Abstract
Mast cells are mobile granule-containing secretory cells that are distributed preferentially about the microvascular endothelium in oral mucosa and dental pulp. The enzyme profile of mast cells in oral tissues resembles that of skin, with most mast cells expressing the serine proteases tryptase and chymase. Mast cells in oral tissues contain the pro-inflammatory cytokine tumour necrosis factor-α in their granules, and release of this promotes leukocyte infiltration during evolving inflammation in several conditions, including lichen planus, gingivitis, pulpitis, and periapical inflammation, through induction of endothelial-leukocyte adhesion molecules. Mast cell synthesis and release of other mediators exerts potent immunoregulatory effects on other cell types, while several T-lymphocyte-derived cytokines influence mast cell migration and mediator release. Mast cell proteases may contribute to alterations in basement membranes in inflammation in the oral cavity, such as the disruptions that allow cytotoxic lymphocytes to enter the epithelium in oral lichen planus. A close relationship exists among mast cells, neural elements, and laminin, and this explains the preferential distribution of mast cells in tissues. Mast cells are responsive to neuropeptides and, through their interaction with neural elements, form a neural immune network with Langerhans cells in mucosal tissues. This facilitates mast cell degranulation in response to a range of immunological and non-immunological stimuli. Because mast cells play a pivotal role in inflammation, therapies that target mast cell functions could have value in the treatment of chronic inflammatory disorders in the oral cavity.
Introduction
Mast cells are mobile, bone-marrow-derived, granule-containing immune cells that are found in all connective tissue and mucosal environments, and in the peripheral and central nervous systems (Purcell and Atterwill, 1995; Silver et al., 1996). Following activation by immunologic or non-immunologic stimuli, mast cells release via their granules a range of pre-formed mediators, including cytokines, vasoactive amines, and enzymes. At the light microscope level, the secretory granules of mast cells give a characteristic metachromatic staining pattern with toluidine blue. Typical histological sections stained with toluidine blue give the impression that mast cells are rounded (Fig. 1); however, when they are examined by transmission electron microscopy or immunohistochemistry, a more stellate or dendritic character becomes evident (Kaminer et al., 1991; Walsh et al., 1991c).
Mast cells are found in all tissues of the oral cavity, including the dental pulp (Farnoush, 1984; Walsh et al., 1995). In dental pulp, their presence is more difficult to detect, because tissue injury and neuropeptide release during removal of the dental pulp elicits degranulation, making the cells difficult to see by traditional staining approaches based on histochemistry (Farnoush, 1984). In contrast, both immunohistochemical detection of mast cell granule proteases and transmission electron microscopy are able to demonstrate degranulated mast cells that would otherwise elude detection (Kaminer et al., 1991).
Mast Cell Mobility
In oral mucosa and skin, mast cells are distributed preferentially about the microvascular bed, close to the basement membranes of blood vascular endothelial cells and nerves (Walsh et al., 1990a,c) (Fig. 2). This localization of mast cells in both normal and inflamed sites results from their interaction with the laminin component of neural and vascular basement membranes via the α6/β1 integrin (CD49f), that serves as a specific laminin receptor (Walsh et al., 1991a; Zhao et al., 1998). Because adhesion of mast cells to laminin-containing basement membranes in vivo is influenced by down-regulation of this receptor during degranulation, mast cells should be recognized as a mobile cell population, capable of migration within tissues. Direct assessment of mast cell positioning in normal and inflamed tissues has demonstrated the plasticity of the mast cell network (Walsh et al., 1991a; Zhao et al., 1997). The physical movement of degranulating mast cells away from endothelia and away from laminin has been documented (Walsh et al., 1991a), and in this context, the ability of mast cell proteases to induce stromelysins that degrade laminin (Welle, 1997) has direct relevance.
A key mediator that influences mast cell migration is mast cell growth factor (MGF). This molecule can be synthesized by endothelial cells (Weiss et al., 1995) and epidermal keratinocytes (Kligman and Murphy, 1996), and is thought to direct the homing of mast cell precursors to epithelial tissues. Of note, MGF expression is not altered by degranulation of mast cells (Weiss et al., 1995). Alterations in the production of mediators such as MGF and RANTES (regulated on activation normal T-cell expressed and secreted) may underlie diseases such as urticaria pigmentosum, in which mast cell traffic is altered (Lazarus et al., 1991), resulting in intense accumulation of mast cells in the dermis of the skin (Mirowski et al., 1990; Walsh et al., 1991a).
The significance of the distribution of mast cells in tissue compartments relates to the potential for mast-cell-derived mediators to influence nearby cells, with resulting stimulatory, inhibitory, or toxic effects. Because degranulation of mast cells can be induced by a range of stimuli (Table 1), mast cells provide a common pathway for the development of inflammation both in the oral cavity and in other sites.
Of particular importance in this regard is the contribution of neuropeptides. As will be discussed further below, the neuropeptides substance P and calcitonin gene-related peptide (CGRP) stimulate mast cell degranulation and cause cytokine release in vitro, and such mediators are implicated in the ongoing degranulation that is a feature of chronic inflammation at both the light (Walsh et al., 1995) and electron microscope levels (Jontell et al., 1986). There is strong evidence for functional interactions between mast cells and nerves (Blennerhassett et al., 1991; Walsh et al., 1991b; Marshall and Waserman, 1995; Theoharides, 1996), and spatial interactions between mast cells and nerves have been characterized in skin (Church et al., 1989; Walsh et al., 1991a; Murphy, 1993; Pujic et al., 1998), oral mucosa (Walsh et al., 1990c; Zhao et al., 1997; Bartold et al., 2000), and in the respiratory, urogenital, and gastrointestinal tracts (Heine and Forster, 1975; Skofitsch et al., 1985; Bienenstock et al., 1987; Stead et al., 1989; Ferrante et al., 1990; Myers et al., 1991; Undem et al., 1995).
