, Y115F, V118I, Q151M, M184V/I, L210W, T215Y/F, and K219Q/E; and for NNRTI—K101E, K103N, V106A, V108I, Y181L/C, Y188L/C, G190A, P225H, and M230L. Laboratory and demographic data were obtained retrospectively from the patients’ medical records and analyzed for the following characteristics: age at time of resistance assay, gender, WHO immunologic category, plasma HIV-1 RNA level, duration of ART prior to resistance assay, and time from virologic failure to resistance assay. Patients were excluded from the study if timing of the resistance assay in relation to first-to-second-line switch could not be confirmed. Patients with protease inhibitor–based first-line regimens were also excluded.
Ethical Approval
This study was approved by the Health Research and Development Committee (HRDC), Ministry of Health, Botswana, and the Institutional Review Board, Baylor College of Medicine, Houston, Texas.
Results
Demographic Characteristics
A total of 45 pediatric and adolescent patients met the inclusion criteria and had results of resistance assays performed at the time of their first-line failure analysis. While all 45 patients had notation documentation of virologic failure on first-line ART, specific VL results at the time of resistance assay were missing for some patients; 35 had results of VL prior to resistance assay available to review; and 18 had a documented date of VL and date of resistance assay available to review. There were 6 patients who did not have a documented date of ART initiation. Table 1 shows demographic, clinical, and laboratory data for these patients.
Demographic, Clinical, and Laboratory Data for 45 Patients Failing First-Line ART at the Botswana-Baylor Children’s Clinical Center of Excellence with a Documented Resistance Assay

Abbreviations: ART, antiretroviral therapy; WHO, World Health Organization; VL, viral load.
a WHO Paediatric Guidelines.
b n = 35 with documented VL prior to resistance assay.
c n = 39 with documented date of ART initiation.
d n = 18 with documented date of VL and date of resistance assay.
Genotypic Resistance Testing Results
Table 2 details the mutations seen in each patient as well as their first-line regimen. At least one resistance mutation was noted in all patients (n = 45). At least 2 resistance mutations were noted in all but 1 patient who was on d4T/3TC/NVP and had M184V as the only mutation (n = 44; 97.7%). In all, 42 patients (93.3%) had at least 1 NRTI mutation and 1 NNRTI mutation. Table 3 presents the frequency of occurrence of each NRTI and NNRTI mutation in the reverse transcriptase gene.
Age, Gender, Time on ART, WHO Immune Status, VL, Duration of VF, and First-Line ART Regimen Prior to Resistance Assay as Well as All NRTI and NNRTI Mutations for 45 Patients Failing First-Line ART at the Botswana-Baylor Children’s Clinical Center of Excellence with Documented Resistance Assay


Abbreviations: I, no significant immunosuppression; II, mild immunosuppression; III, advanced immunosuppression; IV, severe immunosuppression; M, male; F, female; 3TC, lamivudine; ART, antiretroviral therapy; ddI, didanosine; d4T, stavudine; EFV, efavirenz; n/a, data not available; NS, not significant; NRTI, nucleoside reverse transcriptase inhibitor; NS, never suppressed; NNRTI, nonnucleoside reverse transcriptase inhibitor; NVP, nevirapine; WHO, World Health Organization; VF, virologic failure; VL, viral load; ZDV, zidovudine.
a WHO Paediatric Guidelines, immunologic categories, I, II, III, and IV.
