Abstract
The Immune Reconstitution Inflammatory Syndrome (IRIS) in Ethiopian HIV-infected patients coinfected with tuberculosis (TB) was studied. HIV-infected outpatients initiating antiretroviral therapy (ART) at an HIV clinic in northern Ethiopia from January 2007 through September 2008 were identified (n = 1977). Patients with TB-IRIS occurring within 6 months of starting ART (n = 143) were compared with a control group of patients with HIV who began ART but did not develop TB-IRIS (n = 277). ART was not interrupted in any patient. Eleven (8%) patients with TB-IRIS died. New or “unmasked” TB with accompanying IRIS occurred in 132 or 92% of the cases. Worsening or “paradoxical” TB (ie, already known to be present and treated) was accompanied by IRIS in 11 (8%) patients. There was no significant difference between “unmasked” and “paradoxical” cases with respect to presentation of disease and outcome. Only a low baseline CD4 count (mean: 102 cells/μL) and a past history of World Health Organization (WHO) Clinical Stage 3 or 4 were associated with TB-IRIS (P < .05). The clinical manifestations of TB-IRIS were diverse, requiring a high index of suspicion. For example, pleural disease occurred in 13 patients, TB lymphadenitis in 17, intracranial TB in 9 patients, and disseminated TB in 15 patients. The majority of patients (88%) responded to continuation of ART and TB therapy. Thus, TB-IRIS is common in Ethiopian patients beginning ART, occurring in 7% of patients initiating antiretroviral therapy.
Introduction
Since its introduction, potent antiretroviral therapy (ART) has led to significant improvements in the quality of life of patients with HIV coinfected with tuberculosis (TB) by decreasing the incidence of TB and reconstituting immunity.1–3 However, some patients initiating ART experience a paradoxical clinical deterioration due to opportunistic infections that worsen or arise during immune recovery, thus leading to the Immune Reconstitution Inflammatory Syndrome (IRIS). 4 IRIS may be due to infectious or noninfectious causes. The symptoms are the result of a dysregulated inflammatory response to a variety of opportunistic infections including TB. 5 In general, IRIS is defined as a clinical worsening due to either (a) a previously unrecognized subclinical opportunistic pathogen arising after beginning ART (“unmasking” IRIS) or (b) a previously known and treated opportunistic pathogen that worsens in the setting of effective ART (“paradoxical” IRIS).6,7 IRIS can present any time after the initiation of ART, but the majority of cases occur within 90 days of initiation of ART. 8
Tuberculosis (TB) is a common disease associated with IRIS in developing countries, especially sub-Saharan Africa. Clinical manifestations of TB-IRIS include fever and worsening of previous pulmonary symptoms. Pleural effusions and mediastinal lymphadenopathy are common. 9 Other symptoms are nonspecific and include fever, weight loss, and worsening of pulmonary symptoms. 5 Rare forms of TB-IRIS include intracranial tuberculoma and disseminated tuberculosis. Paradoxical central nervous system TB has been reported to occur later as compared to other TB-IRIS, often 5 to 10 months after beginning ART. 10
There is a high burden of HIV-TB coinfection in Ethiopia 11 and the number of patients beginning ART is increasing. 12 Patients with HIV infected with TB constitute one of the most difficult medical management problems. Despite this fact, there is limited data addressing TB-IRIS in Ethiopia. Knowledge gained from studying TB-IRIS may lead to a change in patient management that could decrease the incidence of TB-IRIS and thus decrease the burden on health facilities caring for these patients. Thus, we set out to determine the incidence of TB-IRIS in our HIV-infected patient population and when it arose after beginning ART.
Methods
This study was performed at Dessie Referral Hospital, the largest hospital in the East Amhara region and a site with the fourth largest number of patients on ART in Ethiopia. As of November 2008, the cumulative number of patients enrolled with HIV was 10,374, of which 7685 of the patients began ART. 12
Study Population
This was a retrospective case-control study. All HIV patients who were at least 15 years of age and began ART at the Dessie Referral Hospital according to Ethiopian guidelines 13 were included. Patients initiating ART between January 1, 2007, and September 30, 2008, were identified and their medical records reviewed to identify clinical events consistent with TB and IRIS within 6 months of initiation of ART.