Mast Cell Mediators
Following degranulation, mast cell mediators are deposited in large quantities in the extracellular environment, where they exert effects on endothelial cells and other cell types. Mast cells may subsequently synthesize and secrete additional mediators that are not pre-formed in their granules. Key mediators that are pre-formed in mast cells are the serine proteases tryptase, chymase, and cathepsin G (Irani et al., 1986), histamine, heparin, serotonin (Soter and Austen, 1976), acid hydrolases (Stevens and Austen, 1989), and the cytokines tumor necrosis factor-α (TNF) (Walsh et al., 1991b) and interleukin-16 (Galli, 1993) (Fig. 3). Following activation, mast cells can synthesize a range of mediators, including the interleukins IL-1, IL-3, IL-4, IL-5, IL-6, IL-8, IL-10, IL-13, and IL-16, together with granulocyte-macrophage colony-stimulating factor (GM-CSF), platelet-activating factor (PAF), RANTES, macrophage inflammatory protein (MIP-1α), and the arachidonic acid metabolites prostaglandin 2 and leukotriene C4 (reviewed in Galli, 1993; Sutton and Gould, 1993; Kobayashi et al., 2000; Zhao et al., 2002b). Autocrine and paracrine cytokine regulatory networks influence mast cell synthesis of mediators. For example, IL-5 provides an autocrine influence by promoting the secretion of TNF, IL-5, IL-13, MIP-1α, and GM-CSF after mast cell degranulation, without increasing histamine release (Ochi et al., 2000), while TNF can increase mast cell secretion of IL-8 (Abraham and Arock, 1998).
Each mast cell typically contains between 80 and 300 granules (Eady, 1976). When activated, mast cells may either undergo explosive degranulation and then re-synthesize their granules (Chakravarty, 1983), or they may release solitary granules into their environment on an ongoing basis, a process termed ’piecemeal degranulation’ that has been observed in both the oral mucosa (Walsh et al., 1995) and skin (Kaminer et al., 1991). Even under conditions of rapid, anaphylactic degranulation, some granules within mast cells show gradual ’piecemeal’ release, with micro-vesicular transport of pieces of granules to the cell surface (Kaminer et al., 1995).
Ultrastructurally, the granules in mast cells of the oral mucosa and skin have a complex form, with amorphous regions located next to crystalline regions. The latter show a range of configurations, such as scrolls, lattices, and gratings (Kaminer et al., 1991). Immuno-electron microscopy has confirmed that the key mediators of chymase, tryptase, TNF, and cathepsin G are packaged separately within the granules (Walsh et al., 1991b; Whitaker-Menezes et al., 1995).
Mast Cell Proteases
The historical distinction of mast cell populations in tissues into "connective tissue" and "mucosal" phenotype relates to their tinctorial properties, which reflect the nature of the charged molecules found within mast cell granules. The expression of mast-cell-specific serine proteases (tryptase and chymase) is the current "gold standard" for determining mast cell phenotypes in various tissue compartments (Irani et al., 1986; Kaminer et al., 1991). Thus, "mucosal" mast cells with granules containing scroll-like structures are reactive with antibodies directed against tryptase but not chymase (MCT), whereas "connective tissue" mast cells with granules containing lattice or grating-like structures express both tryptase and chymase (MCTC).
These two mast cell subpopulations show important differences with respect to intracellular stores of mediators, responses to secretagogues, and sensitivities to cytokines that promote mast cell differentiation. Thus, an evaluation of mast cell phenotype is critical to an understanding of the contributions of mast cells in different sites. In human oral mucosa and dental pulp, the predominant mast cell population is MCTC (Walsh et al., 1990c, 1995). MCTC in general contain stores of histamine and TNF that exceed those of their MCT counterparts (Walsh et al., 1990c).
Tryptase is a trypsin-like proteinase that is found in the granules of both mast cells and basophils (Schwartz et al., 1985, 1987; Craig et al., 1988). Major functional activities of mast cell tryptase include serving as a mitogen for epithelial repair, and facilitating the recruitment of granulocytes by inducing epithelial cell secretion of IL-8 (Caughey, 1994; Welle, 1997). Mast cell tryptase can contribute to connective tissue breakdown by activating pro-collagenase directly (Gruber et al., 1988), and by setting in motion a cascade of matrix metalloproteinases (MMP) by activating prostromelysin-1 to stromelysin (Gruber et al., 1989). Breakdown of the connective tissue matrix induced by mast cell tryptase may facilitate the infiltration of tissues by leukocytes during the development of inflammation.
Chymase is a mast-cell-specific mediator that is not found in basophils. Chymase can function as an IL-1β convertase, cleaving inactive precursor IL-1β to yield the active molecule (Kupper et al., 1990). This has direct relevance to inflammation in terms of mucosal inflammation, where the IL-1β precursor may be secreted by activated keratinocytes (Walsh et al., 1990b,c). Chymase also contributes to a negative feedback control mechanism for neurogenic activation of mast cell secretion, since chymase (but not tryptase) can degrade substance P, CGRP, and other neuropeptides, and thereby interrupt axon-reflex-mediated neurogenic inflammation (Caughey et al., 1988; Welle, 1997).
Mast-cell-derived chymase and tryptase are thought to play a role in the breakdown of basement membrane structural proteins in conditions such as oral lichen planus (Zhou et al., 2002). Enzymatic disruption of the epithelial basement membrane may facilitate the migration of cytotoxic CD8+ T-lymphocytes into the epithelium through breaks in type IV collagen created by tryptase (Zhao et al., 2002b). Both tryptase and chymase activate matrix metalloproteinase 3 (MMP-3), and indirectly activate pro-collagenase (pro-MMP-1) (Welle, 1997). Since MMP-3 is a stromelysin that cleaves type IV collagen and laminin, enhanced expression of MMP-3 in inflammatory lesions will facilitate degradation of the extracellular matrix (Gruber et al., 1989; Lees et al., 1994). Direct evidence exists for enhanced expression of MMP-3 in the epithelium overlying degranulated mast cells in oral lichen planus (Zhou et al., 2001). Mast-cell-derived TNF has also been implicated as a major stimulant of T-lymphocyte secretion of the gelatinase MMP-9 in the same condition (Zhou et al., 2001). MMP-9 cleaves type IV collagen, which again would facilitate the migration of leukocytes between tissue compartments.
Changes in epithelial basement membranes induced by mast cell proteases may have an impact on the population of Langerhans cells (LC) in the overlying epithelium. Phenotypic transformation of dermal macrophages to LC in vivo has been documented (Murphy et al., 1986), and the reconstitution of epidermal LC from dermal cells requires focal disruptions of the epidermal basal lamina (Murphy et al., 1985). As will be discussed further below, the process of development of LC from macrophage precursors can be promoted by TNF (Sueki et al., 1993; Anasthanas-Platsis et al., 1995), again implying a contributory role for mast cells in this process.
