Abstract
Radial arterial line placement is often utilized for hemodynamic monitoring in critically ill patients or during invasive procedures. This is generally considered safe with a low incidence of complications compared with catheters placed in the brachial and femoral arterial locations. One of the rare adverse outcomes associated with radial artery access is hemorrhage resulting in compartment syndrome. In the following case, duplex ultrasound played a pivotal role in the diagnosis of the cause of forearm compartment syndrome. An 83-year-old male presented to the vascular laboratory 9 days after undergoing a cardiac procedure via left radial artery access. He reported persistent forearm pain, swelling, and reduced finger mobility. Physical examination revealed extensive bruising along the forearm, extending to the medial upper arm above the elbow. Duplex ultrasound revealed a patent left radial artery with evidence of extraluminal arterial flow consistent with pseudoaneurysm and an associated hematoma. He underwent urgent surgical intervention, including left forearm fasciotomy, carpal tunnel release, and radial artery repair. Forearm compartment syndrome due to radial artery injury is a rare but a serious complication of radial line placement for monitoring or procedural access. Delayed recognition can result in severe complications such as irreversible nerve damage and muscle necrosis. This case highlights the important role of duplex ultrasound in the detection of arterial pseudoaneurysm, which can then prompt referral for appropriate surgical management.
Keywords
Introduction
Radial arterial line placement is often performed for hemodynamic monitoring in critically ill patients or in patients undergoing invasive procedures. Studies suggest that radial arterial line placement is relatively safe, with lower complication rates compared with lines placed in the femoral or brachial arteries. 1 One of the rare adverse outcomes associated with radial arterial line placement is hemorrhage resulting in the development of compartment syndrome. This case demonstrates the role of duplex ultrasound in the diagnosis of a complicated radial artery injury that required surgical intervention.
Case Report
An 83-year-old man presented to his primary care provider (PCP) with complaints of dizziness, syncope, and chest tightness. After evaluation, the PCP noted that the patient was in atrial fibrillation (AF) with rapid ventricular response (RVR). The patient was referred directly to the Emergency Department (ED) for further evaluation, where he was confirmed to have AF with RVR and new heart failure with reduced ejection fraction (HFrEF) of 35%.
Of note, he had a known prior diagnosis of AF on apixaban as an outpatient. His complex past medical history included diagnoses of hypothyroidism, hypertension, hyperlipidemia, deep venous thrombosis and pulmonary embolism, coronary artery disease status post 4-vessel coronary artery bypass (CABG) as well as a subsequent CABG revision, atrial septal defect repair, aortic valve disease status post-replacement with bovine pericardial valve with subsequent aortic valve replacement revision, aortic root replacement, and tricuspid valve repair.
The patient was admitted to the cardiology service. Additional testing included a cardiac stress test to rule out reversible ischemic causes for his reduced ejection fraction, which was negative. The patient was ultimately recommended for cryoablation, a minimally invasive cardiac procedure that utilizes extreme cold to disable heart cells that create irregular rhythms, thus restoring regular heart rhythm. 2 The patient agreed to undergo scheduled AF cryoablation. His apixaban was held 2 days prior to the procedure and he was initiated on an enoxaparin bridge, which was continued until the evening prior to his procedure.
During the procedure, a radial arterial line was placed for continuous blood pressure monitoring. Three attempts were made to place a 20-gauge, and, subsequently, a 22-gauge arterial line via the left radial artery. Line placement was done under ultrasound guidance. Intraluminal access was confirmed by a flash of arterial blood into the reservoir. After each unsuccessful attempt, manual pressure was held for hemostasis. The time duration of this pressure hold was not recorded. Ultimately, a 24-gauge arterial line was successfully placed in a single attempt after switching to the right radial artery. After this, the procedure was performed without apparent complication.
The patient was given 18000 units of heparin for anticoagulation during the procedure, and 40 mg of protamine was given for reversal after the procedure. Post-reversal, the final activated clotting time (ACT) was 152 seconds. In addition, he was noted to have palpable radial and brachial pulses bilaterally prior to the procedure, although an Allen’s test to confirm palmar arch patency was not documented. The post-procedural pulse exam was also not documented.
Two hours post-procedure, the patient complained of pain and swelling in the left forearm. Bedside evaluation demonstrated mild swelling of the left forearm but soft compartments. Sensation was noted to be intact in the radial nerve distribution but decreased in the median and ulnar nerve distributions. In addition, the left radial pulse was noted to be nonpalpable, but a monophasic arterial signal was identified in the radial and ulnar arteries at the wrist. Based on this clinical evaluation, the symptoms were attributed to median and ulnar nerve neuropraxia, possibly due to compression or irritation due to procedural positioning, soft tissue edema, or a small hematoma. As his neurological symptoms appeared to be improved on serial exams, the decision was made to manage the patient expectantly with elevation of the arm with a Murphy sling and continue monitoring.
The patient was admitted overnight for observation. Twelve hours post-procedure, evaluation revealed improved sensation and intact motor function of the left hand. Capillary refill was noted to be normal with no evidence of distal malperfusion on clinical exam, although a repeat pulse exam was not documented. The forearm compartments were evaluated, and only the volar compartment was noted to remain edematous, but it was soft and improved compared with prior examinations.
