Abstract
Objectives:
Monitoring antiretroviral treatment (ART) outcomes is essential for assessing the success of HIV care and treatment programs in resource-limited settings (RLS).
Methods:
Longitudinal analyses of clinical and immunologic parameters in HIV-infected adults initiated on ART between November 2004 and June 2008 at Management and Development for Health (MDH)-Presidents Emergency Plan For AIDS Relief PEPFAR supported HIV care and treatment clinics in Tanzania.
Results:
A total of 12 842 patients were analyzed (65.9% female, median baseline CD4 count, 106 cells/mm3). Significant improvements in immunologic status were observed with an increase in CD4 count to 298 (interquartile range [IQR] 199-416), 372 (256-490) and 427 (314-580) cells/mm3, at 1, 2, and 3 years, respectively. Overall mortality was 13.1% (1682 of 12 842). Male sex, World Health Organization (WHO) stage III/IV, CD4 <200 cells/mm3, hemoglobin (Hgb) <8.5 g/dL, and stavudine (d4T)-containing regimens were independently associated with early and overall mortality.
Conclusions:
Closer monitoring of males and patients with advanced HIV disease following ART initiation may improve clinical and immunologic outcomes in these individuals.
Introduction
In 2008, an estimated 33.4 million people were living with HIV. Sub-Saharan Africa remains the most heavily affected region, accounting for over two thirds of all new infections worldwide. In recent years, unprecedented efforts have been made by national governments and international organizations to scale-up HIV care and treatment services in low- and middle-income countries. By the end of 2008, as a result of these efforts, at least 4 million HIV-infected individuals in sub-Saharan Africa were receiving antiretroviral therapy (ART), an estimated 44% of those who are eligible. 1
Since ART rollout began, several studies examining ART outcomes in resource-limited settings (RLS) have been performed. 2 The majority have reported encouraging clinical, immunological, and virologic responses to therapy with sustained benefit over time. Mortality rates, the most important measure of ART effectiveness, have been estimated to range from 8% to 26% at 1 year after ART initiation in RLS, with the highest rates occurring among those with more advanced HIV at treatment initiation and shortly after ART initiation. 3 Recently, high rates of mortality have been reported among patients lost to follow-up, 4,5 suggesting that mortality in programs without active patient tracking systems may be significantly underreported. Findings such as these have important implications for public health policy since determination of a program’s effectiveness is often based on survival.
In 2007, about 2 million persons were estimated to be living with HIV and AIDS in Tanzania, with approximately 600 000 persons with AIDS in need of ART. 6 Over the last 5 years, a nationwide HIV care and treatment program aimed at providing HIV care and treatment services for all HIV-infected persons was implemented with support from international development partners, including Management and Development for Health (MDH)-Presidents Emergency Plan For AIDS Relief (PEPFAR). To date, very few studies have evaluated the treatment outcomes among HIV-infected individuals initiated on ART in Tanzania. 7 In this article, we report mortality and immunological outcomes in response to ART and examine predictors of overall and early mortality among HIV-infected patients enrolled in a large urban HIV care and treatment program in Tanzania that provides ART within all levels of public health delivery system.
Methods
This prospective observational study was conducted at 12 MDH-PEPFAR supported HIV care and treatment clinics in Dar es Salaam, Tanzania, between November 2004 and June 2008. The MDH-PEPFAR HIV Care and Treatment Program (previously known as the Muhimbili University of Health and Allied Sciences-Dar City-Harvard School of Public Health-PEPFAR HIV Care and Treatment program) was established in 2004 and provides infrastructure, laboratory, and technical support to the 3 municipalities of Dar es Salaam (Temeke, Ilala, and Kinondoni). Inclusion criteria for this study were age >15 years, not yet initiated on ART, and not pregnant at the time of enrollment. At the MDH program, patients are evaluated monthly following enrollment and ART initiation. At each visit, they are examined by a physician and receive adherence and nutrition counseling and ART refills. Monitoring laboratory tests including hemoglobin (Hgb) and alanine aminotransferase (ALT) are performed 2 weeks after ART initiation and every 4 months thereafter. Immunonologic monitoring with CD4 counts is also performed at 4-month intervals. Viral load testing is not performed routinely. Pulmonary tuberculosis (PTB) screening with a chest X-ray and sputum smear for acid fast bacilli is performed at baseline on all patients and repeated in patients who have symptoms suggestive of PTB at follow-up visits. A comprehensive patient tracking system is in place to ensure all patients who are eligible for tracking (ie, missed visit, lost to follow-up, reported to be sick, and abnormal laboratory results) are contacted by phone or in person to encourage them to return to clinic. A network of community-based health workers and volunteer persons living with HIV and AIDS work with the MDH program to actively trace and ascertain the vital status of all patients who are lost to follow-up.