Mast Cell Histamine
Mast-cell-derived histamine mediates important changes in vascular permeability via structural changes in the microvascular endothelium, such as endothelial contraction and intercellular gap formation (Majno and Palade, 1961). In addition, histamine promotes the adhesion of platelets by mobilizing the adhesion molecule P-selectin (CD62P, GMP-140) from Weibel-Palade bodies to the luminal surfaces of endothelial cells (Johnson et al., 1989). In a recently developed microplate histamine assay, direct evidence for histamine release from oral mucosal mast cells has been presented (Zhao et al., 2001).
Mast-cell-derived histamine is responsible for wheal formation in the classic triple response. The effects of histamine in this response are amplified, but not mimicked, by mast cell chymase, via a pathway involving the H1 histamine receptor (Rubinstein et al., 1990). This suggests that when mast cells of the MCTC phenotype degranulate, the vasoactive effects of histamine may be modulated by the chymase that is delivered from the same mast cell granules.
Mast Cell TNF
Considerable attention has been directed toward the contribution of mast-cell-derived TNF in inflammation. Mast-cell-derived TNF induces endothelial expression of E-selectin (CD62E, ELAM-1) (Walsh et al., 1991b). Studies using neutralizing antibodies have shown that for the "connective tissue"-type mast cells (MCTC) that predominate in the oral cavity and skin, TNF is the sole mediator responsible for the induction of E-selectin (Walsh et al., 1991b). E-selectin is critical for the rapid adhesion of neutrophils, T-lymphocytes, monocytes, and other leukocytes to endothelial cells (Bevilacqua, 1993).
In oral inflammatory lesions, degranulation of mast cells can be identified by externalization of granules and depletion of intracellular stores of mediators, as determined by toluidine blue staining and chymase immunohistochemistry. At the ultrastructural level, the pattern of granule enlargement, lucency of granule matrices, and fusion of granules is typical of chronic or piecemeal degranulation (Jontell et al., 1986; Walsh et al., 1995).
Chronic synthesis and release of TNF from mast cells may maintain leukocyte migration and promote chronicity in inflammatory lesions, as has been shown in oral lichen planus (Walsh et al., 1990c, 1995). TNF exerts modest chemotactic effects on neutrophils; however, its major effects are mediated through its effects on adhesion molecule expression. Through the latter effects, mast cell TNF can orchestrate the migration of neutrophils and T-lymphocytes into tissues (Walsh et al., 1990a; Waldorf et al., 1991). A clear causal relationship among mast cell degranulation, TNF release, and the development of inflammation in the oral mucosa and skin has been documented in vitro, by both immunological and non-immunological stimuli (Klein et al., 1989; Walsh et al., 1991b; Walsh, 1995), and the same pathways have been demonstrated in vivo (Waldorf et al., 1991; Walsh and Murphy, 1992a; Walsh, 1995). These studies provide a clear explanation and a molecular mechanism for earlier observations that linked mast cell degranulation with the initiation of inflammation and the development of chronic inflammation (Jontell et al., 1986; Kowalski and Kaliner, 1988).
In addition to the stimulatory effects of mast-cell-derived TNF on endothelium, TNF exerts a range of effects on other cell types (Table 2). A major target of interest in the oral cavity is the keratinocyte. At low concentrations, TNF can arrest the growth of keratinocytes, while high concentrations are cytotoxic (Philip and Epstein, 1986; Walsh et al., 1990c; Sugerman et al., 1996). Prolonged release of TNF from mast cells has been implicated in stem cell damage and epithelial injury in oral lichen planus (Walsh et al., 1990c) and chronic graft-vs.-host disease (Murphy et al., 1994). Because TNF can induce cytotoxic T-lymphocyte differentiation (Robinet et al., 1990), and can enhance the cytotoxicity mediated by monocytes and other cell types (Ostensen et al., 1987), mast-cell-derived TNF can play an important role in augmenting cytotoxic responses that are mediated by other cell types. While mast cells and basophils are the only cells capable of storing and releasing pre-formed TNF (Walsh et al., 1991b), it is important to bear in mind that other cell types in inflammatory lesions can make considerable contributions to local TNF production. Such cells include macrophages, Th1 T-lymphocytes, endothelial cells, and keratinocytes (Walsh et al., 1995; Sugerman et al., 1996).
A second major target for mast-cell-derived TNF is the T-lymphocyte. Recent studies have demonstrated that, in oral lichen planus, TNF induces lesional T-cells to secrete the chemokine RANTES, which then is able to stimulate mast cell degranulation, with the accompanying secretion of histamine (Zhao et al., 2002a). This positive feedback mechanism provides an explanation for how T-lymphocytes in the infiltrate are able to promote a chronic pattern of mast cell degranulation and TNF secretion (Walsh et al., 1990c, 1995). It has been suggested that the expression of RANTES and its receptor (CCR1) on populations of mast cells and T-lymphocytes may prolong the survival of these cell types and thus promote the development of chronic inflammation (Zhao et al., 2002a). Nevertheless, because of the plethora of cytokines and chemokines that can serve as paracrine signals, inflammatory reactions that are driven by interactions between mast cells and T-lymphocytes have a high degree of redundancy, and a contribution from other mediators is likely.
In addition to the influences described above, mast-cell-derived TNF can exert a range of effects on other cells (Table 2). Of immediate interest in the context of inflammation in the oral cavity are the following TNF effects: activation of Langerhans cells (LC) (Ishii et al., 1990; Ioffreda et al., 1993), stimulation of LC precursors (Anasthanas-Platsis et al., 1995), activation of perivascular dendritic cells (Walsh et al., 1991c; Sueki et al., 1993), and inhibition of epithelial cell proliferation (Walsh et al., 1990c; Sugerman et al., 1996). Thus, chronic synthesis and release of TNF from mast cells can be recognized as an important factor in promoting chronicity of inflammation. While these pathways have been studied in most detail for oral lichen planus, there is opportunity to apply similar concepts to other inflammatory conditions of the oral mucosa and skin (Walsh and Murphy, 1992a; Sugerman et al., 2000).
