Abstract
Keywords
Introduction
Pediatric HIV infection can be acquired from the mother during pregnancy, delivery, and breast-feeding. In developing countries, without interventions, 25% to 45% of HIV-poistive pregnant women transmit HIV infection to their children. A notably high number of infants are being infected with HIV every year; in 2008 alone, 430 000 children were infected worldwide, 90% of them through mother-to-child transmission (MTCT). Worldwide, 2.1 million children were living with HIV infection in 2008, and 90% of them in developing countries.1,2
Progress made in knowledge regarding HIV infection has demonstrated that the risks of MTCT can significantly be reduced. As a result, an increasingly high number of developing countries are integrating programs to prevent MTCT into their routine mother and child health care services. 3
In recent years, the international community has investigated the impact and effectiveness of prevention of MTCT (PMTCT) in real life. 4 Current data on the effectiveness of PMTCT programs are emerging, with most of the reports looking at the role of antiretroviral (ARV) drugs.5,6
The objective of this study is to explore how nonchemoprophylaxis factors, including infant feeding practices, mother’s HIV status disclosure, mode and place of delivery, infant gender, and maternal age, are related to MTCT in PMTCT program settings in Zambia.
Methods
Context
Since 2005, the President’s Emergency Plan for AIDS Relief (PEPFAR) through the United States Agency for International Development funded Zambia Prevention, Care, and Treatment Partnership (ZPCT) has been supporting scale-up of PMTCT services in 5 of the 9 provinces in Zambia, at all levels of the health care systems, from rural health centers to tertiary hospitals. All 210 PMTCT-supported health facilities have access to CD4 testing for pregnant women attending antenatal clinics (ANC), who test positive either on-site where there is a laboratory with CD4 capacity, or off-site through a sample referral system for the smaller health facilities with no such capacity. Based on the CD4 results, the health care workers are able to triage the HIV-positive pregnant women to receive antepartum ARV regimen per the national guidelines. The national guidelines during the study period stipulated that pregnant women with CD4 less than 350 cells/mm3 should receive highly active antiretroviral treatment (HAART), while those with CD4 more than 350 cells/mm3 should receive zidovudine (AZT) from 28 weeks with intrapartum single-dose nevirapine (sd NVP) and lamivudine (3TC) added on. Single-dose NVP is offered as the last choice option when women, for one reason or another, are unable to get AZT short-course prophylaxis. 7 Prior to the change in guidelines, sd NVP was the main choice and the standard of care for PMTCT. In August 2007, ZPCT, in close collaboration with the Ministry of Health and the provincial health office, established a DNA polymerase chain reaction (PCR) laboratory at the Arthur Davison Children Hospital, a tertiary facility located in the Copperbelt Province. This extension of the PMTCT program supports early infant diagnosis and strengthens pediatric HIV care. Dried blood spot (DBS) samples are collected at the facilities by trained staff and then transported periodically to a central hub using a motorcycle provided at the district for specimen referral of laboratory samples for investigation. At the central hub, the DBS packages are examined before being sent to the PCR laboratory using expedited mail service of the Zambia postal services. Test results are sent back to health facilities using the same process. Dried blood spot samples ideally should have been collected at 6 weeks, but due to the lack of the PCR testing facility until recently coupled with delays in attending the under-five clinics has led to DBS samples being collected from infants for for DNA PCR, at different time points. Thus, to allow for possible age, history, and cohort effects, we chose to summarize our data according to 3 age groups: 0 to 6 weeks, 6 weeks to 6 months, and 6 to 12 months.
Study Design
This descriptive observational study analyzed all DNA PCR results and client information on all DBS samples from perinatally exposed infants 0 to 12 months of age sent to the Arthur Davison Children’s Hospital PCR laboratory from the provinces of Copperbelt, Central, Northern, North-Western and Luapula, from September 2007 to January 2009. The breast-feeding and nonchemoprophylactic component was a substudy of the main study to assess the effectiveness of PMTCT intervention. 6
Inclusion criteria
All infants, aged 0 to 12 months, born to HIV-positive mothers, and who underwent a DNA PCR test, were eligible to be included.