Patients are initiated on ART according to the National AIDS Control Program (NACP) ART initiation criteria (CD4 count <200 cells/mm3, clinical World Health Organization (WHO) stage IV and clinical WHO stage III with CD4 count of <350 cells/mm3). 6 At the time of this study, standard first-line ART regimens included stavudine (d4T) or zidovudine (ZDV) plus lamivudine (3TC) and efavirenz (EFV) or nevirapine (NVP). To avoid drug interactions, EFV is substituted for NVP in patients receiving rifampin-containing antituberculous therapy. Recommended second-line regimens for patients failing to respond to first-line regimen include the combination of abacavir (ABC) + 3TC and lopinavir/ritonavir (LPV/r) or saquinavir/ritonavir (SQV/r). Antiretroviral drugs and co-trimoxazole are provided free of charge by the Tanzanian government; and since the inception of the MDH program, no patients have experienced treatment interruptions owing to drug shortages.
Patients were recruited for participation and enrolled in the MDH HIV Care and Treatment program following written informed consent, which was subject to ethical review by the Muhimbili University of Health and Allied Sciences and the Harvard School of Public Health.
Data Collection and Management
Patient demographic, clinical, laboratory, and therapeutic data are collected by physicians and nurses on standard case report forms and National Care and Treatment Center forms (CTC 2) which are completed at enrollment and at each follow-up visit. Data reviewers are stationed at each clinic to ensure adequacy and completeness of data recording by the health care workers. Data collected is then entered into a secure computerized database designed solely for the purpose of data collection and analysis. Unique patient identifiers are used. The database is updated daily by professional data entry clerks. Weekly quality assurance checks of the database are performed by the data management team to ensure data accuracy. Data collected for these analyses included baseline demographics, age, weight, height, body mass index (BMI), median upper arm circumference (MUAC), WHO clinical stage, history of prior or current PTB (the latter defined as receiving treatment for PTB within 1 month after ART initiation), ART regimen at initiation, enrollment and visit dates, date of death, and Hgb and CD4 count.
Treatment Outcomes
Primary outcomes of interest included death occurring in the first 90 days after ART initiation (early mortality), overall mortality and immunologic response to ART. Deaths were recorded after notification by family members, friends, patient tracking teams, or community-based health care workers. If the date of death was unknown but a patient was known to have died, the date of the last encounter with the patient was used as the date of death. Immunologic outcomes observed were the mean change in CD4 count over time and the proportion of patients with immunologic failure as defined by the following Tanzanian national criteria: (a) CD4 count less than pretherapy CD4 count after at least 6 months of therapy or, (b)3 50% drop from the peak CD4 count value.
Statistical Methods
Proportions or median (interquartile range [IQR]) were calculated for baseline characteristics at the time of ART initiation. In this study, deaths were followed from date of ART initiation to date of death or date of last visit, whichever happened first. The assessment of immunological failure was also determined from the date of ART initiation to the date of first failure or date of last visit, whatever happened first. Kaplan-Meier methods were used to calculate the cumulative incidence of outcomes. To identify predictors of mortality, univariate and multivariate analyses were performed using Cox regression analyses. P values for the trend test were calculated using median scores test for ordinal variables and the likelihood ratio test was used to assess the statistical significance of nominal polytomous variables. The missing indicator method was used as needed. 8 Generalized estimating equations (GEEs) were used to assess the change in CD4 count. Stepwise restricted cubic splines were used to assess nonlinearity of results and produce graphs of associations. The criterion for significance for all analyses was a P value of α = .05. All statistical analyses were performed with the statistical software package SAS, Release 9.1 (Cary, North Carolina).
Results
A total of 12 842 patients were included in this analysis. Baseline characteristics are shown in Table 1. The majority of patients were female (65.9%) and the median age was 36 (IQR 31-43) years. In all, 6855 (78.9%) patients had a baseline CD4 <200 cells/mm3 and 2619 (24.6%) of patients were WHO stage IV. The median BMI was 20.0 (IQR 17.7-22.7); 4052 (33.4%) adults patients had a BMI < 18.5. One third (31.8%) of the patients reported a past history of PTB and 13.4% were currently infected with PTB. All the patients were initiated on a nonnucleoside reverse transcriptase inhibitor (NNRTI)-containing ART regimen; 7451 (79.7%) patients were on regimens containing d4T.
