Abstract
A wide spectrum of drugs can sometimes give rise to numerous adverse orofacial manifestations, particularly dry mouth, taste disturbances, oral mucosal ulceration, and/or gingival swelling. There are few relevant randomized double-blind controlled studies in this field, and therefore this paper reviews the data from case reports, small series, and non-peer-reviewed reports of adverse drug reactions affecting the orofacial region (available from a MEDLINE search to April, 2003). The more common and significant adverse orofacial consequences of drug therapy are discussed.
Introduction
A wide spectrum of drugs can sometimes give rise to numerous adverse oral manifestations, particularly dry mouth, taste disturbances, oral mucosal ulceration, and/or swelling. A review of the 200 most frequently prescribed drugs (in the USA in 1992) showed the most frequent oral adverse drug reactions (ADRs) to be dry mouth (80.5%), dysgeusia (47.5%), and stomatitis (33.9%) (Smith and Burtner, 1994). However, few randomized double-blind controlled studies have been conducted, and therefore most of the evidence available at the present time is of low level, originating only from case reports, small series, or non-peer-reviewed reports of adverse drug reactions.
This paper reviews the literature based on a MEDLINE search to April, 2003, and highlights the more common and significant adverse oral consequences of drug therapy. When relevant, it provides sources of other suitable reports: It does not include details on the oral side-effects of foodstuffs, tobacco, alcohol, or recreational drugs, or the adverse effects of drugs upon the dentition.
Drug-related Disorders of the Salivary Glands
(1) Drug-related xerostomia
Dry mouth has a variety of possible causes (Scully, 2003) (Table 1). Common habits such as tobacco smoking, alcohol use (including in mouthwashes), and the consumption of beverages containing caffeine (coffee, some soft drinks) can cause some oral dryness. Drugs are the most common cause of reduced salivation (Table 1, Fig. 1) (Scully, 2003). The drugs most commonly implicated include: alpha receptor antagonists for treatment of urinary retention; amphetamines; anticholinergics; antidepressants (serotonin agonists, or noradrenaline and/or serotonin re-uptake blockers); antihistamines; antihypertensive agents; antimigraine agents; antipsychotics such as phenothiazines; appetite suppressants; atropinics; benzodiazepines, hypnotics, opioids, and drugs of abuse; bronchodilators; cytokines; cytotoxics; decongestants and ‘cold cures’; diuretics; histamine H2 antagonists and proton pump inhibitors; muscarinic receptor antagonists for the treatment of overactive bladder; opiates; protease inhibitors; radioiodine; retinoids; and skeletal muscle relaxants (Scully, 2003).
There appear to be multiple mechanisms whereby drugs can produce xerostomia (Madinier et al., 1997). Although a wide range of drugs can give rise to oral dryness (Di Giovanni, 1990; Sreebny and Schwartz, 1997; Wynn and Meiller, 2001; Scully, 2003), the most dramatic effect is with radio-iodine (Mandel and Mandel, 2003).
Dry mouth is a common complaint in patients treated for hypertensive, psychiatric, or urinary problems (Streckfus, 1995) and in the elderly (Vissink et al., 1992; Loesche et al., 1995), mainly as a consequence of the large number of drugs used (Fox, 1998; Närhi et al., 1999) and polypharmacy (Loesche et al., 1995; Nederfors, 1996; Fox, 1998). For example, 63% of hospitalized patients and 57% of outpatients complained of dry mouth, and in all patients, the use of psychiatric drugs was the main cause (Pajukoski et al., 2001).
Age and medication seem to play a more important role in individuals with objective evidence of hyposalivation, while female gender and psychological factors are important in individuals with subjective oral dryness (Bergdahl and Bergdahl, 2000).
Hypnotics are also commonly used by the elderly, and most users report ADRs, mainly dry mouth (30%) (Wishart et al., 1981; Busto et al., 2001). In the elderly using non-prescription products—most frequently, dimenhydrinate (21%), acetaminophen (paracetamol) (19%), diphenhydramine (15%), alcohol (13%), and herbal products (11%)—mild ADRs were reported by 75%, the most common complaint being dry mouth (Sproule et al., 1999).
In a large survey of 3311 evaluatable questionnaires, 21.3% of men and 27.3% of women reported dry mouth, with women statistically reporting higher prevalence of dry mouth than men (Nederfors, 1996). Dry mouth was significantly age-related, and there was a strong co-morbidity between reported prevalence of dry mouth and ongoing pharmacotherapy. Unstimulated salivary flow rates were lower among older persons who were female or taking antidepressants, and higher among smokers or people who were taking hypolipidemic drugs (Thomson et al., 2000). It is clear that medication is a better predictor of risk status for dry mouth than either age or gender (Field et al., 2001).
Even in elderly patients with advanced cancer, dry mouth was the fourth most common symptom (78% of patients), but the usual cause was drug treatment, and there was an association with the number of drugs prescribed (Davies et al., 2001). That is not to say that disease can always be excluded as a cause of dry mouth; for example, saliva secretion is more affected by xerogenic drugs and autonomic nervous dysfunction in patients with non-insulin-dependent diabetes than in non-diabetic controls (Meurman et al., 1998).
Furthermore, the cause for which the drug is being taken may also be important. For example, patients with anxiety or depressive conditions may complain of dry mouth even in the absence of drug therapy or evidence of reduced salivary flow. It is thus important to recognize that some patients complaining of a drug-related dry mouth have no evidence of a reduced salivary flow or a salivary disorder, and there may then be a psychogenic reason for the complaint.
Several different mechanisms account for drug-related dry mouth, but an anticholinergic action underlies many: The M3-muscarinic receptors (M3R) mediate parasympathetic cholinergic neurotransmission to salivary (and lacrimal) glands, but other receptors may also be involved (Kawaguchi and Yamagishi, 1995).
(a) Antidepressants
Early antidepressant medications such as tricyclic antidepressants (TCAs) unfortunately also blocked histaminic, cholinergic, and alpha1-adrenergic receptor sites, causing ADRs such as dry mouth. Men and women may differ in their pharmacokinetic responses to TCAs, in several autonomic indices, and in various adrenergic receptor-mediated responses. Emerging evidence also suggests that women, relative to men, may have a lower rate of brain serotonin synthesis and a greater sensitivity to the depressant effects of tryptophan depletion (Pomara et al., 2001). Finally, the ultrarapid metabolizer phenotype of the enzyme cytochrome P4502D6 (Laine et al., 2001) may be a cause of non-responsiveness to antidepressant drug therapy, and the subsequent prescribing of high doses of antidepressants to such patients leads to an increased risk for ADRs; however, normalization of the metabolic status of ultrarapid metabolizers by the inhibition of cytochrome activity, such as with paroxetine, could offer a solution (Laine et al., 2001).