At 24 hours post-procedure, the patient was discharged to home after a final evaluation confirmed resolved median and ulnar neuropraxia and soft compartments, with no concern for compartment syndrome. Capillary refill was noted to be normal in the left hand, but a pulse exam was not documented.
At 9 days post-procedure, the patient was seen urgently by a cardiac nurse due to swelling and persistent intense pain that was not resolving with acetaminophen or ibuprofen, as well as difficulty with finger mobility. On physical examination, there was extensive ecchymosis throughout the left forearm, extending proximally to the medial upper arm, above the elbow (Figure 1). An arterial duplex was requested to evaluate for vessel injury. The patient was seen in the vascular laboratory on post-operative day 13 for arterial duplex evaluation.
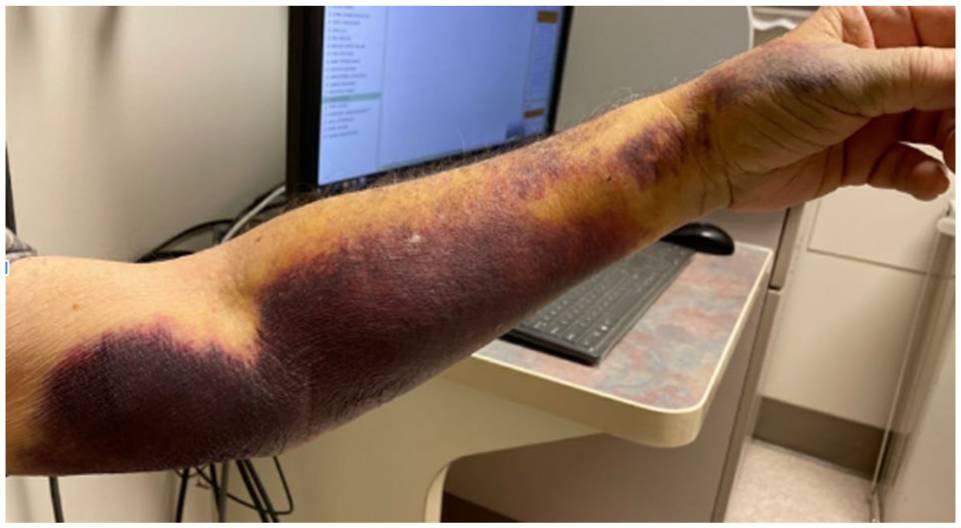
Left arm swelling and ecchymosis extending from the wrist to the mid-upper arm noted on clinical exam on post-operative day 9.
Duplex ultrasound revealed patent flow through the left subclavian, axillary, brachial, and ulnar arteries with normal biphasic and triphasic high resistive waveforms. Flow patterns in the proximal radial artery