Mast Cell IL-1 and IL-4
Activated mast cells can secrete both Interleukin-1 (IL-1) and Interleukin-4 (IL-4), although neither is present as a pre-formed mediator in MCTC (Burd et al., 1989; Klein et al., 1989). Since activation of mast cells (following degranulation) is implicated as a stimulus for mast cell IL-1 production, mast-cell-derived chymase, which is deposited in the extracellular environment following degranulation (Kaminer et al., 1991; Waldorf et al., 1991), is placed ideally to activate released IL-1β.
IL-1 can be synthesized by macrophages and monocytes in response to endotoxin from Gram-negative anaerobes, such as those implicated in pulpal and periapical pathology (Walsh et al., 1989). Because such synthesis and release take from 6 to 24 hours to occur, IL-1 secreted by macrophages (or activated mast cells) is unlikely to serve as a "primary" mediator in the chronological sense, in that its effects will likely follow those of TNF. It should be noted that TNF is more potent and results in more rapid and stronger expression of E-selectin than IL-1 (Pober et al., 1985; Bevilacqua, 1993). In the dental pulp, release of TNF from mast cells would be a key early event in eliciting a neutrophil influx into the dental pulp. However, at subsequent timepoints, IL-1 secreted by mast cells or macrophages would contribute to sustained activation of the microvascular endothelium.
IL-4 secreted by activated mast cells is likely to influence inflammation in terms of the progression from acute to chronic inflammation. IL-4 primes endothelium for adhesion of lymphocytes and monocytes, and suppresses adhesion of neutrophils (Patel, 1999; Kotowicz et al., 2000). These changes are mediated by fluctuations in the adhesion molecule repertoire of endothelial cells, viz. suppression of E-selectin and P-selectin, and induction of vascular cell adhesion molecule 1 (VCAM-1, CD106) (Walsh and Murphy, 1992a; Gemmell et al., 1993; Patel, 1999). Because CD106 binds strongly to lymphocytes, but only weakly to monocytes and not to granulocytes (Osborn et al., 1989), chronic synthesis and release of IL-4 from mast cells would promote a transition from acute to chronic inflammation.
Other molecules involved in leukocyte adhesion to endothelial cells include CD31 (PECAM-1) and CD54 (ICAM-1). These molecules stabilize leukocyte attachment to endothelia to permit trans-endothelial migration (Bevilacqua, 1993). Fluctuations in the expression of these molecules in inflammation have been observed and have been linked potentially with release of cytokines including TNF, IL-1, and IL-4 from mast cells and other cell types (Walsh and Murphy, 1992a; Gemmell et al., 1993).
Interactions between Mast Cells and Other Immune Cell Types
Through the release of mediators, mast cells can exert potent chemotactic and stimulatory effects on other cell types, such as macrophages and neutrophils (Ming et al., 1987). Likewise, mast-cell-derived mediators may affect connective tissue cells and extracellular matrix, and thereby influence wound healing and repair (Walsh and Murphy, 1992a). Interactions between mast cells and other cell types are reciprocal, in that certain mediators produced by endothelial cells and "resident" perivascular macrophages, viz. interleukins 1 and 8, can potentiate or inhibit mast cells’ histamine release.
Antigen Presentation
An important interaction between mast cells and other cell types is that of antigen presentation to T-lymphocytes. Mast cells express both MHC class I and class II molecules (Love et al., 1996; Malaviya et al., 1996), as well as the CD80 (B7-1), CD86 (B7-2), and CD54 molecules that serve as the second signal for T-lymphocyte activation during antigen presentation (Frandji et al., 1996). They are also able to process endogenous viral and bacterial antigens for MHC class-I-restricted presentation to T-cells (Malaviya et al., 1996). While mast cells are not ’professional’ antigen-presenting cells, the antigen presentation and co-stimulatory signals delivered by mast cells may contribute to the development of a specific T-lymphocyte response in the induction phase of inflammation, in conditions such as oral lichen planus (Zhao et al., 2002b). Mast cells share a strategic perivascular location with dendritic antigen-presenting cells (Walsh et al., 1991c), and their production of cytokines in this location may be equally as important as the expression of accessory molecules on their cell surfaces.
Mast cells are able to contribute to the innate immune response to microbial infection, and this is of critical importance in the response to helminths. Recent studies have revealed that mast cells can bind a range of bacteria in the gastrointestinal tract in the absence of antibody or other host-derived opsonins (reviewed in Abraham and Arock, 1998). Mast cells can phagocytose and kill microbes, and also present bacterial antigens. Despite the importance of these roles in the gastrointestinal tract, they appear to be of little or no relevance in the oral cavity.
Mast Cell Influences on T-lymphocytes
Because mast cells have a long lifespan and can produce a large panel of cytokines, it is plausible that they could positively influence the survival of T-lymphocytes. This is of particular relevance given the perivascular clustering of lymphocytes that occurs in many inflammatory lesions in the oral mucosa (Wynne et al., 1988). Mast-cell-derived IL-6 and TNF have been shown to provide alternative co-stimulatory survival signals to CD8+ T-lymphocytes, in the absence of the usual B7-CD28 and IL-2 co-stimulatory pathways (Sepulveda et al., 1999). This positive influence on survival is thought to have direct relevance to disease chronicity in oral lichen planus (Zhao et al., 2002b), and may explain the limited T-cell receptor repertoire of lesional T-lymphocytes in that condition (Zhou et al., 1996).
Mast cells are able to exert an influence on the migration of T-lymphocytes at several junctures: the endothelium (via enhanced adhesion molecule expression), the extracellular matrix (directly through proteases, and indirectly through the induction of gelatinases and stromelysins), and within tissue compartments (via chemotaxis). In addition to mast cell IL-8 (Moller et al., 1993), mast-cell-derived lymphotactin can serve as a chemo-attractant for CD8+ T-lymphocytes, and mast-cell-derived IL-16 is a chemoattractant for CD4+ T-lymphocytes (reviewed in Zhao et al., 2002b). Anchorage signals for T-lymphocytes can be provided by mast cell CD54, RANTES, and TNF (Franitza et al., 2000).
Mast Cells and the Neural-immune Network
As already mentioned, there is a well-known spatial affinity between mast cells and nerves in mucosal tissues of the gastrointestinal tract (Stead et al., 1989), mesentery (Skofitsch et al., 1985), and airways (Undem et al., 1995), as well as in the bronchial ganglia (Myers et al., 1991) and meningeal membranes (Ferrante et al., 1990). Close approximations between mast cells and nerves have been documented in human skin (Walsh et al., 1991a; Pujic et al., 1998), human oral mucosa (Walsh et al., 1990c; Zhao et al., 1997), and rat palatal mucosa (Matsson et al., 1995).
