Abstract
In addition to anti-tumor effects, ionizing radiation causes damage in normal tissues located in the radiation portals. Oral complications of radiotherapy in the head and neck region are the result of the deleterious effects of radiation on, e.g., salivary glands, oral mucosa, bone, dentition, masticatory musculature, and temporomandibular joints. The clinical consequences of radiotherapy include mucositis, hyposalivation, taste loss, osteoradionecrosis, radiation caries, and trismus. Mucositis and taste loss are reversible consequences that usually subside early post-irradiation, while hyposalivation is normally irreversible. Furthermore, the risk of developing radiation caries and osteoradionecrosis is a life-long threat. All these consequences form a heavy burden for the patients and have a tremendous impact on their quality of life during and after radiotherapy. In this review, the radiation-induced changes in healthy oral tissues and the resulting clinical consequences are discussed.
Introduction
In addition to anti-tumor effects, ionizing irradiation causes damage in normal tissues located in the field of radiation. This becomes particularly evident in the head and neck region, a complex area composed of several dissimilar structures that respond differently to radiation: mucosal linings, skin coverings, subcutaneous connective tissue, salivary gland tissue, teeth, and bone/cartilage. Acute changes produced by radiotherapy are observed in the oral mucosa (erythema, pseudomembrane-covered ulceration), salivary glands (hyposalivation, changed salivary composition), taste buds (decreased acuity), and skin (erythema, desquamation). Late changes can occur in all tissues (Cooper et al., 1995; Taylor and Miller, 1999). Although thorough protocols have been developed to minimize or manage the early and late oral sequelae of radiotherapy of the head and neck region (Jansma et al., 1992; Scully and Epstein, 1996; Schiødt and Hermund, 2002), the consequences of radiation-induced salivary gland injury and the other oral sequelae of head and neck radiotherapy are still difficult to manage.
In this review, the radiation-induced changes in healthy oral tissue and the resulting clinical consequences are discussed. The radiation-related changes in the oral mucosa, salivary glands, taste, dentition, periodontium, bone, muscles, and joints are discussed in the order that they appear. They can be divided into early (mucosa, taste, salivary glands), intermediate (taste, salivary glands), and late (salivary glands, dentition, periodontium, bone, muscles, joints) effects.
Radiotherapy
Radiotherapy plays an important role in the management of head and neck cancer. The majority of new cases of invasive head and neck cancer will need radiotherapy as a primary treatment, as an adjunct to surgery, in combination with chemotherapy, or as palliation (Dobbs et al., 1999). The radiation dose needed for the treatment of cancer is based on location and type of malignancy, and whether or not radiotherapy will be used solely or in combination with other modalities. Most patients with head and neck carcinomas, treated with a curative intent, receive a dose between 50 and 70 Gy. This dose is usually given over a five- to seven-week period, once a day, five days a week, 2 Gy per fraction (Dobbs et al., 1999). The total dose for pre-operative radiotherapy or radiotherapy for malignant lymphomas is usually lower.
Fractionated radiation is used because there is a difference in the responses of tumor tissue and normal tissue. In general, normal tissue repairs sublethal DNA damage, especially in the low-dose range, better than tumor tissue. Giving radiation in 2-Gy fractions magnifies the differences in responses between tumor and normal tissues. The sparing effect of fractionated radiation is the largest for late-responding tissues, whereas early-responding tissues respond more like tumor tissue. Next to DNA-repair advantages, fractionated irradiation allows for the repopulation of tissue between fractions (especially during the weekend, when the tumor and normal tissues are not radiated), thereby reducing early effects. This, however, also applies for rapidly proliferating malignant tissue. Another advantage is that fractionated irradiation allows for re-oxygenation of radio-resistant hypoxic tumors between fractions, leading to a higher percentage of radiosensitive oxygenated cells (Steel, 2002; Hall, 2000).
The most important dose-limiting factor is the tolerance of the adjacent normal tissues. Depending on stage and location of the primary tumor and affected lymph nodes, the oral cavity, salivary glands, and jaws of most head and neck cancer patients may be located in the radiation portals. Even with the most optimal radiotherapeutic schedule, unwanted radiation-induced changes will occur in these tissues. Tissues with rapid turnover rates show acute reactions to radiotherapy (early effects), while in tissues with slower turnover rates, damage may not become evident for months or years after therapy (late effects) (Steel, 2002; Hall, 2000). Since the overall five-year rate for oral cancer survival is about 80% for the early stages of oral cancer and about 35% for advanced stages, the objective of effective cancer therapy includes preservation of normal tissue function and reducing injury as much as possible. Several strategies to increase the tumor control probability (TCP) without increasing or even reducing the normal tissue complication probability (NTCP) have been developed and tested in clinical practice or are currently the subject of clinical trials. Based on the above-mentioned radiobiological assumptions, alternative fractionation schemes like hyperfractionation and accelerated fractionation, techniques that reduce the irradiated volume (3D conformal radiation therapy and intensity-modulated radiotherapy; Horiot et al., 1994; Eisbruch et al., 1996; Russell, 2000; Wu et al., 2000), and schemes which increase oxygenation of tumor tissues (Kaanders et al., 2002) have been developed.
Accelerated fractionation and hyperfractionation seem to be effective strategies for improving tumor control. Hyperfractionation makes use of the difference between the repair capabilities between tumor and normal tissue by further fractionation of the dose to a 1.15 Gy-fraction, for example. For overall conventional treatment time to be maintained, 2 fractions per day are given. With this type of fractionation, the total absorbed tumor dose can be increased while not adding to late toxicity. In contrast, accelerated schedules reduce overall treatment time. Accelerated fractionation is based on the observation that radiation injury causes accelerated proliferation of tumor tissue, and shortening of the treatment time would overcome this problem. The fractions typically are given twice a day. Both approaches have been shown to result in a modest gain in curing head and neck cancer when tested in randomized trials (Garden, 2001). Also, combinations of hyperfractionated and accelerated schedules have been shown to be especially successful for rapidly dividing tumors (Awwad et al. 2002). The disadvantage of these new treatment techniques is the higher rates of acute toxicity, especially mucositis. The addition of chemotherapy may, next to systemic cytotoxicity, introduce an exacerbated local tissue reaction (Bensadoun et al. 2001). To reduce chronic and acute tumor hypoxia, investigators have developed "ARCON", which combines accelerated radiotherapy to counteract tumor repopulation with carbogen breathing and nicotinamide to increase oxygenation. Very high local and regional tumor control rates were observed, with an increase in acute toxicity and late morbidity within acceptable limits, although there is some concern regarding late complications in bone (Kaanders et al., 2002). Further reduction of the irradiated volume of normal tissue by means of new computerized planning techniques seems to be the next step.
Three-dimensional (3D) conformal radiation therapy is a treatment technique designed to shape the spatial distribution of the high radiation dose to the target volume, thereby reducing the dose delivered to the normal tissues. Intensity-modulated radiotherapy (IMRT) is even more conformal than the 3D conformal techniques, since this new technique optimally assigns weights to individual rays of a beam as opposed to a single weight to the beam as a whole. The latter makes it possible to produce dose distribution patterns that are much closer to the desired patterns than previously possible, thus optimally minimizing the dose to normal tissues. Completely different treatment plans are constructed in which a smaller volume of tissue receives a high dose of irradiation and a large volume (mostly the whole organ) receives a low dose. Current investigations in our laboratories address the question of whether there are regional differences in radiosensitivity within a particular tissue. Another issue that warrants study is the effect of a low dose to a large volume vs. a high dose to a small volume. Mathematical modeling of the data may lead, in the future, to a prediction of the NTCP with each treatment plan (Schilstra and Meertens, 2001).