demonstrated a brisk systolic upstroke but a prominent diastolic flow reversal component (Figure 2). In the mid-distal left radial artery, an injury was identified with evidence of extraluminal arterial flow into a semi-contained sac, consistent with a pseudoaneurysm measuring 1.5 × 2.0 cm (Figure 3). At the neck, the flow patterns demonstrated a classic to-and-fro pattern with a large diastolic flow reversal component secondary to large volume pseudoaneurysm cavity filling during diastole. In addition, there appeared to be mixed-density extraluminal flow contained within the muscle compartment inferior to the left radial artery; these echoes were mobile in real time (and saved as cine loop clips) (Figure 4).
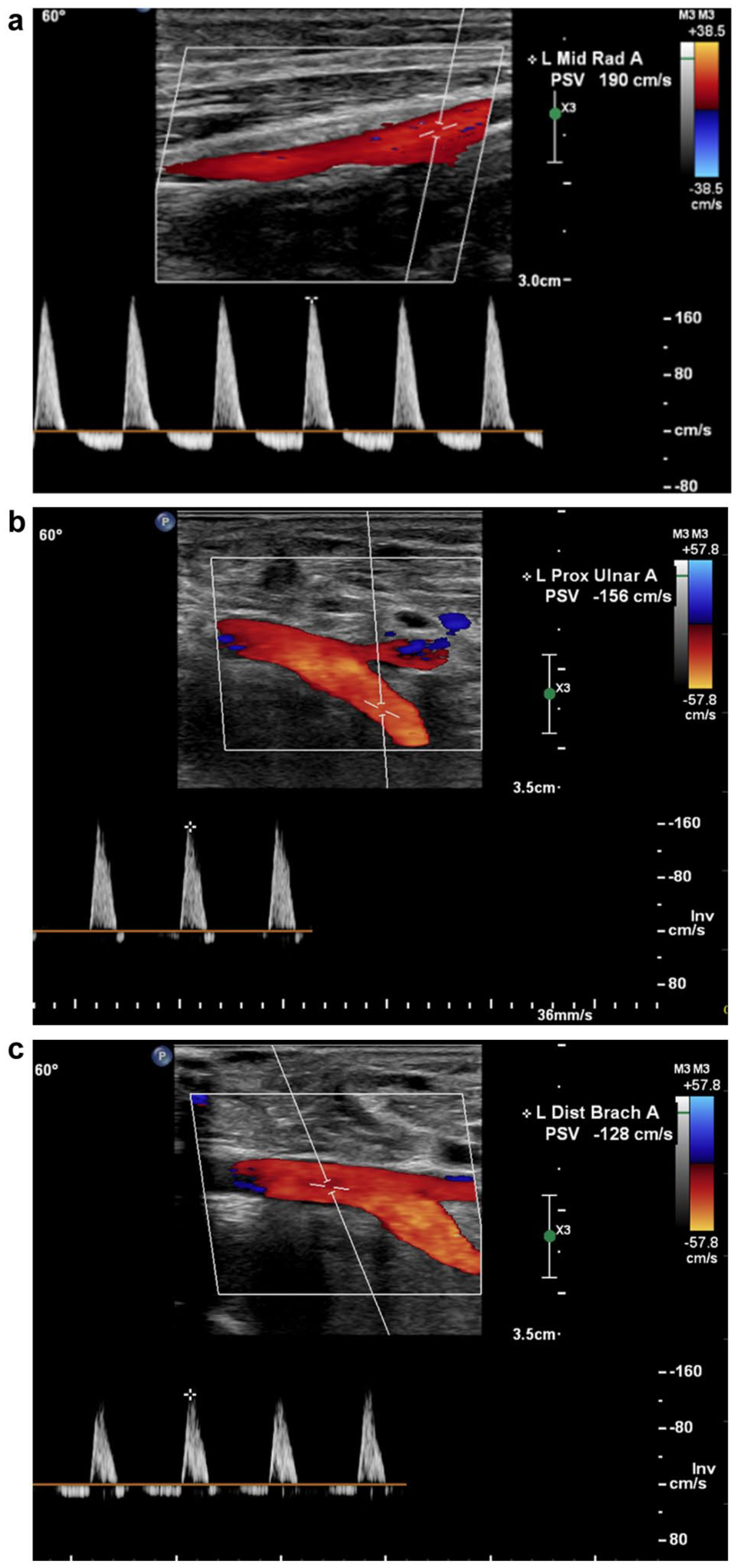
(a) Duplex ultrasound depicts the proximal-mid left radial artery, proximal to the site of the injury, in a sagittal plane. Flow is notably bidirectional. Duplex ultrasound depicts the mid-left ulnar artery (b) and the distal brachial artery (c) in a sagittal plane. Flow is notably highly resistive with minimal flow reversal during diastole.
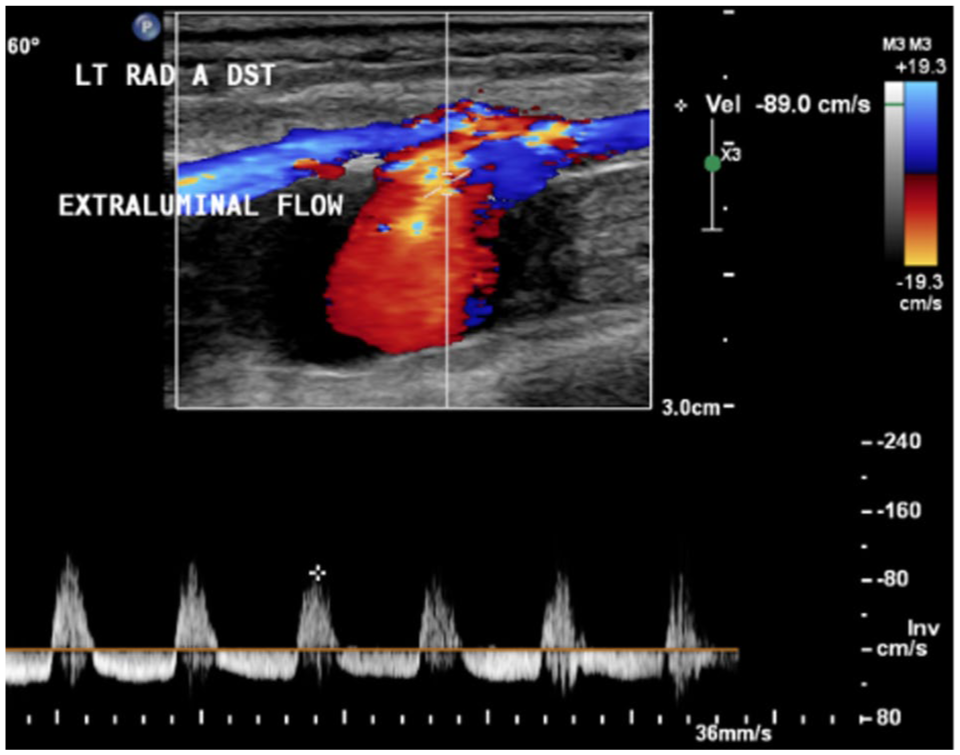
Duplex ultrasound depicts the mid-distal left radial artery in a sagittal plane.
