Abstract
Introduction:
Duplex ultrasound (DUS) is commonly used to diagnose and grade arteriovenous fistula (AVF) stenosis. Duplex ultrasound may also be used to examine stenosis type based on measurement of intimal-medial thickness (IMT). There are, as yet, no criteria in use to define the different stenosis types based on measurement of percentage IMT (%IMT). In this study, we have examined stenosis morphology using ultrasound. We have examined consecutive DUS examinations to assess the incidence and degree of IMT and correlated %IMT with volume flow as an indicator of AVF dysfunction.
Methods:
Duplex ultrasound was performed as per standard of care. Volume flow was measured within the ipsilateral brachial artery. Measurements of luminal diameter and vessel wall diameter were used to calculate %IMT.
Results:
The study group included 156 consecutive DUS examinations demonstrating a greater than 50% AVF stenosis. Mean %IMT was 45%. There were 17 (11%) lesions where there was no measurable IMT; the remainder had measurable IMT. On receiver-operating characteristic (ROC) analysis, a %IMT of 54% predicted low flow with 62.5% sensitivity and 62.7% specificity.
Conclusion:
Intimal-medial thickness has a moderate predictive value in predicting volume flow. Based on our results, there are 4 lesion types: (1) lesions with no measurable intimal hyperplasia, (2) a predominantly intimal hyperplastic lesion, (3) those with nonpredominant intimal hyperplasia, and (4) those with a valve type stenosis. A value based on correlation with volume flow could be considered based on our results at 54%IMT or based on morphology at 45%IMT to distinguish between predominant and nonpredominant intimal hyperplasia groups. Including measurements of %IMT in clinical trials could further our knowledge of how lesions with no measurable IMT and those with different degrees of intimal hyperplasia respond to different percutaneous interventions. This could potentially improve outcomes for patients and target more costly treatment options where there is most benefit.
Keywords
Introduction
The native autologous arteriovenous fistula (AVF) is a widely used and reliable method of vascular access in dialysis patients with end-stage kidney disease (ESKD). When compared with central venous catheters, they have consistently been shown to result in lower infection rates, hospitalizations, and costs.1-3 They are also preferred by patients due to the absence of external components that can affect their activities of daily living. 4
However, AVFs are not without complication, and vascular access dysfunction is one of the leading causes of morbidity and mortality among ESKD patients.5,6 Common problems include failure to mature, stenosis formation, and thrombosis. Autologous arteriovenous fistula stenosis is the most common cause of dialysis access dysfunction and can be diagnosed using noninvasive imaging techniques, such as duplex ultrasound (DUS). This allows timely interventions to be planned and performed. Duplex ultrasound is a cost-effective and noninvasive method of imaging AVFs for the investigation of failure to mature and dialysis fistula dysfunction. Ultrasound assessment allows for diagnosis and assessment of the severity of the stenosis and provides a reproducible and objective measure of blood flow. 7 Studies have shown volume flow in the brachial artery to be the most reliable ultrasound parameter indicative of fistula function.8-10
The range of volume flow values in clinically functioning AVFs is broad, and 500 to 1500 ml/min has been regarded as acceptable.11,12 Most sources agree that rates below 500 ml/min are associated with increased risk of future thrombosis.8,9 It should be noted that even amongst well-functioning AVFs, there are a significant number of asymptomatic stenoses. For example, in a study of 139 clinically functional AVFs with normal volume flow, 64% had stenoses. 11 This indicates a large subclinical component and suggests that the relationship between stenosis, volume flow, and fistula function is complex and requires further investigation.
Characterization of AVF stenosis type in terms of stenosis morphology using ultrasound is an emerging area. Yamamoto et al 13 described 3 types of venous stenoses using ultrasound: (1) a neointimal hyperplasia type, (2) a vascular constriction type, and (3) a combination/mixed type. That study also revealed that different lesion types responded differently to endovascular intervention. The neointimal hyperplasia type had lower secondary patency rates following bare metal stent placement. These differences suggest that further analysis of stenosis morphology is needed as it may influence treatment selection in the future.
