Abstract
The Obstructive Sleep Apnea-Hypopnea Syndrome (OSAHS) is a common sleep-related breathing disorder characterized by repetitive obstructions of the upper airway during sleep. Modification of pharyngeal patency by Oral Appliance (OA) therapy has been suggested as an alternative to various treatment modalities for OSAHS. To determine the evidence base with respect to the efficacy and co-morbidity of OA therapy in OSAHS, we conducted a systematic review of the available literature. Primary outcome measures were the reduction in number of upper-airway obstructions and co-morbidity related to the craniomandibular or craniofacial complex, respectively. Eligible studies regarding efficacy were independently assessed by two assessors using a quality assessment scale. Effect sizes of methodologically sound studies were calculated. In identical interventions, effect sizes were pooled with the use of a random-effects model. Given the scarcity of controlled studies related to co-morbidity, appraisal was confined to a description of eligible studies. Sixteen controlled trials related to efficacy were identified. With respect to the primary outcome measure, OA therapy was clearly more effective than control therapy (pooled effect size, −0.96; 95% confidence interval [CI], −1.49 to −0.42) and possibly more effective than uvulopalatopharyngoplasty. Although patients generally preferred OA therapy, improvement of respiratory variables, such as the number of upper-airway obstructions, was usually better in Continuous Positive Airway Pressure (CPAP) therapy (pooled effect size, 0.83; 95% CI, 0.59 to 1.06). Moreover, specific aspects related to OA design may influence patient-perceived efficacy and preference. Twelve patient-series and one controlled trial related to co-morbidity were identified. Analysis of the data suggests that OA therapy may have adverse effects on the craniomandibular and craniofacial complex. Although CPAP is apparently more effective and adverse effects of OA treatment have been described, it can be concluded that OA therapy is a viable treatment for, especially, mild to moderate OSAHS. Controlled studies addressing the specific indication and co-morbidity of OA therapy are warranted.
(1) Introduction
The Obstructive Sleep Apnea-Hypopnea Syndrome (OSAHS), a common sleep-related breathing disorder, is characterized by repetitive upper-airway obstructions and disruptive snoring during sleep (Guilleminault et al., 1976). Upper-airway obstructions in OSAHS can be either partial or complete and often result in (possibly severe) oxygen desaturations. When a complete or partial airway obstruction is manifested by a complete cessation or substantial reduction (i.e., > 50%) in oronasal airflow of at least 10 sec, the respiratory event is defined as apnea or hypopnea, respectively (AASM, 1999). When associated with an oxygen desaturation (> 3%) or brief awakening from sleep (i.e., arousal), a moderate reduction in airflow (i.e., < 50%) of 10 sec or longer is also defined as hypopnea. Normal upper-airway patency is usually re-established after an increased respiratory effort in response to hypoxia and hypercapnia (abnormal increase in PaCO2) (Gleeson et al., 1990). These increased respiratory efforts result in brief awakenings from sleep (i.e., arousals) that usually go unnoticed by patients (Berry and Gleeson, 1997). Recurrent arousals are associated with sleep fragmentation and a depletion of REM and slow-wave sleep (non-REM stages 3 and 4), ultimately resulting in Excessive Daytime Sleepiness. Other consequences of sleep fragmentation include reduced neurocognitive functioning, an impaired quality of life, and an increased risk of motor vehicle and occupational accidents (Teran-Santos et al., 1999; Lindberg et al., 2001). Hemodynamic consequences of upper-airway obstruction include sustained periods of hypertension (Bananian et al., 2002), and an increased risk for cardiovascular diseases such as myocardial infarction, cerebrovascular accidents, and congestive heart failure (Shahar et al., 2001). Moreover, analysis of the available data suggests that OSAHS, especially when severe, is associated with increased mortality (Redline, 2002).
According to the recommendations of the American Academy of Sleep Medicine, OSAHS is defined by a combination of symptoms (such as Excessive Daytime Sleepiness) and laboratory findings (AASM, 1999). Laboratory findings should demonstrate a Respiratory Disturbance Index (RDI) of five or more obstructed breathing events per hour of sleep. These events include any combination of apneas, hypopneas, and Respiratory Effort Related Arousals (RERAs). On the basis of the RDI, OSAHS may be classified as mild (RDI 5–15), moderate (RDI 15–30), or severe (RDI > 30) (AASM, 1999). Because RERAs are included in the RDI, the Upper Airway Resistance Syndrome, a condition with somewhat similar pathophysiology lacking marked airway obstructions, is classified as OSAHS (AASM, 1999). However, since the detection of RERAs requires more sensitive diagnostic monitoring techniques, the number of breathing events is usually quantified by the number of apneas and hypopneas per hour of sleep (i.e., Apnea-Hypopnea Index; AHI) (AASM, 1999). When the above-stated recommendations are adopted, OSAHS can be diagnosed in 2% of women and 4% of middle-aged men (Young et al., 1993). In contrast to these imposing figures, it is estimated that, in the general population, approximately 80 to 90% of patients meeting the criteria of at least moderate OSAHS remain undiagnosed (Young et al., 1997a). Since untreated OSAHS is likely to deteriorate over time and rarely disappears (Young and Peppard, 2002), this is of serious consequence for unrecognized patients.
It is assumed that both anatomical and neuromuscular factors are of significance in the pathogenesis of upper-airway obstruction in OSAHS (Gleadhill et al., 1991). However, other variables, such as lung volume and individual variability in ventilatory control, may also be of significance (Malhotra and White, 2002). The increased risk of OSAHS in males has been attributed to gender differences in airway morphology (e.g., fat distribution and craniofacial dimension) and protective effects of female hormones on upper-airway patency (Manber and Armitage, 1999; Schwab, 1999). This latter hypothesis is confirmed by the fact that the menopausal state entails a risk for developing OSAHS (Young and Peppard, 2002). Although OSAHS prevalence is shown to increase with age (Ancoli-Israel et al., 1991), it is unclear whether this can be attributed to an accumulation of cases or to an increase in incidence. OSAHS also appears to be more common in several endocrine disorders, like hypothyroidism, acromegaly, Cushing Syndrome, and diabetes mellitus (Rosenow et al., 1998). Other risk factors for OSAHS include familial aggregation and Afro-American racial origin (Redline et al., 1995, 1997). Besides gender and age, obesity is probably the most important risk factor for OSAHS (Flemons et al., 1994). It is hypothesized that obesity can influence breathing during sleep by inducing hypoxemia, and altering (upper) airway structure and function (Strobel and Rosen, 1996). Various abnormalities of the bony and soft-tissue structures of the head and neck may also compromise the upper airway during sleep. Whether related to a genetic syndrome or not, these abnormalities include craniofacial abnormalities (e.g., retro- or micrognathia), macroglossia, adenotonsillar hypertrophy, and palatal enlargement (Strohl and Redline, 1996; Kushida et al., 1997). Moreover, nasal congestion due to allergic rhinitis, acute upper-airway infection, or anatomical abnormalities (e.g., a deviated septum, conchal hypertrophy, or nasal polyps) has been identified as risk factors for OSAHS (Young et al., 1997b). Finally, several intoxications may predispose to upper-airway obstruction during sleep, including the use of tobacco, alcohol, and respiratory depressant or sedative medication (Malhotra and White, 2002).
Despite its labor-intensive character, full-night polysomnography is regarded as the standard diagnostic technique for OSAHS (Malhotra and White, 2002). This comprehensive sleep recording, performed either in a sleep laboratory or ambulatory in a home setting, generally incorporates recording of electroencephalogram, electro-oculogram, chin electromyogram, snoring, thermistor, electrocardiogram, pulse oximetry, and tibialis anterior electromyogram. Polysomnography allows for assessment of sleep architecture and quantification of upper-airway obstructions, arousals, and oxygen desaturations. Other diagnostic instruments may be needed to provide additional information with regard to sleepiness. Although a standard technique for measuring sleepiness is not available at present, the Epworth Sleepiness Scale is probably the most adequate and inexpensive test of all (Johns, 1991). The Epworth Sleepiness Scale is a simple, self-administered questionnaire in which patients rate their propensity to fall asleep in eight different situations. Scales that objectify sleepiness are the Multiple Sleep Latency Test and the Maintenance of Wakefulness Test (AASM, 1999). OSAHS should be discriminated from Central Sleep Apnea Syndrome, Cheyne-Stokes Respiration, and several other conditions characterized by excessive sleepiness, including narcolepsy, insufficient sleep, periodic leg movements, non-respiratory arousal disorders, and alcohol or drug abuse (AASM, 1999). Moreover, OSAHS should be distinguished from simple snoring, which is associated with a physiological number of airway obstructions and the absence of OSAHS-related symptoms.
(2) Current Treatment Modalities for OSAHS
When treating OSAHS, clinicians may consider various non-invasive, surgical and pharmacological modalities. OSAHS-related symptoms (e.g., Excessive Daytime Sleepiness or cardiovascular sequelae) are decisive when a treatment modality is being selected. OSAHS treatment is preferably associated with minimal co-morbidity while optimally relieving symptoms and reducing mortality. The various treatment modalities for OSAHS will be discussed in the next sections.