Newer antidepressants are essentially serotonin (5-hydroxytryptamine: 5HT) agonists, or block re-uptake of noradrenaline (norepinephrine) and/or serotonin. Members of the newer generation of antidepressants—including the selective serotonin re-uptake inhibitors (SSRIs) and multiple-receptor antidepressants (such as venlafaxine, mirtazapine, bupropion, trazodone, and nefazodone)—target one or more specific brain receptor sites without, in most cases, activating unwanted sites such as histamine and acetylcholine (Feighner, 1999; Feighner and Overo, 1999). The SSRI produce no significant changes in salivation (Hunter and Wilson, 1995), but dry mouth may still be seen (e.g., fluoxetine) (Ellingrod and Perry, 1994; Shrivastava et al., 1994; Hunter and Wilson, 1995; Davis and Faulds, 1996; Boyd et al., 1997; Ravindran et al., 1997; Trindade et al., 1998). However, some SSRIs, such as paroxetine, appear to result in less dry mouth than seen with TCAs (Ravindran et al., 1997). Citalopram, the most selective SSRI, is well-tolerated (Feighner and Overo, 1999). Venlafaxine, a mixed-re-uptake inhibitor, may produce dry mouth (Gelenberg et al., 2000). The incidence of ADRs in recipients of venlafaxine XR is similar to that in patients receiving treatment with well-established SSRIs (Wellington and Perry, 2001). Duloxetine hydrochloride, a dual-re-uptake inhibitor of serotonin and norepinephrine, also can produce dry mouth (Detke et al., 2002), as can mianserin, a mainly post-synaptic serotonin 2A agonist (Dolberg et al., 2002). Nefazodone, which has moderate serotonin selective re-uptake blocking properties and direct 5-HT2 antagonism, can produce dry mouth in 19% vs. 13% in placebo (Lader, 1996). Reboxetine, a selective norepinephrine re-uptake inhibitor (selective NRI), causes less dry mouth than TCAs, though it produces dry mouth more frequently than placebo (36% vs. 16%) (Schatzberg, 2000; Versiani et al., 2002). Mirtazapine, a noradrenergic and specific serotonergic antidepressant (NaSSA), can also induce dry mouth (Anttila and Leinonen, 2001).
Bupropion hydrochloride, originally developed as an antidepressant, is a selective re-uptake inhibitor of dopamine and norepinephrine which was found to reduce nicotine withdrawal symptoms and the urge to smoke, but commonly produces dry mouth (Zwar and Richmond, 2002). Dry mouth is positively associated with mean bupropion metabolite concentrations, and is inversely related to patient weight (Johnston et al., 2001).
(b) Antipsychotics
Long-term drug treatment of schizophrenia with conventional phenothiazine antipsychotics such as fluphenazine is commonly associated with dry mouth (Adams and Eisenbruch, 2000).
However, newly developed antipsychotic drugs with more potent and selective antagonistic activity against the dopamine D(2) receptor may not necessarily be associated with a lower incidence of dry mouth. Olanzapine is an atypical antipsychotic which appears to produce dry mouth (Duggan et al., 2000). Significant correlations have been found in in vitro affinities of antipsychotics toward dopamine or other neuronal receptor systems and ADRs: for example, between the K(i) values for dopamine D(1) receptor, alpha (1)-adrenoceptor and histamine H(1) receptor), and the incidence of dry mouth (Sekine et al., 1999). Other atypical antipsychotics—including tiapride (Roger et al., 1998), lithium (Christodoulou et al., 1977; Chacko et al., 1987; Friedlander and Birch, 1990; Tohen et al., 2002), pipamperone dihydrochloride (Potgieter et al., 2002), quetiapine and risperidone (Srisurapanont et al., 2000; Mullen et al., 2001)—may produce dry mouth.
(c) Antihistamines
The therapeutic effects of most of the older antihistamines were associated with sedating effects on the central nervous system (CNS) and antimuscarinic effects including dry mouth (Gwaltney et al., 1996). Non-sedating antihistamines—most of which are histamine H1 receptor antagonists, such as acrivastine, astemizole, cetirizine, ebastine, fexofenadine, loratadine, mizolastine, and terfenadine, however—are not entirely free from ADRs, though there may be less dry mouth (Mattila and Paakkari, 1999).
(d) Muscarinic receptor antagonists used to treat overactive bladder
Overactive bladder (OAB)—a chronic, distressing condition characterized by symptoms of urgency (sudden overwhelming urge to urinate) and frequency (urinating more than eight times daily), with or without urge urinary incontinence (sudden involuntary loss of urine)—is treated with muscarinic receptor antagonists, which can lead to troublesome dry mouth.
Dry mouth is a common side-effect seen with immediate-release oxybutynin (Hay-Smith et al., 2002), but a significantly lower proportion of patients taking controlled-release oxybutynin have moderate to severe dry mouth or any dry mouth compared with those taking immediate-release oxybutynin (Versi et al., 2000). Tolterodine, a competitive muscarinic receptor antagonist, produces dry mouth (Jacquetin and Wyndaele, 2001; Layton et al., 2001), though few patients (< 2%) experience severe dry mouth (Zinner et al., 2002), and there is a 23% lower incidence of dry mouth reported with once-daily extended-release tolterodine capsules than with twice-daily immediate-release tablets (Clemett and Jarvis, 2001). Propiverine hydrochloride, a safe and effective antimuscarinic drug as effective as oxybutynin, has a lower severity and incidence of dry mouth (Noguchi et al., 1998; Madersbacher et al., 1999).
(e) Alpha receptor antagonists used to treat overactive bladder
Tamsulosin, a selective alpha 1A-adrenoreceptor antagonist, produces significantly less dry mouth than terazosin, a less selective alpha antagonist (Lee and Lee, 1997).
(f) Diuretics
Diuretic agents and psychotropics were the most commonly used xerostomic medications in one study of elderly patients, and were almost equally potent in reducing mean salivary flow rate (Persson et al., 1991). Thiazides may cause dry mouth (McCarron, 1984), but there appear to be few reports showing a relationship between diuretic use and dry mouth. Subjectively, xerostomia was experienced 10 times more frequently after ingestion of furosemide than placebo (Atkinson et al., 1989).
(g) Antihypertensives
The ganglion blockers and particularly the beta-blockers (beta-adrenoceptor antagonists) may cause dry mouth (Nederfors, 1996) thought to be associated with activation of CNS and salivary gland alpha 2-adrenergic receptors. Such antihypertensive drugs, or sympatholytics (reserpine, methyldopa, and clonidine), are now little used because of such prominent ADRs as dry mouth.
Newer centrally acting antihypertensives, with selective agonist effects on the imidazoline I1 brainstem-receptors in the rostral ventromedulla (RVLM), appear to modulate sympathetic activity and blood pressure without affecting salivary flow: Moxonidine and rilmenidine are examples of this new class.
Moxonidine can produce dry mouth, more frequently than placebo (Dickstein et al., 2000) but only in a minority (< 10%), and significantly less than with the older antihypertensives (Schachter, 1999). Rilmenidine produces little dry mouth (Reid, 2001).
ACE inhibitors, which block the ACE enzyme in the renin-angiotensin-aldosterone system, produce dry mouth in about 13% of patients (Mangrella et al., 1998).
(h) Appetite suppressants
Several appetite suppressants can cause dry mouth, including sibutramine (Richter, 1999; Bray, 2001), fenfluramine plus phentermine (Weintraub et al., 1992), and the herbal Ma Huang and Kola nut supplement (containing ephedrine alkaloids/caffeine) produce dry mouth (Boozer et al., 2002).
(i) Decongestants and “cold cures”
Decongestants such as pseudoephedrine (Wellington and Jarvis, 2001) and loratadine plus pseudoephedrine sulphate can result in dry mouth (Kaiser et al., 1998).
(j) Bronchodilators
Anti-asthma drugs can be associated with dry mouth (Thomson et al., 2002). The most common reported ADR after use of the bronchodilator tiotropium is dry mouth (Casaburi et al., 2000).
(k) Skeletal muscle relaxants
Tizanidine, an alpha 2-adrenoceptor agonist used for the relief of spasticity, can be associated with significant dry mouth (Taricco et al., 2000).
(l) Antimigraine drugs
Rizatriptan, a serotonin agonist used for the treatment of migraine, can induce dry mouth (Winner et al., 2002).