Oral Mucosa
Damage to oral mucosa is strongly related to radiation dose, fraction size, volume of irradiated tissue, fractionation scheme, and type of ionizing irradiation (Maciejewski et al., 1991; Scully and Epstein, 1996; Denham et al., 1999; Handschel et al., 1999). Oral side-effects develop early during radiotherapy (Dörr and Kummermehr, 1990; Denham et al., 1999). The acute mucosal response to radiotherapy is a result of mitotic death of epithelial cells, since the cell cycle time of the basal keratinocytes is about four days (Scully and Epstein, 1996).
Mucositis induced by radiotherapy is defined as the reactive inflammation of the oral and oropharyngeal mucous membrane during radiotherapy in the head and neck region. It is characterized by atrophy of squamous epithelial tissue, absence of vascular damage, and an inflammatory infiltrate concentrated at the basement region (Handschel et al., 1999). Radiation mucositis is an inevitable but transient side-effect (Spijkervet et al., 1989; Maciejewski et al. 1991; Scully and Epstein, 1996; Denham et al., 1999). It is an integral part of radiotherapy in terms of morbidity, since during a course of curative radiation about 80% of the patients will develop pseudomembranous mucositis. The early radiation reaction causes local discomfort as well as difficulties in drinking, eating, swallowing, and speech. Therefore, it can give rise to nutritional problems, and in severe cases nasogastric feeding, which is very uncomfortable, may become necessary (Donaldson, 1977; Beumer et al., 1979a,b; Wood et al., 1989; Jansma et al., 1992; Lees, 1999; Mekhail et al., 2001). About 20-30% of the patients will need artificial feeding. Severe mucositis may necessitate an interruption of the course of radiotherapy and thus can serve as a dose-limiting factor (Denham et al., 1999; Sonis et al., 1999). Such interruptions must be prevented, because they may result in prolongation of treatment time and thus a reduction in therapeutic effect (Fowler, 1986). As mentioned before, hyperfractionation, accelerated fractionation, and radiochemotherapy, although especially successful for the treatment of rapidly dividing tumors, result in higher rates of acute toxicity, especially mucositis (Bensadoun et al., 2001; Awwad et al., 2002).
Various signs of mucositis may emerge during radiotherapy (Spijkervet et al., 1989; Scully and Epstein, 1996; Riesenbeck et al., 1998; Denham et al., 1999; Handschel et al., 1999). The first clinical signs of mucositis occur at the end of the first week of a conventional seven-week radiation protocol (daily dose of 2 Gy, five times a week). There is no consensus regarding what is the first sign of mucositis. Some authors describe a white discoloration of the oral mucosa, which is an expression of hyperkeratinization as the first symptom, followed by or in combination with erythema (Spijkervet et al., 1989; Scully and Epstein, 1996). Others consider erythema to be the first reaction (Dreizen et al., 1977b; Riesenbeck et al., 1998; Sonis et al., 1999). Using a mouse model, Dörr and Kummermehr (1990) explain the mucosal changes following radiation as follows. The lack of formation of new basal cells caused by radiotherapy leads to a gradual, linear decrease in cell numbers. If the cellularity of the mucosa drops below 70% of the normal level, the cell production rate from the surviving cells increases dramatically (a possible cause for the whitish aspect of oral mucosa). As radiotherapy continues, a steady state between mucosal cell killing and mucosal cell regeneration may occur and favor an acute reaction in the form of a prominent erythema. Around the third week of radiotherapy, more severe symptoms of mucositis, such as the formation of pseudomembranes and ulceration, may appear (Dreizen et al., 1977b; Spijkervet et al., 1989). Some authors consider pseudomembranes to be ulcers covered by fibrinous exudate (Maciejewski et al., 1991; Riesenbeck et al., 1998). Others suggest that pseudomembranous mucositis is related to yeast stomatitis (Ramirez-Amador et al., 1997) or to colonization of the oral cavity with Gram-negative bacilli (Spijkervet et al., 1990, 1991; Martin and van Saene, 1992). In their mouse model, Dörr and Kummermehr (1990) explained the development of pseudomembranes, when radiotherapy commences, as a cell regeneration process that cannot keep up with cell killing. As a result, partial or complete epithelial denudation develops, which presents as spotted or confluent pseudomembranous mucositis. Healing eventually occurs from the surviving mucosal stem cells. Similar changes have been observed in humans, in whom the mucositis is characterized by loss of epithelial cells, absence of vascular damage, and an inflammatory reaction at the epithelial-connective tissue interface (Handschel et al., 1999). Briefly, mucositis generally persists throughout radiotherapy, is maximum at the end of the irradiation period, and continues for one to three weeks after treatment has ceased (Scully and Epstein, 1996).
The severity of mucositis varies considerably between patients (Denham et al., 1999) and may relate to the fractionation schedule applied. Accelerated fractionation results in a more rapid onset of mucositis (Maciejewski et al., 1991; Denham et al., 1999). Furthermore, the mucosa of the oral cavity does not react in the same manner at all locations. Mucositis is most severe in the soft palate, followed, in order, by the mucosa of the hypopharynx, floor of the mouth, cheek, base of the tongue, lips, and dorsum of the tongue. Patients with compromised oral mucous membranes secondary to alcoholism and/or excessive smoking exhibit the most severe mucosal changes (Beumer et al., 1979a,b; Rugg et al., 1990).
Mucositis is basically a tissue reaction to the trauma of radiation (Maciejewski et al., 1991; Scully and Epstein, 1996; Denham et al., 1999) or chemotherapy (Bensadoun et al., 2001). Other factors that may contribute to the development of mucositis include: the increase in the inflammatory mediator, platelet-activating factor in saliva of irradiated patients (McManus et al., 1993); leukocyte adhesion to E-selectin or endothelial intercellular adhesion molecule-1 (ICAM-1) which promotes the radiation-induced inflammatory response in squamous epithelium (Handschel et al., 1999); a decrease in the level of salivary epidermal growth factor (Dumbrigue et al., 2000); and an increase in the carriage rate of Gram-negative bacilli in the oropharynx (among others Enterobacteriaceae, Pseudomonaceae) (Spijkervet et al., 1989; Martin and van Saene, 1992; Scully and Epstein, 1996). This marked increase in oral Gram-negative enterobacteria and pseudomonads has particularly been shown as a possible aggravating factor for development of oral mucositis (Spijkervet et al., 1990, 1991). Less than 10% of healthy individuals exhibit colonization of the oral cavity with these non-indigenous Gram-negative bacilli (Yourassowsky et al., 1987). This is due to the oropharyngeal colonization defense, which is determined by the integrity of the anatomical structures, physiology, motility, secretions, secretory immunoglobulin A, mucosal cell turnover, and the indigenous flora. These factors are impaired by radiotherapy for head and neck cancer and are negatively influenced by more generalized factors, such as advanced age, medical interventions (e.g., surgery), and underlying disease. Selective elimination of Gram-negative bacilli was associated with a reduction of pseudomembranes and ulceration (Spijkervet et al., 1990, 1991). These authors postulated that Gram-negative bacilli or endotoxin released by Gram-negative bacilli could play a major role in the development of the advanced stages of radiation mucositis, while the initial signs are basically related to irradiation only (Spijkervet et al., 1990, 1991).