Patient Sampling
Sample sizes were calculated using Epi Info version 3.3.2 (www.cdc.gov/epiinfo/epiinfo.htm) for an unmatched case-control study. The largest sample size that could detect at least 15% difference between cases and controls with 95% confidence, 80% power, and a control to case ratio of 2:1 was chosen. The required sample size was 274 controls and 137 cases. Consecutive treatment-naïve patients initiating ART who fulfilled the inclusion criteria and without exclusion criteria were included in the sample until the sample size for cases and controls was satisfied.
Tuberculosis
Diagnosis of TB was based on the Ethiopian TB prevention and control guidelines. 11 Pulmonary tuberculosis was established when sputum was acid-fast bacillus (AFB) smear-positive or if AFB smear-negative, patients had 3 negative sputum examinations for AFB and no response to a course of broad spectrum antibiotics with radiological signs suggestive of TB and a decision by the physician to treat TB. Extrapulmonary TB was diagnosed by histopathological findings suggestive or diagnostic of tuberculosis or by the clinical presentation as allowed by the Ethiopian guidelines.
Tuberculosis-IRIS
Charts of patients were reviewed for worsening of TB or onset of TB within 6 months of the initiation of ART. Patients were categorized as new or “unmasked” cases of TB or worsening or “paradoxical” cases of TB. New or “unmasked” cases were patients who began ART without any evidence of TB. Worsening or “paradoxical” cases were patients who initiated ART while on standard TB medications for at least 2 weeks. Patients on TB medications were eligible for the study if they were adherent to their TB medications and if the decision to initiate ART was made after their response to the anti-TB medication was assessed as adequate. Worsening or “paradoxical” TB was confirmed if a patient who responded to standard TB medications redeveloped clinical features and/or new radiological signs suggestive of TB after initiating ART and no other cause was identified after clinical and laboratory evaluation. The control group for the TB-IRIS patients consisted of HIV-infected patients begun on ART during the same time period who did not develop TB-IRIS within 6 months of beginning ART.
Patients who developed TB or worsening of previously diagnosed TB while on ART and whose CD4 count at the time of or within 6 months of beginning ART was equal to or above the pretreatment baseline value were considered TB-IRIS cases. Exclusion criteria for TB-IRIS were documentation of ART treatment failure over the 6-month period; progression of HIV/AIDS with a drop in CD4 count, or with the development of an opportunistic infection (other than TB) or malignancy after initiation of ART; documented noncompliance with ART or diagnosis of multidrug-resistant TB. Furthermore, patients were excluded from the study if they did not complete the observation time for any reason before the diagnosis of TB, died before diagnosis of TB, were lost to follow-up, or transferred out to another health facility before the diagnosis of TB during the observation period.
Statistical Analysis
Patients who initiated ART and developed IRIS were identified and the demographic and clinical characteristics of those cases and controls were compared. Chi-square or Fisher exact test were used for categorical variables. Student t-test was used for baseline CD4 count and the Mann Whitney U test for baseline weight. Logistic regression modeling was used for such variables as weight, baseline CD4 count, the WHO Clinical Stage, a prior history of TB, and use of isoniazid (INH) prophylaxis. All analyses were performed using SPSS 15.0 (SPSS, an IBM Co, Chicago, Illinois).
Ethical Issues
Ethical clearance of this study was obtained from the Gondar University Ethical Board and Dessie Referral Hospital. Confidentiality of the medical records of patients was maintained and data abstracted anonymously.
Results
From January 1, 2007, to September 30, 2008, a total of 2662 patients were initiated on ART. Of the total, 685 or 26% were ineligible for inclusion (168 patients transferred in or out of the clinic within 6 months of initiation of ART; 109 died; 60 were nonadherent to treatment and 348 patients had incomplete or lost records). Of the 1977 patients eligible for the study, 143 (7%) developed TB-IRIS within 6 months of initiation of ART. The first 277 patients who did not develop TB-IRIS within 6 months of beginning ART were selected as the control group, making a total sample of 420 patients for the study. The demographic and clinical characteristics of TB cases and controls are shown in Table 1 . There is no statistically significant difference between cases and controls with regard to gender, age, or weight.
Demographic and Clinical Characteristics of TB-IRIS Cases and Controls

Abbreviations: ART, antiretroviral therapy; NS, not significant; TB-IRIS, tuberculosis−Immune Reconstitution Inflammatory Syndrome; TB, tuberculosis.