(m) Opioids, benzodiazepines, hypnotics, and drugs of abuse
Opioids are well-recognized causes of dry mouth (Bruera et al., 1999), and atropine, hyoscine, and atropinics have long been used to reduce secretions pre-operatively. Dry mouth is the only significant side-effect of transdermal scopolamine (hyoscine) (Gordon et al., 2001). Morphine (Bruera et al., 1999; Zacny, 2001), dihydrocodeine (Freye et al., 2001), and Tramadol (Scott and Perry, 2000) can produce dry mouth.
Benzodiazepines (such as diazepam) (Conti and Pinder, 1979; Elie and Lamontagne, 1984) and zopiclone, a cyclopyrrolone chemically unrelated to benzodiazepines (Wadworth and McTavish, 1993), can produce dry mouth.
Cannabis (Consroe et al., 1986; Grotenhermen, 1999) and Ecstasy (‘XTC’ or ‘E’: 3,4 methylenedioxy-methamphetamine; MDMA) (Peroutka et al., 1988; Milosevic et al., 1999) can produce dry mouth.
(n) H2 receptor antagonists and proton-pump inhibitors
H2-receptor antagonists produce dry mouth in 41% of recipients (Teare et al., 1995; Kaviani et al., 2001).
(o) Cytotoxic drugs
Dry mouth can be a consequence of exposure to agents used for chemotherapy of malignant neoplasms, such as retinol (Goodman et al., 1983), or 5-fluorouracil (McCarthy et al., 1998), but this is not invariable (Laine et al., 1992), and there is a paucity of data in this area.
(p) Retinoids
Systemic retinoids such as etretinate (Wishart et al., 1981), 13 cis-retinoic acid (Hennes et al., 1984), and isotretinoin (Oikarinen et al., 1995) can produce dry mouth.
(q) Anti-HIV drugs
Didanosine (Dodd et al., 1992; Valentine et al., 1992) and HIV protease inhibitors (Greenwood et al., 1998) can cause dry mouth.
(r) Cytokines
Alpha interferon therapy for the treatment of chronic hepatitis C infection (Cotler et al., 2000) can cause significant dry mouth. Interleukin-2 (rIL-2), when used, for example, for control of nasopharyngeal carcinoma, can also impair salivation (Chi et al., 2001), and major salivary gland dysfunction has been described in patients with hematological malignancies given interleukin-2-based immunotherapy after autologous stem cell transplantation (Nagler, 1998).
(2) Drug-related salivary gland swelling
Some drugs have been related to salivary gland swelling (Table 2). Painless, usually bilateral, salivary gland enlargement (resembling sialosis) may be an occasional side-effect of phenylbutazone (Cohen and Banks, 1966; Murray-Bruce, 1966; Chimenes, 1971; Herman et al., 1974; Kaufman et al., 1977), oxyphenbutazone (Bosch et al., 1985), or chlorhexidine (Rushton, 1977). The bilateral sialadenitis observed in one adult following naproxen therapy (Knulst et al., 1995) was thought to be allergic in origin, since the affected patient also had a cutaneous rash. A similar mechanism has been proposed to underlie the salivary gland enlargement caused by intravenous radiological contrast media (Gattoni et al., 1991; Ilia et al., 1991). Clozapine, a novel antipsychotic agent, may cause transient salivary gland swelling (Vasile and Steingard, 1995) as well as sialorrhea (see below).
(3) Drug-related salivary gland pain
Antihypertensives, anti-thyroid agents, chlorhexidine, cytotoxics, ganglion-blocking agents, iodides, phenothiazines, and sulphonamides may cause salivary gland pain, as may drugs causing dry mouth (Glass, 1989). Salivary gland pain is rarely associated with guanethidine or guanacline (Parker, 1975) therapy and can be a side-effect of several other drugs (Table 2).
(4) Drug-related hypersalivation
Anticholinesterases are the main cause of hypersalivation (Table 3). Clozapine, an atypical antipsychotic drug claimed to have superior efficacy and to cause fewer motor adverse effects than typical antipsychotics for people with treatment-resistant schizophrenic patients, can cause hypersalivation (Wahlbeck et al., 2000). This may be ameliorated with atropine eye drops (Comley et al., 2000).
(5) Discoloration of saliva
Discoloration of saliva (red or orange saliva) as well as other body fluids may be seen in patients treated with clofazimine, levodopa, rifampicin, and rifabutin therapy (British National Formulary, 2002) (Table 4).
Drug-related Disorders of Taste
Drug-induced taste disorders are reviewed in detail elsewhere (Griffin, 1992; Henkin, 1994) but are reviewed and updated here (Table 5).
Drugs commonly impair taste. Drugs may cause a loss of taste acuity (hypogeusia), distortion of taste (dysgeusia), or loss of taste sense (ageusia), though this is rare (Ackerman and Kasbekar, 1997). Drugs act either by interfering with the chemical composition or flow of saliva, or by affecting taste receptor function or signal transduction (Femiano et al., 2003).
ACE inhibitors, anti-thyroids, beta-lactam antibiotics, biguanides, chlorhexidine, opiates, and protease inhibitors are particularly implicated. Up to 4% of patients treated with ACE inhibitors may have dysgeusia, although this adverse effect is self-limiting and reversible within a few months, even with continued therapy (Henkin, 1994). Newer therapies—such as the anti-HIV protease inhibitors (Scully and Diz, 2001), therapy with tripotassium dicitrato bismuthate chelate, clarithromycin, and lansoprazole therapy for H. pylori infection (Scott, 1998), terbinafine (Stricker et al., 1996), intravenous pentamidine (Glover et al., 1995), and isotretinoin (Halpern et al., 1996)—may cause some degree of loss of taste or altered taste.
There is preliminary evidence that alpha lipoic acid might improve dysgeusia, at least in idiopathic cases (Femiano et al., 2002), but the condition is best improved by reducing use of the offending drug.
Drug-related Mucosal Disorders
(1) Oral ulceration
A wide range of etiologies, from burns to vesiculobullous disorders of various kinds, can result in oral ulceration. Many reports of mouth ulceration following drug use have been from non-specialists, and therefore their description as ‘aphthous’-like or other fairly specific entities can often be questioned, leading to some difficulty in accurately ascribing cause and effect.
(a) Drug-related oral burns
Oral desquamation or ulceration may follow burns from the accidental ingestion of caustics such as lime (Schier et al., 2002), from the local application of aspirin or toothache preparations, potassium tablets, pancreatic supplements, and other agents such as trichloracetic acid or hydrogen peroxide or from the use of various oral health care products (Kuttan et al., 2001). Sodium lauryl sulfate, present in some oral health care products, particularly dentifrices, may cause mucosal irritation or ulceration (Herlofson and Barkvoll, 1994; Scully et al., 1999; Kuttan et al., 2001). Application of cocaine may produce similar lesions (Parry et al., 1996).
(b) Drug-related aphthous-like ulceration
Sodium lauryl sulfate may predispose to ulcers similar to aphthous ulceration (Herlofson and Barkvoll, 1994). There are also case reports of aphthous-like ulceration arising following the use of beta-blockers such as labetalol (Pradalier et al., 1982), alendronate (Gonzalez-Moles and Bagan-Sebastian, 2000), captopril (Seedat, 1979; Nicholls et al., 1981; Corone et al., 1987; Montoner et al., 1990), nicorandil (Boulinguez et al., 1997; Reichart, 1997; Agbo-Godeau et al., 1998; Cribier et al., 1998; Desruelles et al., 1998; Roussel et al., 1998; Marquart-Elbaz et al., 1999; Shotts et al., 1999), some non-steroidal anti-inflammatory drugs (NSAIDs) (Siegel and Balciunas, 1991; Healy and Thornhill, 1995), mycophenolate or sirolimus (van Gelder et al., 2003), protease inhibitors (Scully and Diz, 2001), tacrolimus (Hernandez et al., 2001), and sulfonamides, though the exact pathogenic mechanisms are unclear in all of these.
