Abstract
Oral fungal infections (mycoses) have come into particular prominence since the advent of infection with Human Immunodeficiency Virus (HIV), and recognition of the Acquired Immune Deficiency Syndrome (AIDS), as well as the phenomenal increase in world travel with increased exposure to infections endemic in the tropics. Paracoccidioidomycosis is a rare mycosis worldwide but common in Brazil and some other areas in Latin America. It can be life-threatening and can manifest with a spectrum of clinical presentations, including frequent oral lesions. This paper reviews the more recent information on Paracoccidioidomycosis, emphasizing those areas most relevant in dental science.
Introduction
Oral fungal infections (mycoses) have come into particular prominence since the advent of the Acquired Immune Deficiency Syndrome (AIDS), but concurrently, increasing globalization and world travel have increased exposure to deep mycoses endemic, mainly in the tropics. Candidosis is the most common oral mycosis worldwide, but others must be considered, particularly in immunocompromised patients such as those infected with Human Immunodeficiency Virus (HIV). Paracoccidioidomycosis is a rare infection worldwide but a common deep mycosis in Brazil and some other endemic areas in Latin America. It is often a subclinical infection but can be life-threatening and can manifest with a wide spectrum of clinical presentations, frequently involving the mouth. In many cases, the first and main clinical manifestations are oral lesions, and the dental surgeon can play an important role in diagnosis and treatment. Imported paracoccidioidomycosis must also be considered in non-endemic areas, because it can easily be misdiagnosed and therefore incorrectly treated (Ajello and Polonelli, 1985).
The topic has been reviewed elsewhere (Lacaz and Rosa, 1979; Del Negro et al., 1982; Franco et al., 1994). This review considers the more recent information on the general and oral aspects of paracoccidioidomycosis.
Mycology
Paracoccidioidomycosis (Pmycosis, formerly termed Lutz’ disease) is caused by Paracoccidioides brasiliensis, a fungus found in the soil of certain areas of Latin America, from Mexico in the north to Argentina in the south, but mainly between latitudes 23° to 34°, and its precise natural history is incompletely understood (Restrepo et al., 2001). P. brasiliensis is a dimorphic fungus, but in humans presents only in the yeast form, usually from 2 to 10 μm in diameter, though cells up to 30 μm or more are also common. The principal characteristic in its parasitic state is the formation of new yeast by evagination of the mother cell wall, resembling a helm. The image in scanning electron microscopy is compared with a “grenade”. Temperature is the key factor for yeast (37°C) or mycelial (25°C) expressions. The fungus is a eukaryote, with one or more nuclei and nucleoli. The cytoplasm contains vacuoles, endoplasmic reticulum, and ribosomes, but Golgi apparatus and lysosomes have not been found. Mitochondria are numerous in young cells, mainly in the periphery, and vacuoles increase with age. The cells have a double refractile wall form 0.2 to 1 μm thick. The cell wall shows two layers. The external layer is electron-dense, in the yeast phase formed by α-1-3-glucan and in the mycelium by β-1-3 glucan. The internal layer of the cell wall is electron-lucent, thick, formed by chitin and juxtaposed to the cytoplasmic membrane.
The size of the fungus depends on the phase of its development. The small forms are frequent when proliferation is fast. In vitro at 25°C, the fungus is seen in the mycelial form, while at 37°C it changes into the yeast. Pathogenicity and invasiveness seem to be associated with α-1-3 glucan, in addition to proteases such as Gp43 (Mendes-Giannini et al., 1990). Glucans and chitin of the cell wall are not antigenic: Gp43, a 43-kDa glycoprotein of the fungus wall, secreted extracellularly, is the main known antigen (Puccia et al., 1986). By molecular techniques, P. brasiliensis was recently classified in the phylum Ascomycota, order Onygenales, and family Onygenaceae. Traditionally, it was included in the phylum Deuteromycota, class Hyphomycetes (San-Blas et al., 2002) [Taxonomy of P. brasiliensis: Kingdom, Fungi; Phylum, Ascomycota; Order, Onygenales; Family, Onygenaceae; Genus, Paracoccidoides; and Species, brasiliensis].
Epidemiology
Lutz first described the disease in 1908 in two patients from the Sta Casa of São Paulo state, Brazil, with oral lesions and cervical lymphadenopathy. In 1912, Splendore called the fungus isolated by Lutz Zymonema brasiliensis. In 1930, Floriano Almeida demonstrated that the fungus was distinct from Coccidioides immitis, and named the genus Paracoccidioides brasiliensis.
In South America, most cases occur in Brazil, with São Paulo, Paraná, Rio Grande do Sul, Goiás, and Rio de Janeiro the most prevalent areas (Franco et al., 1989), but it is also seen in other countries, particularly in Colombia and Venezuela and also in Chile, Guayana, French Guayana, and Surinam (Londero and Del Negro, 1986; Brummer et al., 1993). Interestingly, Pmycosis is rare both in the tropical rain forests of the Amazon and in the dry Northeast of Brazil. Nevertheless, 17 cases of pulmonary Pmycosis were reported from the Amazon area during the period of 1992–1994, and it was the most common pulmonary mycosis in that region (Ferreira et al., 1995).
In Central America, cases have been described in all countries except Belize and Nicaragua (Londero and Del Negro, 1986; Brummer et al., 1993).
Up to 1990, 49 non-indigenous cases of Pmycosis have been reported, mostly in Europe and the USA (Ajello and Polonelli, 1985; Lazow et al., 1990). These have been in travelers who have visited Latin America.