Of the 143 cases of TB-IRIS, 9 (6.3%) patients were in WHO clinical stage 1; 11 (7.7%) in stage 2; 50 (35%) in stage 3; and 73 (51%) in stage 4, whereas, in the 277 control patients, 24 (8.7%), 46 (16.6%), 129 (46.6%), and 78 (28.1%) were in stages 1 to 4, respectively. The difference between cases and controls was statistically significant (P < .005) with regard to WHO clinical stages 3 or 4 (Table 1). In addition, 88 (61.5%) of TB-IRIS cases and 123 (45.1%) of control patients had a history of WHO clinical stage 3 or 4 (P = .002). The mean baseline CD4 count for cases was 102 ± 4 cells/μL and for controls, 132 ± 3 cells/μL (P < .005). There was no significant difference between TB-IRIS cases and controls with regard to when ART was initiated or the use of cotrimoxazole prophylaxis for PCP or INH prophylaxis. However, a history of a previous opportunistic infection occurred significantly more often in TB-IRIS cases than with control cases (P < .05).
Table 2 shows the clinical manifestations of the TB-IRIS cases and treatment outcome. The majority of TB-IRIS cases were new 132 (92%) while worsening cases constituted only 11cases (8%). Pulmonary presentations of TB predominated. However, as shown in Table 2, extra pulmonary TB occurred in many patients, lymphadenitis in 17, intracranial disease in 9, peritoneal in 3, and disseminated disease in 15.
Clinical Manifestations, Treatment, and Outcomes of TB-IRIS Cases

Abbreviations: ART, antiretroviral therapy; NS, not significant; TB-IRIS, tuberculosis−Immune Reconstitution Inflammatory Syndrome; TB, tuberculosis.
Concerning the treatment outcome of patients, 125 (87.4%) of cases recovered while 11 (7.7%) died and the outcome of 7(4.9%) patients was unknown. Steroids were used in treatment in only 8 or ∽6% of TB-IRIS cases and ART was not discontinued on any patient.
“Unmasked” and “paradoxical” cases of TB-IRIS were compared in Table 3 . The median time to onset of new (“unmasked”) TB was 38 days (range 10-106 days) and for worsening (“paradoxical”) cases was 18 days (range 12-69 days; P < .05) and “unmasked” patients improved with therapy and there were no deaths in this group (P < .001).
Clinical Characteristics of New and Worsening TB-IRIS Cases
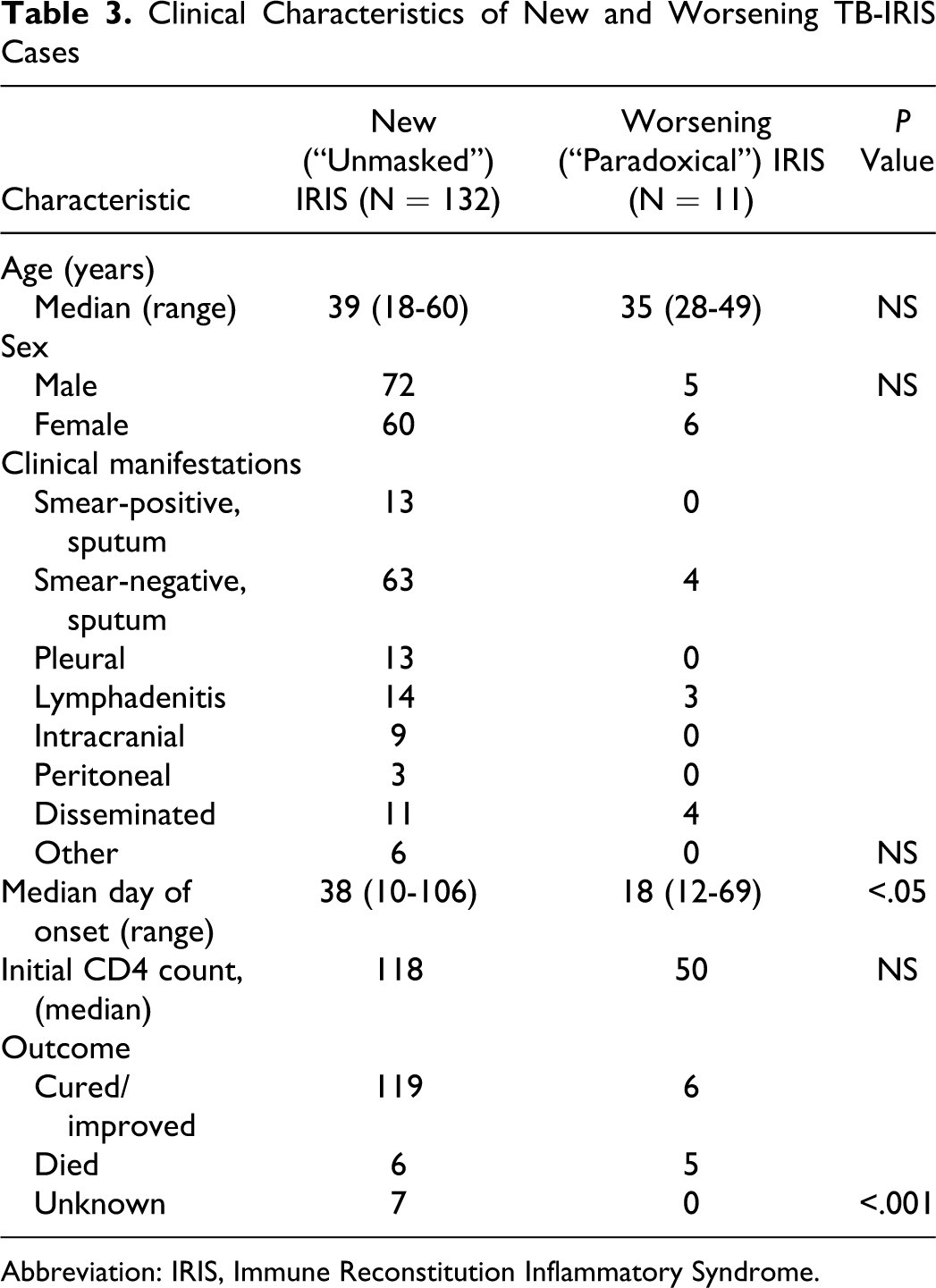
Abbreviation: IRIS, Immune Reconstitution Inflammatory Syndrome.
Discussion