In this study, we have examined stenosis morphology using ultrasound to better understand the different types of AVF stenosis and their possible definition and incidence. We have examined consecutive DUS examinations to assess the incidence and degree of intimal-medial thickness (IMT) and calculated percentage IMT (%IMT), which is the percentage diameter reduction secondary to intimal hyperplasia. The %IMT has been correlated with volume flow as an indicator of AVF dysfunction. We have also attempted to establish a cut-off value for %IMT that predicts low volume flow in AVF access.
Methods
Ultrasound examinations were performed as per standard of care. It is standard practice at our institution to measure IMT for all AVF access stenoses. Patients were referred for investigation of dialysis access dysfunction or follow-up after percutaneous transluminal angioplasty (PTA) procedures. Patients were scanned in a sitting position in a vascular ultrasound room using a GE (General Electric Healthcare, Little Chalfont, Buckinghamshire, UK) Logic E9 and a 15 MHz probe. All images were recorded on the radiology picture archiving and communication system (PACS). Volume flow was measured in the ipsilateral brachial artery using a time-averaged mean calculation of volume flow. The highest value of the volume flow measurements was used.
Near wall and far wall IMT, outer wall to outer wall vessel diameter, and luminal diameter were measured as demonstrated in Figure 1. Intimal-medial thickness was measured in a similar way to measuring IMT in a carotid artery, by measuring the low-level echo-gray band that does not project into the lumen and is part of the vessel wall. The ultrasound examinations were performed by a consultant vascular radiologist and a vascular sonographer. Measurements of luminal diameter and vessel wall diameter were used to calculate the percentage of the lumen occupied by IMT using the formula:
Outer wall to outer wall vessel diameter − Luminal diameter
÷ Outer wall to outer wall vessel diameter × 100 = %IMT
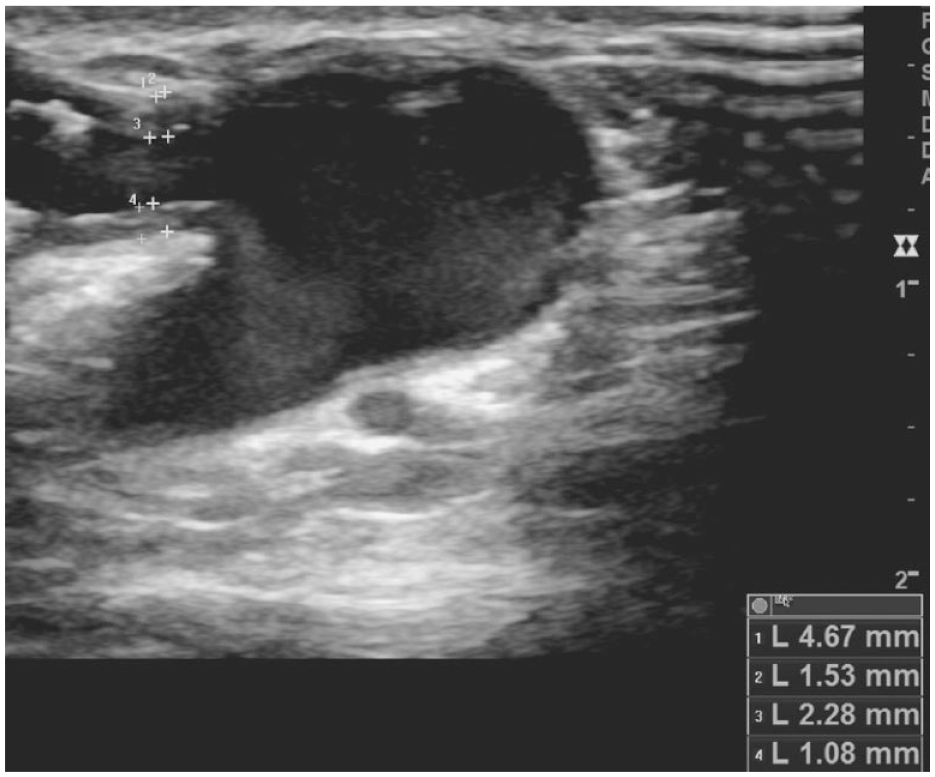
B-mode image of a juxta-anastomotic stenosis in a radiocephalic arteriovenous fistula. Measurements of near and far wall IMT (intimal-medial thickness), outer wall to outer wall vein diameter, and luminal diameter are shown.