(2.1) Conservative management
Conservative management constitutes the correction of a medical problem or a lifestyle that possibly affects OSAHS symptomatology. Successful management of obesity confirms the reversibility of overweight as a risk factor of OSAHS (Smith et al., 1985). However, on a long-term basis, weight loss is often difficult to achieve and not always effective in obese OSAHS patients (Sampol et al., 1998). Sleep deprivation or fragmentation may also predispose to OSAHS (Series et al., 1994). Therefore, when appropriate, OSAHS patients should be encouraged to improve their ‘sleep hygiene’ (e.g., improvement in sleep-wake patterns and avoidance of stimulants in the evening). Patients with ‘supine dependent’ upper-airway obstructions may be successfully treated by changing their sleep position from supine to lateral or more upright (McEvoy et al., 1986; Cartwright et al., 1991). Furthermore, when feasible, patients should be advised to abstain from alcohol, tobacco, and respiratory depressant or sedative medication (e.g., opiates or benzodiazepines). Although conservative measures usually require additional treatment, they should always be considered due to their facilitating effect in OSAHS management.
(2.2) Continuous positive airway pressure
The introduction of Continuous Positive Airway Pressure (CPAP) has been of great significance in the management of OSAHS (Sullivan et al., 1981). Prior to CPAP, symptomatic OSAHS patients were nearly always treated with a tracheotomy. CPAP pneumatically splints the upper airway during sleep by means of a flow generator that is connected to an (oro)nasal mask (Schwab et al., 1996). Treatment usually results in the near-elimination of upper-airway obstructions and notable improvements in oxyhemoglobin saturation, sleep architecture, and snoring (Lojander et al., 1996; Jenkinson et al., 1999). Although a placebo effect has been documented, CPAP is also known to substantially improve Excessive Daytime Sleepiness, quality of life, depression, and neurocognitive performance (Engleman et al., 1999; Jenkinson et al., 1999). Since discontinuation of treatment usually results in a rapid recurrence of symptoms (Kribbs et al., 1993), CPAP is generally a lifelong requisite.
Side-effects of CPAP, although rarely severe, are frequently observed and predominantly relate to the (oro-)nasal mask (Hoffstein et al., 1992; Pépin et al., 1995). Moreover, it is not uncommon for CPAP to cause nasal congestion, rhinorhea, sneezing, and mucosal dryness of the upper airway (Hoffstein et al., 1992; Pépin et al., 1995). Poor compliance has been suggested to be the major drawback of CPAP, with patients troubled by more side-effects generally showing poorer compliance (Engleman et al., 1996). Best compliance is usually seen in patients with severe OSAHS or substantial daytime sleepiness (Pépin et al., 1995, 1999). CPAP should be considered when conservative management is not applicable or when additional treatment is required (White et al., 2002). Individuals failing or refusing CPAP and patients with clearly reversible abnormalities (e.g., anatomical deformities) are candidates for alternative treatment (Malhotra and White, 2002). Moreover, since results of CPAP in mild to moderate OSAHS are conflicting, alternative treatment may also be considered in these patients (Krieger et al., 1996; Engleman et al., 1999).
(2.3) Surgical management
Adenotonsillectomy or surgical correction of a compromised nasal passage usually fails to correct OSAHS (completely) in adult patients (Sher et al., 1996). However, these interventions should always be considered, because they facilitate the treatment of OSAHS with other modalities (e.g., CPAP). For a considerable time, the uvulopalatopharyngoplasty (UPPP) has been the most popular surgical procedure in the treatment of OSAHS. UPPP involves a resection of the uvula and redundant palatal, tonsillar, and mucosal tissues. Despite its initial popularity, it has been shown that only approximately 40% of OSAHS patients are successfully treated with an UPPP (Sher et al., 1996). Long-term results are not more successful because relapse occurs in a significant proportion of initially successfully treated patients (Janson et al., 1997). Moreover, more recently employed techniques for correcting retropalatal airway obstruction, like Laser-Assisted Uvulopalatoplasty or radiofrequency volumetric tissue reduction (i.e., somnoplasty), have also been disappointing in the treatment of OSAHS (ASDA, 1994; Brown et al., 2001).
Better results have been obtained with more ‘aggressive’ surgical interventions, including genioglossal advancement with hyoid myotomy and suspension or maxillomandibular advancement surgery (Riley et al., 2000; Sher, 2002). In genioglossal advancement, the surgeon puts the tongue under anterior traction by performing a limited parasagittal mandibular osteotomy and subsequently advancing the genioid tubercle. In addition to genioglossal advancement, a hyoid myotomy is often performed. The latter procedure is intended to enlarge the retrolingual airway by anterior fixation of the hyoid bone following the release of the infrahyoid muscles. Maxillomandibular advancement provides maximal enlargement of the retrolingual and some enlargement of the retropalatal airway by means of a Le Fort I and a mandibular sagittal split advancement osteotomy. When these surgical procedures were performed according to protocol, successful management of OSAHS has been reported in approximately 60% of patients following UPPP and/or genioglossal advancement with hyoid myotomy, and in 90% of patients after maxillomandibular advancement (Riley et al., 2000). Because generalization of these results is questionable, and widespread acceptance is lacking, randomized controlled trials are needed (Bridgman et al., 2002). A laser midline glossectomy and lingualplasty create an enlarged retrolingual airway by laser excision of a portion of the posterior half of the tongue. These procedures, however, are not frequently used for the management of OSAHS (ASDA, 1996). Despite considerable co-morbidity, a tracheotomy, which produces a bypass of the upper airway, is probably the most predictable surgical intervention for OSAHS (Riley et al., 2000). Although gastric restrictive and bypass procedures are increasing in popularity, the limited experience in OSAHS patients and associated morbidity and mortality restrict application of these procedures to only selected cases (Livingston, 2002). Because OSAHS is associated with an increased operative risk (Esclamado et al., 1989), anaesthesiological precautions and treatment according to protocol are always requisite (Riley et al., 1997). The practice parameters of the American Sleep Disorders Association also provide recommendations for the surgical management of OSAHS patients (ASDA, 1996).
(2.4) Pharmacological and medical management
Various pharmacological agents have been deployed in the management of OSAHS. Although beneficial effects of tricyclic antidepressants and serotonergic agents have been reported, widespread use in OSAHS is mainly constrained by anti-cholinergic side-effects and limited overall efficacy, respectively (Smith et al., 1983; Hedner and Grote, 2002). In selected patients with persisting excessive daytime sleepiness, wake-promoting agents like modafinil may be beneficial (Arnulf et al., 1997). Contrary to the suggested protective effect of sex hormones in OSAHS pathogenesis, steroid treatment (e.g., medroxyprogesterone) is generally worth considering only in patients with awake respiratory failure (Hedner and Grote, 2002). Thyroxine may be successful for the management of OSAHS patients with hypothyroidism (Grunstein and Sullivan, 1988). However, in acromegalic patients, additional treatment of OSAHS is usually required following the pharmacological suppression of growth hormones (Grunstein et al., 1994).
Treatment with nasopharyngeal intubation or mechanical dilation of the anterior nasal valve is usually associated with poor tolerance and inadequate results, respectively (Nahmias and Karetzky, 1988; Hoffstein et al., 1993). However, promising results have been obtained following the stimulation of specific upper-airway musculature during sleep. Especially, unilateral electrical stimulation of the hypoglossal nerve by means of an implanted electrode has been successful in the management of OSAHS (Eisele et al., 1997). However, durable and reliable stimulation systems are required.
(3) Oral Appliances
(3.1) Background
Since their introduction in the 1980s, a variety of dental devices has been developed for the management of OSAHS and simple snoring. These intra-oral devices, commonly known as Oral Appliances, aim at relieving upper-airway obstruction and snoring by modifying the position of the mandible, tongue, and other (oro-)pharyngeal structures. In 1902, the French physician Pierre Robin laid the foundation for Oral Appliance (OA) therapy. With his ‘Monobloc’ appliance, Robin treated children who suffered from breathing difficulties and glossoptosis due to mandibular hypoplasia (Robin, 1934). The first case of an OA that repositioned the mandible in an adult patient with OSAHS was not reported until 1980 (Bear and Priest, 1980). The first patient-series of OA therapy in OSAHS management was reported in 1982 and described the effects of an OA that repositioned the tongue (Cartwright and Samelson, 1982). Currently, well over 50 different Oral Appliances are marketed for the treatment of OSAHS and simple snoring (Lowe, 2000).
(3.2) Types of appliances
Based on the mode of action, Oral Appliances may be roughly divided into Tongue-retaining Appliances and Mandibular Repositioning Appliances. Tongue-retaining Appliances reposition the tongue in an anterior position by securing it with negative pressure in a soft plastic bulb or with a plastic depressor that comes into direct contact with the base of the tongue. The latter device, known as SnorEx® (Depita, Nienhagen, Germany), is limited for large-scale use because of poor results and non-compliance (Schönhofer et al., 1997). The Tongue-retaining Device, which incorporates a plastic bulb into a custom-made dentally retained soft acrylic appliance, has been demonstrated to effectively reduce the number of upper-airway obstructions in OSAHS patients (Cartwright and Samelson, 1982). However, a compromised nasal passage or discomfort and loss of negative pressure in the bulb may hamper full-night application of the appliance. The Tongue-stabilizing Device, an ‘off-the-shelf’ appliance somewhat similar to the Tongue-retaining Device, shows comparable results (Kingshott et al., 2002). Although rarely used because of poor results and patient tolerance, palatal lifting devices, tongue posture trainers, and labial shields are also Oral Appliances that claim to improve snoring and OSAHS (Marklund and Franklin, 1996; Loube, 1998; Barthlen et al., 2000). Our systematic review is limited to the application of the Mandibular Repositioning Appliance (MRA), because it is used most frequently in the treatment of OSAHS.