Data Collection, Entry, and Analysis
Health care workers at maternal, neonatal, and child health care units in the health facilities routinely filled the PCR requisition form for all babies who were perinatally exposed to HIV, during DBS sample collection. The client information collected included age of the infant, maternal age, breast-feeding status, type of ARV regimen given to mother and/or baby, and whether the mother disclosed her HIV status to her spouse. Information on history of the mode of infant feeding up to the time the DBS samples (mixed, formula, or exclusive breast-feeding) were collected from those who had stopped or were still breast-feeding.
A Microsoft Access database in the PCR laboratory was used to store and process data from the PCR request form and the results from the PCR laboratory. Internal consistency of the data was assessed and electronic data were verified against paper records and cleaned where appropriate. Repeat samples were excluded from the analysis.
The vertical transmission rates of HIV were estimated, along with 95% confidence interval (CI), separately for each age group and for specific subpopulations (eg, among mothers who did or did not disclose their HIV status). Mantel-Haenszel methods were used to calculate
Ethical Approval
Ethical approval was granted by the University of Zambia Research and Ethics Committee and Protection of Human Subjects Committee of Family Health International, North Carolina, United States.
Results
Characteristics of Study Population
A total of 8237 samples from infants aged between 0 and 12 months from 206 facilities in 39 districts in the 5 provinces were analyzed. In all, 49% of the infants were males, 18% were aged 0 to 6 weeks, 58% were aged 6 weeks to 6 months, and 24% were aged 6 months to 12 months (Table 1).
Mother and Child Characteristics

Abbreviations: AZT, zidovudine; HAART, highly active antiretroviral treatment; NVP, nevirapine; PCR, polymerase chain reaction; PMTCT, prevention of mother-to-child transmission.
Infant Feeding Status
In all, 84% of the mothers ever breast-fed their children. Among those who never breast-fed and in instances where both mother and infant received intervention, the estimated transmission rates of HIV were 2.3% (95% CI 0.5, 4.2), 5.8% (95% CI 4.0, 7.5), and 4.9%(95% CI 1.8, 8.1) at 0 to 6 weeks, 6 weeks to 6 months, and 6 to12 months, respectively.
Of those who ever breast-fed, 96.2% were still breast-feeding at 0 to 6 weeks with 52.8% still breast-feeding after 6 to 12 months. The estimated transmission rates of HIV among those wherein both infant and mother received intervention and who were still breast-feeding compared to those who had stopped breast-feeding at 0 to 6 weeks were 7.5% (95% CI 5.8, 9.1) and 10.7% (95% CI 0, 23.0), respectively. This difference was not statistically significant (

Estimated HIV transmission rates by infant age group, breast-feeding status, and infant feeding method among infants for whom both mother and infant received PMTCT intervention. PMTCT indicates prevention of mother-to-child transmission.
Disclosure of HIV Status
A majority of the mothers (94.3%) claimed that they had disclosed their sero status. Across all infants, nondisclosure was associated with a higher transmission especially after 6 weeks (Table 2). In instances where both mother and baby received an intervention, the trend remained the same but the differences were not significant.