Basic Characteristics of Study Population (N = 12 842)

Abbreviations: ART, antiretroviral therapy; IQR, interquartile range; d4T, stavudine; 3TC, lamivudine; NVP, nevirapine; EFV, efavirenz; ZDV, zidovudine; ALT, alanine aminotransferase.
Significant improvements in immunologic outcomes were observed in patients on ART, with a median ART duration of 6.0 months (1.9-2.5). There was an increase in CD4 count throughout the 3 years following ART initiation, with no evidence of nonlinearity (P = .08; Figure 1). The median CD4 count 1, 2, and 3 years after ART initiation was 298 (199-416), 372 (256-490), and 427 (314-580) cells/mm3, respectively. The cumulative incidence of immunological failure defined as a CD4 count less than pretherapy CD4 count after at least 6 months on therapy at 1, 2, and 3 years was 10.5%, 17.5%, and 20.8%, respectively. When defined as a 50% drop from the peak CD4 count value, the cumulative incidence of immunologic failure at 1, 2, and 3 years was 5.7%, 16.8%, and 30.8%, respectively (Table 2).

CD4 count by time in months since ART initiation. Overall slope 2.1 cells/mm3 per month (standard error = 0.24, P value <.001). ART indicates antiretroviral therapy.
Cumulative Incidence of Mortality and Immunologic Failure
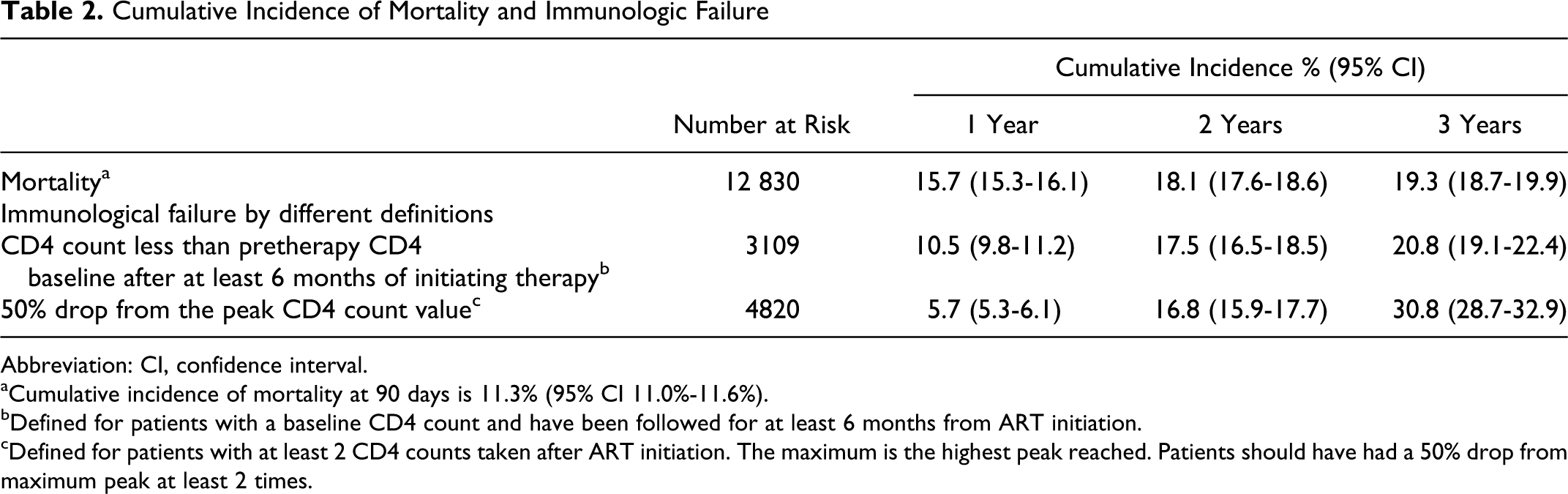
Abbreviation: CI, confidence interval.
aCumulative incidence of mortality at 90 days is 11.3% (95% CI 11.0%-11.6%).
bDefined for patients with a baseline CD4 count and have been followed for at least 6 months from ART initiation.
cDefined for patients with at least 2 CD4 counts taken after ART initiation. The maximum is the highest peak reached. Patients should have had a 50% drop from maximum peak at least 2 times.