Pathogenesis
Cases of Pmycosis diagnosed clinically represent only a small proportion of the individuals infected. Subclinical infections (detected by skin tests with paracoccidioidin antigen) occur in up to 60% of the population in endemic areas (Bagatin, 1986; Franco et al., 1994). Overall, in countries with endemic regions, such as Brazil, it is estimated that 10% of the population is infected, but the incidence of the disease is 1–3 per 100,000 (Marques et al., 1983). Ten million individuals are estimated to be infected with P. brasiliensis, and 2% may present with the disease (McEwen et al., 1995). Therefore, it is evident that humans are fairly resistant to infection with P. brasiliensis: Hormonal, genetic, immunological, and nutritional factors must play a role in the development of the infection and clinical disease. Interestingly, there is a surprisingly low incidence of Pmycosis in HIV patients, probably explained by the higher prevalence of Pmycosis but lower prevalence of HIV in rural areas, and also by the extensive use of antifungals in HIV-infected patients.
There is no firm evidence that HLA antigens are associated with the development of Pmycosis (Dias et al., 2000). However, immune factors are important. There is a correlation of T-cell depression with the severity of Pmycosis. Patients with paracoccidioidomycosis show a decreased cellular immune response, with a decreased CD4+/CD8+ ratio in blood, possibly mediated by increased IL-4 production. In fact, patients with the acute juvenile form of Pmycosis show impaired response to PbAg, and a Th2 response, i.e., lower IFN-gamma and higher levels of Il-4, IL-5, and IL-10 (Marques Mello et al., 2002; Oliveira et al., 2002). Immune complexes are detected in serum, and there is no evidence of deficiency in antibody production (Teixeira et al., 2001). As in infections such as leprosy, immune responses can vary and can influence the natural history of the associated lesions. The immune responses of patients with Pmycosis can be either hyperergic or anergic. The hyperergic type shows a benign course, with preserved cellular immunity, organized epithelioid granulomas, low antibody titers, and a positive paracoccidioidin skin test. The anergic type is characterized by generalized systemic lesions rich in fungi, decreased cellular immunity, high antibody titers, and a negative paracoccidioidin test. Therefore, skin tests are unreliable for diagnosis but can be useful for follow-up treatment.
Female hormones (estrogen) inhibit the in vitro transformation of the filamentous phase to yeasts (Salazar et al., 1988; Aristizabal et al., 1998). This is considered the main reason for the low incidence in adult females. It is postulated that after menopause, women are more susceptible to the disease. A 60-kDa glycoprotein has been identified as the receptor for estradiol on the cell membrane of P. brasiliensis (Loose et al., 1983).
Since the disease is polymorphic, various classifications were used until in 1986, when a new classification was proposed (Franco et al., 1987), outlined as follows:
Infection—healthy individuals, both genders, positive cutaneous test. Disease
Acute or subacute (juvenile)—youngsters of both genders, with
involvement of the mononuclear phagocytic system, depressed cellular
immunity, and increased antibodies. Chronic (adult)—mainly adult males, commonly affecting lungs, lymph
nodes, and oropharynx. Subclassified into mild, moderate, and severe
forms.
(b1) unifocal (b2) multifocal Residual (sequelae)
Transmission
In soil, the fungus P. brasiliensis grows as a mycelium. Conidia, formed from the mycelium when nutrition and water are poor, are mononucleated structures. Man seems to be susceptible to infection with conidia, and is the only host susceptible to the disease, although nine-banded armadillos can harbor the fungus. There is speculation that armadillos can be an important reservoir of P. brasiliensis and that transmission is perhaps via armadillo feces in the soil (Bagagli, 1998). The natural habitat is not yet established, but the most favorable characteristics of the soil are described as humid, rich in organic materials, without drastic temperature variations (around 20°C), and covered by vegetation (Franco et al., 2000). There are some reports of isolation of P. brasiliensis from soil, particularly in areas in Brazil cultivated for coffee (Silva-Vergara et al., 1998). Inhalation of the conidium form of the fungus and transformation into a yeast in the tissues, with a primary infection of the lungs and dissemination via lymphatic and blood vessels, are the most usual mechanism of infection. There is no evidence of human-to-human transmission of P. brasiliensis.
Clinical Aspects
When Pmycosis was first described, it was almost uniformly fatal. The disease was, at that time, called Lutz disease, Lutz-Splendore-Almeida disease, Blastomicose brasileira, and later, Blastomicose Sul-americana. Then, in 1971, the term Paracoccidioidomycosis (Pmycosis) became accepted (Lacaz, 1994).
Pmycosis can manifest with mucocutaneous, lymphatic, and visceral manifestations. In endemic areas, primary pulmonary infection probably occurs in the 1st and 2nd decades of life, usually with a benign and often symptomless course. Reactivation of quiescent lesions is considered the main mechanism of adult chronic manifestations (Bethlem et al., 1999). In this context, it is important to note that, in non-endemic areas (imported Pmycosis), such as cases reported in the USA and Europe, the dormant period has been described as lasting as long as up to 60 years (Ajello and Polonelli, 1985).
The disease is classified in acute, subacute, and chronic forms (Franco et al., 1987). The acute and subacute forms affect mainly children and adolescents of both genders, rapidly and severely involving the monocytic phagocytic system (liver, spleen, lymph nodes) (Benard et al., 1994). Lung lesions are rarely present.
Patients with chronic forms are typically male, middle-aged (from 29 to 40 yrs), in a ratio of 15 males to one female. Of the 584 cases reviewed by Blotta et al.(1999), 84% were males between the ages of 40 and 50 yrs. Most of these patients were positive for serum antibodies, and although antibodies do not protect against disease, they can be useful to monitor treatment.