The most common infection in the oral cavity during or shortly after radiotherapy is candidiasis (Epstein, 1990; Ramirez-Amador et al., 1997). Many patients become colonized intra-orally with Candida albicans during radiotherapy (Chen and Webster, 1974). Ramirez-Amador et al.(1997) showed that the prevalence of positive Candida cultures increased from 43% at baseline to 62% at completion of radiotherapy and to 75% during the follow-up period. Some authors believe that oral mucositis is aggravated by fungal infections (Beumer et al., 1979a,b; Al-Tikriti et al., 1984). However, treatment of yeast and Gram-positive cocci with topical anti-fungals and disinfectants failed to relieve such complications (Chen and Webster, 1974; Martin and van Saene, 1992; Wijers et al., 2001). Thus, many of the oral lesions observed during treatment do not seem to be due to candidiasis or streptococcal infection. Finally, it should be mentioned that herpes simplex virus infection is not a significant contributing factor in irradiation mucositis. This is in contrast to the commonly seen herpes simplex virus re-activation following chemotherapy and radiochemotherapy patients (Redding et al., 1990; Scully and Epstein, 1996).
In summary, although the etiopathogenesis of radiation mucositis still is not fully clear, it most likely can be considered as a four-step inflammation consisting of an inflammatory/vascular phase, an epithelial phase, a bacterial phase, and a healing phase. This sequence of phases has been proposed by Sonis (1998) for chemotherapy-induced stomato-toxicity, but probably also holds true for radiation mucositis. This, however, does not necessarily imply that the treatments for chemotherapy- and radiation-induced mucositis are similar, since many of the treatments to alleviate stomato-toxicity resulting from chemotherapy have been shown to be ineffective for radiation-induced mucositis (Vissink et al., 2003).
Taste
Alteration in taste is an early response to radiation and often precedes mucositis. Radiotherapy to the head and neck affects taste thresholds, food intake, chewing, the hedonics of tasting (Beidler and Smith, 1991; Spielman, 1998), and may result in weight loss (Erkurt et al., 2000). Most patients experience partial or complete loss of taste acuity during radiotherapy (Beumer et al., 1979a, b). Conger (1973) found that taste sensation decreases exponentially with a cumulative dose of about 30 Gy (3 weeks), 2 Gy per fraction, after which it becomes virtually absent. The loss in perception of all flavors rarely occurs (Toljanic and Saunders, 1984). Perception of bitter and acid flavors is more susceptible to impairment than perception of salt and sweet flavors (Mossman et al., 1982). The loss of taste is not only a result of the effect of irradiation on the taste buds, but is also related to the reduction in salivary flow rate. A reduced salivary flow decreases transport and solubilization of gustatory stimulants, reduces the ability of saliva to protect the mucosa against bacteria, fungi, and variation in the oral pH, alters the ionic composition of saliva which is important for taste, and affects mastication, nutrition, and the hedonic aspects of tasting (Spielman, 1998).
Direct radiation damage to the taste buds or their innervating nerve fibers has been reported as the main cause of taste loss (Conger, 1973; Dreizen et al., 1977b; Mossman, 1986). Histologically, taste buds showed signs of degeneration and atrophy at 10 Gy (2 Gy per day), while at therapeutic levels the architecture of the buds was almost completely destroyed (Conger, 1973).
Loss of taste is usually transient (Tomita and Osaki, 1990). Taste gradually returns to normal or near-normal levels within one year after radiotherapy, although it can take as long as five years. The degree of taste recovery and the recovery time depend on the radiation dose received. Some patients may retain a residual reduction in taste acuity (hypogeusia), or even a permanent impairment in sensation (dysgeusia) (Conger, 1973; Dreizen et al., 1977b; Mossman et al., 1982; Toljanic and Saunders, 1984), but near-normal suprathreshold levels of taste in irradiated patients have been reported as well (Schwartz et al., 1993; Spielman, 1998). These obvious discrepancies between measurable taste loss and subjective awareness of taste loss may be due to adaptation of the patient to the sensory loss (Mossman et al., 1982).
Taste impairment has profound effects on the nutritional status of the patient and is associated with weight loss through reduced appetite and altered patterns of food intake. This is due not only to the loss of taste per se, but to the non-equal impairment of the perception of the various flavors as well. The result is that food tastes different, and often unpleasant, after radiation therapy, a fact which, of course, many patients do not appreciate. Also, further studies are needed to evaluate the effect of irradiation on von Ebner’s glands and the residual flow from these glands. This may be sufficient to obscure the interpretation of currently available data (Spielman, 1998). If so, radiotherapy treatment plans should attempt to spare these glands if possible.
Salivary Glands
Based on the slow turnover rates of their cells, the salivary glands are expected to be relatively radio-resistant. Yet the changes in quantity and composition of saliva that occur shortly after radiotherapy indicate that the gland tissue is an acutely responding tissue (Vissink et al., 1992; Taylor and Miller, 1999; O’Connell, 2000; Burlage et al., 2001; Nagler, 2002). It is not clear whether the direct effects of radiation on the secretory and ductal cells cause radiation damage of salivary gland tissue, or if it is secondary to injury of the fine vascular structures, increased capillary permeability, interstitial edema, and inflammatory infiltration. In a human post mortem study, it has been assessed that, in the lower dose range (< 30 Gy), damage is reversible to a certain level, but after cumulative doses (> 75 Gy), extensive degeneration of acini is observed, along with inflammation and fibrosis in the interstitium (Dreyer et al., 1989). As treatment continues, there is progressive degeneration of the acinar epithelium and development of interstitial fibrosis. Serous acinar cells appear to be more readily affected by irradiation than mucous acinar cells and ductal cells (Kashima et al., 1965; Seifert and Geier, 1971; Dreyer et al., 1989). From non-human primate experiments, it was concluded that the acute functional impairment was caused directly by serous acinar apoptotic cell death rather than being the result of inflammatory processes and circulatory compromise due to vascular injury (Stephens et al., 1989). Also, the later loss of function is not thought to be due to chronic inflammation or fibrosis of the glandular tissue (O’Connell et al., 1999). The most likely course of radiation-related events that occur in rat parotid salivary gland tissue has been recently described by Coppes et al.(2001). They observed four phases in the radiation-induced loss of salivary gland function. The first phase (0-10 days) was characterized by a rapid decline in flow rate without changes in amylase secretion or acinar cell number. The second phase (10-60 days) consisted of a decrease in amylase secretion and was paralleled by acinar cell loss. Flow rate, amylase secretion, and acinar cell numbers did not change in the third phase (60-120 days). The fourth phase (120-240 days) was characterized by a further deterioration of gland function but an increase in acinar cell number, albeit with poor tissue morphology. Comparable changes have been observed in rat submandibular tissue (Zeilstra et al., 2000; Coppes et al., 2002).
The early response in gland function has been studied thoroughly in rats (Vissink et al., 1990; Franzén et al., 1991; Nagler et al., 1993; Coppes et al., 1997a,b, 2000, 2001; Zeilstra et al., 2000). Within three days after irradiation with a single dose of 15 Gy of x-rays, a decrease in salivary flow of nearly 50% can be observed (Vissink et al., 1990; Peter et al., 1995; Coppes et al., 1997a,b, 2001; Zeilstra et al., 2000). Lack of obvious quantitative morphological alterations (Franzén et al., 1991; Henricksson et al., 1994; Zeilstra et al., 2000; Coppes et al., 2001), a rather quick recovery of the morphological changes if they occur (Vissink et al., 1991), and a lack of increase in apoptotic cells early after radiotherapy (Paardekooper et al., 1998) point to the presence of altered cell membranes (Sodicoff et al., 1974; El Mofty and Kahn, 1981; Vissink et al., 1992) and/or a disturbed intracellular signaling (Vissink et al., 1991; Coppes et al., 1997b) as the cause of the early effect of radiation on the parotid gland. This was confirmed in an in vitro study showing that muscarinic receptor-induced calcium mobilization and protein kinase C activation were affected (Coppes and Kampinga, 2001). The late effects of radiation on the parotid and submandibular glands have been studied less extensively, and have been reported as a dose-dependent further decline in function (Nagler et al., 1998; Coppes et al., 2001) and loss of acinar cells (Henricksson et al., 1994; O’Connell et al., 1999). Unfortunately, in the latter studies the whole or half of the head including the glands was irradiated. Therefore, indirect effects due to damage to other organs confound the interpretation with regard to salivary gland function (Nagler, 2001; Konings et al., 2002).