Quantitative Summary of All NRTI and NNRTI Mutations Seen in 45 Patients at the Botswana-Baylor Children’s Clinical Center of Excellence with Documented Resistance Assay
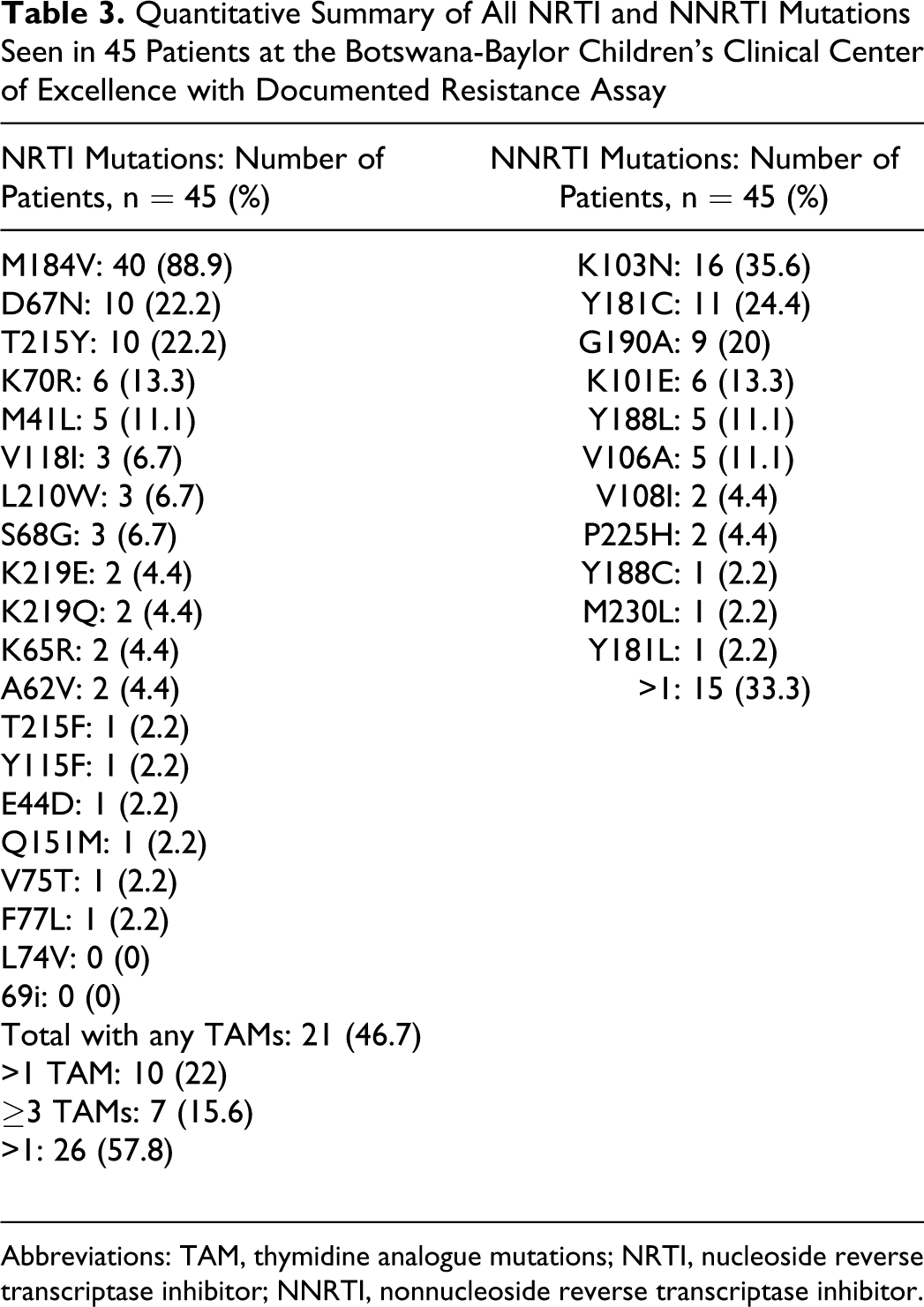
Abbreviations: TAM, thymidine analogue mutations; NRTI, nucleoside reverse transcriptase inhibitor; NNRTI, nonnucleoside reverse transcriptase inhibitor.
Nucleoside reverse transcriptase inhibitor mutations
The most commonly detected mutations were M184V (in 40 patients, 88.9%) and TAMs (in 21 patients, 46.7%). More than 1 TAM was seen in 10 patients (22%). Seven patients (15.6%) had 3 or more TAMs. Of the 21 patients with TAMs, 11 (52.4%) demonstrated TAMs exclusive to the TAM-1 pathway (41L, 210W, and 215Y), 14 (66.7%) had TAMs exclusive to the TAM-2 pathway (67N, 70R, 215F, and 219Q/E/N/R), and 4 (19.1%) had at least 1 mutation from both TAM-1 and TAM-2 pathways. The mutation K65R was seen in 2 patients (4.4%), one of whom was on ZDV/3TC/NVP (patient 11), the other (patient 35) was on both ZDV/d4T as well as NVP/EFV, at points in time (ZDV/d4T-3TC-NVP/EFV). Of these, patient 11 was the only patient who demonstrated the Q151M mutation. No patients had the L74V mutation.