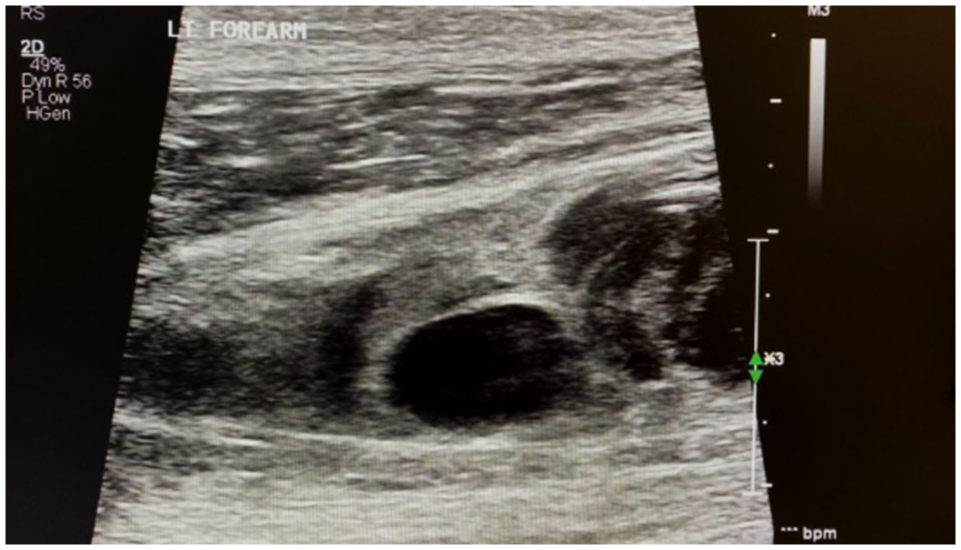
Left forearm in a sagittal view with mixed density “mobile” echoes (active flow) within the muscle compartment.
The findings were immediately communicated with the ordering provider (Figure 5), and upon their recommendation, the patient was sent to the ED. In the ED, computed tomography angiography (CTA) was performed, which correlated the vascular laboratory findings (Figure 6).
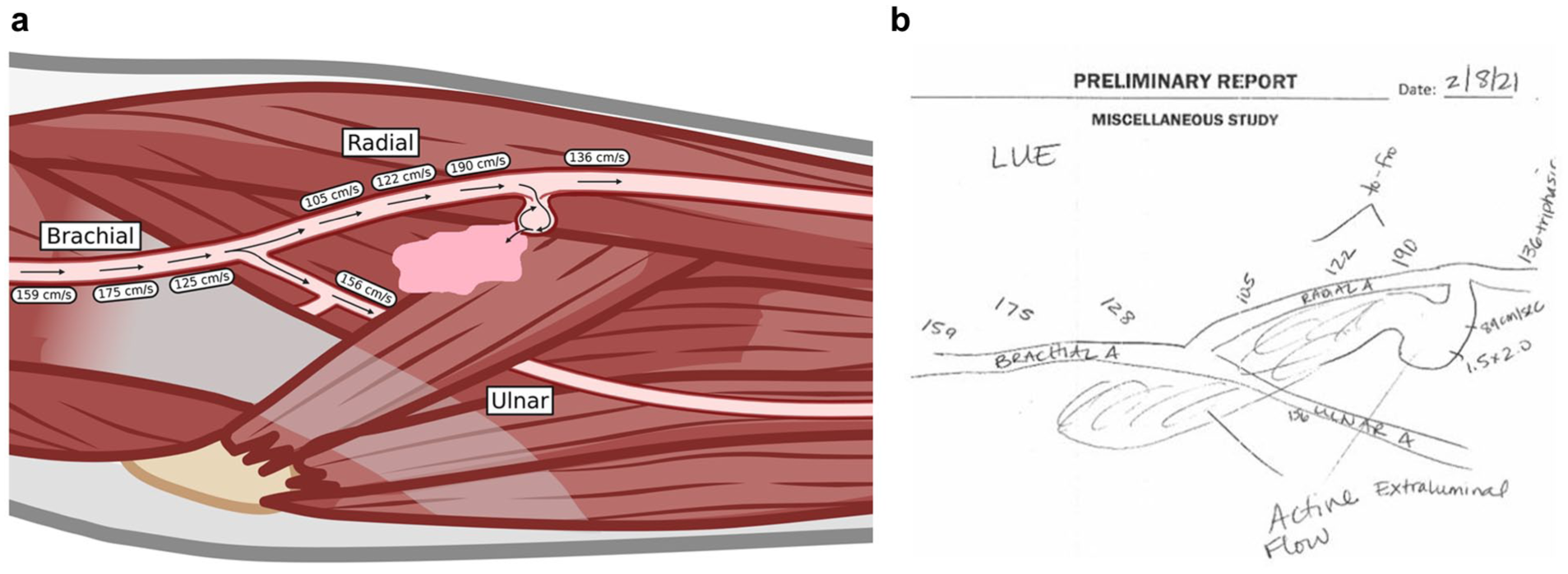
(a) Graphic representation of findings. (b) Vascular laboratory technologist’s preliminary report.