Also, in humans, depending on the localization of the radiation portals, a rapid decrease of the salivary flow rate is observed during the first week of radiotherapy, after which there is a gradual decrease to less than 10% of the initial flow rate (Fig. 1) (Dreizen et al., 1976; Shannon et al. 1978b; Liu et al., 1990; Franzén et al., 1991; Valdez et al., 1993; Jones et al., 1996; Burlage et al., 2001). Although in the older literature the submandibular gland was thought to be less radiosensitive than the parotid gland, both glands have been shown to be as sensitive to radiotherapy, at least with respect to their function (Liu et al., 1990; Valdez et al., 1993; Burlage et al., 2001). In rats, it has been shown that the submandibular gland may be even more sensitive to the late effects of radiation, due to its inability to restore the damage (Coppes et al., 2002), but it remains to be established if it is true for humans. It has been suggested that the final degree of radiation-induced hyposalivation depends on individual patient characteristics, such as pre-irradiation salivary gland activity, age, and sex (Eneroth et al., 1972a,b; Mira et al., 1981). It has been stated that salivary glands with high flow rates before the initiation of radiotherapy show less reduction in salivary flow rate (Eneroth et al., 1972a,b; Mira et al., 1981, 1982), but this observation could not be confirmed in recent dose-volume studies (Eisbruch et al., 1999, 2001; Roesink et al., 2001). Clinically, of more importance is the observation that the irradiated volume of salivary gland tissue correlates directly with the severity of oral complications (Cheng et al., 1981; Mira et al., 1981; Tsujii, 1985; Liu et al., 1990; Hazuka et al., 1993; Jones et al., 1996; Nishioka et al., 1997; Eisbruch et al., 1999, 2001; Wu et al., 2000; Roesink et al., 2001). The implementation of alternative fractionation schedules, like hyperfractionation and accelerated fractionation, to reduce the side-effects of radiotherapy on normal tissues has also been proposed (Leslie and Dishe, 1991, 1994), but its effect on salivary gland function and morphology is negligible (Price et al., 1995; Coppes et al., 2002), which is advantageous for tumor control.
The early (Burlage et al., 2001) and late (Liu et al., 1990; Valdez et al., 1993) human data on the radiation-induced severe drop in flow rate of both the parotid and submandibular gland somewhat contradict the functional data derived from scintigraphic studies (Valdés Olmos et al., 1994; Liem et al., 1996). These authors showed a failure of the major salivary glands to excrete saliva early post-irradiation, and a decreased uptake of 99mTc-pertechnetate together with a loss of secretory function in the post-irradiation stage. This effect was stronger in parotid than in submandibular glands, although the incidence of xerostomia did not correlate with the effects observed in the scintigraphic studies (Liem et al., 1996), once again pointing to the obvious discrepancy between the actual salivary flow and the scintigraphic (Liem et al., 1996) and morphological changes (Vissink et al., 1991) induced by irradiation. Therefore, we strongly suggest that, from a clinical point of view, the combination of objective (measurement of salivary flow rate) and subjective (questionnaires) parameters still provides the best assessment with regard to the pattern of patients’ complaints and the effects of various therapies on these complaints.
Recent prospective studies of salivary flow following non-homogeneous irradiation of the parotid glands with fractionated radiotherapy have utilized dose-volume histograms and various models to assess these relationships. These studies found that the mean dose to the gland is correlated with the reduction of the salivary output. This is consistent with the suggested parallel architecture of the functional subunits (each functional subunit, e.g., an acinus, functions independently of other subunits, in contrast to serially organized organs, e.g., the spinal cord) of the salivary glands (Eisbruch et al., 2001; Schilstra and Meertens, 2001). The range of the mean doses, which have been found in these studies to cause significant salivary flow reduction, is from 26 to 39 Gy (Kaneko et al., 1998; Eisbruch et al., 2001; Roesink et al., 2001). These calculations, however, have been made with the assumption that the dose on the gland can be averaged disregarding the possibility of regional differences in sensitivity.
Aside from the quantity of saliva, radiotherapy also results in a change of salivary composition. Saliva turns into a very viscous, white, yellow, or brown fluid (Ben-Aryeh et al., 1975; Dreizen et al., 1976). The obvious qualitative salivary changes are a reduced pH and buffering capacity, altered salivary electrolyte levels, and changed non-immune and immune antibacterial systems. The average pH decreases from about 7.0 to 5.0 (Ben-Aryeh et al., 1975; Dreizen et al., 1976). The reduced buffering capacity is primarily due to a reduction of bicarbonate concentration in parotid saliva (Dreizen et al., 1976; Marks et al., 1981). An increase in the concentrations of sodium, chloride, calcium, and magnesium has been reported, while the concentration of potassium is only slightly affected (Ben-Aryeh et al., 1975; Dreizen et al., 1976; Anderson et al., 1981; Valdez et al., 1993). The concentrations of immunoproteins (e.g., sIgA), lysozyme, and lactoferrin are increased (Brown et al., 1976, 1978; Makkonen et al., 1986; Valdez et al., 1993; Almståhl et al., 2001). The decrease in salivary flow rate, however, is greater than the increase in immunoprotein and lysozyme levels, and this results in a significant immunoprotein deficit. Since oral clearance and immunologic mechanisms are potent means of host protection, their compromise is intrinsically related to changes in the oral flora of irradiated patients (Brown et al., 1975). One of the major radiation-induced changes in the oral flora is a pronounced increase in acidogenic, cariogenic micro-organisms, at the expense of non-cariogenic micro-organisms. The most clinically significant changes are the increase of Streptococcus mutans, Lactobacillus species, and Candida species (Llory et al., 1972; Brown et al., 1978; Keene et al., 1981; Ramirez-Amador et al., 1997; Epstein et al., 1998a). Comparable changes in oral flora have been observed in patients with hyposalivation from other causes, indicating that a low salivary secretion rate mainly promotes a flora associated with the development of dental caries (Almståhl and Wikström, 1999). The major changes in the oral flora as a result of hyposalivation after radiotherapy are observed in the period from the onset of radiotherapy to three months after completion. From the sixth month after radiotherapy, the composition of the oral microflora remains constant or partially returns to baseline composition (Brown et al., 1978).