MRAs are of either a one-piece (Monobloc) or a two-piece design (Bibloc), and are either custom-made or pre-fabricated (Eckhart, 1998). A pre-fabricated MRA generally requires only an individual molding of a thermolabile material, while a custom-made appliance usually requires dental impressions, bite registration, and fabrication by a dental laboratory. Retention of the appliances is usually provided by clasps, acrylic, and thermoplastic polymer (Lindman and Bondemark, 2001). The one-piece design fixes the mandible rigidly in an anterior position, whereas the two-piece MRA usually allows for some freedom of mandibular movement (i.e., lateral, vertical, and/or anterior). This latter feature has been suggested to decrease the chance of temporomandibular disorders and improve patient comfort (Henke et al., 2000). Conversely, fixation of the mandible with a one-piece appliance is suggested to prevent suppression of tongue-protruding muscles, resulting in a less collapsible upper airway (George, 2001). Most two-piece appliances are sagittally adjustable, thereby allowing for individual titration and possibly greater mandibular advancement (Pancer et al., 1999). Another variation in design is the degree of bite opening imposed by the appliance. Fluoroscopic recordings suggest that bite opening should be kept to a minimum, since, in awake OSAHS patients, it results in posterior movement of both tongue and soft palate (L’Estrange et al., 1996). However, increased baseline genioglossus muscle activity has been implicated in downward rotation of the mandible (Lowe et al., 1990). Moreover, bite opening may improve upper-airway patency by stretching the palatoglossus- and superior pharyngeal constrictor muscle (George, 2001).
(3.3) Mechanism of action
Forward displacement of the mandible appears to prevent oropharyngeal airway obstruction by (indirectly) moving the suprahyoid and genioglossal muscles anteriorly (Tegelberg et al., 1999). It has been suggested that forward and inferior displacement of the mandible decreases the gravitational effect of the tongue and preserves the velopharyngeal airway by stretching the palatoglossal- and palatopharyngeal arch (Ryan et al., 1999; Tegelberg et al., 1999; Liu et al., 2000). Moreover, stabilization of the mandible and hyoid bone prevents posterior rotation of the jaw and retrolapse of the tongue during sleep (Loube, 1998). Three-dimensional imaging and (supine-)cephalometric studies demonstrated that mandibular repositioning increases oro-, hypo-, and velopharyngeal dimensions (Battagel et al., 1999; Gao et al., 1999; Gale et al., 2000; Liu et al., 2000; Schwab, 2001; Fransson et al., 2002a). Endoscopic studies have demonstrated that mandibular advancement results in, particularly, an increased cross-section of the lateral dimension of the velopharynx (Isono et al., 1995; Ryan et al., 1999). Beside anatomical changes, the effect of an MRA has also been attributed to a normalization in physiological properties of the upper airway (Yoshida, 1998). For example, mandibular rotation and advancement have been associated with increased upper-airway muscle activity (Lowe et al., 1990; Yoshida, 1998). Conflicting results and the fact that most imaging studies are performed while subjects are in the wake state require further elucidation of the precise biomechanical mechanism of MRA therapy.
(3.4) Effectiveness
Based on subjective reports of patients and their bed partners, MRA therapy generally results in improvements of snoring in a high proportion of patients (Schmidt-Nowara et al., 1995; Lindman and Bondemark, 2001). Other reported benefits of MRA treatment include substantial decreases of daytime sleepiness, improvements in work performance, and improved sleep quality of both patient and bed partner (Arai et al., 1998; Cameron et al., 1998; Mehta et al., 2001; Gotsopoulus et al., 2002). Sleep registration generally confirms the patient-perceived benefits by demonstrating a decrease in snoring frequency and intensity, AHI or RDI, oxygen desaturation frequency and intensity, and number of arousals (Bloch et al., 2000; Mehta et al., 2001; Gotsopoulus et al., 2002; Johnston et al., 2002). Moreover, MRA treatment has been associated with significant increases in slow-wave and REM sleep (Clark et al., 1996; Bloch et al., 2000). Although the initial effect of an MRA has been reported to be stable over a five-year period (Marklund et al., 2001a), there are studies suggesting a gradual decline in treatment effect in both the short (i.e., six weeks) and long term (i.e., four years) (Randerath et al., 2002; Walker-Engström et al., 2002). Despite an unsatisfactory change in the number of breathing events, patients may report fewer symptoms (Lowe et al., 2000). Moreover, an increased AHI after MRA therapy has been reported in approximately 13% of patients (Schmidt-Nowara et al., 1995). Because of this risk of an increased or suboptimal AHI, a follow-up sleep study should always be conducted in MRA treatment.
Although it has been suggested that MRA therapy is less effective in severe OSAHS (Marklund et al., 1998a; Liu et al., 2000), others postulate that severity should be no reason to exclude patients from treatment (Henke et al., 2000). Cephalometric and MRI variables associated with a favorable response to MRA therapy include a cranial position of the hyoid bone, a smaller mandibular plane angle, a reduced lower anterior face height, a longer anterior cranial base, an increased maxillary length, a shorter soft palate, and a relatively ‘normal’ airway diameter or soft-palate and tongue proportion (Eveloff et al., 1994; Gao et al., 1999; Liu et al., 2000; Johal and Battagel, 2001; Liu et al., 2001; Skinner et al., 2002). However, because of conflicting findings and unclear clinical significance, the relevance of these cephalometric predictors should be questioned. Treatment success has also been associated with a lower Body-Mass Index (i.e., patient’s weight [kg]/square of patient’s height [m]), smaller neck circumference, younger age, and a lower baseline AHI (Liu et al., 2001; Mehta et al., 2001). Furthermore, successful treatment has been reported to be more likely in patients with ‘supine dependent’ airway obstructions and in patients who are able to protrude their mandible for 5 mm or more (Marklund et al., 1998a,b). However, it should be noted that not all of these predictors of treatment success are uniformly reported.
(3.5) Side-effects, complications, and compliance
Although side-effects are frequently reported with MRA therapy, these are usually mild and acceptable, with most symptoms subsiding when treatment is continued (Fritsch et al., 2001; Lindman and Bondemark, 2001). Tenderness of the teeth and jaws, myofascial pain, gum irritation, excessive salivation, and xerostomia are commonly reported in the initial period of use (Ferguson et al., 1996; Pantin et al., 1999; Mehta et al., 2001). In exceptional cases, treatment may be complicated by involuntary removal of the device, an exaggerated gag reflex, periodontal damage, or fractured teeth and fillings (Pantin et al., 1999; Rose et al., 2002a,b). It has been suggested that advancement of the mandible for considerable periods may have adverse effects on the stomatognathic system. Mild complaints of pain and strain of the masticatory muscles and the temporomandibular joint frequently occur at the initiation of treatment (Pantin et al., 1999; Tegelberg et al., 1999). Some studies have observed an increase in bruxism in response to MRA therapy (Mehta et al., 2001). In the long term, MRA treatment has been suggested to initiate or aggravate temporomandibular joint disease in individual patients (Walker-Engström et al., 2002). A temporary bite change in the morning after removal of the appliance occurs in almost all patients (Lindman and Bondemark, 2001). This phenomenon has been attributed to a partially contracted lateral pterygoid muscle and accumulation of retrodiskal blood in the temporomandibular joint area as a result of full-night mandibular protrusion (George, 2001). However, to date, this hypothesis has never been scientifically supported. In individual cases, permanent occlusal alterations have been observed after long-term treatment periods (Rose et al., 2001). These observations emphasize the importance of regular follow-up examinations in MRA therapy. It should be noted that the type of appliance, patient compliance, and the amount of mandibular protrusion may affect the frequency and severity of side-effects.
Although generally well-accepted by patients, some studies report poor initial tolerance to MRA therapy (Smith and Stradling, 2002). Patient-reported compliance with MRA therapy is generally high, with studies reporting regular use in 75 to 100% of patients (Lindman and Bondemark, 2001). Long-term compliance has been reported to decrease over time. After a four-year period, one study reported appliance use as prescribed in only 32% of patients (Rose et al., 2002c). Conversely, others suggest that long-term compliance with MRA therapy is similar to CPAP (Eveloff, 2002). Although patient-reported compliance may be an overestimate of actual use, covert compliance monitoring has shown excellent agreement between objective and subjective compliance (Lowe et al., 2000). Discontinuation of MRA treatment is generally related to side-effects, complications, or the lack of perceived benefits (McGown et al., 2001). Data on the impact of side-effects on long-term compliance are conflicting. Some studies observe similar frequencies of side-effects in compliant and non-compliant patients, whereas others report a higher number of side-effects in patients who discontinued treatment (Clark et al., 2000; McGown et al., 2001).
(3.6) Indications and contraindications
Several exclusion criteria should be taken into account when MRA therapy is considered. These include an insufficient number of teeth, (extensive) periodontal disease or dental decay, active temporomandibular joint disorders, and restrictions in mandibular opening or protrusion. In one study adopting similar exclusion criteria, MRA therapy was contraindicated in 34 out of 100 consecutive OSAHS patients (Petit et al., 2002). However, although some consider a minimum of ten sound teeth in each of the maxillary and mandibular arches a requisite in MRA treatment, the location rather than the number of teeth may be more important (i.e., posterior teeth provide more adequate retention) (Petit et al., 2002). According to recommendations of the American Sleep Disorders Association, OA therapy should be considered in patients with simple snoring or mild OSAHS who do not respond to or are not appropriate candidates for conservative management (ASDA, 1995). In moderate to severe OSAHS, the recommendation is to consider OA therapy when patients do not tolerate or refuse CPAP, and when patients are not candidates for or refuse surgical intervention. Recent reports demonstrating the effectiveness of Oral Appliances in moderate and severe OSAHS probably necessitate redefinition of these recommendations (Eveloff, 2002).