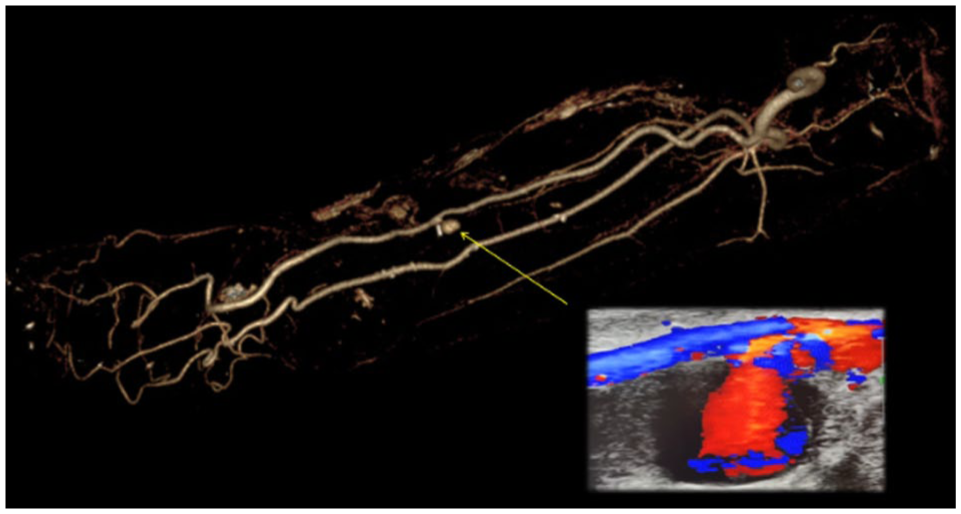
CTA demonstrated a radial artery pseudoaneurysm and mid-forearm hematoma, correlating with vascular laboratory findings.
The patient was seen by the plastic surgery hand service, who recommended urgent surgical intervention the same day (post-operative day 13). Of note, palmar arch evaluation was not performed as a part of the routine protocol for arterial duplex in our vascular laboratory, although it may be considered for cases like this one to guide surgical decision-making if the radial artery needed to be ligated. In this case, the patient was able to undergo primary repair of the left radial artery injury. In addition, he underwent left forearm fasciotomy and left carpal tunnel release during the same procedure. Retention sutures were placed along the length of the incision to prevent wound dehiscence (Figure 7). The patient was discharged home, 2 days after surgical repair.
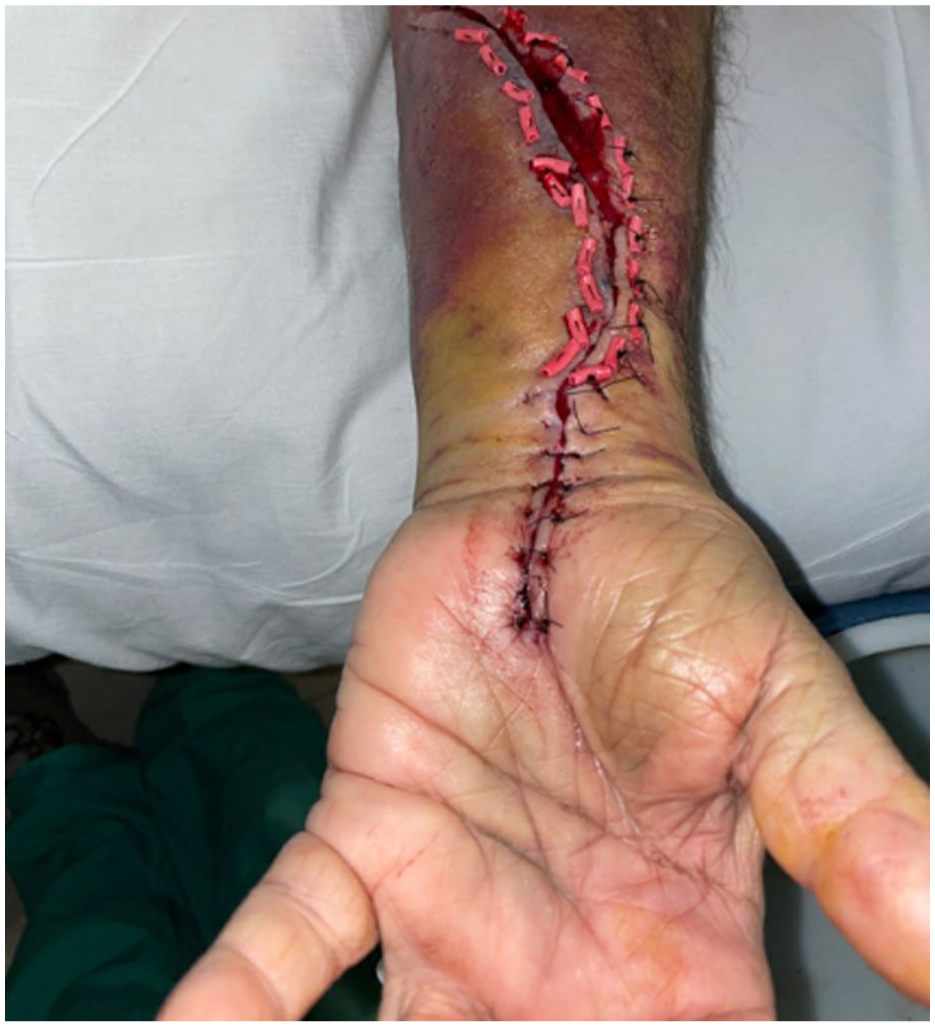
Image depicting left forearm and hand post-fasciotomy, left carpal tunnel release, and repair of the left radial artery.