Saliva is an important host defense component of the oral cavity (Nieuw Amerongen and Veerman, 2002). Thus, the quantitative and qualitative salivary changes predispose the irradiated patient to a variety of problems that develop either directly or as an indirect result of the diminished salivary output. The Table provides a list of the consequences of radiation-induced hyposalivation. Oral function (speech, chewing, and swallowing) is hampered because of, e.g., insufficient wetting and lubrication of the mucosal surfaces. Moreover, swallowing and chewing are impeded because of insufficient moistening of food by saliva (Hamlet et al., 1997). The increased viscosity and reduced flow of saliva cause intolerance to prosthetic appliances. Saliva is an effective lubricant at the denture-mucosal interface. With lesser amounts of saliva present, retention of the denture is poor and more friction is produced during function, which may easily traumatize the vulnerable irradiated oral mucosa. Many patients suffer from nocturnal oral discomfort. They are often awakened at night by a serious dryness of the mouth or have to get up frequently because of polyuria due to polydipsia throughout the day. The oral mucosa can have a dry, atrophic, pale, or hyperemic appearance. The mucosa of the tongue can exhibit similar features or appear fissured. The lips may be dry, cracked, or fissured. These changes in the oral mucosa are, in general, typical for xerostomia of any origin. The shift in oral microflora toward cariogenic bacteria, the reduced salivary flow (oral clearance), and the altered saliva composition (buffer capacity, pH, immunoproteins, oral clearance) may result in rapidly progressing radiation caries, along with a greater incidence of periodontal infections. The caries susceptibility is further increased by altered eating habits. Due to the radiation-mediated changes such as mucositis, atrophy of oral mucous membranes, hyposalivation, and taste loss, the diet of irradiated patients shifts to softer, sticky, carbohydrate-rich foods, with an increase in the frequency of intake—all of which promote caries. The average daily energy intake is about 300 kcal lower in irradiated patients with dry mouth symptoms (Bäckström et al., 1995).
In summary, the salivary glands show, in contrast to the other tissues, both early
Dentition
During and following a full course of radiotherapy, many patients experience an increased dental sensitivity to temperature changes and to sweet- and sour-tasting foods which possibly is related to the loss of the protective layer of saliva (Toljanic and Saunders, 1984). The most threatening complication for the dentition, however, is radiation-related caries. Radiation caries is a highly destructive form of dental caries which has a rapid onset and progression (Del Regato, 1939; Frank et al., 1965; Karmiol and Walsh, 1975; Jansma et al., 1993). Dental caries may become evident as early as three months following the initiation of radiotherapy. In severe cases, a previously healthy dentition can be completely lost within a year (Dreizen et al., 1977b).
Clinically, three types of caries lesions can be observed (Del Regato, 1939; Frank et al., 1965; Karmiol and Walsh, 1975). All three types of lesions can be observed within the same mouth. In view of the rapid progression, it is surprising that there is rarely any acute pain associated with radiation caries, even in its most severe manifestations. The histological features of the initial radiation caries lesions are similar to those observed in normal incipient dental caries lesions (Jongebloed et al., 1988; Jansma et al., 1993), but erosive types of lesions can be observed as well (Jansma et al., 1993).
The first type is a frequently observed lesion that starts on the labial surface at the cervical area of the incisors and canines. Initially, the lesion extends superficially around the entire cervical area of the tooth, and then progresses inward, often resulting in complete amputation of the crown. In the region of the molars, complete amputation of the tooth occurs less frequently; however, the caries tends to spread over all surfaces of the molar with changes in translucency and color leading to increased friability and breakdown of the tooth. Occasionally, only a rapid wearing away of the incisal and occlusal surfaces of the teeth is seen either with or without cervical lesions.
The second type of lesion is a generalized superficial defect that first affects the buccal and later the lingual or palatal surfaces of the tooth crowns. The proximal surfaces are less affected. This lesion often begins as a diffuse, punctate defect and then progresses to generalized, irregular erosion of the tooth surfaces. In this type of lesion, decay localized at the incisal or occlusal edges is often observed. The result is a destruction of the coronal enamel and dentin, especially on the buccal and palatal surfaces.
The third type is less frequently observed. It consists of a heavy brown-black discoloration of the entire tooth crown, accompanied by wearing away of the incisal and occlusal surfaces.
Besides the rapid onset and progression, radiation caries is most commonly found on tooth surfaces (buccal, labial, lingual, palatal, incisal, occlusal) that are normally relatively immune to dental caries. The areas just below the contact points seem to be the last areas to be affected by radiation caries. Furthermore, the mandibular anterior teeth, which normally are the teeth most resistant to caries, are equally if not more affected by radiation caries (Karmiol and Walsh, 1975). The characteristic attack on normally caries-immune, self-cleansing areas may be caused by changes in salivary flow and consistency that give rise to accumulation of a highly acidogenic dental plaque on these surfaces, and the result is a rapid decalcification of enamel.
It has always been a matter of debate whether radiation caries is due to a direct or indirect effect of irradiation on teeth, or to both. Several investigators have reported that the development of radiation caries was not dependent on the presence of teeth in the field of irradiation, but that the determining factor was whether the main salivary glands were within the radiation field (Del Regato, 1939; Frank et al., 1965; Karmiol and Walsh, 1975; Brown et al., 1976; Dreizen et al., 1976). Notwithstanding the study by Grötz et al.(1997), which showed that irradiation also resulted in dentinal changes in vital teeth, the current opinion still is that radiation caries is mainly due to salivary gland damage resulting in hyposalivation (Jansma et al., 1989; Joyston-Bechal et al., 1992; Spak et al., 1994; Al-Nawas et al., 2000; Kielbassa et al., 2001). Thus, collectively, hyposalivation-related alterations in microbial, chemical, immunologic, and dietary parameters of cariogenicity contribute to an enormous increase in the caries challenge in irradiated patients (Dreizen et al., 1977a,b). This enormous caries challenge becomes even more obvious since both loss of enamel (type II lesion) and severe destruction at the dentin-enamel junction (type I lesion) can be observed within a few weeks of exposure of enamel slabs in the oral cavities of patients with radiation-induced hyposalivation (Jansma et al., 1988b). The changes observed were similar to the changes occurring in natural hyposalivation-related dental caries (Jansma et al., 1993). So both the coronal enamel and the cervical area, where cementum or dentin is directly exposed to the oral environment, are areas at risk in dry-mouth patients. Clinically, the most striking and most difficult to treat phenomenon is the type I, wrapping around, caries lesion at the base of the crown which often results in an amputation of the crown. The mechanism behind and treatment of this type of lesion need further study.
Whether a direct effect of irradiation on teeth, other than the already-mentioned dentinal changes in vital teeth, also contributes to the development of radiation caries has not been fully elucidated, and reports are contradictory. Some investigators have reported that irradiated teeth decalcify more readily than non-irradiated teeth (Castanera et al., 1963), while others noted no differences in decalcification rates in vitro (Wiemann et al., 1972; Walker, 1975; Shannon et al., 1978a) or even reported decreased enamel and dentin solubility after therapeutic radiation (Joyston-Bechal, 1985; Jansma et al., 1988a; Kielbassa et al., 1999, 2002). Also, it has been shown that ionizing irradiation of dental enamel, at a therapeutic level, has no influence on its permeability and thus on the organic component of enamel (Jansma et al., 1990). In addition, there are some indications that the mechanical properties of enamel and, to a lesser extent, dentin deteriorate after exposure to radiation (Al-Nawas et al., 2000), but again, this effect is of minor significance, and indeed xerostomia-related changes constitute the major contributory factor in the development of radiation caries.
High levels of radiation exposure can markedly affect tooth development. The extent of the effect is dependent on the radiation dose and the stage of tooth development (Gorlin and Mishkin, 1963). In general, there is agreement that odontogenic cells in the pre-formative and differentiation phases are more radiosensitive than cells in the secretory or mature stage. If exposure to irradiation occurs before calcification, the tooth bud may be destroyed. Radiation at a later stage of development may arrest further growth and result in irregularities in enamel and dentin together with shortened roots (Scheibe et al., 1980; Dahllöf et al., 1994b; Kaste et al., 1994). According to Scheibe et al.(1980), tooth eruption is mostly delayed but not hindered, but this phenomenon still needs further study.