Lesions proximal to the cephalic arch were included in the study, because in our experience, IMT is only reliably seen and accurately measured on ultrasound where the vein is suitably superficial. To accurately correlate with volume flow, 118 AVFs with a single stenotic lesion were included in the correlation analysis. Twelve lesions with no detectable IMT were also excluded from the correlation analysis. The lesions within AVFs with multiple stenotic lesions were included in the analysis of stenosis characteristics and %IMT measurements.
The continuous variables were expressed as mean ± SD and categorical variables were expressed as proportion or percentages. Spearman’s rank correlation was used to estimate the correlation between percentage intimal hyperplasia and brachial artery volume flow as the volume flow had a skewed distribution. Receiver-operating characteristic (ROC) analysis was used to identify the %IMT at which the AVF volume flow drops below the threshold value. Analysis was performed using IBM SPSS software version 17.0.
Results
There were 156 consecutive DUS examinations that revealed a significant (greater than 50%) AVF or graft-vein stenosis that were include in the analysis. A significant stenosis of greater than 50% was determined by the operator performing the ultrasound examination. Measurements of luminal diameter and peak systolic velocity comparing the stenosis with an adjacent normal nonaneurysmal vein segment were used to grade the stenosis. The examinations were performed on 92 patients. Patient demographics and results are summarized in Table 1. The majority of patients were white (70%). There were slightly more male patients at 60% when compared with female patients, and approximately half the patients (51%) had diabetes.
Demographics and Results Summary for 92 Patients With 156 Stenotic Lesions.

Note. AVF = arteriovenous fistula; IMT = intimal-medial thickness; %IMT = percentage intimal-medial thickness.
Of the 156 DUS examinations, 81 (52%) were proximal (upper arm) AVFs and 75 (48%) were distal (forearm) AVFs. Of the stenoses identified, 67 (43%) were at a juxta-anastomotic location, 79 (51%) had stenosis in the outflow vein, and 10 (6%) had a stenosis at the graft-vein anastomosis involving an interposition graft. Of the total number of lesions, 92 (59%) were recurrent lesions that had undergone a previous PTA procedure, and 64 (41%) were de novo lesions. There were 17 (11%) lesions where there was no measurable IMT, 3 of which were a valve-type stenosis. Of the de novo lesions, 10 (16%) had no measurable IMT, 2 of which were valve-type stenoses. Of the recurrent lesions post-PTA 7 (8%) had no measurable IMT, one of which was a valve-type stenosis.
The mean %IMT was 45% with a standard deviation of 20%, and the range of %IMT was 0% to 83%. Figure 2A demonstrates the frequency distribution of %IMT. Median volume flow measured in the ipsilateral brachial artery was 799 ml/min. Figure 2B demonstrates the ROC curve analysis that was used to determine the cut-off for %IMT that has the highest sensitivity and specificity to predict a drop in brachial artery volume flow below a threshold of 500 ml/min in distal AVF and 600 ml/min in proximal AVF. There was a significant but moderate inverse correlation between percentage intimal thickening and volume flow (P = .0003, Rho = −0.28). On ROC analysis, a %IMT of 54% predicted low flow (for both proximal and distal AVFs) with 62.5% sensitivity and 62.7% specificity. Intimal-medial thickness has a moderate predictive value in predicting volume flow.