By post-operative day 27, he had near-complete healing of the wound but continued to complain of persistent decreased sensation to light touch in the left hand as well as tenderness of the left forearm (Figure 8). A referral was placed to occupational therapy. At his 6-month follow-up, he had continued weakness of the left-hand grip and finger extension as well as numbness and neuropathy.
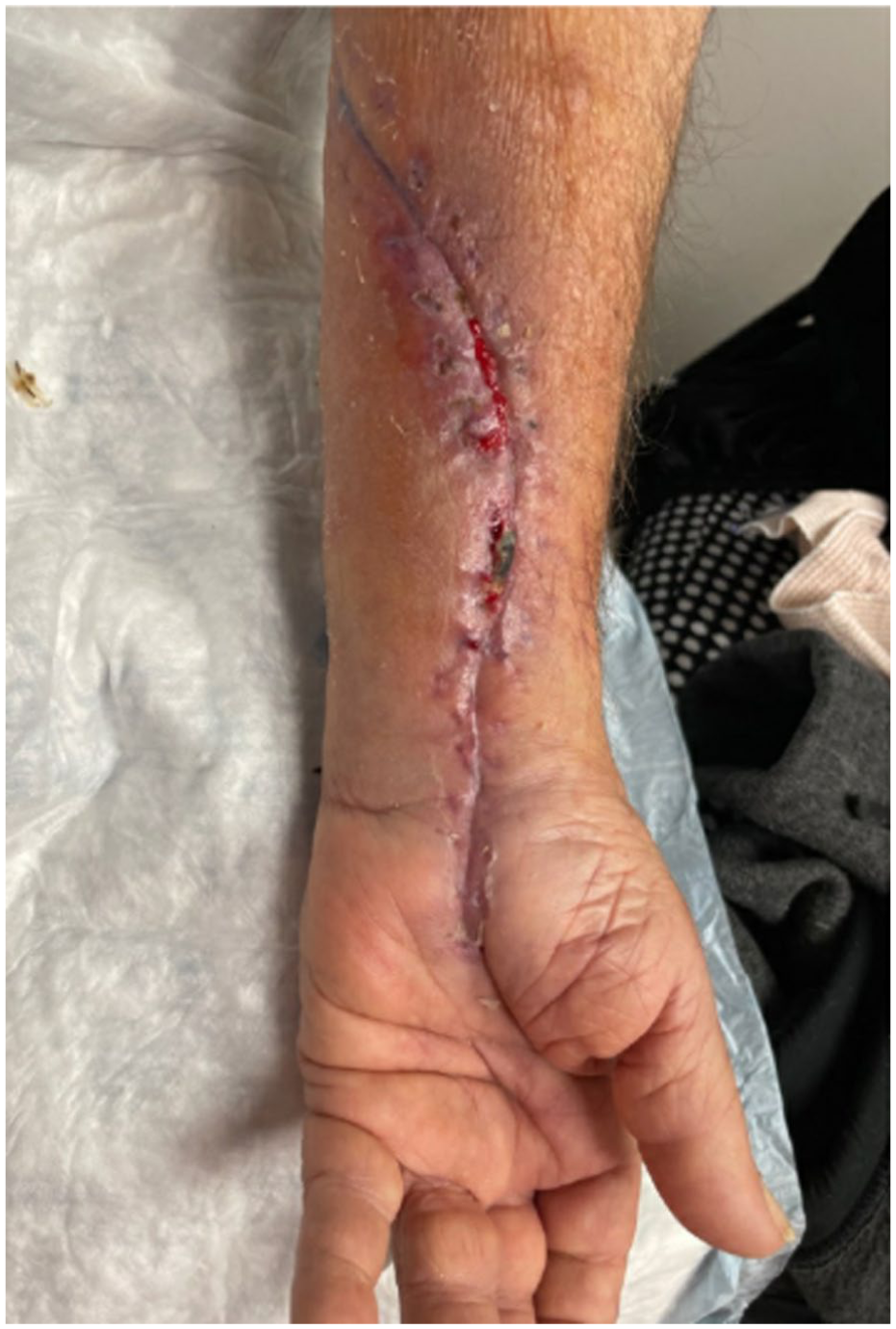
Image of left forearm and hand post-fasciotomy, with a nearly healed incision.