The overall crude mortality rate was 13.1% (1682 of 12 842). The majority of deaths (76.4%) occurred in the first 3 months. The cumulative incidence of death at 1, 2, and 3 years was 15.7%, 18.1%, and 19.3%, respectively (Table 2). In multivariate analyses, predictors of early and overall mortality were very similar (Table 3). Males had a significantly higher risk of both early (hazard ratio [HR] 1.19; 95% confidence interval [CI] 1.06-1.34; P < .001) and overall (HR 1.19; 95% CI 1.07-1.32; P <.001) mortality. Patients with clinical and immunological parameters associated with advanced HIV at the time of enrollment were also at higher risk of early and overall mortality with a higher WHO clinical stage being the strongest predictor; CD4 <200 cells/mm3: (HR 1.62, 95% CI 1.33-1.97 [early mortality]; HR 1.42, 95% CI 1.20-1.67 [overall mortality]); Hgb <8.5g/dL: (HR 1.94, 95% CI 1.71-2.20 [early mortality]; HR 1.87, 95% CI 1.67-2.08 [overall mortality]); and WHO stage IV: (HR 4.99, 95% CI 3.81-6.53 [early mortality]; HR 4.61, 95% CI 3.66-5.82 [overall mortality]). The cumulative risk of mortality by CD4 count strata (
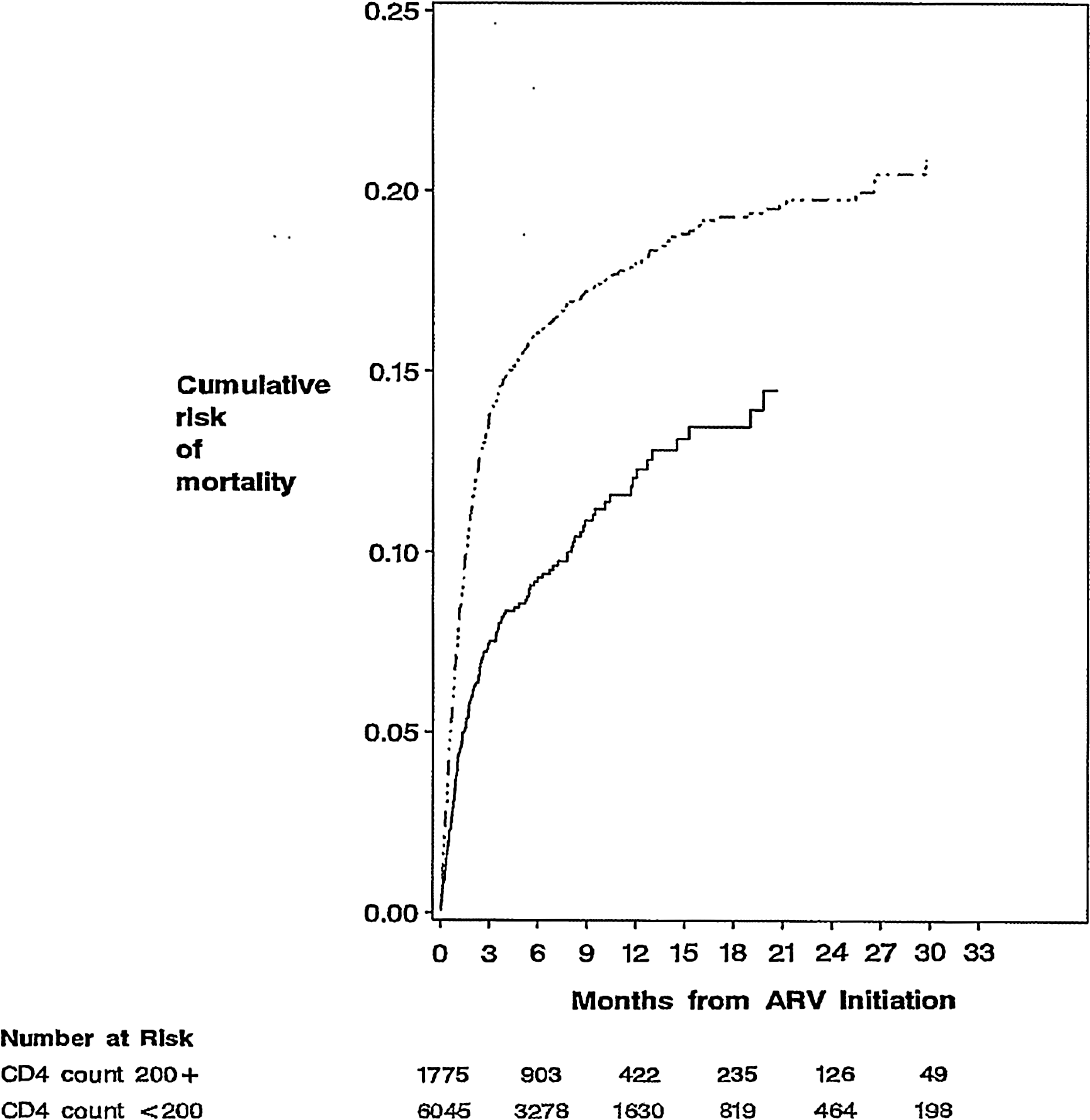
Cumulative risk of mortality within strata of baseline CD4 count.