Most cases of Pmycosis have a chronic evolution, with granulomatous lesions mainly in the lungs but also in other tissues, such as the oral mucosa and cervical lymph nodes. In 173 patients with paracoccidioidomycosis in São Paulo, 74% had pulmonary involvement, 48% the oropharynx, 36% lymph nodes, and 13% the digestive tract (Franco et al., 1989). Affected lymph nodes become large, hard, coalescent, and sometimes fistulous. Although in some patients lung lesions are not evident, they must always be considered; for example, Restrepo et al.(1989) found that, in three patients with Pmycosis without lung involvement as shown by clinical signs, symptoms, and radiography, it was possible to isolate the fungus from the sputum of all three patients, indicating the existence of a latent primary pulmonary infection. Rarely, the patient can present an asymptomatic pulmonary lesion called a paracoccidioidoma (Santos et al., 1997).
Hematogenous dissemination of Pmycosis to abdominal lymph nodes, spleen, liver, adrenal glands, bones, skin, or brain can result in life-threatening complications. Adrenal involvement in Pmycosis is one of the main causes of hypoadrenocorticism in South America, and fine-needle aspiration can be particularly helpful in the diagnosis (Faical et al., 1996).
Eventually, the disease can manifest in the central nervous system, bones, or other tissues. Latent infections can flourish after many years (Restrepo, 2000), and relapse is a hallmark of Pmycosis.
Paracoccidioidomycosis in the Immunocompromised Person
Pmycosis has been described in immunosuppressed patients on cancer treatment and after renal transplantation (Sugar et al., 1984; Shikanai-Yasuda et al., 1995). There are reports of Pmycosis in patients using corticosteroids, azathioprine, and antineoplastic drugs (Londero et al., 1987). Some cases have been described in HIV-positive patients, and at least one person has died with acute disseminated Pmycosis (Pedro et al., 1989; Marques et al., 1995). In HIV-infected patients, the disease can present acute lymphohematogenous dissemination, but, as in the chronic form, mucocutaneous lesions can also occur (Benard and Duarte, 2000). Giovani et al.(2000) described a 29-year-old HIV-positive female patient with Pmycosis of the gingiva. It is unknown if oral Pmycosis in immunocompromised patients is related to reactivation of latent disease in the lungs, but this is possible as in other mycoses such as histoplasmosis. Surprisingly, however, Pmycosis is not as common as might be expected in immunocompromised patients (Goldani and Sugar, 1995; Marques et al., 2000).
Oral Lesions
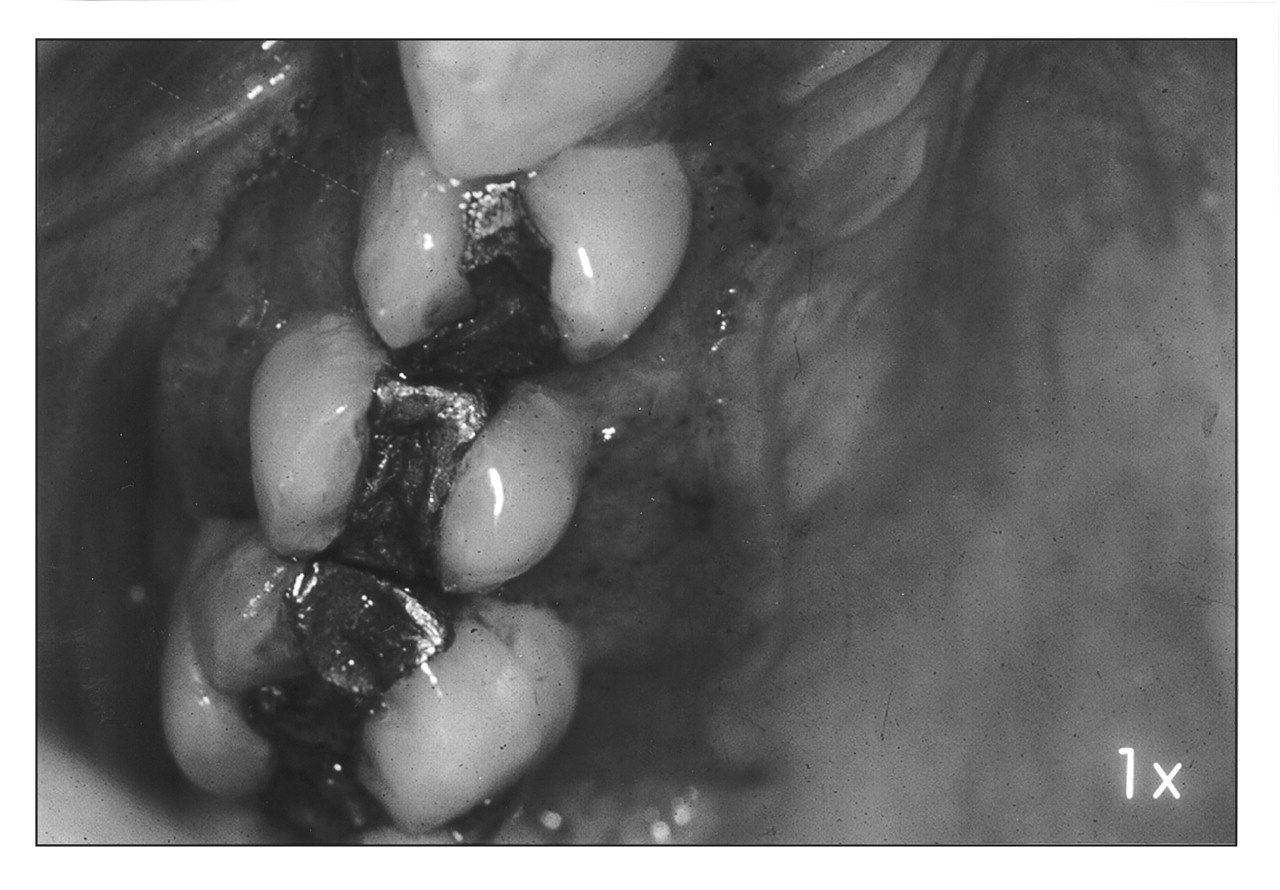
Oral involvement in Pmycosis was first described by Lutz in 1908, in two patients who also had cervical lymphadenopathy. The oral lesions typically show an erythematous finely granular hyperlasia, speckled with pinpoint hemorrhages, and a mulberry-like surface called “moriforme” stomatitis (Figs. 1, 2). Areas of ulceration are common (Almeida and Scully, 1991; Scully et al., 1998). Involvement of the lips causes a pronounced increase in thickness and consistency. The juvenile form can cause alveolar bone destruction and tooth loss (Migliari et al., 1998).