The results of studies that have failed to find any mast-cell/nerve interactions in normal buccal mucosa (Ruokonen et al., 1993) can be explained by technical factors, in that such studies used toluidine blue staining to identify mast cells. This method is less sensitive than the immunohistochemical localization of mast cells by anti-tryptase antibodies, and underestimates the total number of mast cells as well as the number of degranulated mast cells (Zochodne et al., 1994). The preferred method in the author’s laboratory is a sensitive double- or triple-labeling immunohistochemical technique that allows for the co-localization of two or three antigens in frozen tissue sections (Zhao et al., 1997; Pujic et al., 1998), since this is believed to result in an accurate assessment of mast-cell/nerve interactions. Useful markers to reveal the delicate neural plexus include neurofilaments, neural cell adhesion molecule, neuropeptides, and nerve-cell-specific clathrin (reviewed in Egan et al., 1998).
The spatial association of nerves and mast cells facilitates the effects of neuropeptides that are secretagogues for mast cells. In an organ culture system, neuropeptides found in dental pulp nerves (substance P [SP] and calcitonin gene-related peptide [CGRP]) have been shown to cause degranulation of dental pulp mast cells, and thereby to elicit the induction of E-selectin and the up-regulation of CD54 on post-capillary venular endothelium in the sub-odontoblastic plexus (Gentner et al., 1996; Thomas and Walsh, 1997). SP and CGRP often co-exist in nerve fibers (Gibson et al., 1984; Lundberg et al., 1986) and frequently are released simultaneously (Saria et al., 1986; Payan et al., 1984). As well as inducing degranulation of mast cells (Piotrowski and Foreman, 1986), CGRP inhibits the degradation of SP (Le Greves et al., 1985).
SP and CGRP are important mediators in neurogenic inflammation, and both are present in considerable quantities in nerves within the oral cavity (Wakisaka, 1990). SP is associated with numerous biological events, ranging from bronchoconstriction, nociception, and plasma extravasation to neurogenic inflammation. In the dental pulp, stimulation of type A nerve fibers results in increased pulpal blood flow and permeability of microvessels. This increased blood flow occurs predominantly via axon reflexes rather than by the direct action of vasomotor nerves. Antidromic stimulation leads to release of SP, which promotes vasodilation and inflammation. In contrast, CGRP plays a lesser role in neurogenic inflammation in the oral cavity than in the skin (Payan, 1992).
A major pathway for SP-mediated inflammatory responses is the mast cell. For exploration of the actions of SP on mast cells in human dental pulp, a useful experimental approach has been to obtain tissues from extracted third molars, and maintain these in short-term organ culture in the presence of SP or SP agonists. SP agonists include septide ([pGlu6, Pro9]-substance P [6-11], an NK-1 selective agonist) and senktide (succinyl-[Asp6, N-Me-Phe8]-substance P [6-11], an NK-3 selective agonist). In these studies, endothelial E-selectin expression was found to be increased following treatment with either SP or septide, whereas in senktide-treated pulp tissue, expression was very weak, and was not different from that in control samples maintained in medium alone. This indicates that the SP/mast-cell/endothelial response of E-selectin expression is mediated through the NK-1 receptor expressed on dental pulp mast cells, rather than by other classes of tachykinin receptor (Thomas and Walsh, 1997). This is consistent with the known role of SP and mast cells in neurogenic inflammation in the skin (Matis et al., 1990; Payan, 1992).
It has been suggested that mast cell degranulation in response to release of neuropeptides is a key event in the immunopathogenesis of oral lichen planus (Walsh et al., 1990c). Formal analysis of spatial interactions between mast cells and nerves in normal oral mucosa and oral lichen planus by double-labeling techniques indicates that the density of mast cells is greater by two-fold in lichen planus than in normal buccal mucosa. Moreover, the frequency of spatial associations between nerves and mast cells is twice as high in lichen planus (Zhao et al., 1997). The most superficial region of the lamina propria is the area with the highest number of interactions with nerves, as predicted earlier (Walsh et al., 1990c).
Useful parallels can be drawn between the physical arrangement of mast cells in the sub-odontoblastic plexus of the dental pulp and their positioning in the lamina propria of the oral cavity and in the superficial dermis of the skin. In all three sites, the mast cells form a network that places them close to neural elements (’upstream’) and to post-capillary venular endothelium (’downstream’). The downstream events (release of TNF, histamine, serine proteases, etc.) have been studied intensively for many years (Walsh et al., 1990a; Walsh and Murphy, 1992a), and the phenomenon of activation of endothelial cells induced by mast cells has been characterized in detail (Klein et al., 1989; Matis et al., 1990; Walsh et al., 1991b).
More recently, attention has turned to the ’upstream’ side, specifically how mast cells may interface with other cells to form a ’neural-immune network’ (NIN). That such a network should exist in oral mucosa and skin is not surprising, given that close apposition between immune cells and nerves has been shown in other tissues—for example, in the spleen (Felten et al., 1987). The genesis of the NIN concept was mapping studies that used multiple labeling to determine the spatial interactions between mucosal and cutaneous nerves and cells of the immune system. Close approximations of vertically ascending, unmyelinated nerve fibers with mast cells and Langerhans cells (LC) were demonstrated, initially in human skin (Walsh and Murphy, 1992b; Murphy, 1993; Hosoi et al., 1993; Walsh et al., 1996; Pujic et al., 1998) and subsequently in primate and murine skin (Egan et al., 1998) and human oral mucosa (Bartold et al., 2000). Of note, these nerves did not come into contact with stem cells, Merkel cells, or melanocytes, but rather were confined to the LC, immune surveillance cells that form a complex three-dimensional network by means of their extensive dendrites that traverse intercellular spaces between keratinocytes. These studies confirmed that the formation of spatial interactions of mast cells and LC with these nerve fibers was not a chance phenomenon. Both mast cells and LC are highly mobile cell populations, while nerves are stationary elements within tissues. The existence of trophic effects was suggested by the alignment of newly arrived LC and mast cells with these same nerve fibers (Walsh et al., 1996). The occurrence of neuropeptides such as CGRP, both in nerve fibers and on the cell surface of LC, is functionally relevant, as demonstrated by effects of CGRP on LC function (Hosoi et al., 1993; Asahina et al., 1995).