In summary, the effects of radiation on the dentition are predominantly thought to be indirect, mainly caused by the reduced salivary flow rate and its related consequences. Prevention therefore has to be directed to the treatment of xerostomia-related complaints, meticulous oral hygiene, change of diet, control of cariogenic flora, and prevention of caries with frequent fluoride applications (Vissink et al., 2003).
Periodontium
Decreased vascularity and acellularity of the periodontal membrane with rupturing, thickening, and disorientation of Sharpey’s fibers and widening of the periodontal space have been reported after irradiation (Silverman and Chierici, 1965; Anneroth et al., 1985). Others, however, found normal alignment of periodontal fibers (Scheibe et al., 1980). The cementum appears completely acellular, and its capacity for repair and regeneration is severely compromised (Silverman and Chierici, 1965). Early changes include radiographic widening of the periodontal ligament spaces and destruction of bony trabecules (Fujita et al., 1986).
The changes in cementum and periodontal ligament may predispose individuals to infection (Schüle and Betzhold, 1969). The risk of periodontal infection is also increased due to radiation-induced hyposalivation, the concomitant increased plaque accumulation and shift in oral microflora (Markitziu et al., 1992; Position paper, 1997; Leung et al., 1998). However, the major components of the subgingival flora of shallow pockets in head- and neck-irradiated individuals are similar to those of gingivitis sites in the normal population, although they may contain bacterial or fungal species that are uncommon in normal subjects (Leung et al., 1998). That the prevalence of (advanced) periodontal disorders is somewhat lower in irradiated patients than expected is probably related to the development of dental caries: If radiation caries develops, its progression is often so fast that the affected teeth are lost before (advanced) periodontal pathosis can occur.
The direct and indirect effects of high-dose radiotherapy on the periodontium result in an increased risk of periodontal attachment loss and tooth loss, and even in an increased risk for the development of osteoradionecrosis (Yusof and Bakri, 1993; Epstein et al., 1998b). This underscores the need for proper pre- and post-irradiation treatment planning (Jansma et al., 1992; Position paper, 1997; Epstein et al., 1998b; Epstein and Stevenson-Moore, 2001; Schiødt and Hermund, 2002).
Bone
The gross changes in the bone matrix after irradiation develop relatively slowly. The initial changes in bone result from injury to the remodeling system (osteocytes, osteoblasts, and osteoclasts). Osteoblasts tend to be more radiosensitive than osteoclasts; thus a relative increase in the lytic activity may occur. Whether the altered bone remodeling activity is the result of direct irradiation injury to the cells of the remodeling system or the indirect result of irradiation-induced vascular injury, or a combination of both phenomena, is still a matter of debate. Radiation injury to the fine vasculature of bone and its surrounding tissues first leads to hyperemia, followed by endarteritis, thrombosis, and a progressive occlusion and obliteration of small vessels. Within bone, this results in a further reduction of the number of cells and progressive fibrosis. With time, the marrow exhibits marked acellularity and hypo- or avascularity, with significant fibrosis and fatty degeneration. Some lacunae may become devoid of osteocytes. The endosteum atrophies, with significant loss of active osteoblasts and osteoclasts. The periosteum demonstrates significant fibrosis, with a similar loss of remodeling elements (Silverman and Chierici, 1965; Dreizen et al., 1977b; Beumer et al., 1979a,b; Marx and Johnson, 1987; Constantino et al., 1995). Marx and Johnson (1987) found hypovascularity and fibrosis to be the common end-stage of irradiation-induced tissue injury. Taken together, these observations lead to the obvious conclusion that irradiated bone is likely to respond poorly to trauma and infection.
The most severe potential complication of bone irradiation is osteoradionecrosis. The incidence of osteoradionecrosis of the mandible varies from 2.6% to 22%; the range is most commonly from 5% to 15% in recent reports (Constantino et al., 1995; Epstein et al., 1997; Thorn et al., 2000). The incidence of osteoradionecrosis of the maxilla is much lower (Curi and Dib, 1997; Tong et al., 1999; Thorn et al., 2000).
The definition of osteoradionecrosis is "bone death secondary to radiotherapy" (Marx and Johnson, 1987; Constantino et al., 1995). Some authors have advocated using the more general term "osteonecrosis", since necrosis of bone and soft tissue can also occur in other conditions, including cancer patients receiving chemotherapy and in diabetics (Epstein et al., 1987a,b). The latter authors have stressed that in radiotherapy, the exposure of soft and hard tissues—with subsequent hypoxia, hypovascularity, and hypocellularity—markedly increases the risk of necrosis. For those cases, they proposed the term "post-radiation osteonecrosis", but in this paper the more commonly used term "osteoradionecrosis" is used. The diagnosis of osteoradionecrosis is based mainly on patient history and clinical signs such as severe pain, non-healing (exposed) bone within the treatment area after completion of radiotherapy, and repeated infections. This process may progress to fistula or sequester formation and eventual spontaneous fracture (Marx, 1983a,b; Epstein et al., 1987a,b, 1997; Constantino et al., 1995; Thorn et al., 2000). The presenting lesion (e.g., superficial involvement vs. localized or diffuse involvement of the mandible) dictates the treatment protocol to be followed and stresses the need for an effective clinical staging system (Epstein et al., 1997; Schwartz and Kagan, 2002; Vissink et al., 2003).
In the early literature, the pathogenesis of osteoradionecrosis of the jaws was regarded as the inevitable triad sequelae of radiation, trauma, and infection (Watson and Scarborough, 1938; Meyer, 1958, 1970). In this concept, trauma serves as a portal of entry for oral bacteria into the underlying bone. Osteoradionecrosis is thus considered to be an infectious process, which progresses rapidly and spreads throughout the bone that cannot wall off the infection because of compromised vascularity and minimal regenerative capabilities. The source of trauma may be anything, including denture irritation, sharp or hard food particles, and sharp bony ridges. Tooth removal is said to be the most common cause of trauma (Meyer, 1970). Later, Marx (1983a,b) suggested that the underlying problem in osteoradionecrosis is a compromised wound-healing rather than an infection. Furthermore, osteoradionecrosis is as much a disease process of the covering soft tissues as that of the underlying bone (Epstein et al., 1987a,b, 1997; Constantino et al., 1995; Curi and Dib, 1997; Thorn et al., 2000). According to Marx (1983a,b), the sequence in the development of osteoradionecrosis is:
radiation; hypoxic-hypovascular-hypocellular tissue: the ability of bone to replace normal collagen loss or normal cellular loss is severely compromised or non-existent; tissue breakdown: unrelated to micro-organisms but related to the degree of radiation damage and the rate of normal or induced cellular death (Collagen lysis and cell death exceed synthesis and cellular replication.); and chronic non-healing wounds: energy, oxygen, and metabolic demands exceed the supply.