Some studies have reported the epidemiological aspects of oral Pmycosis (Sposto et al., 1993; Bicalho et al., 2001). In 173 São Paulo patients with paracoccidioidomycosis, 48% had lesions in the oropharynx, 36% in lymph nodes (Franco et al., 1989). Usually, the oral lesions are multiple, involving the lip, gingiva, buccal mucosa, palate, tongue, and floor of the mouth. Although not common, oral paracoccidioidomycosis can result in perforation of the hard palate (Castro et al., 2001).
It is well-established that most patients with oral Pmycosis are adult males with the chronic mucocutaneous form of the disease. For example, Villalba (1998), who studied 64 cases of patients with Pmycosis, found that 93% were male, with a mean age of 43 yrs, and a male:female ratio of 15:1. In 36 cases of Pmycosis with oral involvement, 89% were in males; 69% had multiple lesions in the mouth, with 78% on the gingiva, 47% on the palate, 36% the lips, and 25% the buccal mucosa (Sposto et al., 1993). Most studies show a preponderance for oral paracoccidioidomycosis to affect white patients (Almeida et al., 1991; Sposto et al., 1994; Bicalho et al., 2001). Frequent oral involvement has also been found in autopsy studies (Montenegro and Franco, 1994).
Oral lesions are usually secondary to lung involvement, though dissemination can also occur via lymphatic and blood vessels, possibly into inflamed or traumatized areas. Involvement of the mouth, epiglottis, pharynx, and larynx may cause hoarseness and dyspnea, and eventually tracheostomy may be needed due to scarring of the larynx (Do Valle et al., 1995). Cervival and submandibular lymph node involvement can be secondary to mucosal lesions, or be primary in acute forms.
Diseases that should be considered in a differential diagnosis include carcinoma, lymphoma, tuberculosis, sarcoidosis, syphilis, Wegener’s granulomatosis, granuloma inguinale, actinomycosis, histoplasmosis, cryptococcosis, blastomycosis, coccidioidomycosis, and leishmaniasis (Scully and Almeida, 1992).