The dynamic and specific nature of the nerve-LC interaction was demonstrated by two different experimental approaches: by studying regeneration of the epithelial LC-neural plexus following ablation of LC during allogeneic bone marrow transplantation in humans (Walsh et al., 1996), and by evaluating changes in the expression of protein gene product 9.5 by epidermal LC following transection of the sciatic nerve in rats, and its subsequent re-innervation (Hsieh et al., 1996).
The same nerves that come into contact with LC were shown to form close associations with mast cells (MCCT) (Walsh and Murphy, 1992b; Murphy, 1993; Egan et al., 1998; Pujic et al., 1998), and this raises the intriguing issue of the function of the LC/mast-cell connection, in the context of immune surveillance.
Several investigations have shed light on this issue. Studies of the sunburn response from short-wavelength ultraviolet B (UVB) radiation have revealed that UVB, both in vitro and in vivo, induces degranulation of cutaneous mast cells, with release of TNF and serine proteases, and subsequent induction of E-selectin on microvascular endothelium (Walsh, 1995). Removal of the epidermis and the use of dermal explants maintained in organ culture confirmed that all TNF released was derived from mast cells in the superficial dermis (Walsh, 1995). UVB is also known to impair the induction phase of delayed-type hypersensitivity (DTH) in particular strains of mice by a mechanism triggered by cis-urocanic acid, which is dependent upon TNF (Bacci et al., 1996). Indeed, cis-urocanic acid has been shown to elicit mast cell degranulation, TNF release, and the consequential expression of E-selectin on endothelium (Wille et al., 1999). TNF has been shown to alter LC surface antigen expression (Ishii et al., 1990), and to alter the LC cytoskeleton by reducing the expression of vimentin (Bacci et al., 1996). More recently, TNF has been shown to regulate the migration of LC from epithelia, causing them to accumulate in draining lymph nodes (Cumberbatch and Kimber, 1992; Cumberbatch et al., 2001). Thus, TNF released from mast cells in the response to acute UVB irradiation is able to alter the function not only of the endothelium, but also of the overlying LC network. Mast-cell-driven changes in connective tissue elements such as fibroblasts have also been described with UVB irradiation (Kligman and Murphy, 1996).
A second insight into the functional interplay between mast cells and LC has been gained from studies of DTH in humans. LC serve as the first line of responder cells in oral mucosal and cutaneous inflammation. Elegant studies of ultrastructural events in DTH have demonstrated the ability of LC to serve as a "messenger" between skin and lymph nodes (Silberberg et al., 1976). Uptake of antigens or haptens by LC is known to be an early event in this reaction, and occurs within 1-2 hours of challenge (Shelley and Juhlin, 1976).
The important possibility exists that when LC interact with antigen, the release of SP or CGRP in the lamina propria or dermis elicits mast cell degranulation, with the consequential events of TNF release and endothelial activation (Murphy, 1993). Mast cell degranulation, as witnessed by ultrastructural alterations and discharge of pre-formed mediators, also occurs in the first two hours following challenge (Waldorf et al., 1991; Walsh et al., 1991b). Mast cells are the only dermal cell type to display ultrastructural alterations at this juncture. In the ensuing four-hour period, expression of E-selectin on microvascular endothelium can be observed, and "pioneer" CD4+/CD45RO+/CD29+ T-lymphocytes begin to form perivascular aggregates (Waldorf et al., 1991). Mast cells, which have depleted their stores of TNF by four hours post-challenge, subsequently express enhanced levels of TNF mRNA, and resynthesize this pre-formed mediator during the ensuing 24 hours (Walsh et al., 1991b).
It is noteworthy that while these changes precede clinical evidence of inflammation, they are frequently accompanied by urtication, a sign tentatively related to neuropeptide release and mast cell degranulation (Murphy, 1993). More importantly, the key observation from these studies—that mast cells degranulate at a time when hapten is still within the epidermis—has until recently lacked any cogent explanation. The existence of a "neural immune network" (NIN) which links epidermal "surveillance" cells (LC) and dermal regulatory cells (mast cells) provides an explanation for coordinated LC and mast cell responses in delayed-type hypersensitivity.
Additional proof of the functional role of the NIN was provided by studies that compared mast-cell-driven responses in human skin in vivo, with an intact NIN, with full-thickness human skin grafted to immuno-deficient SCID mice. The latter lacked the unmyelinated axons of the NIN, but otherwise had intact mast cell and endothelial cell responses (Christofidou-Solomidou et al., 1996). Mast cell degranulation with chymase release and endothelial expression of E-selectin were used as markers of NIN function. Topical application of capsaicin, an agent that causes explosive release of substance P and other neuropeptides from neural elements, to forearm skin elicited mast cell degranulation and E-selectin induction, whereas these events failed to occur in xenografts treated with capsaicin (Egan et al., 1998). From the foregoing, it can be concluded that neuropeptide release within an intact NIN exerts a functional role in regulating inflammation in the skin and oral mucosa.
Therapeutic Implications
Based on the concept that mast cells play a critical role in the induction of inflammation, it is logical to use therapeutic agents to alter mast cell function and secretion, to thwart inflammation at its earliest phases. For cutaneous inflammation, this has been accomplished in vitro and in vivo with the mast cell stabilizing agent disodium cromoglycate and a related compound, proxicromilin (Klein et al., 1989; Walsh et al., 1990c; Walsh, 1995). Abrogation of inflammation by first purging the challenge site of granulated mast cells with the secretagogue compound 48/80 or by depletion of neuropeptide (substance P, CGRP) stores with the agent capsaicin has also been reported (Payan et al., 1984). Blockade of dental pulp mast cell responses in vitro has been achieved with neutralizing antibodies to TNF (Gentner et al., 1996), and with substance P receptor antagonists (Thomas and Walsh, 1997). Similarly, neutralizing anti-TNF antibodies have been shown to abrogate mast-cell-driven graft-vs.-host disease in both murine and human systems (Herve et al., 1992; Murphy et al., 1994).
An additional stratagem is the use of soluble receptors to inactivate the TNF released from mast cells. The p55 and p75 soluble TNF receptors can compete with cell-bound receptors for binding TNF, and can bind directly to TNF to block its biological activity (Engelmann et al., 1990; Leeuwenberg et al., 1994). Accordingly, it has been suggested that soluble TNF receptors applied locally may abrogate the pro-inflammatory effects of TNF (Sugerman et al., 1996).