(3.7) Objective review
Since the introduction of Oral Appliances in the 1980s, their use in the treatment of OSAHS has gained considerable popularity as an alternative to current modalities because of their simplicity and supposed reversibility. Since patients generally prefer OA therapy to CPAP (White et al., 2002), some patients are preferably treated with this alternative treatment modality. Despite a possible favorable outcome of the OA in the treatment of OSAHS, comparative studies regarding efficacy and co-morbidity of this dental treatment modality are scarce. In the current article, the available literature regarding the efficacy and safety of the OA as a treatment modality for OSAHS is systematically reviewed.
(4) Methods
(4.1) Study selection
To identify studies related to the efficacy and co-morbidity of OA therapy for OSAHS, we conducted a highly sensitive search in the databases of MEDLINE (1966–2002), EMBASE (1989–2002), and Cinahl (1982–2002). The search was supplemented with a systematic search in the ‘Cochrane Central Register of Controlled Trials’ (CENTRAL) (1800–2002). The search strategy regarding the applied thesaurus (MeSH) and text words in the above-mentioned databases is summarized in Table 1. To ensure that eligible studies were not overlooked, we contacted several experts in the field of Oral Appliance therapy for unpublished or ongoing studies. Checking references of relevant review articles and eligible studies for missing publications complemented the search. No language restrictions were used throughout the study selection procedure.
On the basis of title and abstract, we decided whether an identified article was relevant to the topic under study (i.e., OA therapy for OSAHS). We then retrieved a full-text document of each ‘relevant’ article to decide whether the study was eligible for methodological appraisal. Studies regarding the efficacy of OA therapy in the treatment of OSAHS were eligible for further methodological appraisal when they met the following criteria: (1) studied patients diagnosed with OSAHS (i.e., AHI/RDI > 5); (2) studied patients at least 21 years of age; (3) intervention group treated with an MRA; (4) control group treated with any conservative, surgical, or non-invasive treatment modality for OSAHS (including none or placebo intervention); and (5) main outcome measure of treatment being the AHI (or RDI) assessed during a full-night sleep study (i.e., no split-night studies). In common with studies related to efficacy, studies related to co-morbidity of OA therapy had to meet the first three of the above-mentioned criteria. However, contrary to studies related to efficacy, studies related to co-morbidity were still eligible for further appraisal if the studied patients represented a non-homogenous group (e.g., OSAHS and snoring patients). Studies related to co-morbidity were eligible for further methodological appraisal when the main outcome measure objectively identified side-effects of OA therapy related to the craniomandibular or craniofacial complex. Therefore, studies regarding patient-perceived co-morbidity of OA therapy were excluded from further analyses. Moreover, articles in Hebrew or Asian languages, case reports, abstracts, or letters with respect to the subjects under study were not considered for further analysis. Fig. 1 outlines the algorithm of the study selection procedure.
(4.2) Methodological appraisal
Eligible studies included for methodological appraisal were independently assessed by two observers (AH, BS). So that observer bias would be minimized, all included papers were blinded with respect to title, authors, and journal name. Prior to the appraisal, the observers discussed all relevant methodological items to reach consensus about their content. When the observers could not agree on a subject during the methodological appraisal, consensus was reached by consultation with a third party (LGMdB).
Efficacy
The methodological quality of all eligible papers related to efficacy was evaluated with the ‘quality of study tool’ developed by Sindhu et al.(1997). With the use of a Delphi technique, this quality tool was especially developed to rate the methodological quality of randomized clinical trials to be included in a meta-analysis. The ‘quality of study tool’ consists of 53 items in 15 different dimensions, with each dimension having a specific weight (W). The 15 dimensions evaluate the following variables of a study: control group (max. W = 15), randomization (max. W = 10), measurement outcomes (max. W = 10), study design (max. W = 8), conclusions (max. W = 8), ‘intention-to-treat’ analysis (max. W = 8), statistical analysis (max. W = 6), adherence to study protocol (max. W = 6), blinding (max. W = 5), research question (max. W = 5), loss to follow-up (max. W = 4), outcomes (max. W = 4), reporting of findings (max. W = 4), patient compliance (max. W = 4), and remaining variables (max. W = 3). The observers scored each of the included trials according to the 15 dimensions. Agreement on the weight of each dimension was reached in a consensus meeting. In sum, of the 15 dimensions, a study can theoretically score a maximum of 100 points. On the basis of this total score, it was decided whether a study should be considered for inclusion in a meta-analyses. For this purpose, a threshold value was set. The two observers independently determined the minimum number of items required in each dimension for considering a study ‘methodologically sound’. In a consensus meeting, agreement was reached on the required weights in each dimension. Subsequently, the sum of the required weights in the 15 dimensions resulted in a threshold value of 47 points.
Co-morbidity
Based on the eligibility criteria related to co-morbidity, studies without a concurrent control group could also be included. Thereby, the methodological quality of included studies (possibly) did not meet with the most important parameter in the design of observational studies (West et al., 2002). The methodological appraisal of studies related to co-morbidity was limited to an overall impression of the study (i.e., poor, adequate, or good). Agreement on the overall impression of each study was reached in a consensus meeting.
(4.3) Presentation of data
Two reviewers independently performed the data extraction. Consensus was reached in cases of disagreement.
Efficacy
The methodological quality of each included paper is presented according to the total score. If possible, we report data on the study design, type of MRA (including mean amount of mandibular advancement), type of control, number of patients included, number of patients completing, and the reported success percentage of both MRA and control treatment. With respect to the main outcome measure (i.e., AHI/RDI) and the Epworth Sleepiness Scale (ESS), effect sizes and approximate 95% confidence intervals (CI) of ‘methodologically sound’ trials were calculated. Furthermore, relevant outcomes related to other physiological parameters, quality of life indicators, sleepiness scores, and behavioral- or cognitive-function indices are reported. A meta-analysis was carried out on the effect sizes of ‘methodologically sound’ trials with comparable (control) interventions.
Co-morbidity
The methodological quality of each included paper is presented according to the overall impression of the study. The following data are presented: study design, type of MRA (including mean amount of mandibular advancement), types of patients studied, number of patients completing the trial, mean duration of treatment, and patient-reported compliance. With each included trial, the co-morbidity of MRA treatment related to the craniomandibular and craniofacial complex is reported.
(4.4) Statistical analysis
All data were computer-analyzed with the StatsDirect software package, version 2.2.3 (Cheshire, UK). The degree of agreement with respect to the methodological appraisal of eligible studies before the consensus meeting is expressed as percentage of agreement and weighted Cohen’s kappa. Because clinical heterogeneity between and among the included trials was expected, effect sizes of trials with comparable control interventions were pooled with the use of a random-effects model (DerSimonian-Liard random effects analysis), in which smaller studies (with larger variances) contribute less than larger studies to the pooled effect.
(5) Results
The MEDLINE search yielded 1004 publications, the EMBASE search 618, the Cinahl search 131, and the CENTRAL search 548. Systematic assessment of this output revealed 289 relevant publications (Fig. 1). Although reference-checking of relevant review papers and included studies did not reveal additional articles, contact with experts in the field yielded one eligible article ‘in press’ related to co-morbidity (Robertson et al., in press).
(5.1) Efficacy
Using the specified criteria, we considered 17 trials related to efficacy as eligible for further appraisal. Because one trial reported on the four-year follow-up of another eligible study (Walker-Engström et al., 2002), 16 studies were included for methodological appraisal (Fig. 1, Table 2). Four trials compared MRA therapy with an ‘inactive’ control device (Hans et al., 1997; Mehta et al., 2001; Gotsopoulos et al., 2002; Johnston et al., 2002). Two trials studied the effects of anterior and vertical mandibular displacement in MRA treatment, respectively (de Almeida et al., 2002; Pitsis et al., 2002), whereas three trials compared several different Oral Appliances (Barthlen et al., 2000; Bloch et al., 2000; Rose et al., 2002a). In one trial, MRA therapy was compared with UPPP (Wilhelmsson et al., 1999), and six trials compared MRA therapy with CPAP (Clark et al., 1996; Ferguson et al., 1996, 1997; Engleman et al., 2002; Randerath et al., 2002; Tan et al., 2002). Patient baseline characteristics of the included trails were comparable with respect to male-to-female ratio, age (means ranging from 44.0 to 57.6 yrs), and Body-Mass Index (means ranging from 26.9 to 32.0).