Nonnucleoside reverse transcriptase inhibitor mutations
Among NNRTI exposures, 6 patients had only been exposed to EFV, while 12 had been exposed to both EFV and NVP, 27 had been exposed to NVP only. The most commonly detected NNRTI mutations were K103N (n = 16; 35.6%) and Y181C (n = 11; 24.4%). In all, 41 patients (91.1%) were found to have at least 1 mutation associated with intermediate-to-high level resistance to EFV and/or NVP (K103N, Y181C, G190A, K101E, Y188L, V106A, Y188C, M230L, and Y181L). 27 A total of 13 patients (28.9%) exhibited 2 or more NNRTI mutations, conferring intermediate-to-high level resistance to NVP/EFV, and 21 patients (46.7%) had at least 1 mutation associated with etravirine (ETR) resistance as reported in the DUET studies (K101E, Y181C, and G190A). 28,29 Of these 21 patients, 20 had been exposed to NVP, with only 1 of 6 patients exposed to just EFV, developing 1 of these 3 mutations, that is, K101E. In all, 37 patients (82.2%) were noted to have both M184V and at least 1 mutation conferring intermediate-to-high-level resistance to an NNRTI.
Discussion
Low genetic barrier mutations (M184V, K103N, and other NNRTI associated) are common in our cohort, as are TAMs, including more than 1 TAM. This is consistent with the results from many other studies in sub-Saharan Africa, evaluating resistance mutations seen after failure on WHO-recommended first-line ART. 12 –18,20 It has been suggested that where VLs are unavailable and ARV treatment failure is determined using only immunologic and/or clinical criteria—as in much of sub-Saharan Africa—extensive ARV drug resistance is common at the time of line switch. 20 Justifiably, improved access to VLs is advocated for such settings.
Our data demonstrate that even where VLs are widely available, such as in Botswana, extensive ARV drug resistance at the time of switch from first-line ART may be common, potentially affecting the response to second-line antiretrovirals. This suggests that in such settings—where ART options beyond second-line remain few—wider availability of resistance assays at the time of switch from first-line ART be considered for inclusion into standard ART programming.
The high frequency of TAMs, including 7 patients (15.6%) with 3 or more TAMs, seen in our study is noteworthy for several reasons. As the presence of multiple TAMs predicts wider NRTI resistance and the possible compromise of empirically selected second-line ART, the potential role of resistance assay in selection of second-line backbones is heightened. Standard of care in well-resourced settings and access to resistance genotypes prior to initiating second-line ART may hold even more value where ART choices are limited.
It has been shown previously that in Botswana, adult patients with HIV-1C treated with ZDV/didanosine (ddI) first-line backbones may demonstrate pathways of TAM development distinct from HIV-1B, specifically the 67N/70R/215Y genotype. 11 The variety of TAM pathways seen in our study, including 67N/70R/215Y in 1 patient and other hybrid mutation patterns distinct from HIV-1B, indicate that distinct TAM pathway development may be seen in children with HIV-1C as well. While data to date do not suggest a need to modify the recommended first- and second-line ART combinations based on subtype, the concept deserves further study, including genotypic and phenotypic resistance patterns induced in different HIV-1 subtypes by particular ART regimens as well as additional studies in children.