A case-control study has now confirmed the associations of oral ulceration with NSAIDs and beta-blockers (Boulinguez et al., 2000) (Fig. 2).
(c) Fixed drug eruptions
Fixed drug eruptions (contact stomatitis or stomatitis venenata) comprise repeated ulceration at the same site in response to a particular drug and may be caused by anesthetics, antibiotics, antiseptics, barbiturates, chewing gum, cosmetics, dental materials, dentifrices, mouthwashes, phenacetin, sulphonamides, or tetracyclines. The lesions may be localized to the mouth or can be associated with lesions at other mucocutaneous sites, and manifest as ulceration, bullae, erythematous patches, or superficial erosions. Initially, the lesions are solitary, but with repeated drug exposure, they may become multiple. A wide range of drugs may cause fixed drug eruption, particularly paracetamol, barbiturates, phenacetin, pyrazolone derivatives, sulphonamides, and tetracyclines (Abdollahi and Radfar, 2003), as may agents such as cinnamon (Miller et al., 1992).
(d) Drug-related mucositis
Cytotoxic drugs are very commonly associated with mucositis and ulceration, which arises consistently with many chemotherapy regimens (Table 6), particularly those involving methotrexate, 5-fluorouracil, doxorubicine, melphelan, mercaptopurine, or bleomycin (Femiano et al., 2003). Such reactions can be so severe as to be treatment-limiting on occasion (Bell et al., 2001). Widespread sloughing and ulceration arise within days of commencement of therapy, the associated pain often requiring opioid therapy and/or alteration or cessation of chemotherapy. The ulceration may be a portal of entry for infection and hence a potential cause of septicemia. Drugs such as phenylbutazone that can cause agranulocytosis may also induce oral ulceration (Fig. 3).
Immunosuppressive agents may also cause ulceration. Ulcers in iatrogenically immunocompromised individuals may have a herpesvirus etiology, or occasionally other infective causes (Greenberg et al., 1987; Schubert et al., 1993). Opportunistic infection secondary to cytotoxic chemotherapy may cause oral ulceration. In particular, herpes simplex virus 1, varicella zoster, and cytomegalovirus give rise to oral ulceration, while, less commonly, ulceration may be due to Gram-negative bacterial infections (e.g., pseudomonas, klebsiella, Escherichia coli, enterobacter, or proteus) or to exogenous bacteria such as tuberculosis (Toren et al., 1996), or to fungi such as mucormycosis (Femiano et al., 2003) or even candidosis (see below).
(e) Drug-related neoplasms and potentially malignant lesions
There is an increased prevalence of dysplastic and malignant lip lesions in immunosuppressed renal-transplant recipients (King et al., 1995) and liver transplant recipients (Haagsma et al., 2001). Oral leukoplakia has progressed rapidly to squamous cell carcinoma in some immunosuppressed patients (Hernandez et al., 2003), and oral squamous cell carcinoma has been reported in immunosuppressed patients without any recorded precursor lesion (Varga and Tyldesley, 1991).
Post-transplant lymphoproliferative disease (Raut et al., 2000), non-Hodgkin’s or MALT lymphoma (Hsi et al., 2000), usually manifesting as ulceration of the gingivae, fauces, or palate (Bilinska-Pietraszek et al., 2001; Mandel et al., 2001), or, rarely, Kaposi’s sarcoma (Meyers et al., 1976; Qunibi et al., 1988) may be complications of long-term immunosuppressive therapy, and there have even been reports of the resolution of malignancies where immunosuppression has been reduced (Keogh et al., 2002).
(f) Drug-related pemphigoid-like reactions and other bullous disorders
At least 30 drugs can give rise to conditions resembling bullous or mucous membrane pemphigoid (Vassileva, 1998) (Table 7). These drugs belong to a variety of pharmacological (thiol and non-thiol) and therapeutically targeted groups, including ACE inhibitors, furosemide, NSAIDs, penicillamine, psoralens, sulphonamides, cardioactive agents, and penicillin-related antibiotics. The oral mucosa is frequently affected in drug-induced pemphigoid, particularly penicillamine-induced disease, and can be the only affected mucosal surface, although patients often also have cutaneous lesions (Troy et al., 1981; Shuttleworth et al., 1985; Velthuis et al., 1985; Gall et al., 1986; Rasmussen et al., 1989). Other than the high frequency of oral mucosal lesions, the only other clinically distinguishing features of drug-induced pemphigoid are the younger age of affected patients compared with idiopathic (autoimmune) pemphigoid, and the resolution of disease following withdrawal of the causative agent.
Drug-induced pemphigoid may be due to thiol-induced local epithelial damage, drugs acting as haptens, or drug-induced immunological dysfunction. Affected patients can have circulating antibodies to the same antigens as idiopathic pemphigoid, hence making diagnosis of drug-related disease so reliant upon the recording of an accurate drug history.
Linear IgA disease (LAD) can be drug-induced, and affected patients have IgA antibodies to bullous-pemphigoid-associated antigen 1 (BPAG or BP1) or other antigens (Palmer et al., 2001). LAD is especially commonly induced by vancomycin (Kuechle et al., 1994), but other drugs such as angiotensin-converting enzyme inhibitors may be involved (Femiano et al., 2003).
(g) Drug-related pemphigus
Drug-induced pemphigus is not uncommon (Brenner et al., 1997). Traditionally, drugs that are capable of inducing pemphigus are divided into two main groups according to their chemical structure—drugs containing a sulfhydryl radical (thiol drugs or SH drugs) and non-thiol or other drugs, the latter often sharing an active amide group in their molecules (Wolf and Brenner, 1994).
Pemphigus vulgaris may occasionally be associated with drugs with active thiol groups in the molecule (Scully and Challacombe, 2002). Drugs implicated include penicillamine (Wolf et al., 1991; Laskaris and Satriano, 1993; Shapiro et al., 2000), phenol drugs (Goldberg et al., 1999), rifampicin (Gange et al., 1976), diclofenac (Matz et al., 1997), and, rarely, captopril (Korman et al., 1991), other ACE-inhibitors (Kaplan et al., 1992; Ong et al., 2000), and other drugs (Table 8).
The clinical features of drug-induced pemphigus mimic those of pemphigus vulgaris or foliaceus, and affected individuals can have variable levels of circulating antibodies to epithelial components and to expected antigens (e.g., desmoglein 1 and 3) (Kuechle et al., 1994). Aside from epithelial damage being due to the action of these antibodies, some of the implicated drugs are thiols (Wolf and Ruocco, 1997) that may induce a fall in local levels of plasminogen activator inhibitor, leading to increased plasminogen activation and epithelial damage (Lombardi et al., 1993). Thiols such as penicillamine may also interfere in cell membrane cysteine links, potentially leading to antibody generation and epithelial damage (Wolf et al., 1991).
The role of diet in the etiology of pemphigus is reviewed elsewhere (Brenner et al., 1998; Tur and Brenner, 1998), but garlic in particular may cause occasional cases of pemphigus (Laskaris and Nicolis, 1980; Korman et al., 1991; Ruocco et al., 1996).