HIV Transmission Rate by Mother’s Disclosure Status and Age Group across All Infants and among Those Wherein Both Mother and Infant Received PMTCT
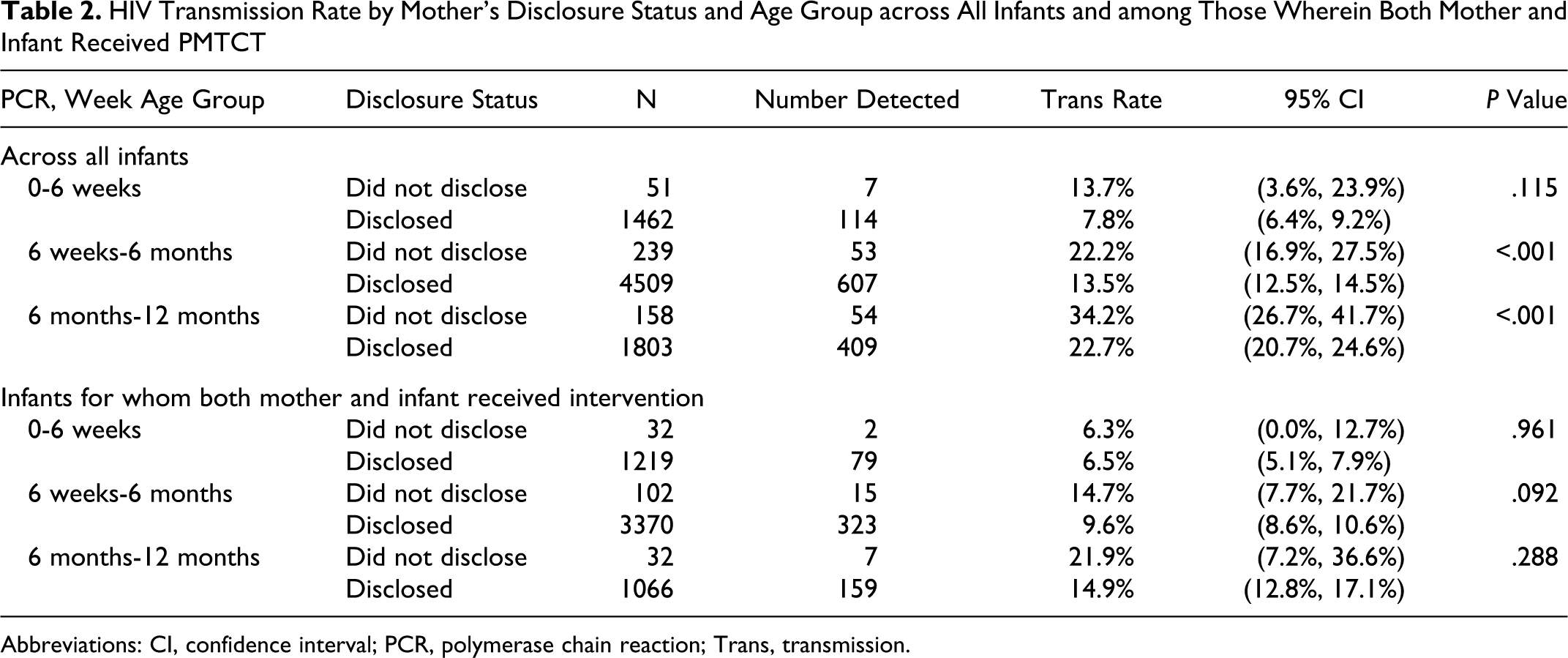
Abbreviations: CI, confidence interval; PCR, polymerase chain reaction; Trans, transmission.
Transmission Rate of HIV by Infant Gender
Among infants for whom both mother and infant received intervention (where intervention is defined as receiving ARV drugs), the estimated transmission rates for male babies was 6.3% compared to 6.7% in females at 0 to 6 weeks and 8.9% versus 10.5% at 6 weeks to 6 months, respectively. At 6 to 12 months, the estimated transmission rates for males and females were both 15.1%. These differences were not statistically significant.
Age of Mother and Transmission Rates of HIV
Maternal age of 30 years and older was associated with a lower HIV transmission rate across all age groups and significantly after 6 weeks (

Estimated HIV transmission rates by infant age group and maternal age along with 95% confidence intervals among infants for whom both mother and infant received PMTCT intervention. PMTCT indicates prevention of mother-to-child transmission.
Location and Mode of Delivery
The estimated transmission rates of HIV were similar among mothers who delivered at home compared to those who delivered in a health facility, and the differences were not statistically significant when both the mother and infant received PMTCT interventions (Table 3).
Transmission Rates by Location of Delivery and Age Group

In the multivariable logistic regression model, only breast-feeding status was consistently associated with vertical transmission across all ages (Table 4). Maternal age of less than 30 years was associated with a high vertical transmission rate for infants aged 6 to 12 months but not for the younger age groups.