Discussion
Access to the radial artery is also often performed when continuous cardiac monitoring is necessary for patients who are critically ill or undergoing a surgical procedure. Studies have demonstrated that the risk of procedural complications for radial line placement is low (0.03%) compared with the femoral (0.08%) and brachial (0.12%) approaches. 1 Radial artery access for sheath placement during cardiac or vascular procedures is also safe and is associated with a lower risk of bleeding, reduced rest time post-procedure, reduced costs, decreased incidence of hematoma, and lower rates of morbidity and mortality compared with femoral access. 3 However, despite the safety of radial line placement and radial access for cardiovascular procedures, adverse outcomes may still occur. These include infection, nerve damage, radial artery occlusion, allergic reactions, vessel spasm, hematoma, pseudoaneurysm, arterial perforation, arteriovenous fistula, and compartment syndrome. 4
The most common complication of radial arterial line placement is occlusion of the radial artery, which has been reported in 1.5% to 35% (mean 19.7%) of patients. 5 Recanalization is common and patients are typically asymptomatic due to collateral circulation through the palmar arch. As such, determination of palmar arch patency, either through physical exam or vascular evaluation, can help mitigate the risk of ischemic complications from radial artery occlusion due to line placement. Pseudoaneurysm is rare, occurring in 0.09% of patients. 5 Factors associated with radial artery thrombosis after line placement include small radial artery diameters, use of larger catheter sizes, low cardiac output, multiple punctures, and prolonged duration of cannulation, while use of aspirin and heparin may be protective.1,5 Factors that predispose the development of a pseudoaneurysm after radial arterial line placement is not well studied, likely due to the rarity of this complication. Proposed risk factors include multiple attempts at cannulation, inadequate compression for hemostasis, large-sized cannula, and use of anticoagulation or antiplatelet therapy. 6 The patient discussed in this case did have some of these risk factors for pseudoaneurysm development—including multiple attempts at cannulation and therapeutic anticoagulation. However, the size of the access needles used, 20 and 22 gauges, is not unusually large. The adequacy of the compression between each attempt at line placement was not documented or confirmed with imaging. Compartment syndrome is quite rare and occurs in less than 0.05% of cases after radial artery access for cardiovascular procedures. 4 It is not well described as a complication after radial arterial line placement. Compartment syndrome following radial artery access may be secondary to perforation, unsuccessful compression or hemostasis at the puncture site, or excess anticoagulation or glycoprotein IIb/IIIa inhibitor administration. 4
The radial artery typically resides deep to the brachioradialis and flexor carpi radialis muscles on the proximal aspect of the volar forearm. At the level of the distal forearm, both of these muscles thin into tendons and the radial artery becomes more superficial. For this reason, radial artery access is typically performed at the distal aspect of the forearm, where the radial artery can be safely and easily punctured (Figure 9).
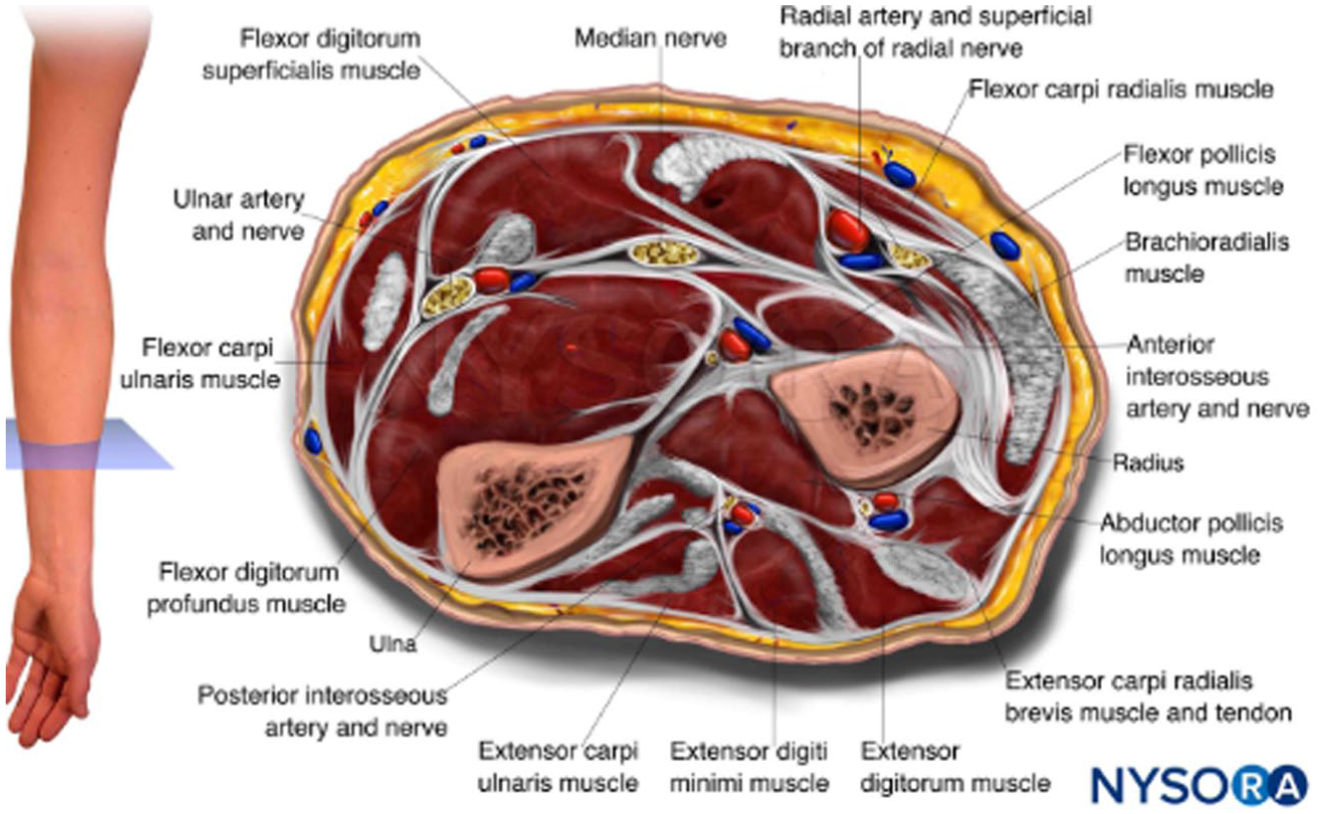
Cross-section depiction of anatomy in the forearm. 7
Compartment syndrome occurs when the pressure within a muscle compartment exceeds the tissue’s capacity to maintain normal perfusion. This is largely due to the limited compliance of the surrounding fascia. Fascia is a soft tissue structure that encases muscle to form compartments. The rigidity and strength of fascia allow it to provide structural integrity to each muscle compartment. However, when inflammation or swelling occurs within a compartment, often secondary to bleeding or edema, the rigid fascia cannot expand to accommodate the increased volume of its contents. 8
There are 2 forms of compartment syndrome: acute and chronic. Acute compartment syndrome develops rapidly, often following traumatic events such as motor vehicle accidents, fractures, surgical complications, or crush injuries. 8 It is a medical emergency that requires a fasciotomy to relieve pressure, as the swelling within the compartment can compress capillaries, nerves, and muscles—impairing blood flow and potentially leading to irreversible nerve and muscle damage. In contrast, chronic compartment syndrome develops gradually, typically as a result of repetitive physical activity or exercise. It is often seen in athletes and presents with exertional pain that subsides with rest. 9