(h) Drug-related erythema multiforme
A wide range of drugs—especially barbiturates, cephalosporins, NSAIDs, estrogens, phenothiazines, progestogens, protease inhibitors, sulphonamides, sulphonylurea derivatives, and tetracyclines—may give rise to erythema multiforme (Table 9), and it may be clinically impossible to distinguish drug-induced erythema multiforme from disease due to other causes (Roujeau, 1997; Ayangco and Rogers, 2003). The distinction of severe erythema multiforme from toxic epidermal necrolysis is quite unclear.
Lesions of erythema multiforme typically affect the oral mucosa, the lips, and bulbar conjunctivae. Initial bullae rupture to give rise to hemorrhagic pseudomembrane of the lips and widespread superficial oral ulceration. Other mucocutaneous surfaces less commonly affected include the nasopharyngeal, respiratory, and genital mucosae.
(i) Drug-related toxic epidermal necrolysis
Toxic epidermal necrolysis (TEN; Lyell syndrome) is clinically characterized by extensive mucocutaneous epidermolysis preceded by a macular or maculopapular exanthema and enanthema (Lyell, 1979; Rasmussen et al., 1989). Intra-orally, there is widespread painful blistering and ulceration of all mucosal surfaces. Toxic epidermolysis may be associated with antimicrobials (sulphonamides, thiacetazone), analgesics (phenazones). anti-epileptics, allopurinol, chlormezanone, rifampicin, fluconazole, and vancomycin (Ayangco and Rogers, 2003).
(j) Drug-related lupus-like disorders
Systemic lupus erythematosus (SLE) may be induced by a wide variety of different drugs. Indeed, over 70 agents have been implicated in causing drug-induced lupus (Rich, 1996) (Table 10). The most commonly implicated agents of drug-induced SLE are procainamide and hydralazine, although drugs less commonly associated include chlorpromazine, isoniazid, methyldopa, penicillamine, and quinine, as well as whole groups of drugs such as anticonvulsants, beta-blockers, sulphonamides, and others (Price and Venables, 1995).
The possible pathogenesis of drug-related SLE is reviewed in detail elsewhere, but it may have an immunogenetic basis, and affected patients have some of the immunological features of classic SLE (Burlingame and Rubin, 1996).
(2) Drug-related white lesions
(a) Burns (see above) (b) Lichenoid eruptions
Since the advent of antimalarial therapy, there have been an ever-increasing list and spectrum of drugs that may give rise to mucocutaneous lichen planus (LP)-like eruptions (lichenoid reactions) (McCartan and McCreary, 1997; Scully et al., 1998). However, many of the reports claiming associations have been single case reports, and many of the drugs implicated in cutaneous lichenoid reactions have not been shown to be associated with oral lesions.
The possible association of drugs with lichenoid reactions was noted when quinacrine and mepacrine, used as antimalarials during World War II, were seen to cause lichenoid lesions. Apart from these drugs, gold was probably the most common agent recognized as initiating a lichenoid reaction (Penneys et al., 1974). Gold salts can cause a range of mucocutaneous lesions (Hakala et al., 1986) of which oral lichenoid lesions may be the first (Brown et al., 1993; Laeijendecker and van Joost, 1994).
The drugs now most commonly implicated in lichenoid reactions are the non-steroidal anti-inflammatory drugs and the angiotensin-converting enzyme inhibitors (Potts et al., 1987; Firth and Reade, 1989; Robertson and Wray, 1992; Van Dis and Parks, 1995). Lichenoid reactions also may follow the use of HIV protease inhibitors (Scully and Diz, 2001), antihypertensive agents, antimalarials, phenothiazines, sulphonamides, tetracyclines, thiazide diuretics, and many others (Table 11) (Dinsdale and Walker, 1966; Roberts and Marks, 1981; Chau et al., 1984; Hogan et al., 1985; Colvard et al., 1986; Markitziu et al., 1986;Torrelo et al., 1990), but the list of drugs implicated lengthens almost weekly and, interestingly, includes several agents which have also been used in the therapy of lichen planus, particularly dapsone (Downham, 1978), levamisole (Kirby et al., 1980), tetracycline (Mahboob and Haroon, 1998), and interferon (see below). Occasionally, there are lichenoid reactions to multiple drugs (Wiesenfeld et al., 1982).
Several questions remain regarding drugs as causal agents of these reactions. For example, why can the same drug bring about different clinical manifestations? How can quite different chemical structures coincide in the clinical expression of their side-effects? and How can some drugs belonging to the same family (such as antimalarials) both produce a lichenoid reaction and at the same time find some use in the treatment of oral lichen planus (LP)? (Eisen, 1993).
The exact pathogenic mechanism by which drugs may cause LP-like disease are not known. Some of the agents implicated (e.g., penicillamine, captopril, and gold sodium thionalate) are thiol-like and hence implicated in pemphigus-like disease (see below). However, in LP, quite different immunological mechanisms are involved. It is likely that Grinspan’s syndrome simply represents a drug-induced disorder (Lamey et al., 1990), and drug therapy may occasionally account for the co-occurrence of LP with lupus erythematosus or bullous-like disease (Flageul et al., 1986). Clinical identification of lichenoid drug reactions has been based largely on subjective criteria: There does seem to be sometimes a tendency for these oral lesions to be unilateral (Lamey et al., 1995a) and erosive (Potts et al., 1987), but these features are by no means invariable. Histology may help; lichenoid lesions may have a more diffuse lymphocytic infiltrate and contain eosinophils and plasma cells, and there may be more colloid bodies than in classic LP, but there are no specific features (Van der Haute et al., 1989), and immunostaining is usually non-contributory, though basal cell cytoplasmic antibodies may be found (Lamey et al., 1995b), but this has not been confirmed (Ingafou et al., 1997) and surely occurs less reliably than in cutaneous drug reactions (van Joost, 1974; McQueen and Behan, 1982; Gibson et al., 1986).
The most reliable means to diagnose lichenoid reactions is if the reaction remits with drug withdrawal and returns on rechallenge, but frequently this is not possible because of the need to ensure patient safety.
Dental restorative materials may also be associated with lichenoid lesions. Most patients with OLP have no evidence of any association with dental restorative materials (Hietanen et al., 1987). However, contact with or proximity to restorations involving amalgams or other materials causes some lichenoid reactions—that is, lesions that clinically and histologically resemble LP closely, but have an identifiable etiology (Lind et al., 1986; Bolewska et al., 1990). These reactions are presumably due to allergic or toxic reactions to compounds released or generated, the Koebner phenomenon, or possibly due to plaque accumulated on the surfaces of the restorations (Holmstrup, 1991).
Some of these oral lesions may improve after substitution of the amalgam by other materials (Finne et al., 1982; Jolly et al., 1986; Lind et al., 1986; Bolewska et al., 1990; Jameson et al., 1990; Skoglund and Egelrud, 1991; Laine et al., 1992; Bircher et al., 1993; Ostman et al., 1994; Henriksson et al., 1995; Smart et al., 1995; Bratel et al., 1996; Ibbotson et al., 1996), though this is often not the case with gingival lesions (Henriksson et al., 1995).