Diagnosis and Histopathology
Fungal infections are typically diagnosed by culture, serology, or histopathology. Pulmonary involvement should be confirmed by imaging. However, P. brasiliensis may require weeks to grow, serology may be inconclusive, and histopathology, although more practical, does not always lead to a final diagnosis. Molecular techniques are promising to facilitate reaching a definitive diagnosis, since they have higher specificity and sensitivity, particularly with the use of DNA probes (Sandhu et al., 1997; Lindsley et al., 2001). Molecular identification of Paracoccidioides brasiliensis has been done successfully by immunohistochemistry and polymerase chain-reaction (PCR) amplification of ribosomal DNA (Motoyama et al., 2000).
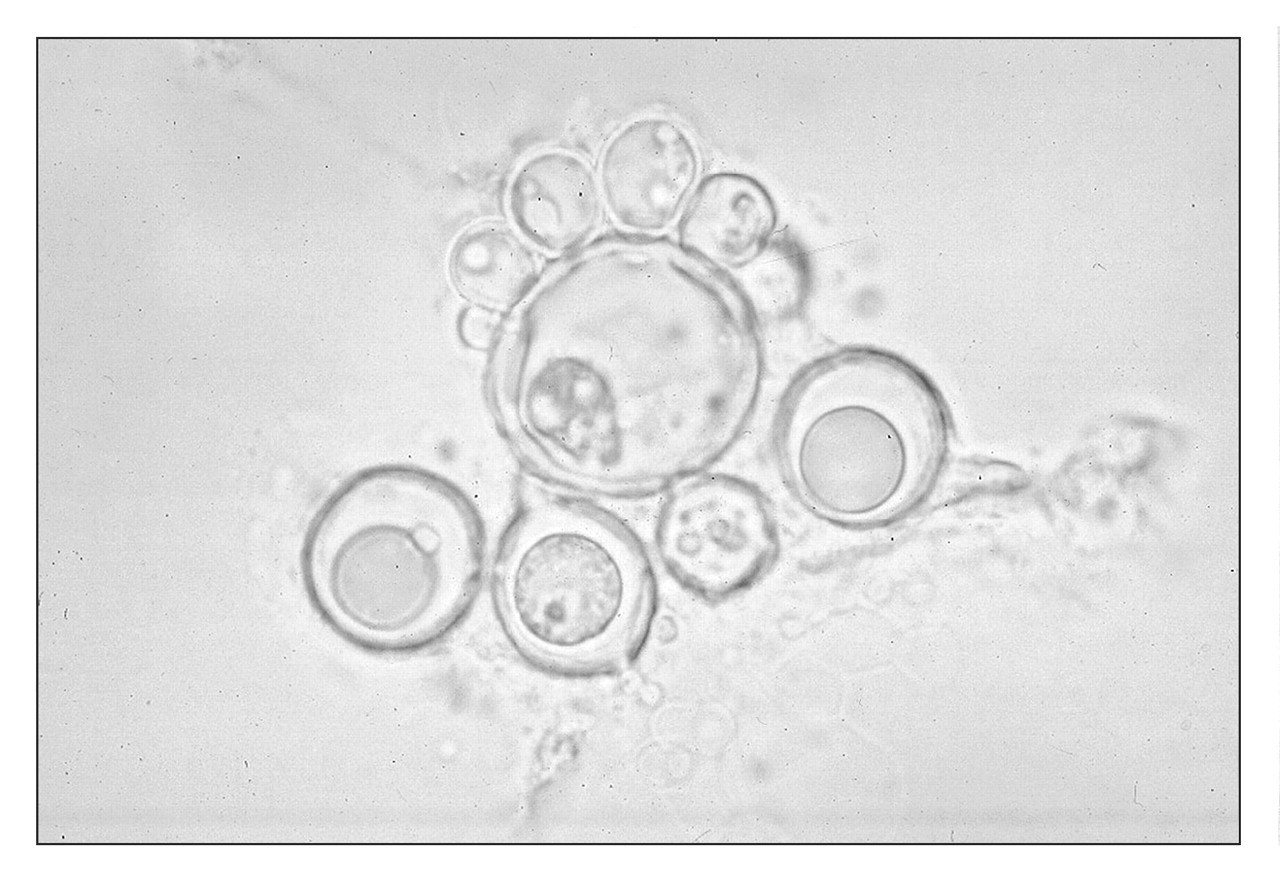
Currently, the final diagnosis of Pmycosis depends on the results of clinical evaluation and cytological diagnosis, supported by histopathology. Material scraped from the mouth can be stained with periodic acid Schiff (PAS) or placed in 10% potassium hydroxide (Fig. 3). The yeasts vary from buds 2 to 10 μm to cells up to 30 μm or more. Invariably, the fungus is found in these preparations. Cardoso et al.(2001) evaluated exfoliative cytology for the diagnosis of oral Pmycosis in 40 cases. Histologically, 28 cases out of 40 were confirmed as Pmycosis. Cytologically, nine cases out of the 28 were negative (23%), and of the 12 cases that were not histologically confirmed as Pmycosis, one was positive in the smears. Therefore, the sensitivity was 67.9% and specificity 91.7%. In another report, all ten cases studied were positive in cytologic smears (Araujo et al., 2001). The fungus has also been detected in smears and cell-block preparations of sputum (Mattos et al., 1991). In the smear preparations, the fungus when present is easily recognized, either isolated or within giant cells, as birefringent yeasts with a diameter varying from 1 to 30 μm. Polymerase chain-reaction with oligonucleotide primers of the Gp-43 antigen has recently been used to detect P. brasiliensis in sputum (Gomes et al., 2000).