Compartment syndrome is estimated to affect 10 out of every 100 000 individuals in the United States each year. 8 Compartment syndrome classically presents with pain out of proportion with examination, as well as symptoms of nerve injury—paresthesias and paralysis—and/or distal ischemia such as pallor and pulselessness. On examination, patients often have firm compartments with tenderness to palpation as well as significant pain with passive stretching. 10 Although less common than lower extremity compartment syndrome, compartment syndrome of the forearm can lead to significant morbidity due to the important role the hand plays in day-to-day function. If not treated in time, forearm compartment syndrome can ultimately lead to loss of digit perfusion, resulting in the need for digit amputation and loss of hand motor and sensory function. Acute sensory changes develop after 30 minutes of ischemia, while acute irreversible muscle necrosis occurs in 3 to 8 hours, and acute irreversible nerve damage occurs within 12 to 24 hours. 10
In this case, the time course of the patient’s compartment syndrome is not clear. While there was initial concern for compartment syndrome, his improving edema, resolving digit numbness, and soft compartments on serial exams were reassuring, albeit potentially falsely so. Once home, the patient noted persistent pain, but providers were not made aware of this until 9 days post-procedure, so the time course for his worsening swelling and neurological compromise is not well established. It is possible that his compartment syndrome developed acutely after his initial procedure and persisted over the course of the 9 days. However, given his reassuring exam during his initial hospitalization, it was thought that late bleeding from his arterial injury led to the gradual development of a large intramuscular hematoma that then resulted in compartment syndrome.
Forearm compartment syndrome is typically treated with fasciotomy, often with concurrent carpal tunnel release. Forearm fasciotomy seeks to release the 3 forearm compartments and alleviate compression of their associated nerves. This is performed via a volar “lazy S” incision to release the volar compartment, decompressing the median, ulnar, and anterior interosseous nerves, and the mobile wad, followed by a dorsal curvilinear incision to release the anterior compartment, decompressing the radial and posterior interosseous nerves.
Carpal tunnel release is a surgery performed to mitigate median nerve compression at the wrist. The median nerve and tendons are responsible for flexion of the digits and pass through the carpal tunnel at the wrist level. This tunnel is small in caliber, so even minimal swelling can compress the nerve. 11 Carpal tunnel release is typically performed via a volar incision overlying the wrist to divide the transverse carpal ligament. Successful release of the carpal tunnel is confirmed by visual and tactile confirmation of no persistent bands or areas of compression of the median nerve.
Finally, arterial repair is determined by the extent of the injury and can range from primary closure to patch repair and interposition graft placement. In this case, after the fasciotomy, the left radial artery arteriotomy was identified and was able to be repaired primarily. In patients where there is no associated compartment syndrome, radial artery pseudoaneurysms can potentially be repaired via minimally invasive means, such as thrombin injection or endovascular embolization.
In this case, duplex ultrasound was critical in identifying the radial artery injury and prompting referral for admission and surgical intervention. A low index of suspicion for possible arterial injury after radial artery cannulation and early utilization of vascular ultrasound, possibly when the patient was initially noted to have edema, pain, and sensory changes, might have allowed for earlier diagnosis and prevented permanent neurological compromise from compartment syndrome.
Conclusion
Forearm compartment syndrome resulting from radial artery injury is a rare but potentially limb-threatening complication following radial artery access. If not promptly recognized and treated, it can cause permanent nerve damage as well as necrosis of the muscle and soft tissues. This case study highlights the crucial role of duplex ultrasound in aiding in the diagnosis of arterial injury and compartment syndrome and underscores the importance of timely reporting, which enabled immediate surgical intervention.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