Estimated Adjusted Odds Ratio a and 95% Confidence Interval for Maternal and Infant Characteristics, by Age Group
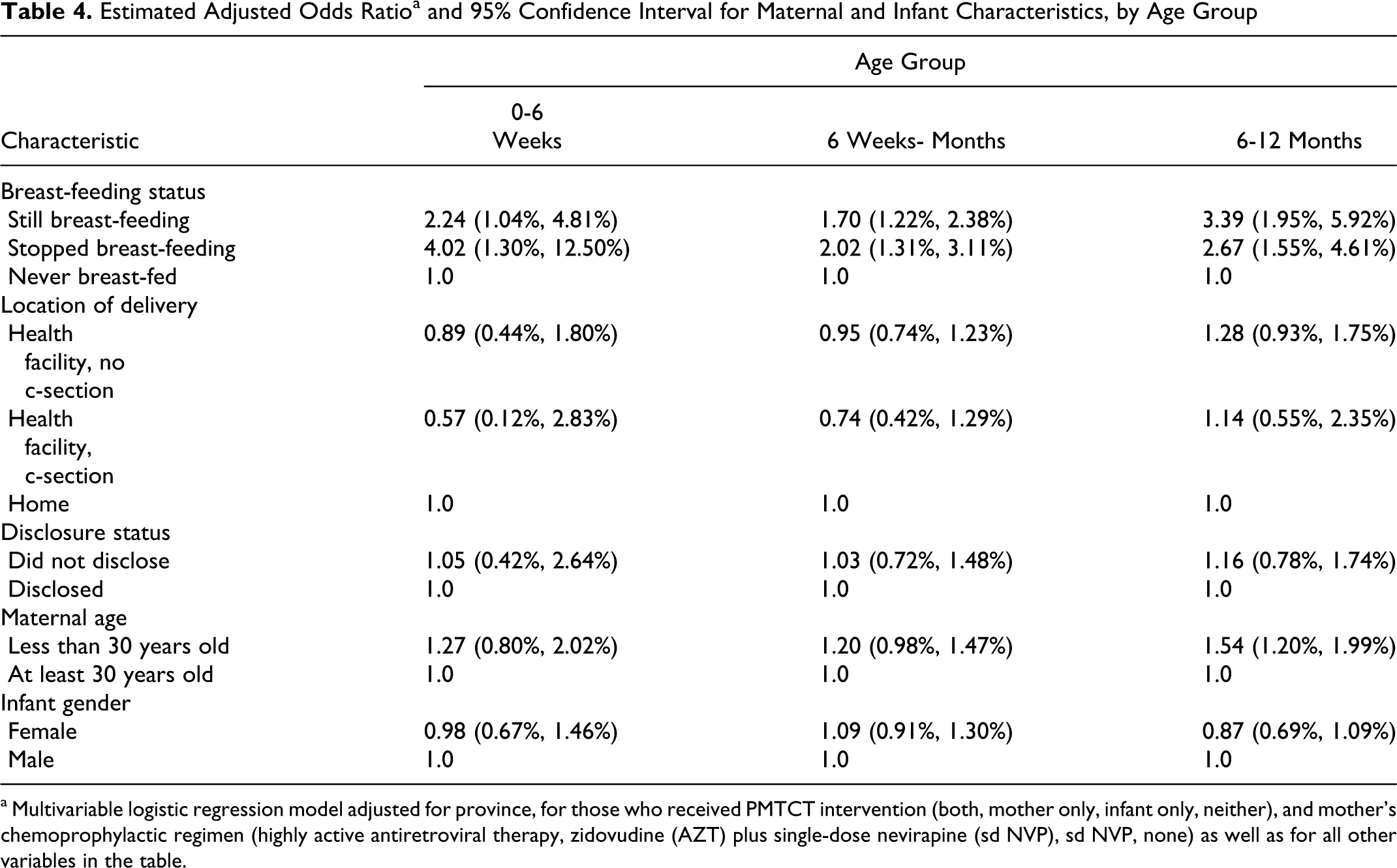
a Multivariable logistic regression model adjusted for province, for those who received PMTCT intervention (both, mother only, infant only, neither), and mother’s chemoprophylactic regimen (highly active antiretroviral therapy, zidovudine (AZT) plus single-dose nevirapine (sd NVP), sd NVP, none) as well as for all other variables in the table.