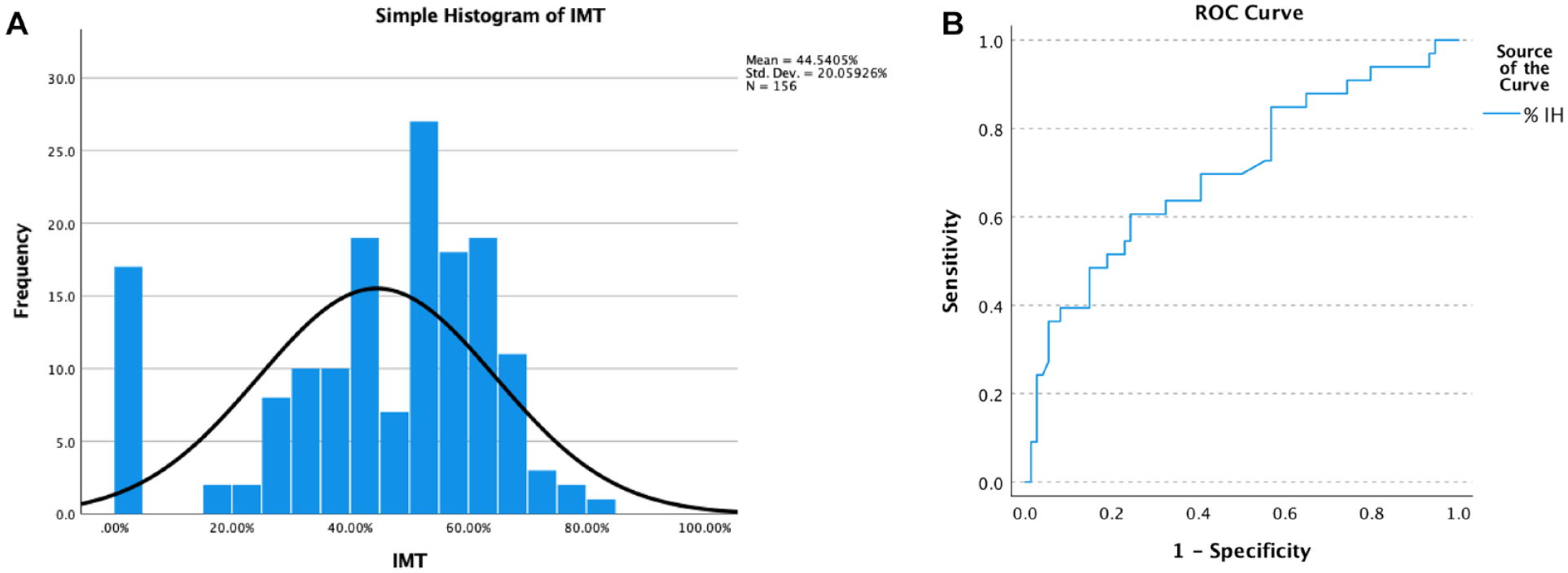
(A) Frequency distribution of percentage IMT (intimal-medial thickness). The mean was 45% with a standard deviation of 20%. (B) ROC (receiver-operating characteristic) curve analysis.
Discussion
We have measured IMT in AVF stenoses in a similar way to measuring IMT in carotid arteries. Measuring IMT in carotid arteries has been validated using histology correlation and may be used to predict cardiovascular risk.14,15 It has previously been demonstrated in a small series looking at AVF stenotic lesions that IMT appears to correspond to intimal hyperplasia on histology. 16 It can therefore be assumed that we are measuring the depth of intimal hyperplasia when measuring the equivalent of IMT in dialysis access stenosis. This is the low-level echo-gray band that does not project into the lumen and is part of the vessel wall, in contrast to thrombus that projects into the vessel lumen and has a different morphological appearance on B-mode ultrasound.
The majority of the lesions had a degree of measurable IMT, but in 10% of lesions, there was no measurable IMT. These lesions appear to be a distinct type where there is luminal narrowing but no intimal hyperplasia. Interestingly, excluding one valve-type stenosis, 6 lesions demonstrated no measurable IMT post-PTA. During PTA, there is vessel wall trauma resulting in endothelial and smooth muscle cell injury that results in migration of smooth muscle cells and myofibroblasts to the intima where they proliferate resulting in neointimal hyperplasia. It is therefore surprising to see recurrent lesions post-PTA with no intimal hyperplasia. These lesions may be those where there has been elastic recoil post-PTA or they are predominantly fibrotic lesions.
The limitations of this study include that there is a degree of intraobserver variability in measurements made with ultrasound. The majority of the studies were performed by the consultant radiologist but should be reproducible with similar accuracy in suitably trained hands. Measurements of IMT were taken only at the site of a stenosis, and the majority of stenoses have a uniform degree of intimal thickening along their length. Measuring IMT where there is a tortuous segment of nonstenotic vein or aneurysmal dilation is more challenging and less likely to be reproducible.