Conceptually, spontaneous and trauma-induced osteoradionecrosis are different entities. Spontaneous osteoradionecrosis, which has been reported to occur in almost 35% of all cases of osteoradionecrosis, is related to increased age, high radiation dose (> 65 Gy), field of radiation (volume of the mandible included in the field and proximity of maximal dosing to bone), hyperfractionation, use of implant sources too close to the bone, and combined interstitial and external beam irradiation (Murray et al., 1980a; Marx, 1983a,b; Marx and Johnson, 1987; Kluth et al., 1988; Constantino et al., 1995; Glanzmann and Grätz, 1995; Curi and Dib, 1997; Tong et al., 1999; Thorn et al., 2000). It represents a greater outright cellular kill of normal tissue elements, and an inability of soft and hard tissue to sustain cell turnover and collagen synthesis. This type of necrosis usually occurs within the first 2 years after radiotherapy (Marx, 1983a,b; Marx and Johnson, 1987; Thorn et al., 2000), but it can occur at any time following irradiation (Epstein et al., 1997; Thorn et al., 2000). However, late cases mostly occur as a result of trauma (Thorn et al., 2000).
Trauma-induced osteoradionecrosis represents a mixture of cell death and cell injury. As the years pass after irradiation, the tissue becomes more fibrotic and more hypovascular. If the tissue is traumatized by surgical procedures (e.g., extractions) or by persistent infection, it is suddenly required to meet the demands of wound healing. The reduced healing capacity may result in osteoradionecrosis—a risk which increases with time (Marx and Johnson, 1987; Constantino et al., 1995; Curi and Dib, 1997; Thorn et al., 2000). Several pre- and post-irradiation factors may increase the risk of osteoradionecrosis. Pre-irradiation extraction followed by inadequate healing time is known to predispose to osteoradionecrosis (Marx and Johnson, 1987; Constantino et al., 1995; Tong et al., 1999). In dentulous patients, the osteoradionecrosis risk is increased after radiotherapy if there is a trauma in the radiation field, such as tooth removal or other surgical procedures (periodontal procedures, biopsies), poor oral hygiene and inadequate home care, and ongoing periodontal or periapical infection (Murray et al., 1980b; Marx and Johnson, 1987; Epstein et al., 1997; Tong et al., 1999). In edentulous patients, trauma induced by prosthetic appliances is regarded as a predisposing factor (Dreizen et al., 1977b), especially when related to certain mastication and parafunctional habits (Marunick and Leveque, 1989). However, the use of implants can minimize the trauma induced by prosthetic appliances. To date, no cases of osteoradionecrosis related to dental implants have been reported. In both the irradiated mandible and the maxilla, the placment of implants seems to be a reliable procedure, at least in the short term (Esser and Wagner, 1997; Niimi et al., 1998).
In summary, osteoradionecrosis is a very unfortunate complication of head and neck radiotherapy that may result in loss of a significant volume of bone, mostly mandibular. Prevention of this condition has to be achieved by all means, since its treatment is difficult and time-consuming and causes much discomfort to the patient (Vissink et al., 2003).
Muscles and Joints
Trismus, or limited jaw opening, may develop due to tumor invasion of the masticatory muscles and/or the temporomandibular joint (TMJ), or be the result of radiotherapy if masticatory muscles and/or the TMJ is included in the field of radiation, or a combination of both (Steelman and Sokol, 1986; Ichimura and Tanaka, 1993; Dahllöf et al., 1994a; Goldstein et al., 1999). The limited jaw opening interferes with oral hygiene, speech, nutritional intake, examination of the oropharynx, and dental treatment, and can be particularly discomforting to the patient.
Trismus occurs with unpredictable frequency and severity. Generally, trismus develops three to six months after radiation treatment is completed and frequently becomes a lifelong problem (Ichimura and Tanaka, 1993). Trismus is attributed to muscle fibrosis and scarring in response to radiation injury as well as to fibrosis of the ligaments around the TMJ and scarring of the pterygo-mandibular raphes (Steelman and Sokol, 1986; Dahllöf et al., 1994a). Besides tumor growth and surgical procedures, the severity of trismus is dependent on the configuration of the radiation field (unilateral or bilateral), the radiation source, and the radiation dose (Wollin et al., 1976; Goldstein et al., 1999). It has been reported that trismus develops after high radiation doses to the TMJ only (Ichimura and Tanaka, 1993), while other authors reported that trismus may already develop after low doses and increases with increasing doses (Dahllöf et al., 1994a; Goldstein et al., 1999). The most decisive factor which determines whether trismus will develop is probably the inclusion of the pterygoid muscles in the treatment portals (Goldstein et al., 1999). This may explain the differences observed among the various studies reported in the literature.