Discussion
Our finding is in line with the literature in clinical trial settings: the risk of MTCT is higher when breast-feeding is prolonged and not exclusive; the uniqueness of our studies is that it showed the same trend but in a real-life setting. In the Mashi study, the 7-month HIV-infection rate was 5.6% in the formula fed group compared to 9% in the breast-fed group. Cumulative infant mortality was, however, significantly higher in the formula fed group at 7 months. 8 A number of studies have also shown that mixed feeding is associated with significant HIV transmission risk compared with exclusive breast-feeding and exclusive formula feeding.6,9,10 This is confirmed by our study findings where mixed feeding was consistently associated with higher transmission rates of HIV across all age groups. Replacement feeding, however, is not without challenges; preparation of formula under hygienic conditions, contamination, clean water source, and stigma makes alternative feeding difficult.11,12
Clearly, prolonged breast-feeding is associated with increased postnatal infection, and this is evident by the high transmission rates of HIV after 6 months.
In a study by Shapiro et al, it was demonstrated that the use of ARV drugs during the breast-feeding period has the potential of reducing vertical transmission. 13 This intervention has the potential of making breast-feeding, which is a major cause of postnatal transmission, safer and hence should be considered in PMTCT programming.
In a subset of the Zambia Exclusive Breast-feeding study, maternal nonadherence to ARV prophylaxis was associated with home births. 14 Other studies in Malawi and Kenya have shown that delivery in the hospital is associated with ingestion of the intrapartum dose of ARV chemoprophylaxis compared to home deliveries.15,16 However, this finding is not supported from the results of our study. The transmission rates of HIV did not vary significantly by location of delivery (facility vs home), provided both the mother and the infant received ARV prophylaxis. This can probably be explained by the strategy of offering sd NVP at first contact, which offers a safety net for women who attend the ANC once and do not come back to deliver at the health facility.
Disclosure of sero status to a partner is a challenging element in PMTCT programming. Disclosure may possibly lead to adherence to and support for appropriate infant feeding options. Unfortunately, available studies have documented women’s experience to disclosure to their partners have led to stigmatization, family rejection, breach of confidentiality, or accusations of infidelity. 17 In this study, disclosure was associated with decreased transmission rates of HIV across all categories but more significant after 6 weeks. Disclosure, however, did not seem to play a role in cases where both mother and infant received intervention.
In our study, women over 30 years of age had lower transmission rates across all age groups. When adjusted for other factors, maternal age of less than 30 was associated with higher transmission rates only in infants aged 6 to 12 months. Older women are more likely to be more economically empowered and more experienced in infant feeding issues, thereby indirectly reducing vertical transmission.
This study confirms the role of infant feeding in reducing vertical transmission of HIV. It underlines the importance on the use of ARV drugs remains a predominant factor in PMTCT. In instances where both infants and mother receive prophylaxis, the role of location, mode of delivery, and disclosure have a limited effect.
Our study, though informative, has some limitations. It did not include samples from HIV-exposed infants who died before 6 weeks and those who did not bring their infants to a health facility for their immunization.
Furthermore, given the repeated cross-sectional nature of our data, we are unable to distinguish any effects of age from cohort or history effects; for example, the effect of age is masked with any changes in PMTCT regimens that occurred over time. In addition, using observational data precludes making strong causal statements when comparing estimates of HIV transmission rates.
Conclusion
Among the nonchemoprophylaxis factors, infant feeding option remains an important modifiable factor to prevent pediatric infection. Addressing this in PMTCT programming will help reduce pediatric HIV infections.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Support for this paper was provided by Family Health International (FHI)/Zambia Prevention Care and Treatment Partnership with funds from the U.S. President's Emergency Plan for AIDS Relief (PEPFAR) through U.S. Agency for International Development (USAID) Cooperative Agreement No. 690-A-00-04-00319-00. The views expressed in this publication do not necessarily reflect those of FHI.