Previous reports of resistance mutations in HIV-1C after first-line failure have suggested that K103N is the most commonly seen NNRTI mutation 13 –16,20 ; the same was seen in our study. However, the exclusive presence of V106A and the absence of V106M in our study counter previous suggestions that V106M is the favored mutation at position 106 in HIV-1C. 16 Rather, the development of V106A versus V106M in HIV-1C likely depends on the particular NNRTI exposure, with exposure to EFV yielding V106M and to NVP yielding V106A. 30 Indeed, all 5 of our patients who developed V106A had been exposed to NVP alone or to NVP prior to EFV. Both V106A and V106M are clinically significant mutations which may confer high-level multi-NNRTI resistance. 30
Also notable is the high frequency of 1 or more NNRTI mutations associated with resistance to ETR in HIV-1B subtypes. 28,29 Whether these substitutions will compromise ETR in HIV-1C subtypes is not yet known; currently ETR is not US Food and Drug Administration (FDA)-approved and available for children, although pediatric studies are ongoing. 3 Although not yet available in Botswana, ETR has potential for utility in future treatment approaches. 3,31 In light of this, the high frequency of multiple NNRTI mutations in our study is causing concerns and may reflect delayed switch from first- to second-line therapy; our data for this variable are limited but suggests it for the 18 patients with documented time from virologic failure to resistance assay.
A high frequency of likely ETR-resistant virus after failure of NNRTI-based first-line therapy has been seen elsewhere in resource-limited settings 32,33 ; and several studies have specifically noted reduced susceptibility to ETR after failure of NVP-based ART as compared with ART based on EFV, 34 –36 including with non-B HIV-1 subtypes. 36 Similarly, in our study, 20 of 21 patients with an ETR resistance-associated mutation had been exposed to NVP, when compared with only 1 of 6 patients exposed to EFV alone. While patients who virologically fail ART while on EFV likely have better odds of maintaining susceptibility to ETR than those who fail while on NVP, 35 in both cases prompt switch from failing regimens is essential to prevent the accumulation of NNRTI mutations and preserve future treatment options, including ETR.
There are limitations to our study, including its retrospective nature, lack of data on perinatal NNRTI exposure (particularly sd-NVP), lack of data on individual patient adherence to ART prior to treatment failure and resistance assay being performed, and incomplete data on duration of virologic failure prior to resistance assay. These limitations are discussed below. Also, our study was not designed to evaluate the use of resistance assays on improvements in patient outcomes; a study along such lines would be of interest.
It is well appreciated that a substantial fraction of HIV-infected individuals exposed to even single doses of NVP develop resistance to NNRTIs, including high-grade class resistance–inducing mutations such as K103N (up to 25% for HIV-1A and HIV-1D and 39% for HIV-1C). 37 Exposure, therefore, of our patients to sd-NVP as part of PMTCT or to maternal NNRTI as part of treatment or PMTCT would be of interest as it could explain some proportion of NNRTI-associated mutations in our sample. While both data points are lacking in our database, by age only 3 (6.7%) of 45 patients could potentially have received or been exposed to maternal sd-NVP as it was not part of Botswana’s PMTCT program until November 2003. However, 10 (33.3%) of 45 patients were born after Botswana’s ART program was established in November 2001 and may have had NNRTI exposure due to maternal treatment, a variable not present in our database also. Given that NNRTI-containing treatment would have been a triple-drug therapy, this would have predisposed to lower rates of NNRTI resistance than sd-NVP.
Associating adherence data with development of particular mutations would also be of interest. While specific adherence data are not included in our database for the years in study, many of the patients studied have chart notations of poor or less than complete adherence. Pre-ART adherence counseling and ongoing support is critical to achieving good patient outcomes on ART, particularly in pediatrics.
Similarly, complete data on duration of virologic failure prior to resistance assay would be of interest in the analysis of resistance mutation patterns. For the 18 patients for whom this data was available, the median/mean time from virologic failure to resistance assay was prolonged (10/9.6 months, respectively) and suggests delay in the switch of ART lines. The reasons why an individual patient has delay in switching therapy line are, of course, many and beyond the scope of this study.
While our study was not designed to specifically evaluate whether or not the use of resistance assays improves clinical decision making, our results and similar data from other resource-limited settings nonetheless suggest implications for informing decisions on the empiric management of ART among children failing NNRTI-based first-line therapy utilizing WHO-recommended NRTI backbones. Despite the ideal nature of basing the choice of second-line regimen on resistance assay results obtained at the time of first-line failure, the reality is fiscal, and programmatic constraints in many resource-limited settings will mandate continued empiric selection of second-line and further regimens.