Body mass index in relation to mortality risk.
Results of Multiple Cox Regressions for Time to Overall and Early Mortality

Abbreviations: ART, antiretroviral therapy; BMI, body mass index; CI, confidence interval; d4T, stavudine; 3TC, lamivudine; NVP, nevirapine; EFV, efavirenz; ZDV, zidovudine; ALT, alanine aminotransferase; TB, tuberculosis; WHO, World Health Organization.
aHazard ratios, 95% CI, and corresponding P were obtained from Cox proportional hazards models. Missing indicator method was used for missing variables. Only variables significant at P < .05 were included in the multivariate model. The multivariate model was adjusted for patient referral site, calendar month, and calendar year as indicators in addition to variables in the table.
b P value for trend calculated using median score test.
c P value for trend calculated using likelihood ratio test.
Discussion
In this study, we report ART outcomes from one of the largest HIV care and treatment programs in sub-Saharan Africa. Notable findings include robust improvements in CD4 count over time, and a relatively low risk of mortality, despite the advanced stage of HIV disease at ART initiation in most of our patients. Our findings provide yet more evidence that it is feasible to provide effective ART within all levels of public health delivery system in RLS.
Baseline demographics of our cohort were similar to other HIV cohorts in RLS; the majority of patients were female and most patients initiated ART at very advanced stage of HIV disease. Several clinical and laboratory markers associated with advanced HIV disease were observed in high proportions among study patients at enrollment, including a low CD4 count, BMI, Hgb, and higher WHO clinical stage. Advanced HIV disease at enrollment has been reported frequently among other HIV cohorts in Africa 9,10 ; a previous study from Tanzania reported up to 30% of patients starting ART with CD4 counts below 50 cells/mm3. 11 Factors that have been found to be associated with late disease presentation include lower educational level, male gender, stigma, inadequate referral systems, and poor access to treatment centers. 12 How to best promote early uptake of ART-eligible patients to treatment programs is a central challenge facing the scale-up of HIV care and treatment programs in RLS and a focus on reducing factors associated with late treatment access is therefore necessary.
According to Tanzania National guidelines, immunological failure is defined as a 50% drop from peak value in CD4 count or return to pre-ART baseline CD4 count or lower. 6 In this study, a number of patients failed ART therapy according to these criteria. Of concern, an estimated one third of all patients met the definition of 1 of the criteria at 3 years. In prior studies where similar definitions of immunologic failure have been used, the rates of failure have varied. In a study from Ghana 13 which used 3 criteria (2 of which are similar to the Tanzanian criteria), immunologic failure rates were as low as 12%. The insensitivity of immunologic failure criteria when compared to virologic failure criteria has been well described and may result in delays in antiretroviral switching as well as unnecessary treatment switches, both of which have been shown to negatively impact treatment outcomes. 14,15 It is therefore possible that, some of our patients may not have actually failed therapy using these criteria alone. On the other hand, these findings should not be completely dismissed; in a recent randomized trial of monitoring strategies (clinical findings, CD4 count, viral load) by Coutinho et al in Uganda, the addition of viral load measurements did not add any benefit over the use of the CD4 count plus clinical staging in predicting the risk of death. 16
Mortality rates in our study were slightly lower than the rates in some other published studies from RLS. 3,17–19 We observed that the majority (1285 of 1,682, 76.4%) of deaths occurred within 90 days, a similar finding to other studies. 20,21 In a recent review by Lawn et al 3 of 18 published HIV cohort studies from RLS, between 8% and 26% of patients who were not lost to follow-up died after 12 months, with the greatest burden of mortality occurring during the initial months of therapy. In this study, many of the same predictors of mortality were observed, especially those indicative of an advanced stage of disease at ART initiation including low CD4 counts and Hgb and high WHO clinical stage. The striking difference in mortality in patients with CD4 counts <200 cells/mm3 compared CD4 counts above 200 cells/mm3 has also been previously observed.