Different types of stenotic AVF lesions based on ultrasound appearances have been described. In 2012, Yamamoto et al 13 described 3 different types of AVF access stenosis at the arteriovenous anastomosis of arteriovenous grafts based on ultrasound appearances—a neointimal proliferation type, a vascular constriction type, and a mixed type. This group demonstrated higher primary patency rates at 6, 12, 18, and 24 months where bare metal stent placement was used to treat the vascular constriction type of stenosis (100%, 92.3%, 84.6%, and 75%, respectively) compared with bare metal stent placement used to treat the neointimal proliferation type (66.7%, 33.3%, 33.3%, and 25.0%) or the mixed type (90.5%, 52.4%, 38.1%, and 27.2%). 13 In 2017, Suemitsu et al 17 described 3 types of stenosis based on ultrasound appearances—an intimal hyperplasia type, a shrinking type, and a valve type. They reported that a shrinking lumen morphology had a negative impact on primary patency at 6 months following percutaneous angioplasty (HR = 2.05, 95% CI = 1.25-3.36, P = .005) and a venous valve-type stenosis had a positive impact on primary patency (HR = 0.19, 95% CI = 0.04-0.79, P = .023). 17 These studies appear to show that different stenosis types may respond differently to different endovascular interventions.
Yamamoto et al 13 calculated a vascular constriction rate and intimal proliferation rate based on measurements of normal adjacent vein diameter, outer wall to outer wall vein diameter, and inner luminal diameter. They used the mean values as a cut-off for the different stenosis types and described the stenosis type based on % intimal proliferation and % shrinkage. Suemitsu et al 17 measured outer diameter and inner luminal diameter and calculated a percentage of intimal hyperplasia. An intimal hyperplasic lesion was described when % intimal hyperplasia was greater than 50% and a shrinking type when less than 50%. 17 However, neither of these studies was designed to examine the incidence and degree of IMT in a dialysis population being investigated with DUS primarily for dialysis access dysfunction, and it was those patients who were referred for angioplasty who were included in the analysis. In this study, we have correlated IMT with volume flow and demonstrated that 54%IMT predicted low flow with a moderate predictive value. Mean %IMT was 45% with a standard deviation of 20%. The correlation with reduction of volume flow was moderate as there are factors other than IMT that will influence volume flow, including the total vessel diameter and cardiac output.
Stenosis characterization may be performed using various methods. Calculation of %IMT is reproducible and appears to correspond to intimal hyperplasia on histology. 16 Based on our results, a similar method for characterization of lesion type could be used in that described by Suemitsu et al, 17 with the exception of distinguishing those lesions where there is no measurable IMT. These include lesions with no measurable intimal hyperplasia (non-IH lesions), where the stenosis is caused primarily by intimal hyperplasia (predominant IH lesions), and those where the stenosis is not primarily caused by intimal hyperplasia (nonpredominant IH lesions). This terminology reflects the variation in intimal hyperplasia and the distribution across the lesions from 0% to 83%. A value based on correlation with volume flow could be considered based on our results at 54% or based on morphology at 45% to distinguish between predominant IH and nonpredominant IH groups. Different lesion types from our study group based on B-mode ultrasound appearances are illustrated in Figure 3. The formula we have used may be added as a calculation on an ultrasound machine. Figure 4 shows a juxta-anastomotic stenosis, and %IMT has been automatically calculated based on measurements of the outer diameter and inner diameter of the stenosis. This measurement could form part of the routine assessment of AVF stenosis on DUS. It is now routine practice at our institution to measure IMT for all AVF access stenoses.