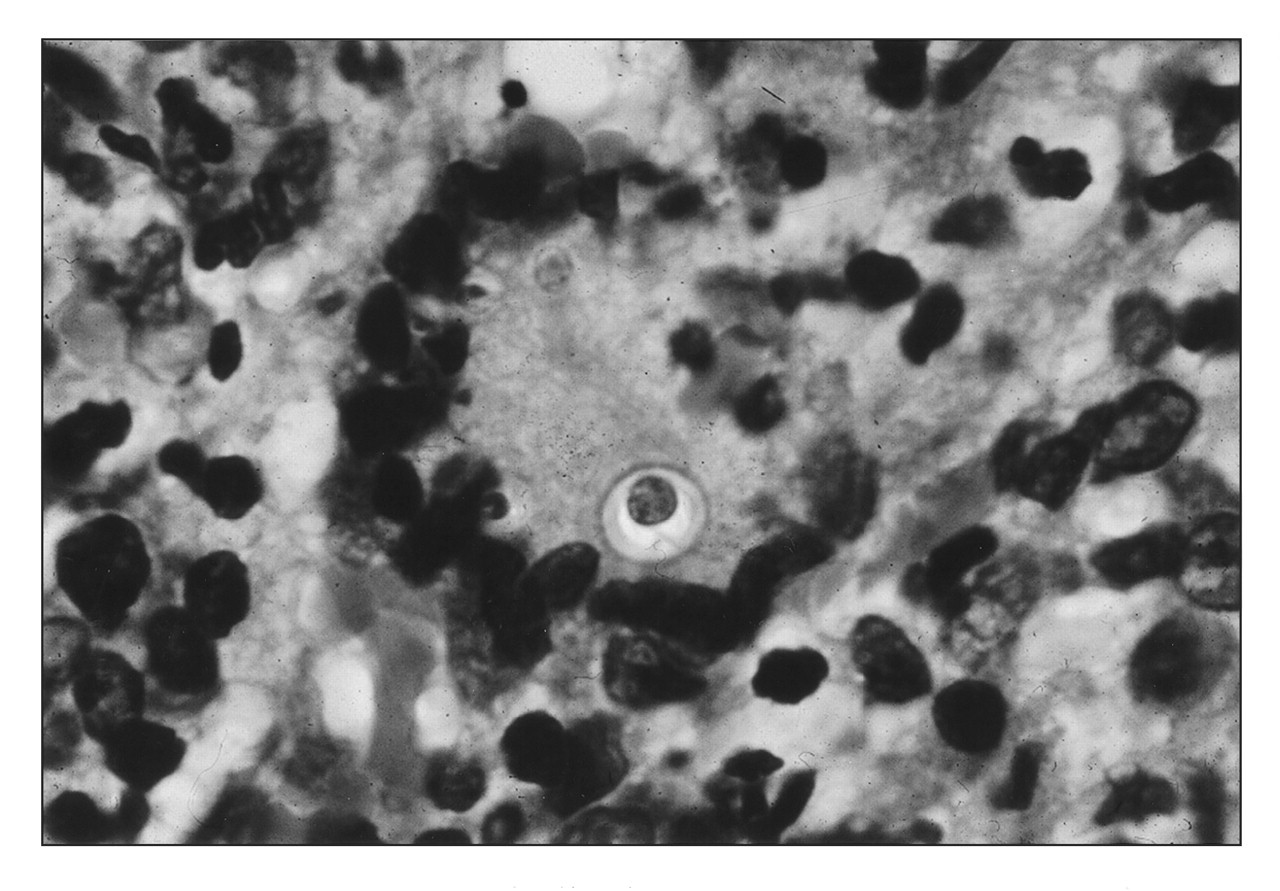
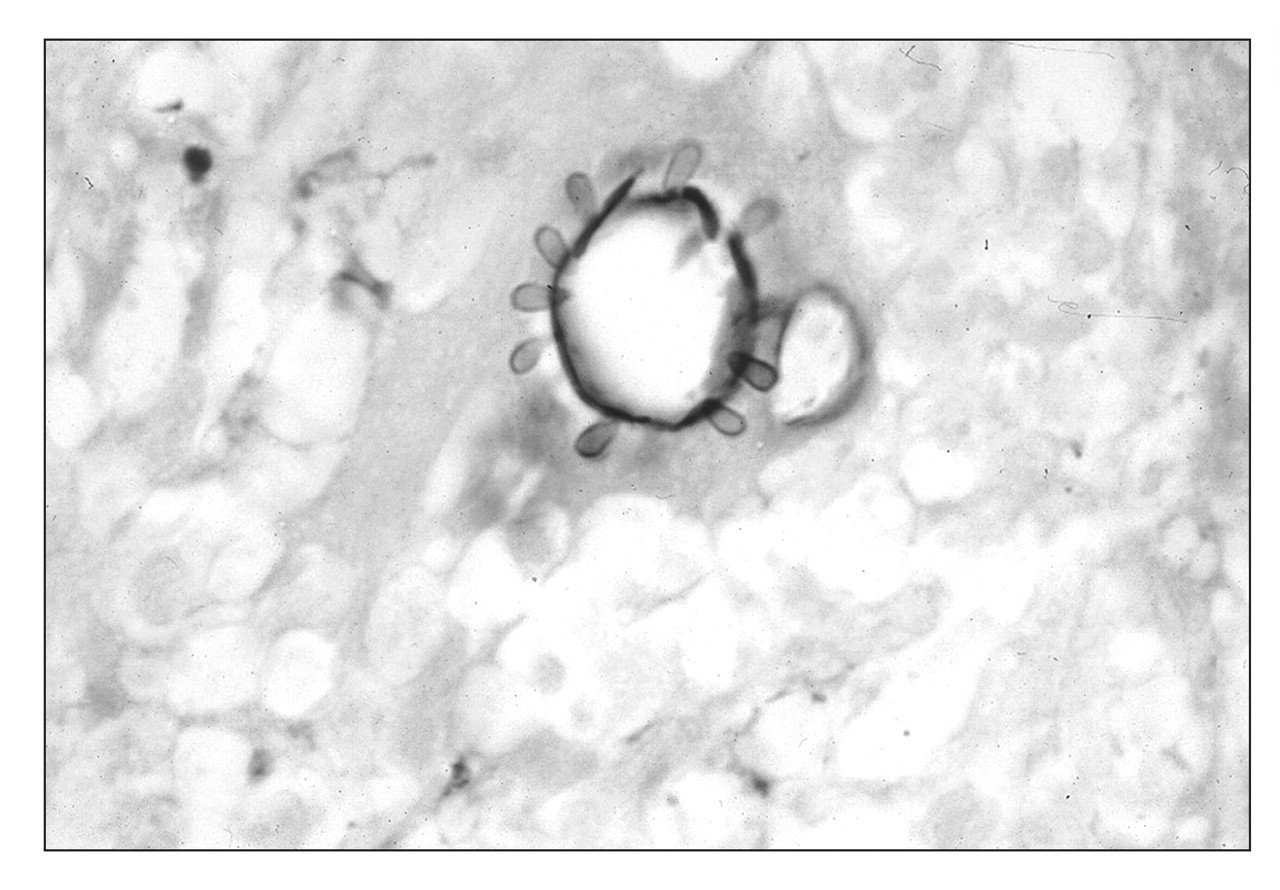
Histology shows pseudoepitheliomatous hyperplasia with intraepithelial microabscesses, and a granulomatous reaction with giant cells, interspersed with PMN, plasma cell, and an eosinophil infiltrate. The granuloma is an immunological response to neutralize and destroy the fungus, found mainly within the multinucleated cells, either undergoing degeneration, normal, or proliferating. Only yeast forms are found in the tissues, frequently in multinucleated giant cells, but also in the microabscesses (Fig. 4). They can be observed in routine H&E sections, but are more readily seen with PAS or methenamine silver nitrate (Fig. 5).
Immunohistochemistry and in situ hybridization can also be used for detection of the fungus (Brito et al., 1999). In contrast to the other mycoses, there are no reports of mycelia in tissue sections. In HIV-positive patients, macrophages, lymphocytes, and multinucleated cells do not form well-organized granulomas, since the cellular immunity is depressed (Marques et al., 1995). The main cell to harbor and destroy the fungus is the macrophage. Neutrophils phagocytose, but fail to digest, the yeasts, unless the cell membrane had been altered by amphotericin B (Goihman-Yahr et al., 1989). In non-activated macrophages, the fungus proliferates, causing cell death, and invades the adjacent tissues. Nevertheless, macrophages activated by IFN-γ can digest P. brasiliensis (Brummer et al., 1989), and the phagosomes show a granular electron-dense material derived from the yeast cytoplasm (Brummer et al., 1990). Eosinophils are common in the cellular infiltrate, but their role is unknown. It has also been suggested that toxic granule proteins, as major basic proteins, participate in the pathophysiology of Pmycosis (Wagner et al., 1998).