Corticosteroids have been utilized for decades in the treatment of inflammatory conditions of the dental pulp and oral mucosa. An important property of corticosteroids that may contribute to their therapeutic efficacy is their ability to deplete mast cells locally at sites of prolonged or occlusive application (Lavker and Schechter, 1985). Corticosteroids have been used successfully to deplete mast cells in the treatment of the cutaneous mast cell disease urticaria pigmentosum (Barton et al., 1985); however, the side-effects of intense therapy with topical steroids (such as epithelial atrophy) limit the usefulness of this therapeutic approach (Walsh et al., 1990a).
Conclusions
Mast cells serve a critical role in the development of inflammation in the oral mucosa and the dental pulp, both in the early, vaso-inductive events and in the transition from acute to chronic inflammation. Because of the unique properties of mast cells, these cells are ideally poised to serve as "gatekeepers" of the microvasculature in the oral cavity. An appreciation of the multiple interactions among mast cells, endothelial cells, nerves, and other cells of the immune system provides a basis for therapies for targeting mast cell responses. In the future, it may be possible to develop novel approaches that influence the release of pro-inflammatory molecules or neuropeptides to ameliorate mast-cell-driven inflammation in the oral mucosa and dental pulp. The recent identification of spatial interactions between nerves and both mast cells and LC in a neural-immune network opens new possibilities for altering the function of these critical immune cells.
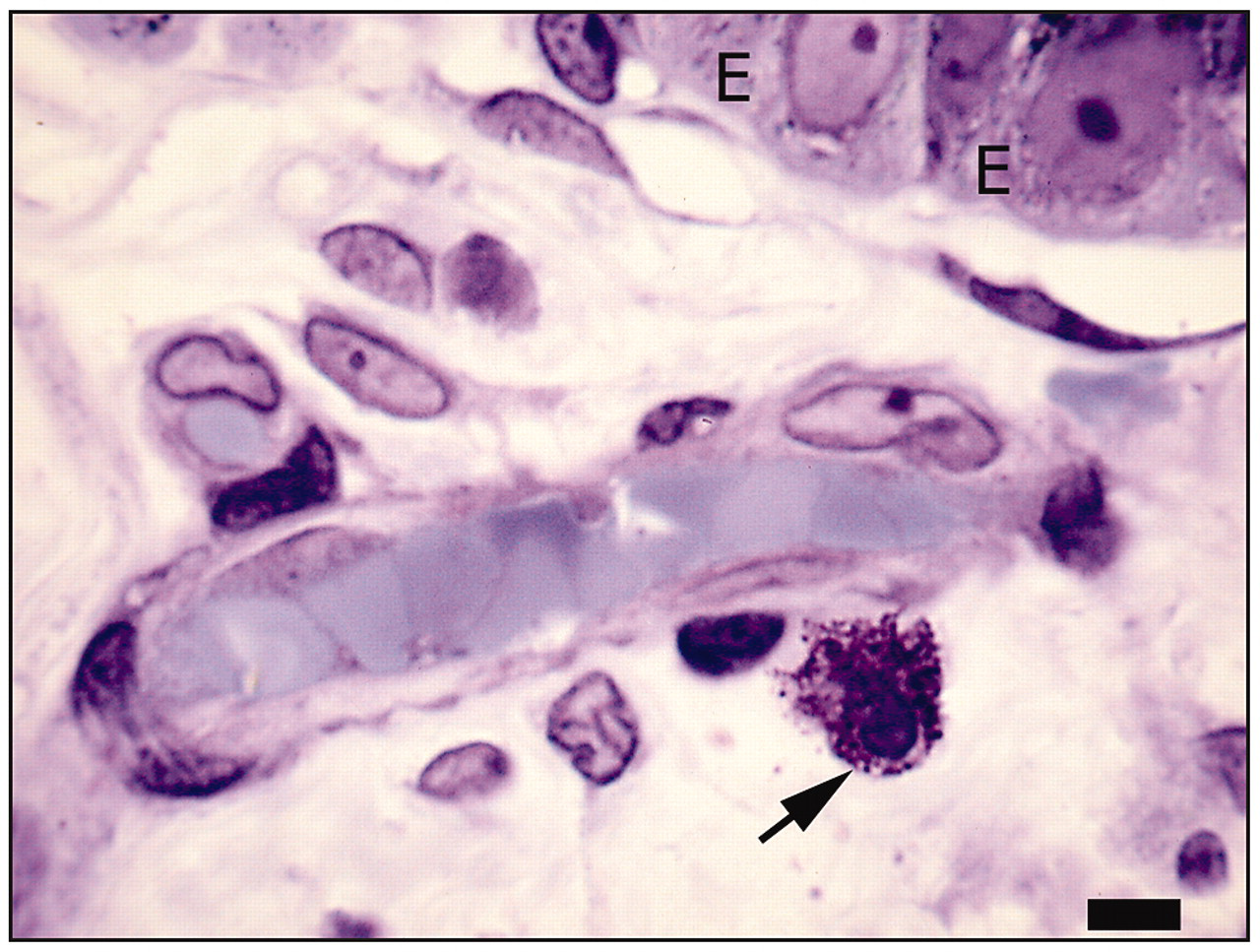
Characteristic morphology and perivascular location of a mast cell (arrow), as seen in a one-micrometer-thick resin section stained with toluidine blue. The mast cell is close to the abluminal surface of the endothelium of this post-capillary venule. The basal epithelial layer (E) is seen in the upper right. Original magnification X1000. Scale bar indicates 5 μm.