In a large retrospective analysis in the United Kingdom examining all manifestations of IRIS, ∽25% of patients exhibited one or more IRIS episodes after ART initiation. 14 A prospective study done in South Africa examined 423 patients and estimated the incidence of all forms of IRIS to be 10%. 8 Studies to determine the incidence of TB-IRIS among HIV-infected patients demonstrated a wide range of 11% to 45% incidence over a duration of 1 year.1,2,7,8,15,16 In a South African prospective study, only baseline CD4 count was an independent predictor of IRIS and a high CD 4 count was protective against IRIS. 8
Ethiopian studies to determine the incidence of IRIS include one in an Addis Ababa, Ethiopia hospital to determine the incidence of IRIS and the pattern of opportunistic infections among 186 HIV/AIDS patients receiving ART. 17 The incidence of IRIS was 17.2%. The mean number of days for IRIS occurrence ranged between 26 and 122 days, with a mean of 80 days. A similar study done in the Dessie Referral Hospital determined that ∽3% of patients with TB-IRIS had severe-enough disease to be hospitalized. 18 Our current study in the same institution revealed that 7% or 143 patients of 1977 outpatients initiating ART developed TB-IRIS within 6 months of beginning ART. A low baseline CD4 count at the initiation ART and a history of WHO Clinical Stage 3 or 4 were identified as independent risk factors. The presence of active TB would classify an HIV patient as WHO Stage 3 or 4 by current definitions, but the patients in this study were classified as stage 3 or 4 before the diagnosis of TB, thus reinforcing the concept that the patients with TB-IRIS were those who were sicker as well as had low CD4 counts.
Fully, 72% of patients developed TB-IRIS within 2 months of initiation of ART consistent with previous studies.1,2,8 However, unlike other reports 10 the majority of our cases were new or “unmasked” (92%) despite the fact that up to 23% of our patients initiated ART while on TB medications. One explanation for this difference may be that TB was not diagnosed in a timely fashion in our HIV-positive patients.
In order to decrease the incidence of TB-IRIS in this patient population, one must start ART earlier in the course of HIV disease when CD4 counts are greater. Furthermore, more rigorous screening for TB at the time of initiation of ART seems prudent, especially with the majority of our cases being new or “unmasked” TB that had a mean onset of disease at 38 days after beginning ART.
TB-IRIS affects a significant proportion of patients initiating ART in Ethiopia and the clinical manifestations are diverse requiring a high index of suspicion for its diagnosis.
Footnotes
Acknowledgements
Yemane Berhane, MD, MPH, PhD, advised KA on the study. This study was performed in partial fulfillment of the requirements for MPH.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