The majority of the 16 included trials used a crossover design, with only two studies applying a parallel study design (Hans et al., 1997; Wilhelmsson et al., 1999). However, in three studies, subjects were not randomly allocated to the treatment groups (Clark et al., 1996; Barthlen et al., 2000; de Almeida et al., 2002). Methodological quality of the included trials, according to the total score on the quality tool, ranged from 38 to 86 points. The overall quality of the 16 trials was adequate, with three studies not meeting the predetermined threshold value of 47 points (Hans et al., 1997; Barthlen et al., 2000; de Almeida et al., 2002). Two of the three studies that did not meet the threshold lacked randomization for treatment allocation. However, studies meeting the threshold value also had methodological deficits. Although most of these studies were described as randomized, the method of randomization was generally not detailed or reported as secure and ‘blind’ to the assessors. Moreover, only one study reported blinding of patients to active and control treatment (Gotsopoulus et al., 2002). Because of the lack of a comparable placebo or control intervention, blinding of patients and therapists was usually not possible in these trials. However, reasons as to why assessment was not blinded were generally not provided, nor was there a discussion of possible bias resulting from non-blind assessment. Conversely, in six trials, the assessor of sleep variables was blinded to treatment (Clark et al., 1996; Wilhelmsson et al., 1999; Mehta et al., 2001; Engleman et al., 2002; Gotsopoulos et al., 2002; Tan et al., 2002,). Finally, in several ‘methodologically sound’ trials, the results could have been biased due to selective dropouts (i.e., no ‘intention to treat’ analysis) (Clark et al., 1996; Ferguson et al., 1996, 1997; Rose et al., 2002a; Tan et al., 2002). Inter-rater agreement on the methodological quality of each trial, according to the assigned weights, was very good (Agreement, 97%; Kappa, 0.91; 95% CI, 0.82 to 0.99). Disagreements were generally caused by slight differences in interpretation and were easily resolved in the consensus meeting.
MRA vs. control devices
Control devices were designed to increase vertical opening minimally without advancing the mandible. They consisted of either a modified MRA or the lower or upper piece of a two-piece MRA. Compared with the control device, MRA treatment was reported to be significantly more effective in improving the AHI in all four trials. This positive effect on physiological parameters was confirmed by studies demonstrating significant improvements with MRA treatment in the mean number of arousals per hour of sleep (arousal index), lowest oxygen saturation during sleep (minSaO2), and snoring frequency and intensity when compared with control devices (Mehta et al., 2001; Gotsopoulos et al., 2002). Moreover, the hourly rate of oxygen desaturations (≥ 4% fall in SaO2) decreased significantly in MRA treatment when compared with the control device (Johnston et al., 2002). Although two trials could not demonstrate a significant difference in the ESS score (Hans et al., 1997; Johnston et al., 2002), one trial reported a small but significant reduction in the ESS with MRA treatment when compared with the control device (Gotsopoulos et al., 2002). Moreover, objective daytime sleepiness according to the Multiple Sleep Latency Test improved significantly with MRA therapy when compared with the control device (Gotsopoulos et al., 2002). In one study, a significant increase in REM sleep was observed when MRA treatment was compared with the control group (Mehta et al., 2001). However, in another study, this phenomenon could not be demonstrated (Gotsopoulos et al., 2002). When compared with the control device, the patient-reported frequency and intensity of snoring significantly improved with MRA treatment in one trial but not in another (Gotsopoulos et al., 2002; Johnston et al., 2002). Although patients generally experienced more side-effects with MRA treatment, poorer patient satisfaction and compliance were reported with control devices (Hans et al., 1997; Gotsopoulos et al., 2002).
Due to inadequate methodological quality, the effect sizes of one trial were not calculated (Hans et al., 1997). Moreover, the effect size of the ESS of a second trial could not be calculated because the scale was not administered when the control device was used for treatment (Mehta et al., 2002). When the remaining effect sizes were pooled, the AHI significantly improved (effect size, −0.96; 95% CI, −1.49 to −0.42), while there was no significant change in ESS (effect size, −0.22; 95% CI, −0.51 to 0.08) in a comparison of MRA therapy with control devices (Fig. 2).
Variability in mandibular advancement and bite opening
In one study, the effect of progressive mandibular advancement on AHI was studied in seven OSAHS patients (de Almeida et al., 2002). By progressively increasing the amount of mandibular protrusion and evaluating the effect in a sleep study, the investigators could identify the amount of protrusion as one factor that decreased the AHI with MRA treatment. In another study, the effect of bite opening on efficacy and side-effects of MRA treatment was evaluated in 24 OSAHS patients (Pitsis et al., 2002). Except for an inter-incisal opening of 4 and 14 mm, patients were treated with two identical appliances. Although with both appliances the AHI and arousal index decreased significantly, no significant differences could be demonstrated between the devices. Moreover, subjective outcomes like the ESS, sleep quality, and improvements in snoring did not differ between the two appliances. Although side-effects and reported compliance did not differ, a significantly higher proportion of patients preferred using the MRA with lower vertical dimension.
The effect size of the AHI of one trial was not calculated due to inadequate overall methodological quality (de Almeida et al., 2002). Although there was a trend toward greater efficacy with the MRA with lower vertical dimension, no significant differences in effect sizes with respect to the AHI (effect size, −0.26; 95% CI, −0.84 to 0.32) and ESS (effect size, 0.00; 95% CI, −0.58 to 0.58) could be demonstrated (Fig. 2) (Pitsis et al., 2002).
Variability in appliance design
In one trial, the effect of an MRA was compared with that of a Tongue-retaining Device and a soft palatal lifting device in eight patients with severe OSAHS (Barthlen et al., 2000). The AHI significantly decreased compared with baseline values with MRA treatment, whereas it did not with the Tongue-retaining Device or the soft palatal lifting device. Moreover, the success of the latter two appliances was seriously compromised by poor patient tolerance. Two other trials compared a one-piece MRA (OSA-Monobloc) with a two-piece MRA (OSA-Herbst) and two other two-piece Mandibular Repositioning Appliances (Karwetzky activator vs. Silensor®), respectively (Bloch et al., 2000; Rose et al., 2002a). In both studies, the amount of mandibular protrusion was identical with either appliance. Although no significant difference in the AHI could be demonstrated when the one- and two-piece MRAs were compared (Bloch et al., 2000), the Karwetzky activator was reported to be significantly more effective with respect to the AHI (Rose et al., 2002a). Both studies could not demonstrate a significant difference between the devices with respect to improvements in oxygen saturation parameters. Moreover, no significant differences in the arousal index, snoring frequency, percentage of slow-wave sleep, or ESS were demonstrated between the Monobloc and Herbst appliances (Bloch et al., 2000). Patient-perceived relief of symptoms and snoring was slightly better with the Monobloc appliance (Bloch et al., 2000), whereas, in this respect, no difference was observed between the Karwetzky activator and the Silensor® (Rose et al., 2002a). Although the prevalence of side-effects was equal with the Herbst and Monobloc appliances, the majority of patients preferred Monobloc treatment (Bloch et al., 2000). Side-effects were more frequent with the Karwetzky appliance, but the majority of patients preferred it to the Silensor® (Rose et al., 2002a).
The effect size of the AHI of one trial was not calculated due to inadequate overall methodological quality (Barthlen et al., 2000). Furthermore, effect size of the ESS could not be calculated in one trial because only median and quartile range were reported (Bloch et al., 2000). The remaining effect sizes were not pooled due to the disparities between the (control) interventions (Fig. 2). Although there was a trend toward greater success with the one-piece MRA with identical protrusion, no significant difference in effect size with respect to the AHI (effect size, −0.10; 95% CI, −0.67 to 0.46) was observed (Bloch et al., 2000). Contrary to the reported significant difference in AHI, the calculated effect size (−0.43; 95% CI, −1.07 to 0.22) did not demonstrate a significant difference between the two-piece appliances (Rose et al., 2002a).
MRA vs. UPPP
The effect of MRA treatment was compared with that of UPPP in one trial (Wilhelmsson et al., 1999). After a one-year treatment period, a significant difference in the AHI in favor of the MRA treatment was observed. However, other physiological parameters, including the hourly rate of oxygen desaturations (≥ 4%) and registered snoring time, did not differ between the two interventions. Although, after six months of treatment, subjective daytime sleepiness was less in the UPPP group, no significant difference in sleepiness was observed after a one-year treatment period. In a separate publication reporting on changes in quality of life, the UPPP group showed a greater level of contentment than the MRA-treated patients after a one-year treatment period (Walker-Engström et al., 2000). Since no other trials compared MRA therapy with UPPP, a pooled estimate could not be calculated. The effect size of the AHI demonstrated that MRA therapy was more effective than UPPP (effect size, −0.47; 95% CI, −0.91 to −0.02) (Fig. 2).
MRA vs. CPAP
Three of the included trials compared a one-piece MRA with CPAP (Ferguson et al., 1996; Engleman et al., 2002; Tan et al., 2002), whereas the other three trials used a two-piece appliance (Clark et al., 1996; Ferguson et al., 1997; Randerath et al., 2002). However, in one trial, patients were randomized to a one-piece MRA with a flexible or rigid construction (Engleman et al., 2002). Moreover, one trial replaced a one-piece appliance with a two-piece MRA halfway through the study, due to nocturnal breathing difficulties (Tan et al., 2002). Since both studies did not observe differences in efficacy as a result of appliance design, data on the different devices were pooled.
Compared with MRA treatment, CPAP resulted in a significant improvement in the AHI in five out of six trials (Clark et al., 1996; Ferguson et al., 1996, 1997; Engleman et al., 2002; Randerath et al., 2002). Although there was no significant difference in the arousal index between the interventions, snoring frequency did significantly differ in favor of CPAP (Randerath et al., 2002; Tan et al., 2002). With CPAP, the minSaO2 improved more significantly when compared with MRA treatment in three trials (Ferguson et al., 1996, 1997; Randerath et al., 2002), whereas it did not in another trial (Clark et al., 1996). Variability in the changes of other parameters of oxygen saturation during sleep was also reported. Two trials demonstrated significant improvements in the hourly rate of oxygen desaturations (SaO2 < 90%) with CPAP but not with MRA therapy (Ferguson et al., 1996, 1997), whereas another trial did not observe significant differences in oxygen desaturation intensity and duration between CPAP and MRA (Tan et al., 2002).