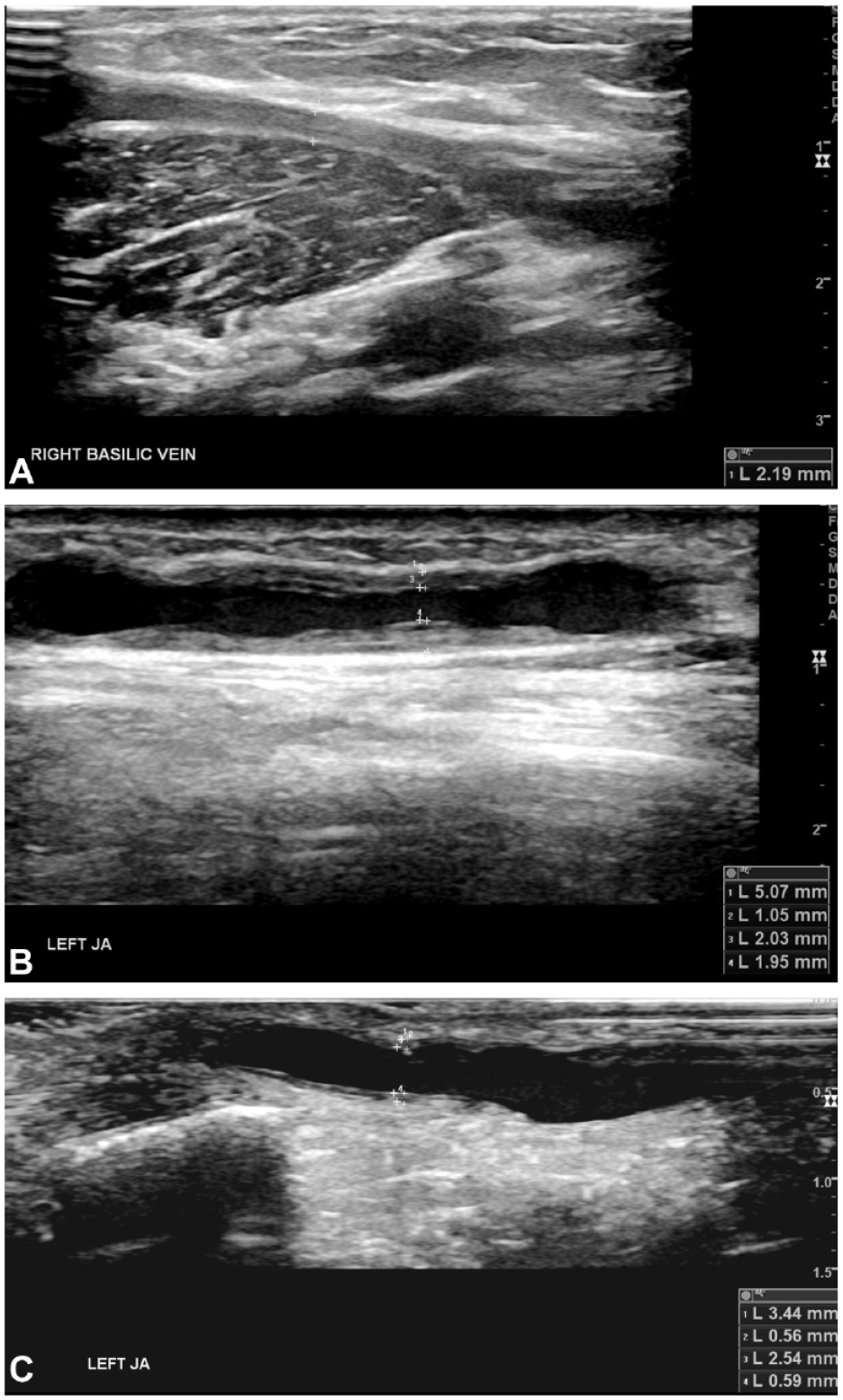
(A) B-mode image of a basilic vein stenosis where there is no measurable IMT (intimal-medial thickness), consistent with a non-intimal hyperplasia lesion (non-IH lesion). (B) B-mode image of a juxta-anastomotic stenosis where there is intimal hyperplasia and percentage IMT is 60%, consistent with a stenosis caused primarily by intimal hyperplasia (predominant IH lesion). (C) B-mode image demonstrating a juxta-anastomotic stenosis with percentage IMT of 26%, consistent with a lesion where the stenosis is not primarily caused by intimal hyperplasia (non-predominant IH lesion).