Males were also found to have a significantly higher risk of early and overall mortality in this study even after controlling for baseline clinical and immunologic status. A higher risk of mortality among men has been increasingly reported in other studies in RLS, although some of the data are conflicting. 22,23 Behavioral factors are likely to have contributed significantly to these outcomes. In recent studies, males have been noted to have higher rates of loss to follow-up and nonadherence than women 24 and report more HIV-associated stigma. 25
A notable finding in our study was the protective effect of current or prior history of PTB at ART initiation on mortality. This finding has also been reported in one other study and warrants further investigation. 26 The incidence of PTB in our patient cohort was much lower than expected, and although screening with chest X-rays (CXR) and sputum are performed routinely on every patient at the time of program enrollment, the low sensitivity and specificity of this type of screening is well known. 27,28 Potentially, more patients had undiagnosed PTB at ART initiation and were at higher risk of adverse outcomes than those who were actually diagnosed, perhaps because they were monitored less closely for other comorbidities such as the immune reconstitution inflammatory syndrome (IRIS) or were less likely to be receiving prophylactic co-trimoxazole.
Another unique finding in our study was the higher risk of overall and early mortality observed among patients on regimens containing d4T. An association between d4T-containing regimens and mortality was recently observed in a collaborative analysis of 12 prospective HIV outcome cohort studies from North America and Europe, 29 however there was no association of d4T and mortality in 2 other studies from RLS. 9,21 A further investigation into this relationship is needed, especially as d4T continues to be widely used in these settings. Stavudine has been found to be associated with numerous and potentially life-threatening side effects which could have contributed significantly to the higher rate of overall mortality seen in these patients. 30
There were several limitations to our study that may have influenced our findings. Several studies report adherence as one of the most important determinants of treatment outcomes, which we were not able to assess. 31,32 As this was an observational study, other potential confounders may have been present that may have affected clinical and immnunologic outcomes that were not accounted for (ie, opportunistic infections). In our study, viral loads were not measured and thus data on virologic outcomes were not presented, which would have been a more sensitive maker of treatment response in this cohort. Finally, the primary data were determined through physician reporting in clinical records and were therefore subject to potential inaccuracies or omissions in data. Despite these limitations, this study is one of the largest cohorts of HIV-infected patients on ART in a RLS and the program has one of the most comprehensive tracking systems ensuring reliable recording of deaths.
In conclusion, this study demonstrates feasibility and the effectiveness of large-scale HIV treatment initiatives in Tanzania and other RLS, where initiation of ART resulted in robust immunological responses and low overall mortality. However, greater efforts are needed to promote early HIV testing as well as prompt access to care and initiation of ART for those most in need of treatment. More intensive follow-up of patients in the first 3 months after ART initiation would help reduce the risk of early mortality as well as closer monitoring of high-risk individuals such as males and patients with advanced HIV disease at presentation. Nutritional interventions that address anemia and low BMI may be important as adjunct therapies to ART and need to be examined. Ongoing efforts to phaseout d4T from antiretroviral formularies must be reinforced, given the significant association between d4T use and mortality.
Footnotes
Acknowledgments
We thank Management and Development for Health (MDH), Dar es Salaam City Council, Muhimbili University of Health and Allied Sciences (MUHAS), Harvard School of Public Health (HSPH), and the Ministry of Health and Social Welfare for the guidance and collaboration in implementing a national HIV care and treatment program in Dar es Salaam, Tanzania. This program is supported by the US President’s Emergency Plan for AIDS Relief (PEPFAR) through the HSPH and by the Ministry of Health and Social Welfare, Tanzania. We thank all the patients and staff of the MDH supported care and treatment sites, who have contributed to these findings.
Authors’ Note
This paper was presented in part as a poster presentation #824 at the Conference on Retrovirus and Opportunistic infections (CROI) February 3-6, 2008, Boston, MA, USA.
Declaration of Conflicting Interests
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Presidents Emergency Plan For AIDS Relief (PEPFAR).