Although in two trials the ESS improved with both CPAP and MRA, no significant difference could be demonstrated between the interventions (Ferguson et al., 1997; Tan et al., 2002). In one trial, CPAP resulted in a more significant improvement in the ESS and the Functional Outcomes of Sleepiness Questionnaire (Engleman et al., 2002). However, the same trial could not demonstrate a significant difference between CPAP and MRA in objective sleepiness according to the Maintenance of Wakefulness Test and home portable sleep time registration. Moreover, none of the other trials found a significant difference between CPAP and MRA therapy in sleep-quality-related variables like the amount of REM or slow-wave sleep. Although mental well-being and health transition according to the Short-form 36 Health survey were significantly better with CPAP, no significant differences between the treatments were observed according to the Hospital Anxiety and Depression Scale or cognitive performance tests (Engleman et al., 2002). Three trials demonstrated more pronounced subjective improvements in OSAHS-related symptoms like snoring or sleepiness with CPAP (Ferguson et al., 1996, 1997; Engleman et al., 2002), whereas the remaining trials could not. Although the severity of adverse events was generally not different between the two interventions, one trial reported more side-effects with CPAP (Ferguson et al., 1996). Patient-reported use generally did not differ between CPAP and MRA therapy (Ferguson et al., 1996, 1997; Engleman et al., 2002). However, in one study patient-reported compliance was greater in MRA therapy (Randerath et al., 2002). Moreover, patients were generally less satisfied with CPAP and found MRA therapy easier to use (Clark et al., 1996; Ferguson et al., 1996, 1997; Randerath et al., 2002; Tan et al., 2002).
When pooling the available effect sizes, we noted a significant improvement in AHI (effect size, 0.83; 95% CI, 0.59 to 1.06) with CPAP when compared with MRA therapy. Conversely, no significant difference in the pooled effect size of the ESS (effect size, 0.32; 95% CI, −0.24 to 0.89) could be demonstrated when MRA therapy was compared with CPAP (Fig. 2).
(5.2) Co-morbidity
Using the specified criteria, we considered 14 articles related to co-morbidity as eligible. Because the journal’s permission to use the article in press was not granted (Robertson et al., in press), 13 articles were included for methodological appraisal (Fig. 1, Table 3). Eight of the included articles studied co-morbidity of MRA therapy on the craniomandibular complex (Bernhold and Bondemark, 1998; Bondemark, 1999; Pantin et al., 1999; Tegelberg et al., 1999; Bondemark and Lindman, 2000; Marklund et al., 2001b; de Almeida et al., 2002; Walker-Engström et al., 2002). Orthodontic side-effects of MRA treatment were assessed in six studies (Tegelberg et al., 1999; Pantin et al., 1999; Bondemark and Lindman, 2000; Fritsch et al., 2001; Marklund et al., 2001b; Rose et al., 2002d). Finally, in six studies dental and skeletal changes resulting from MRA therapy were assessed by means of upright cephalometry (Bondemark, 1999; Fritsch et al., 2001; Robertson, 2001, 2002; Fransson et al., 2002b; Rose et al., 2002d). In six studies, overlap with respect to baseline characteristics was noted. Two studies reported on the one- and four-year follow-up of adverse effects of MRA therapy on the stomatognathic system (Tegelberg et al., 1999; Walker-Engström et al., 2002). Two similar studies reported on the effect of MRA treatment on the craniofacial and craniomandibular complex (Bondemark, 1999; Bondemark and Lindman, 2000). Finally, dental and skeletal changes associated with MRA treatment were studied in one study and were further explored with respect to the upper facial skeleton in a second study (Robertson, 2001, 2002).
In two studies, more than one appliance design was used (Fritsch et al., 2001; Marklund et al., 2001b). The amount of mandibular protrusion with MRA treatment was comparable among the 13 included studies (Table 3). However, the durations of the follow-up periods differed among studies (range: six months to a mean follow-up of four years). Moreover, by taking the review cephalogram at six-month intervals (from 6 to 30 months), two studies tried to establish whether changes in craniofacial characteristics were progressive with continuing treatment (Robertson, 2001, 2002). Although compliance was not reported in all studies, patients generally used their MRA more than five nights per week for five hours or more per night. Except for the male-to-female ratio in two studies (Bondemark, 1999; Bondemark and Lindman, 2000), patient baseline characteristics of the 13 included studies were comparable (when reported) with respect to male-to-female ratio, age (means ranging from 46.7 to 55.3 yrs), and Body-Mass Index (means ranging from 26.3 to 29.2).
The majority of the included studies were patient series, with only one study using a concurrent control group (Marklund et al., 2001b). Despite the non-controlled design of most studies, methodological quality was generally rated as ‘adequate’. The single study that used a concurrent control group was rated as ‘good’, whereas one study was rated as ‘poor’ (Pantin et al., 1999). This was mainly due to the unclear description of materials and methods in the latter study. Inter-rater agreement on the methodological quality of each trial, according to the overall impression, was good (Agreement, 97%; Kappa, 0.65; 95% CI, 0.32 to 0.98).
Craniomandibular complex
MRA treatment did not result in significant changes in maximum mouth opening, laterotrusion, or protrusion in the short or long term in three studies (Tegelberg et al., 1999; Bondemark and Lindman, 2000; Walker-Engström et al., 2002). However, in one study, an increased mouth opening was observed in 28% of patients following a mean treatment period of 31 months (Pantin et al., 1999). Except for individual patients, no significant changes in pain on movement or palpation of the temporomandibular joints and masticatory muscles were detected (Tegelberg et al., 1999; Bondemark and Lindman, 2000). Moreover, changes in joint function as a result of treatment were generally minor (Pantin et al., 1999; Bondemark and Lindman, 2000; Walker-Engström et al., 2002). When changes in these clinical parameters were quantified according to the Helkimo clinical dysfunction index or score, minor and insignificant changes in craniomandibular status were observed (Bernhold and Bondemark, 1998; Tegelberg et al., 1999; Bondemark and Lindman, 2000). Moreover, no changes in the relation between centric occlusion and centric relation could be demonstrated (i.e., no ‘dual bite’) (Bondemark, 1999; Marklund et al., 2001b). Finally, in six out of seven patients, MRI studies of the temporomandibular joint and masticatory muscles did not reveal any changes in function and morphology as a result of MRA therapy after a mean treatment period of one year (de Almeida et al., 2002).
Craniofacial complex
In five studies, plaster cast measurements demonstrated significant decreases in dental overbite and overjet as a result of MRA treatment (Pantin et al., 1999; Bondemark and Lindman, 2000; Fritsch et al., 2001; Marklund et al., 2001b; Rose et al., 2002d). Although patient follow-up was shorter, clinical examination in one study could not demonstrate significant changes in dental occlusion (Tegelberg et al., 1999). In four studies, long-term MRA therapy resulted in a mesial shift of the mandibular first molars relative to the maxillary first molars (mesial shift intermolar relationship) (Bondemark and Lindman, 2000; Fritsch et al., 2001; Marklund et al., 2001b; Rose et al., 2002d). In one of these four studies, the changes were accompanied by a posterior open bite in 26% of patients and a significant reduction in anterior mandibular crowding (Rose et al., 2002d). Although transverse measurements demonstrated a significant decrease in maxillary inter-canine width in MRA users compared with controls (Marklund et al., 2001b), no significant inter-arch changes could be demonstrated in another study (Rose et al., 2002d). The proportion of patients with occlusal changes tended to increase with length of MRA use in up to two years of treatment (Pantin et al., 1999). More than half of the patients with occlusal changes in this latter study were not aware of the changes. No correlation could be demonstrated between orthodontic side-effects and the amount of protrusion, treatment duration, age, gender, or (skeletal) dentofacial pattern (Pantin et al., 1999; Fritsch et al., 2001; Rose et al., 2002d). A correlation was demonstrated between patients’ impressions of tooth movement and a mesial shift in intermolar relationship (Fritsch et al., 2001).
Three cephalometric studies confirmed the results from plaster cast measurements by demonstrating a decreased dental overbite and overjet (Bondemark, 1999; Robertson, 2001; Rose et al., 2002d). Whereas some studies did not observe changes in upper incisor inclination (Bondemark, 1999; Fransson et al., 2002b), others demonstrated a more lingual inclination of the maxillary incisors following MRA therapy (Fritsch et al., 2001; Robertson, 2001; Rose et al., 2002d). In addition, some studies did not observe any changes in lower incisor inclination (Bondemark, 1999; Fritsch et al., 2001), whereas others demonstrated a more labial inclination of the mandibular incisors after MRA treatment (Robertson, 2001; Fransson et al., 2002b; Rose et al., 2002d). Variable results were also reported with respect to changes in mandibular position. As a result of treatment, posterior rotation of the mandible in relation to the skull base was observed in two studies (Fritsch et al., 2001; Fransson et al., 2002b), whereas others did not observe a change in mandibular position (Rose et al., 2002d). In a third study, a relatively forward and downward change in mandibular position, accomplished by an increased mandibular length, was observed following MRA treatment (Bondemark, 1999). Although changes in mandibular posture in the latter study were suggested to result from condylar or glenoid fossa remodeling, more recent studies demonstrating changes in condylar vertical position following mandibular advancement suggest that alterations in mandibular position are causal for changes in mandibular posture (Robertson, 2001, 2002). Changes in anterior face height, mainly resulting from an increased lower anterior face height, were demonstrated in two studies (Robertson, 2001; Fransson et al., 2002b). Although similar changes in posterior face height were demonstrated (Robertson, 2001), these were not reported uniformly (Fransson et al., 2002b). No correlation could be demonstrated between dental side-effects and the amount of mandibular protrusion, treatment duration, patient-perceived side-effects, age, gender, or (skeletal) dentofacial pattern (Bondemark, 1999; Rose et al., 2002d; Fritsch et al., 2001). A correlation was demonstrated between treatment duration and changes in mandibular posture relative to the skull base (Fritsch et al., 2001).