Significant reactions to mercuric salts on skin-testing may be seen in some patients with OLP (Finne et al., 1982; Eversole and Ringer, 1984; Mobacken et al., 1984; James et al., 1987; Ostman et al., 1994), though others have not found this (Hietanen et al., 1987). Finne et al.(1982) demonstrated mercury sensitivity by patch-testing in 62% of 29 patients with OLP and only 3.2% of a control group, and in a few patients their oral lesions regressed on removal of the amalgams (Finne et al., 1982). Reactions to mercuric chloride have been reported (Skoglund and Egelrud, 1991; Smart et al., 1995). These findings have been supported, and recently in studies by Skoglund and co-workers (Ostman et al., 1994), a patch-test positivity against mercury was found in 39.6% of 48 patients. Of those who had a positive patch test to mercury, 94.7% showed regression of lesions after removal of amalgams, but even 82.6% of those who showed no reaction to mercury on patch-testing also showed regression after removal of amalgams. Epicutaneous patch tests are of little prognostic value (Ostman et al., 1994; Ibbotson et al., 1996). Oral mucosal lesions of lichenoid character might not be associated with allergy to mercury, but with mechanical or galvanic insults; their interpretation calls for intra-oral patch-testing (Axéll et al., 1986).
There may also be occasional reactions to gold restorations (Conklin and Blasberg, 1987), although this has not been substantiated. There is also a report of OLP in relation to cobalt in a restoration (Torresani et al., 1994).
Composite restorations have also been implicated in oral lichenoid reactions (Lind and Hurlen, 1988), and so the wholesale replacement of amalgams as a possible treatment is not necessarily warranted.
(c) Lupoid reactions (see above) (d) Candidosis
Pseudomembranous candidosis arises secondary to therapy with broad-spectrum antibiotics (Scully et al., 1994), corticosteroids (both systemic and inhaler preparations), and other immunosuppressive regimens (e.g., ciclosporin) and cytotoxic therapies (Table 12). More rarely, mucormycosis and aspergillosis may cause thrush-like areas or other lesions in patients on long-term immunosuppressive therapies (Dreizen et al., 1992; Scully, 1992; Seymour et al., 1997).
(e) Papillomas
Human papillomavirus infection manifesting as warty-like growths may arise in patients on long-term immunosuppressive therapy (Schubert, 1991).
(f) Hairy leukoplakia
Oral hairy leukoplakia, usually affecting the dorsum and lateral borders of the tongue and floor of mouth, may be a consequence of Epstein-Barr virus infection, associated with therapy with corticosteroids (topical and systemic), ciclosporin, or other long-term immunosuppressive regimens (Triantos et al., 1997).
(g) Leukoplakia
Tobacco and alcohol use are important risk factors for leukoplakia (Pindborg et al., 1980; Sciubba, 1995) and oral epithelial dysplasia (Jaber et al., 1998). An increased frequency of lesions with epithelial dysplasia of the lips (but not oral mucosa) has been observed in some but not all iatrogenically immunosuppressed patients (King et al., 1995; Seymour et al., 1997).
Sanguinarine, the principal alkaloid of the bloodroot plant (Sanguinaria canadensis), which is used in some mouthwashes and dentifrices for its anti-plaque activity, may be associated with the development of oral leukoplakia (Damm et al., 1999; Allen et al., 2001; Mascarenhas et al., 2001, 2002).
Drug-related Mucosal Pigmentation
(1) Drug-related superficial transient discoloration
Superficial transient discoloration of the dorsum of the tongue and other soft tissues and teeth may be of various colors, typically yellowish or brown, and may be caused by some foods and beverages (such as coffee and tea), some habits (such as tobacco, betel, and crack cocaine use) (Mirbod and Ahing, 2000), and some drugs (such as iron salts, bismuth, chlorhexidine, or antibiotics), especially if these also induce xerostomia (such as psychotropic agents) (Ramadan, 1969; Suzuki et al., 1983; Ogunwande, 1989; Heymann, 2000) (Table 13). When such discoloration noticeably affects the posterior dorsum of the tongue, and the filiform papillae are excessively long and stained dark brown or black, the term ‘black hairy tongue’ is used, but this is less common than other superficial discolorations.
Stomatitis has been reported after the use of proton pump inhibitors (PPIs) such as lansoprazole with amoxicillin (Hatlebakk et al., 1995), and discolored tongue or glossitis has been recorded after the use of lansoprazole plus antibiotics such as clarithromycin with or without amoxicillin (Greco et al., 1997) or lansoprazole alone (Scully and Diz, 2001).
(2) Drug-related intrinsic pigmentation
Localized areas of pigmentation of the mucosa may be due to amalgam, while gingival pigmentation can arise secondary to the gold or metal alloys of crowns (Kedici et al., 1995). Heavy metal salts were previously reported to cause pigmentation, particularly of the gingival margin (Tables 13, 14).
Blue, blue-grey, or brown mucosal pigmentation can be an adverse effect of antimalarials, phenothiazines, and phenytoin (Giansanti et al., 1971; Watson and MacDonald, 1974; Manor et al., 1981; McAllan and Adkins, 1986). Amiodarone may cause a grey orofacial and oral mucosal pigmentation (Bucknall et al., 1986).
Minocycline has increasingly been reported to cause widespread blue, blue-grey, or brown pigmentation of the gingivae and mucosae. While much of this pigmentation may reflect discoloration of the underlying bone and roots of teeth, there is also inherent pigmentation of the oral mucosa, including the tongue (Meyerson et al., 1995; Westbury and Najera, 1997).
Oral contraceptives may, albeit rarely, cause melanotic pigmentation (Hertz et al., 1980), as can cyclophosphamide, busulphan, and ACTH (Scully and Porter, 1997). In HIV disease, drug-induced melanotic pigmentation may arise following therapy with clofazimine zidovudine and/or ketoconazole, the pigmentation being variably diffuse or macular-like (Ficarra et al., 1990; Porter et al., 1990).
Kaposi’s sarcoma of the mouth is a rare complication of immunosuppression, manifesting as a blue, red, or purple macule, papule, nodule, or area of ulceration, typically on the palate or gingivae, but able to affect other oral mucosal sites (see above).
Drug-related Swellings
(1) Drug-related gingival enlargement
Gingival enlargement is a well-described oral side-effect of drug therapy (Marshall and Bartold, 1998) (Table 15). The drugs most commonly implicated in causing this enlargement are phenytoin (Seymour and Jacobs, 1992), ciclosporin (Seymour and Jacobs, 1992a), and the calcium-channel-blockers nifedipine (Fattore et al., 1991), diltiazem (Bowman et al., 1988), verapamil (Pernu et al., 1989), and amlodipine (Ellis et al., 1993). Patients receiving therapy with both ciclosporin and calcium-channel-blockers (e.g., post-cardiac or -renal allograft recipients) may be sometimes, but not always, particularly liable to drug-induced gingival enlargement.
In general, the gingival enlargement develops within a few months of the commencement of drug therapy, is usually generalized, is only partly associated with poor oral hygiene and local plaque accumulation, and responds variably to improved plaque control and/or withdrawal or reduction of drug therapy (Cebeci et al., 1996; Thomason et al., 1996; Jackson and Babich, 1997).
Rarely, Kaposi’s sarcoma (Qunibi et al., 1988) or squamous cell carcinoma (Varga and Tyldesley, 1991) may arise within areas of ciclosporin-induced gingival enlargement.
Other drugs that have been occasionally reported to cause gingival enlargement include erythromycin (Valsecchi and Cainelli, 1992), sodium valproate (Syrjanen and Syrjanen, 1979), phenobarbitone (Gregoriou et al., 1996), and vigabatrin (Katz et al., 1997), but these have all been isolated reports.