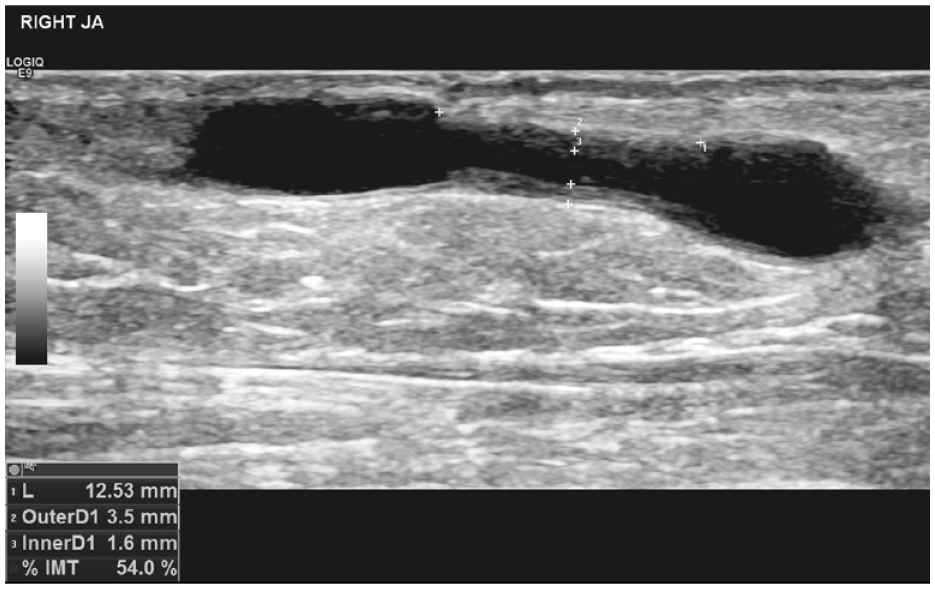
B-mode image demonstrating a juxta-anastomotic stenosis. Percentage IMT (%IMT) has been calculated automatically based on measurements taken of vein diameter and luminal diameter. Stenosis length has also been measured.
Arteriovenous fistula stenosis post-PTA procedures occur with patients often requiring multiple PTA procedures to maintain functional patency. In the past, endovascular treatment options in AVF access were more limited, but now with emerging technologies, there are more treatment options available. These include drug-coated balloons (DCBs) that are angioplasty balloons coated with an antiproliferative agent, usually paclitaxel, and an excipient that aids drug transfer from the balloon into the endothelium and vessel wall. Drug-coated balloons vary in cost between countries, regions, institutions, and manufacturer, but are more costly than standard angioplasty balloons. In the United Kingdom, DCB cost may be between 4 and 8 times the cost of a standard high-pressure balloon. Large multicenter prospective randomized controlled trials comparing DCB angioplasty with plain balloon angioplasty (PBA) in the treatment of AVF access stenosis have been published. The Lutonix AV randomized trial of paclitaxel-coated balloons in arteriovenous fistula stenosis 24-month results demonstrated that fewer interventions were needed to maintain target lesion primary patency in the DCB group compared with PBA at 9 months, where improvement in target lesion primary patency was statistically significantly higher. 18 There were variable differences in patency rates between the groups at other time points within the study.
The IN.PACT AV Access clinical study comparing DCB angioplasty with PBA demonstrated that at 6 months, target lesion primary patency was significantly higher in the DCB group at 82.2% compared with 59.5% in the standard PBA group. 19 The Lutonix AV and IN.PACT AV Access study results appear to demonstrate that the use of DCBs confers a benefit to the patient of less frequent interventions. Other trials have demonstrated no benefit of DCBs over PBA. 20 There are many factors that can lead to mixed results when comparing trial data. These include trial design, patient population, and lesion location. Lesion morphology and type appear also to be a potentially significant variable. No study as yet has examined %IMT and outcome after the use of DCBs, which based on their mechanism of action may be most effective in the treatment of stenosis with significant intimal hyperplasia.
Conclusion
The %IMT has a moderate predictive value in predicting volume flow. There are different appearances on ultrasound to AVF stenoses that can be characterized into 4 distinct types—those with no intimal hyperplasia, those with predominantly intimal hyperplasia, those with nonpredominant intimal hyperplasia, and valve-type stenosis. Based on our analysis, those with nonpredominant intimal hyperplasia can be defined as less than 45%IMT based on the mean value for IMT, or 54% based on correlation with volume flow. Neointimal hyperplasia was not seen on ultrasound in a small percentage of recurrent lesions after angioplasty. Further research with larger numbers based on the methodology in this study could help further characterize lesions and grade %IMT. This would be required to establish that the results we have demonstrated are generalizable.
Two small studies have demonstrated that lesion type may influence outcome after percutaneous interventions. Including ultrasound measurements of intimal hyperplasia and %IMT in clinical trial protocols will further our knowledge of how lesions with no measurable IMT and those with different degrees of intimal hyperplasia respond to different percutaneous interventions. This could potentially improve outcomes for patients and target more costly treatment options where there is most benefit.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