(6) Discussion
Systematic review of the available literature regarding efficacy and co-morbidity of Oral Appliances in the treatment of OSAHS indicates that OA therapy is a viable treatment modality in the adult patient with OSAHS, although CPAP is apparently more effective, and adverse effects of OA treatment have been described. However, definite conclusions with respect to the precise indications of Oral Appliances in the management of OSAHS cannot be drawn. Moreover, the evidence base regarding the co-morbidity of Oral Appliance therapy is generally obscured by methodological limitations of the available literature. Therefore, a discussion of our findings seems appropriate.
When compared with a control device, MRA therapy was clearly more effective in improving the AHI and other physiological indicators. Superior results of MRA treatment with respect to objective daytime sleepiness, patient compliance, and patient satisfaction support the efficacy of MRA therapy in OSAHS. In contrast to these favorable results, the effect of MRA therapy on sleep architecture varied among different reports. However, in the treatment with CPAP, non-significant differences in sleep quality have also been observed when compared with a placebo intervention (Loredo et al., 1999). Variable results in improvements of subjective parameters like the Epworth Sleepiness Scale and reported snoring suggest a placebo effect of Oral Appliances. However, these findings may also be attributed to factors other than mandibular advancement, such as stimulation of neuromuscular reflexes and changes in the bite relationship, in both MRA and control treatment (Mehta et al., 2001). Future studies using a ‘true placebo’ rather than an intra-oral control device may further elucidate the possible placebo effect of OA therapy.
Outcomes of variability in mandibular advancement and bite opening suggest that MRA therapy derives its therapeutic efficacy mainly from the amount of mandibular protrusion imposed by the appliance. Studies observing higher MRA success rates in patients with a greater mandibular protrusion capacity support this suggestion (Marklund et al., 1998a). Moreover, other studies on progressive mandibular advancement observed a similar ‘protrusion-dependent’ effect in MRA treatment (Raphaelson et al., 1998; Kato et al., 2000). However, in some patients bite opening also had a favorable effect on the treatment outcome (Raphaelson et al., 1998). Moreover, in some OSAHS patients, the number of upper-airway obstructions may even increase when the mandible is protruded toward its maximum (Lamont et al., 1998; Loube, 1998). These findings suggest that the optimum in mandibular protrusion in MRA therapy is not always equal to the maximal mandibular protrusion. Although a relationship between the degree of mandibular advancement and the therapeutic efficacy of an MRA seems evident, shortcomings in the available literature and conflicting data do not allow for definite conclusions to be drawn. Variability in MRA bite opening appears to be of no consequence on both physiological and subjective parameters. Therefore, the controversy persists regarding the amount of bite opening indicated with MRA treatment (George, 2001). However, patient preference may be an argument to keep the bite opening in MRA therapy to a minimum.
With respect to both physiological parameters and patient acceptance, MRA therapy proved superior to other types of Oral Appliances in the management of OSAHS. These findings correspond to results of a similar study in snoring patients in which MRA therapy was compared with a palatal lifting device and a mouth shield (Marklund and Franklin, 1996). When compared with an MRA, employability of a Tongue-retaining Device is probably poorer due to clinical limitations and inferior patient acceptance (Cartwright et al., 1991; Barthlen et al., 2000). However, unlike most MRAs, Tongue-retaining Appliances have been reported to be suitable for the edentulous patient as well (Kingshott et al., 2002). Similar to variability in bite opening, MRA design (i.e., one-piece or two-piece) had no serious consequences on the physiological outcomes. Moreover, MRA design generally did not affect patient-perceived symptomatology. These observations correspond to the findings of a review of 21 publications on Oral Appliance therapy (Schmidt-Nowara et al., 1995). In their review, Schmidt-Nowara et al. concluded that, despite considerable variations in appliance design, clinical effects of Oral Appliances are remarkably consistent. However, it should be noted that appliance design may influence therapeutic efficacy by affecting patient preference or patient-perceived symptomatology. The precise benefits of specific features in MRA design, such as adjustable mandibular advancement and freedom of mandibular movement, need to be further elucidated.
The one-year follow-up of patients treated with a MRA or UPPP suggests that the former should be preferred in the treatment of mild to moderate OSAHS. Although success rates of both interventions showed a tendency to decrease over a four-year period, MRA therapy remained more successful than UPPP (Walker-Engström et al., 2002). The additional value of MRA therapy is confirmed by another study which suggested MRA treatment as an adjuvant therapy following unsuccessful UPPP (Millman et al., 1998). However, it should be noted that after a one-year treatment period, UPPP-treated patients generally showed a greater level of contentment than MRA-treated patients (Walker-Engström et al., 2000). Moreover, the number of drop-outs in the MRA group at the four-year follow-up limits definitive conclusions with respect to the long-term results of these interventions in OSAHS.
Results of crossover trials comparing MRA therapy with CPAP indicate that, when physiological outcomes like the AHI are considered, CPAP should be preferred over MRA therapy. The superiority of CPAP is confirmed in another study showing poor patient tolerance to MRA therapy in patients already on CPAP (Smith and Stradling, 2002). However, patient acceptance of the MRA in the latter study could have been affected by negative expectations and specific appliance design. It is suggested that MRA treatment is generally more successful in patients with mild to moderate OSAHS (Marklund et al., 1998a; Johnston et al., 2002). In their crossover study, Engleman et al.(2002) performed a subgroup analysis on treatment efficacy of MRA therapy in patients with mild OSAHS. Although, in this subgroup, changes in AHI did not significantly differ between CPAP and MRA therapy, efficacy and subjective parameters like patient satisfaction and sleepiness were better with CPAP. Contrary to these findings, other included trials did not demonstrate a significant difference in most subjective parameters between CPAP and MRA treatment. Moreover, changes in sleep quality, like the amount of REM or slow-wave sleep, did not differ between the interventions. Because some suggest MRA therapy as first-line treatment in mild to moderate OSAHS (Ferguson et al., 1997; Tan et al., 2002), and others obtain superior results with CPAP in this respect (Engleman et al., 2002; Randerath et al., 2002), the precise indication for MRA therapy in OSAHS management requires further study. Although the non-significant change in subjective sleepiness according to the Epworth sleepiness scale may be related to a placebo effect of OA therapy, a clear patient preference for MRA therapy indicates that CPAP should not be considered ideal in the management of OSAHS.
While the included trials related to efficacy were generally of adequate quality, some general aspects should be taken into consideration. The reported success percentage of MRA therapy in the treatment of OSAHS ranged from 30 to 81% (Table 2). However, among other factors such as amount of mandibular advancement and specific MRA design, these figures probably reflect bias due to the various definitions of treatment success. If different studies of MRA therapy in OSAHS are to be compared, a uniform definition of treatment success is clearly indicated. We suggest defining success as a correction of the RDI to physiological levels (i.e., RDI < 5). A partial response to treatment may be defined as a satisfactory improvement of symptoms combined with a 50% or greater reduction in the RDI. In addition, since this is associated with an increased mortality, the post-treatment RDI in partial responders should not exceed 20 (He et al., 1988). Finally, patients not meeting these criteria or patients unable to use the MRA are defined as treatment and compliance failures, respectively. In some trials, external validity may have been compromised due to the inclusion of patients of generally mild to moderate severity (Ferguson et al., 1996, 1997; Wilhelmsson et al., 1999; Bloch et al., 2000; de Almeida et al., 2002; Pitsis et al., 2002; Randerath et al., 2002; Rose et al., 2002a; Tan et al., 2002). Moreover, in three trials, bias may have been incorporated due to the inclusion of patients who failed or refused CPAP (Barthlen et al., 2000; Bloch et al., 2000; Rose et al., 2002a). Most of the included studies used a crossover design. Although this offers the advantage of efficiency and within-subject comparison, a crossover design may incorporate deficiencies that compromise study validity (Woods et al., 1989). For example, a ‘carry-over of treatment effect’ in crossover studies could result in a treatment-period interaction, thereby yielding a biased estimate of the treatment effect. Moreover, since blinding of patients to treatment is generally not possible, psychological influences cannot be overlooked when treatment efficacy is evaluated in a crossover study. To preclude these methodological deficits, especially when MRA is compared with CPAP, future randomized trials in MRA treatment should preferably be of a parallel-group design. Moreover, a parallel design allows for easier long-term follow-up and determination of the precise indication of MRA therapy in OSAHS management.