(2) Drug-related lip and mucosal swelling
Drug-induced mucosal swelling predominantly affects the lips and tongue (although rare isolated swelling of the uvula [Quinke’s disease] can occur [Mattingly et al., 1993]), and is typically due to type I hypersensitivity reactions. The oral and dental aspects of hypersensitivity are reviewed elsewhere (Rees and Gibson, 1997), but a range of drugs—particularly penicillins, local anesthetic agents, cephalosporin derivatives, angiotensin-converting enzyme inhibitors, aspirin, and barbiturates—may give rise to angio-edema (Table 16). Hypersensitivity to latex is an increasing problem in oral health care and may cause rapid-onset angio-edema in susceptible patients.
Non-allergic oral swelling can also arise in response to angiotensin-converting enzyme (ACE) inhibitor therapy. This adverse effect occurs in 0.1 to 0.7% of patients, typically arising in the early weeks of therapy, although it may occur within a few hours of commencement of treatment, or after long-term therapy. The swelling usually affects the lips, although it can be localized to the tongue, and is occasionally fatal. African-Americans may be at particular risk (Ulmer and Garvey, 1992; Gabb et al., 1996; Sabroe and Black, 1997; Lapostolle et al., 1998; Vleeming et al., 1998). The tissue swelling associated with ACE inhibitor therapy may be due to a rise in levels of bradykinins and/or altered levels or function of C1 esterase inhibitor (Ebo and Stevens, 1997; Seymour et al., 1997).
Plasmacytosis due to ‘tartar control’ toothpastes gives rise to localized enlargements of the gingivae, tongue, and other oral mucosae. This disorder is histopathologically characterized by a polyclonal plasmacytic infiltrate of the upper lamina propria and may rarely affect other mucosal surfaces, such as the larynx and anogenital surfaces (White et al., 1986; Timms et al., 1988).
Drug-related Cheilitis
Cheilitis is commonly caused by contact reactions to cosmetics or foods, but drugs may also be implicated, particularly cytotoxic agents, phenothiazines, protease inhibitors, psoralens, and retinoids (Scully et al., 2000) (Table 17).
Drug-related Neuropathies
(1) Drug-related trigeminal neuropathies
Trigeminal paresthesia, hypoesthesia, or anesthesia can arise following the use of interferon alpha (Read et al., 1995), acetazolamide, labetalol, sulthiame, vincristine (McCarthy and Skillings, 1992), mefloquine (Watt-Smith et al., 2001), and, occasionally, with other agents—most recently by hepatitis B vaccination (Maillefert et al., 1997) and some of the protease inhibitors (Scully and Diz, 2001) (Table 18). Local anesthetics such as articaine and prilocaine may exhibit a small degree of neurotoxicity (Haas and Lennon, 1995).
(2) Drug-related involuntary facial movements
Butyrophenones, phenothiazines, tricyclic antidepressants, and other drugs can occasionally produce several different drug-induced movements which can affect the mouth and face (Jimenez-Jimenez et al., 1997), particularly tardive dyskinesia secondary to antipsychotics (Hansen et al., 1997; Diederich and Goetz, 1998) and dystonias with metoclopramide (Table 19). Although these disorders principally affect the face, there can be abnormal movements restricted to the tongue—for example, the dystonia secondary to carbamazepine therapy.
(3) Drug-related orofacial pain and oral dysesthesia
Several drugs, especially the vinca alkaloids such as vincristine, can cause orofacial pain (Table 20) (McCarthy and Skillings, 1991, 1992). Enalapril (Guasti et al., 1998) and sometimes other angiotensin-converting enzyme inhibitors, such as captopril and lisinopril, may rarely cause a scalded-type sensation of the mouth (Vlasses et al., 1982; Savino and Haushalter, 1992). Orofacial pain may also be a rare consequence of drug-induced tardive dyskinesia (Ford et al., 1994).
Drug-related Oral Malodor (Halitosis)
Oral malodor may be related to drugs causing xerostomia, which may indirectly cause or aggravate this problem, but other drugs, such as isosorbide dinitrate, dimethyl sulphoxide, or disulfiram, can be directly responsible for malodor (Table 21).
Drug-related Tooth Discoloration
Chlorhexidine, fluorides, iron, and habits such as tobacco and betel use are well-recorded as causing superficial tooth discoloration, but some other drugs such as antibiotics and essential oils may also be causal. Intrinsic discoloration is prominent when tetracyclines are given to children under 12 years, but other drugs, especially ACE inhibitors, are also able to produce alterations in the color of the teeth (Table 22).
Summary
A wide spectrum of drugs can give rise to numerous adverse orofacial manifestations. The most common reactions are dry mouth, taste disturbances, and gingival swelling. Drug-induced oral mucosal ulceration is also not uncommon, particularly in cancer chemotherapy. There are many other occasional reactions.
There are few relevant randomized double-blind controlled studies in this field, and the only data available are from case reports, small series, and non-peer-reviewed reports of adverse drug reactions.
The clinician should take a careful drug history and always exclude drugs as a cause of oral and peri-oral symptoms and signs.
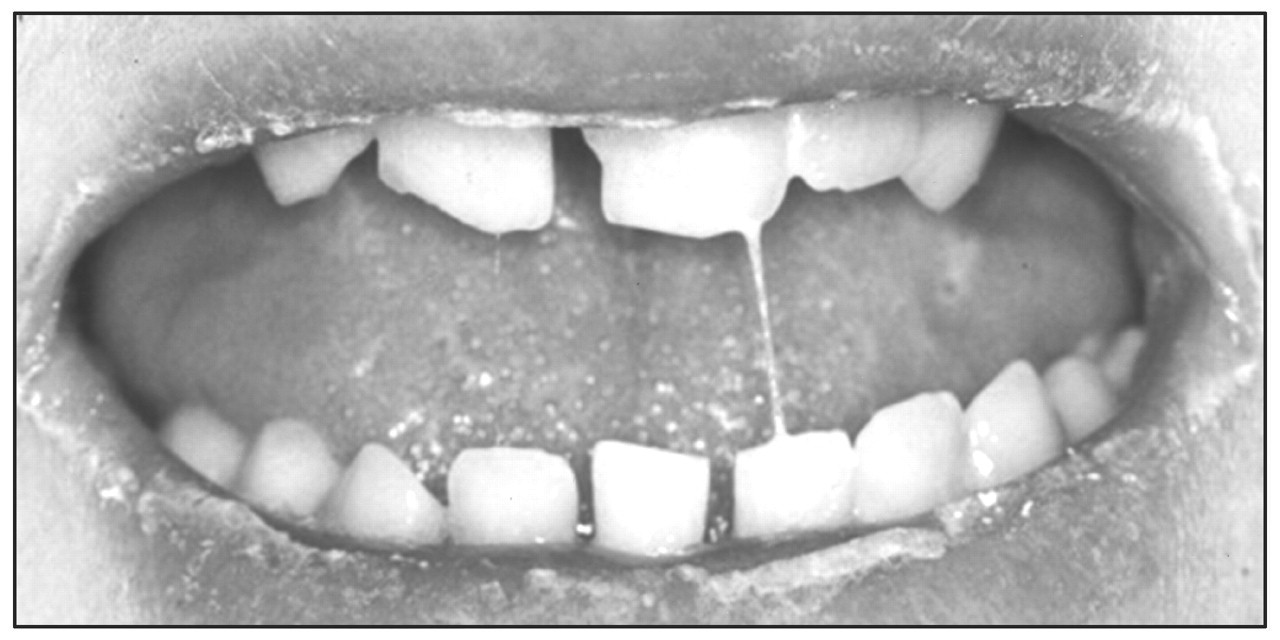
Drug-induced xerostomia.