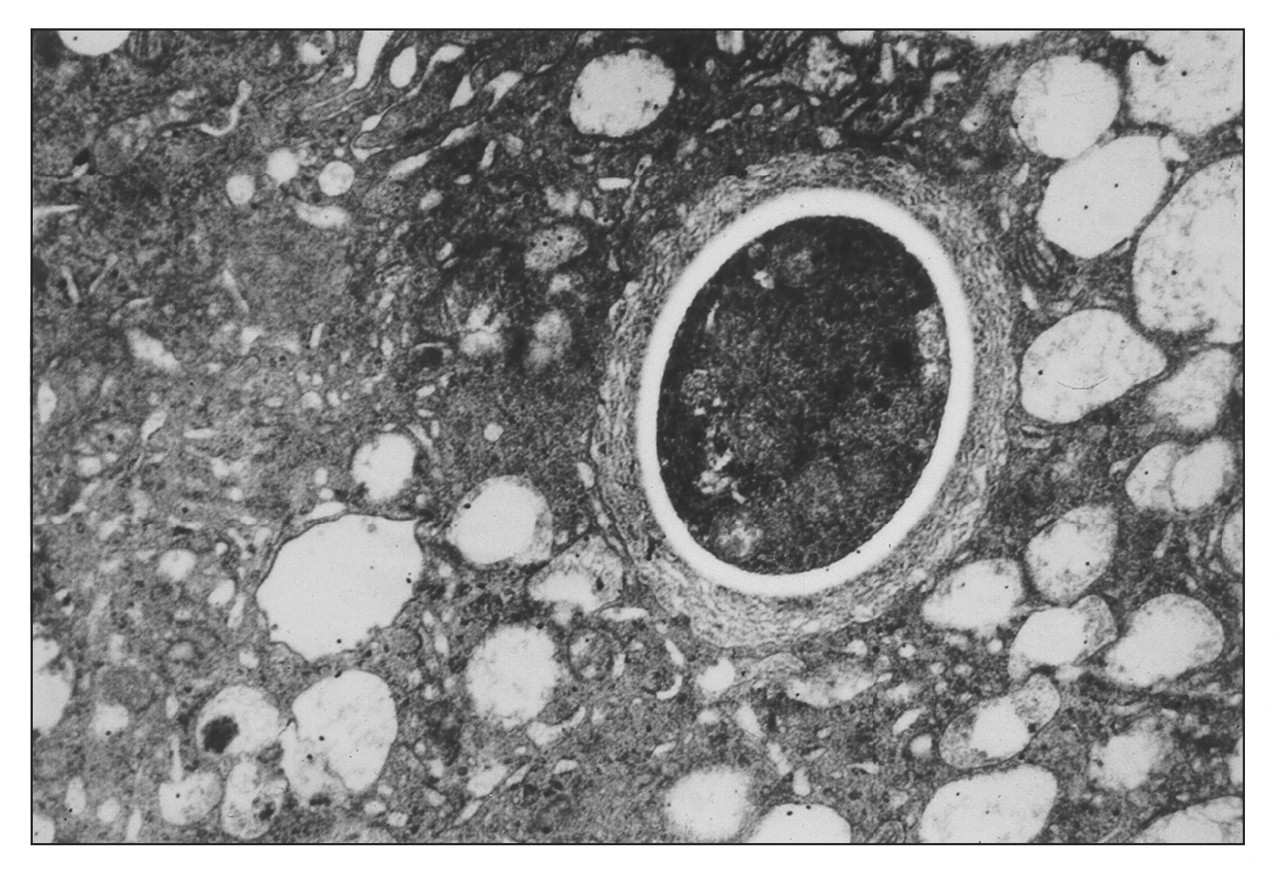
The yeasts are multinucleated (2–5 nuclei), and the cytoplasm contains mitochondria, scarce endoplasmic reticulum, free ribosomes, and vacuoles of various sizes (Borba et al., 1999). By scanning electron microscopy, the yeasts are seen as round to ovoid structures, with a fibrillar surface (Vieira e Silva et al., 1974). The wall of the fungus is 0.1–0.2 μm thick and is formed by three layers: a thin plasma membrane, a thick lamellar medium layer, and an external fibrillar layer (Fig. 6). According to Iabuki and Montenegro (1979), there is an interface between the fungus wall and the membranes of the inflammatory cells that disappears when the fungus is in degeneration.
The numbers and organization of the granulomas are variable. The so-called hyperergic spectrum of the disease is characterized by a localized infection, with cellular immune response and well-organized epithelioid granulomas. The anergic type presents as a disseminated infection, decreased cellular immunity, and poorly organized granulomas rich in fungi (Brito and Franco, 1994; Soares et al., 2000).
Culture can sometimes be useful diagnostically, but P. brasiliensis grows extremely slowly: On Sabouraud agar, colonies start to appear only after 20 days. Serological examination by immunodiffusion or complement fixation and skin tests with paracoccidioidin are used mainly for epidemiological studies or as a parameter to monitor treatment rather than as diagnostic aids (Saraiva et al., 1996). Patients with severe Pmycosis tend to be unresponsive to paracoccidioidin because of a deficiency of cellular immunity, and are therefore termed anergic.