Adverse effects of MRA therapy on the craniomandibular complex appear to be limited. In the clinical situation, signs or symptoms of temporomandibular disorders that result from MRA therapy are also not commonly reported (Bonham et al., 1988; Wilhelmsson et al., 1999). It has been suggested that long-term evaluation of MRA therapy is needed to monitor any changes in craniomandibular status (Bonham et al., 1988). However, a four-year follow-up period of MRA treatment demonstrated only a few adverse effects on the stomatognathic system (Walker-Engström et al., 2002). It has been reported that serious adverse effects on the craniomandibular complex are the chief reasons for patients discontinuing MRA therapy (Pantin et al., 1999; Rose et al., 2002a). Conversely, orthodontic side-effects were observed more frequently in MRA treatment. Although generally minor, a decreased dental overbite and overjet, accompanied by a mesial shift in intermolar relationship, were reported most uniformly. Patient-perceived changes in occlusion may be of additional value in detecting changes in dental occlusion. Other adverse events on dental occlusion, such as a posterior open bite or reduced anterior mandibular crowding, may accompany these changes. The observed changes in dental occlusion are confirmed by cephalometric studies that also reported changes in overbite and overjet. Although not reported uniformly, cephalometric studies also suggest changes in incisor inclination as a result of MRA therapy. These changes may be attributed to a labially directed force against the mandibular incisors and a lingually directed force against the maxillary incisors as the mandible attempts to return to a less constrained position (Rose et al., 2002d). The reported effect of long-term mandibular advancement on mandibular position varies. Since skeletal alterations resulting from mandibular advancement are generally minimal in adult individuals (Pancherz, 1985), changes in dental occlusion are most likely to result from a dento-alveolar effect of MRA therapy. Moreover, the observed shift in occlusion may be attributed to myostatic contracture of the lateral pterygoid muscle and failure of the mandibular condyles to reposition fully following full-night mandibular protrusion (Pantin et al., 1999; George, 2001). This latter phenomenon may also explain the observed changes in anterior face height and condylar vertical position in the cephalometric studies. Rigidity and amount of dental coverage have been implicated in the occurrence of adverse events in MRA treatment. It has been suggested that MRAs with full dental coverage, and both soft elastomeric and rigid acrylic appliances, minimize the chance of occlusal changes (Bondemark, 1999; Marklund et al., 2001b; George, 2001). However, others suggest that occlusal side-effects in MRA treatment are not design-related (Pantin et al., 1999; Rose et al., 2002d). Analysis of the available data suggests that dental and skeletal changes in MRA therapy may progress by becoming more prominent over time (Pantin et al., 1999; Robertson, 2001; Fritsch et al., 2002). Skeletal changes, most likely related to repositioning of the mandibular condyles, have been demonstrated to occur soon after the onset of treatment, whereas dental changes appear to develop as treatment continues (Robertson, 2001). If there is a good patient follow-up, it is thought reasonable to persist with MRA treatment in the presence of acceptable and non-progressive side-effects (Pantin et al., 1999; Rose et al., 2002d).
On the basis of the available literature, it appears that adverse effects of MRA therapy generally involve changes in dental occlusion. Since these changes appear to originate over longer treatment periods, they may go unnoticed by patients (Pantin et al., 1999). Most studies included in this review with respect to co-morbidity of MRA therapy did not use a control group and recruited a non-homogenous group of patients. Moreover, the use of various different appliances may obscure the findings. Despite these limitations, other methodological aspects of the included studies were generally of adequate quality. However, definite conclusions with respect to the adverse effects of long-term MRA therapy on the craniomandibular and craniofacial complex are not possible. Controlled studies are warranted addressing long-term co-morbidity of MRA therapy. Moreover, controlled studies should address the specific effects of MRA design, degree of mandibular protrusion, and treatment duration on the occurrence and progression of adverse effects in MRA treatment.
(7) Summarizing and Concluding Remarks
Randomized trials offer an evidence base for the use of Oral Appliances in the treatment of, especially, mild to moderate OSAHS. Oral appliances are effective in the treatment of OSAHS, although a placebo effect should be considered. MRA therapy generally yields results superior to those achieved with other types of Oral Appliances in the treatment of OSAHS. Although definite conclusions are not possible, efficacy of MRA treatment appears to be related to the degree of mandibular advancement. Moreover, appliance design, like the amount of bite opening or the means of mandibular fixation, may affect subjective parameters of success. Although short-term results indicate that MRA therapy should be preferred to UPPP, definite conclusions cannot be drawn. Superior results with respect to physiological outcomes indicate that CPAP should be preferred to MRA treatment. However, a clear patient preference for MRA therapy indicates that CPAP should not be considered ideal in the treatment of OSAHS. To optimize methodological quality, future randomized trials in MRA treatment should incorporate a parallel-group design, one which may clarify the specific indication of MRA therapy in OSAHS management. Moreover, important outcomes of MRA therapy—like long-term efficacy, performance, cardiovascular status, and objective compliance—should be compared with other treatment modalities like CPAP. To compare different trials in MRA therapy, investigators much reach a consensus on the definition of treatment success. MRA therapy may result in adverse—although generally not serious—effects on the craniomandibular and craniofacial complex. Adverse effects on the craniofacial complex appear most often in MRA treatment and generally involve changes in dental occlusion. However, the lack of controlled studies related to co-morbidity preclude any definite conclusions. Controlled studies are needed to address the long-term adverse effects of MRA therapy. Although it cannot be excluded that efficacy or co-morbidity of Oral Appliance therapy is influenced by the individual appliance design, it can be concluded that MRA therapy is a viable treatment modality for OSAHS. Similar to treatment with CPAP, MRA therapy is usually a life-long requirement in the management of OSAHS. Physicians with experience in the field of sleep-disordered breathing must supervise treatment with Mandibular Repositioning Appliances. To guarantee long-term efficacy and safety, follow-up should be conducted on a regular basis.
Algorithm of study selection procedure. OSAHS, Obstructive Sleep Apnea-Hypopnea Syndrome; MRA, Mandibular Repositioning Appliance; AHI, Apnea-Hypopnea Index; RDI, Respiratory Disturbance Index.
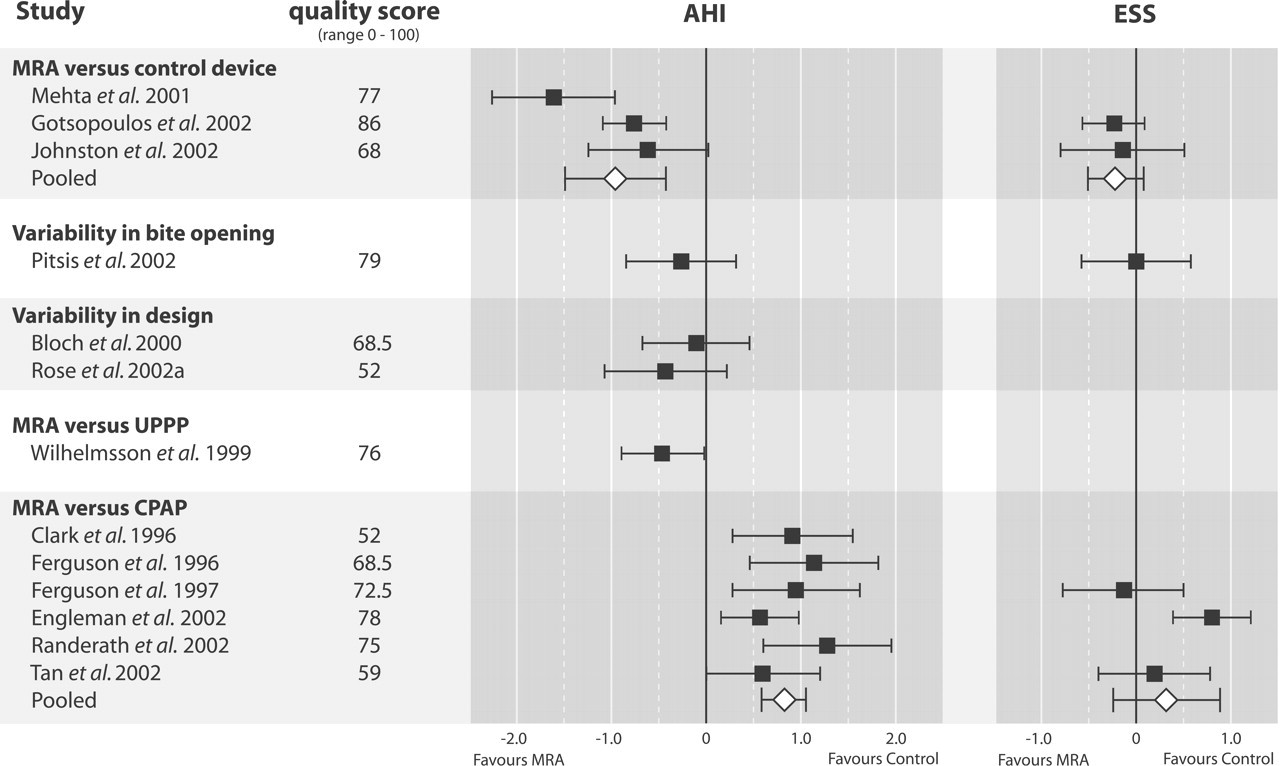
Effect sizes with approximate 95% confidence intervals of methodologically sound trials related to efficacy. AHI, Apnea-Hypopnea Index; ESS, Epworth Sleepiness Scale; MRA, Mandibular Repositioning Appliance; UPPP, uvulopalatopharyngoplasty; CPAP, Continuous Positive Airway Pressure.
Footnotes
Acknowledgements
The authors thank Ms. I.I. Riphagen from the Groningen University medical library for her assistance in the elaboration of our search strategy, and Mr. B.K. Uildriks for his efforts in the graphics lay-out. The authors also thank Drs. F. de Vries and J. Schortinghuis for their critical appraisal of the manuscript. This systematic review was written in partial fulfilment of the requirements for a PhD degree. Financial support for this MD-clinical research traineeship was granted by the Netherlands Organisation for Health Research and Development.