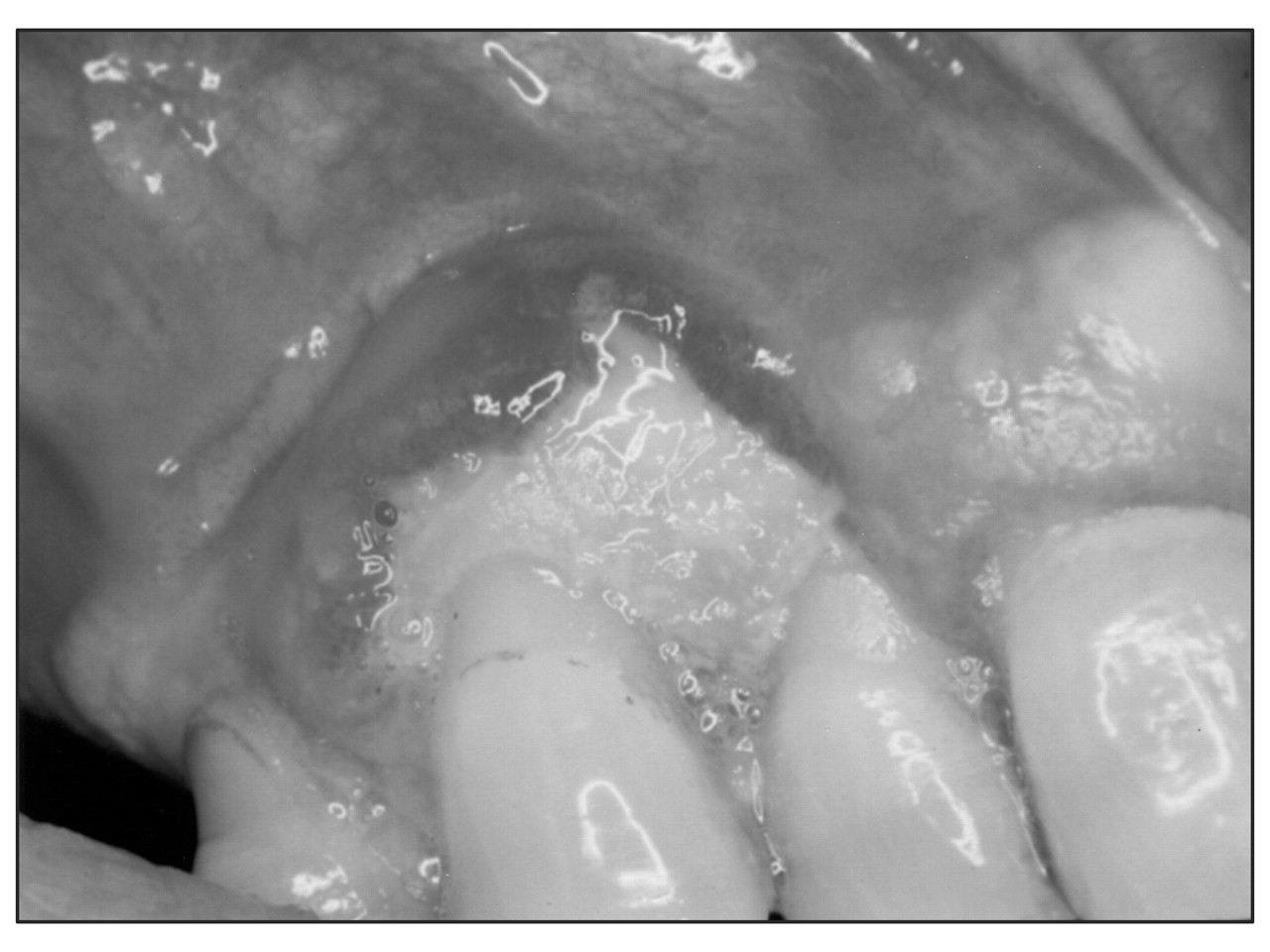
NSAID-induced ulceration.
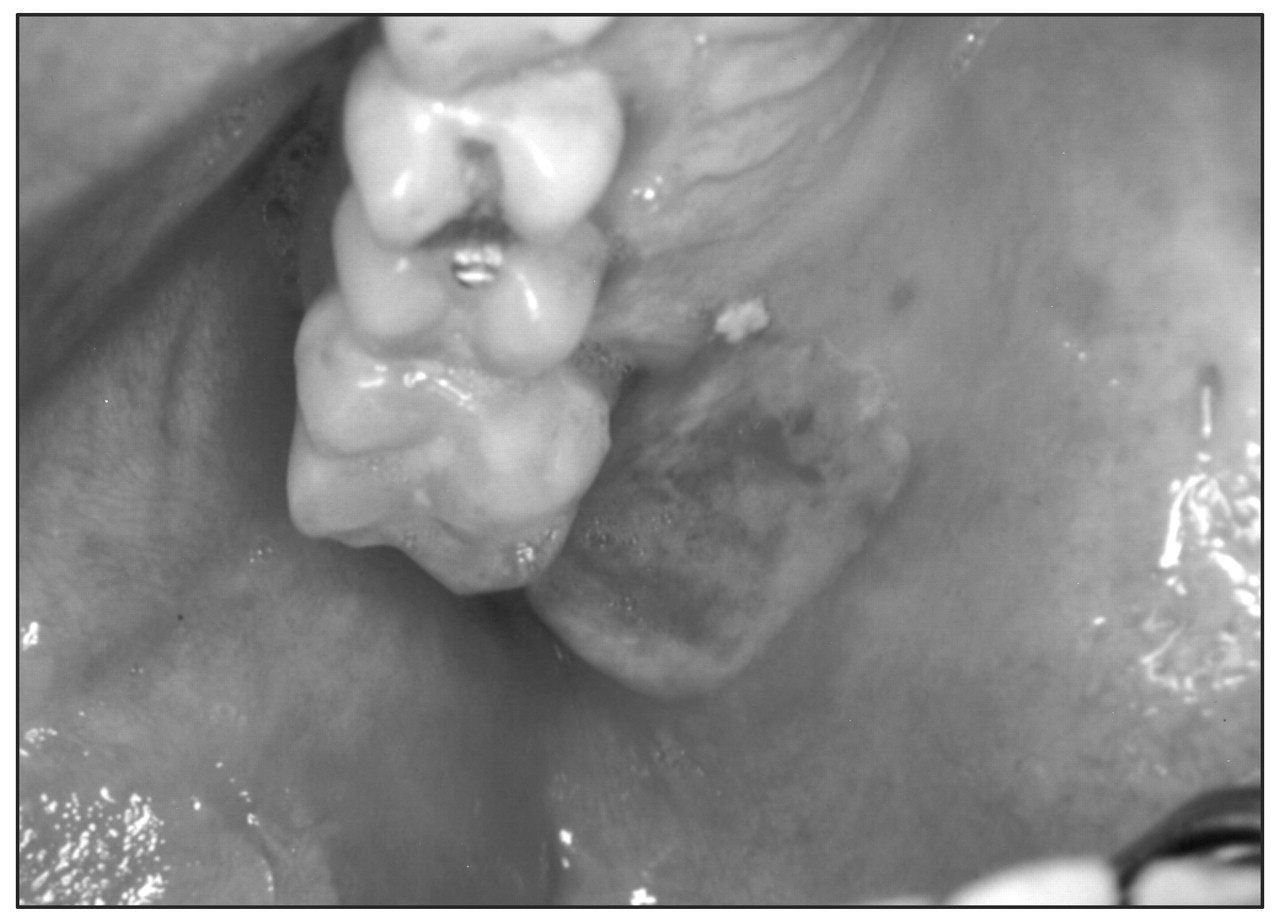
Phenylbutazone-induced ulceration.