Management
Paracoccidioidomycosis is life-threatening, and, even with supposedly adequate treatment, death is not uncommon, in both acute and chronic cases. Underlying predisposing factors should be corrected where possible. Most patients are from rural areas, with low income and presenting difficulties with food ingestion and anorexia due to the disease. Control of malnutrition with an adequate diet is important.
Antifungal therapy is required, though even after treatment there are no guarantees of complete destruction of the fungus (Franco et al., 1989). Initial treatment lasts from 2 to 6 mos, and includes sulfonamides, amphotericin B, or imidazoles. However, although Barravieira et al.(1989) successfully used cotrimoxazole and cotrimazine, sulfonamides are generally not very effective. Amphotericin B can provoke severe adverse effects, particularly nephrotoxicity.
Azoles are therefore preferred. Oral itraconazole (100 mg PO qds for 6 mos) is considered the drug of choice, but fluconazole and ketoconazole are also used (Mendes et al., 1994). After the initial treatment of the disease, maintenance involves sulphadimethoxine or sulphadoxine 500 mg, twice a wk, for about two years. Oral lesions typically show an excellent response after a few weeks of treatment, but if treatment is not followed as described above, relapse is common.
DNA-based vaccination with the gp43 gene has been tested in mice, eliciting a protective immunity against P. brasiliensis (Pinto et al., 2000).
Paracoccidioidomycosis involving the tongue in a 55-year-old woman. Paracoccidioidomycosis on the palatal and buccal gingivae. The “moriforme”
aspect with pinpoint hemorrhages is typical of this disease. This patient also
had skin lesions. Fresh preparation of Paracoccidioides brasiliensis in 10%
potassium hydroxide, showing the refractile wall and the formation of new
fungi. Microscopic view of P. brasiliensis phagocytosed by a
multinucleated giant cell. The fungi are easily seen by H&E staining. Gomori-Grocott silver impregnation showing the typical pilot-wheel aspect,
characteristic of P. brasiliensis. Electron micrograph of P. brasiliensis inside a multinucleated
giant cell.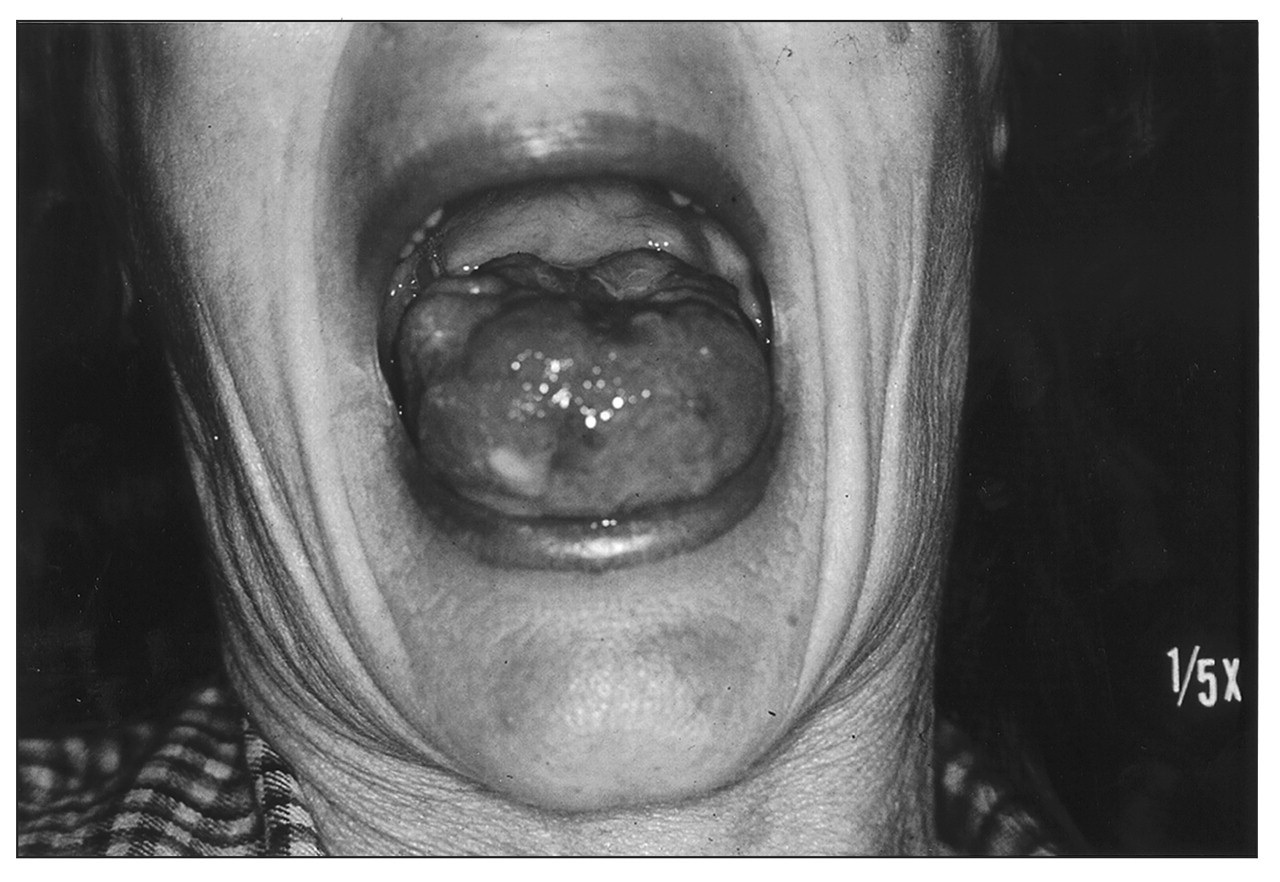
Footnotes
Acknowledgements
This work was supported by FAPESP (Fundação de Amparo a Pesquisa do Estado de São Paulo).