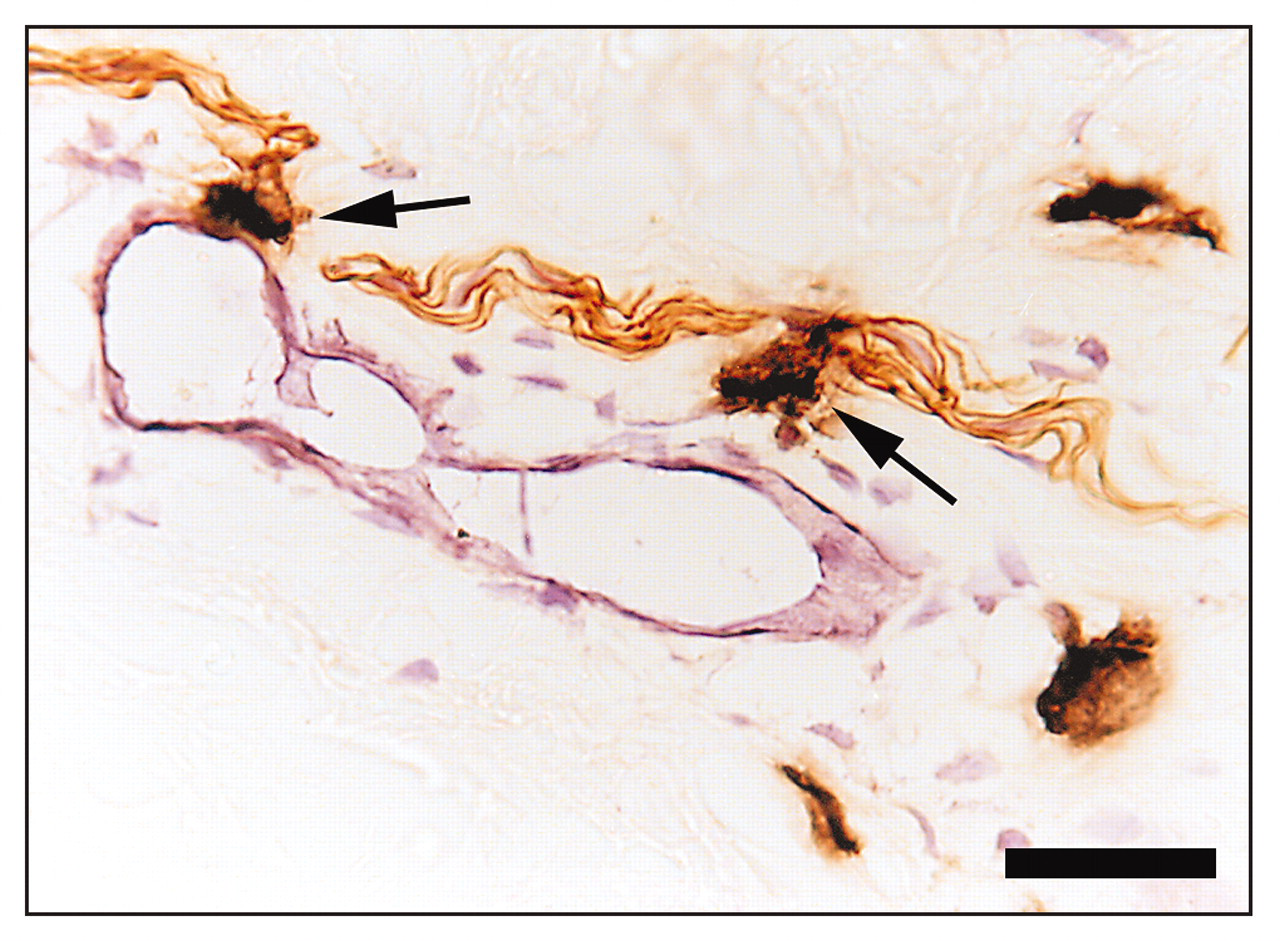
Triple-immunoperoxidase-labeled frozen section showing the relationship among mast cells, blood vessels, and nerves. Peripheral nerves stained with antibodies to neurofilament protein are brown, while the CD31 (PECAM-1) adhesion molecule on the luminal surface of the blood vessel is stained pink. Tryptase in mast cells is stained black. Several mast cells are present in the section. Two of these (indicated by arrows) are juxtaposed between nerves and blood vessels. Original magnification X400. Scale bar represents 20 μm.
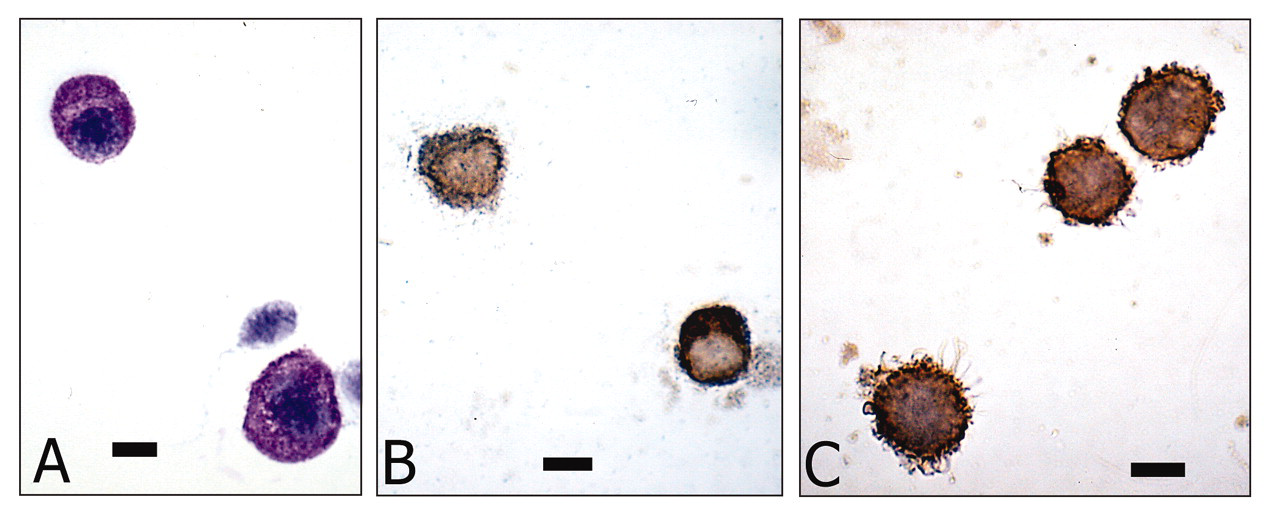
Stained cytocentrifuge preparations of purified mast cells, showing metachromatic staining of the granules with toluidine blue
Footnotes
Acknowledgements
The research work of the author’s laboratory has been supported by grants from the National Health and Medical Research Council of Australia and the Australian Dental Research Foundation. I thank Neil Savage, Zhen Zhen Zhao, Zac Pujic, Mark Bartold, and Philip Sugerman for their contributions to the mast cell biology research program over several years. Particular thanks are due to George Murphy, with whom some of the key concepts presented in this paper were originally developed.