Unlike in many regional studies, 13,20,21 the frequency in our study of mutations compromising nonthymidine analogue backbones (containing tenofovir [TDF], ABC, or ddI) was rare or nonexistent, suggesting empiric change in ZDV- or d4T-containing first-line ART regimens should be to nonthymidine analogue-containing NRTI backbones.
Of particular concern has been the suggestion that the propensity for HIV-1C subtypes to develop the K65R mutation may be considerably higher than for other HIV subtypes, particularly HIV-1B. 12,19,21 Although most supportive data are from adult populations exposed to ART containing ddI or TDF, 21,38,39 other regional studies have shown substantial rates of K65R accumulation after failure of standard thymidine analogue-based (WHO recommended) first-line ART among patients who have not been exposed to nonthymidine analogue NRTIs. 13,20 Stavudine rather than ZDV is associated with the development of K65R mutation, 40 particularly in combination with NVP 41 and for the HIV-1C subtype. 20,21,38,39,42 Especially where both HIV-1C is prevalent and ART regimens containing d4T and NVP remain in widespread use, as in much of southern and east Africa, this has substantial implications for pediatric populations who will transition into adolescent and adult care over time, as updated WHO recommendations for adolescent and adult first-line ART include TDF, 43 the ARV most compromised by K65R.
In our pediatric population, the frequency of developing K65R mutation during the course of ART failure was low (2 of 45 patients), likely reflecting the relatively low exposure to d4T (15 of 45 patients), and very low exposure to ddI (1 of 45 patients) in our population; none of our patients had prior exposure to TDF or ABC. One of the patients (patient 35) in whom K65R mutation developed had been exposed to d4T (along with, over time, 3TC, ZDV, NVP, and EFV), while the other (patient 11) had only been exposed to ZDV/3TC/NVP.
Genotype of patient 11 highlights a number of interesting associations regarding the presence of K65R, including perinatal transmission of virus harboring the K65R mutation 40 or the possibility of the presence of K65R in drug-naive patients, a phenomenon more likely in HIV-1C than in other subtypes. 44 Also, patient 11 was the only patient in our study to have the Q151M mutation, which induces reduced susceptibility to all NRTIs other than 3TC and TDF. 40 Although ZDV and d4T rarely select Q151M, Q151M is the most common mutation coselected or sequential to the development of K65R, 40 and the presence of K65R and Q151M inhibit the development of M184V. 45 Indeed, in genotype of patient 11, M184V was found to be absent. The combination of K65R and Q151M is particularly detrimental, conferring more resistance by itself than either K65R or Q151. 40
No patients in our study had L74V, likely reflecting the very low rate of ddI use in our population. Taken together, then, our results suggest that it is likely that children who fail first-line NRTI backbones containing d4T or ZDV/3TC would still respond to ABC, ddI, and, for adolescents, TDF.
It is of interest that what data, such as ours, suggest about maintaining 3TC in second-line pediatric regimens is now recommended by WHO for all age groups, including children. The reduced viral fitness and hypersensitization to other NRTIs yielded by the M184V mutation is well appreciated, and clinical benefit has been noted with continued use of 3TC in second-line regimens in the presence of M184V. 46 Given the very high frequency with which M184V was seen in our study after failure on 3TC-containing first-line ART, it seems reasonable to consider the empirical continuation of 3TC in second-line regimens; the data from ongoing pediatric studies of this approach in southern Africa and Thailand will be received with great interest. 31
Footnotes
Acknowledgments
The authors would like to acknowledge Dr Mark Kline and Dr Gordon Schutze who gave valuable editorial input on the manuscript. We thank the Botswana-Harvard HIV Reference Laboratory in Gaborone for processing patient resistance assay samples. We also thank our Centre's patients and clinical team, as well as the Government of Botswana for its support of Baylor activities in Botswana. These findings were presented in abstract form at the XVIII International AIDS Conference, Vienna, 18-23 July 2010.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