Nutritional Status
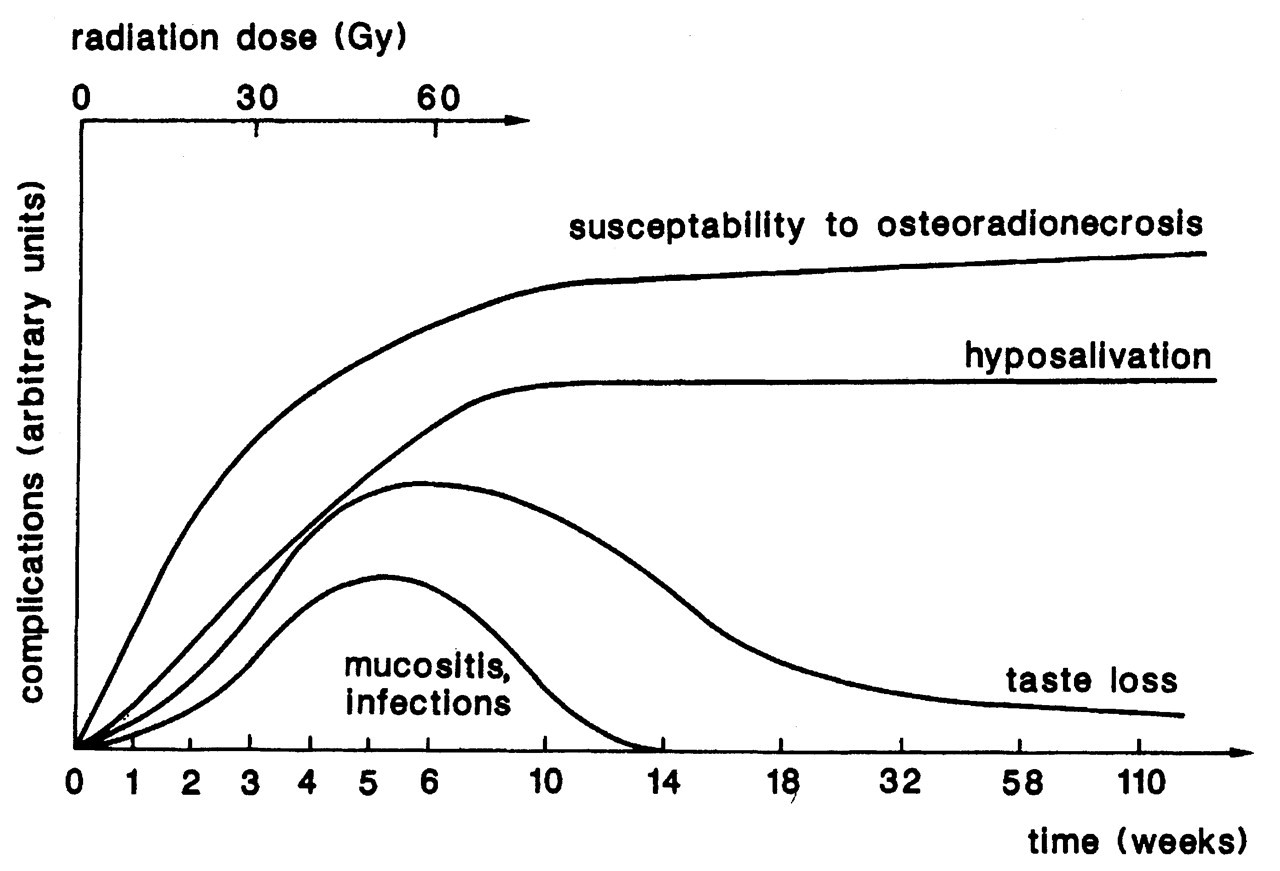
Several studies have shown that up to 60% of head and neck cancer patients were nutritionally compromised at initial diagnosis (Donaldson and Lenon, 1979; Bassett and Dobie, 1983; Wood et al., 1989; Bäckström et al., 1995; Lees, 1999; Van Bokhorst-van der Schueren et al., 2001). A pre-operative weight loss of 10% of body weight has been reported as a predictive risk factor for major post-operative complications (Van Bokhorst-van der Schueren et al., 1997). During radiotherapy, oral intake of food may be impeded due to mucositis, loss of taste acuity, hyposalivation, and changes in viscosity of saliva. Fig. 2 outlines the time frame involved in the development of each particular problem. Pain during chewing and swallowing due to mucositis or yeast stomatitis which predisposes the patient to lose appetite, nausea, and malaise may further decrease the nutritional status and result in significant weight loss (Beumer et al., 1979a,b; Bassett and Dobie, 1983; Logemann et al., 2001). The more frequent use of intensive chemoradiotherapy in head and neck cancer exacerbates this problem, since swallowing dysfunction is prevalent after such therapy (Eisbruch et al., 2002). Donaldson and Lenon (1979) reported that their patients lost about 3.7 kg during the course of radiation therapy, while Beumer et al. (1979a,b) reported weight losses of 7-11 kg not to be uncommon. In general, it can be stated that a 10% loss of body weight is not uncommon following head and neck radiotherapy (Lees, 1999). In severe cases of weight loss, enteral nutrition either by, e.g., a nasogastric tube or a percutaneous endoscopic gastrostomy (PEG) may become necessary (Magne et al., 2001; Mekhail et al., 2001). Patients often prefer a PEG rather than a nasogastric tube, but it has been reported that a PEG is often required for longer periods of time and is associated with more persistent dysphagia and an increased need for pharyngo-esophageal dilatation (Mekhail et al., 2001). These observations need further study. Weight loss leads to weakness, inactivity, discouragement, anorexia, and susceptibility to infection. It has been postulated that patients with a good nutritional and emotional status have improved tumor response to both radiotherapy and chemotherapy (Copeland et al., 1979), but this hypothesis still needs to be validated (Van Bokhorst-van der Schueren et al., 2001). In addition, the early and late morbidity of radiation treatment is less in patients who are in good health (Van Bokhorst-van der Schueren et al., 1997). It is therefore of the utmost importance that a good nutritional and positive emotional status be maintained in the head and neck receiving radiotherapy.
Epilogue
The early and late sequelae of head and neck radiotherapy have a large impact on the quality of life (Vissink et al., 1987; Epstein et al., 1999; Ohrn et al., 2001). Although the pathogenesis of many of the oral sequelae of head and neck radiotherapy is not fully understood, the radiation-induced side-effects often can be reduced with appropriate prevention and/or treatment. Notwithstanding the reduction of these side-effects that currently can be achieved, many patients will continue to experience these side-effects to some extent. In addition, appropriate prevention and treatment of both early and late radiation morbidity also become increasingly important, because more and more patients with head and neck cancer will be cured. In a separate review, the prevention and treatment considerations are discussed (Vissink et al., 2003).
Flow rate of parotid and submandibular-sublingual (SM/SL) saliva as a function of time after start of radiotherapy (conventional fractionation schedule, 2Gy per day, 5 days per week, total dose 60-70 Gy). The parotid, submandibular, and sublingual glands are located in the treatment portal. Initial flow rates were set to 100% (modified after Burlage et al,. 2001). Schematic diagram illustrating time of onset and duration of radiation-induced oral sequelae (modified after Maxymiw and Wood, 1989).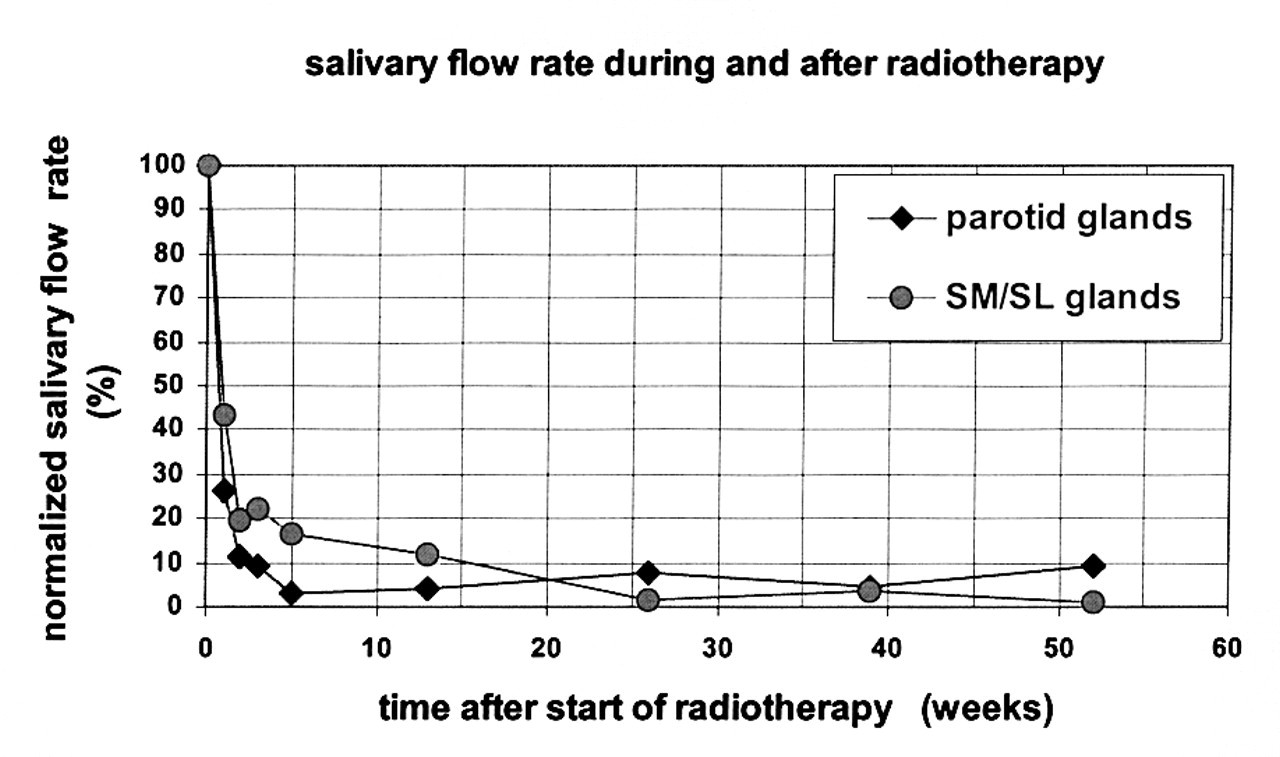
Footnotes
Acknowledgements
Support for the studies performed at our institutes was obtained from Praeventiefonds (grant 28-1290), the Dutch Cancer Foundation Koningin Wilhelmina Fonds (grants GUKC 83-20, RRTI 84-08, GUKC 86-03, GUKC 88-01, RUG 93.504, RUG 98.1658, RUG 02.2673), and the fellowship program of the Royal Dutch Academy of Arts and Sciences (1987-1992).
