Abstract
Fluoride is a well-known G protein activator. Activation of heterotrimeric GTP-binding proteins by fluoride requires trace amounts of Al3+ or Be2+ ions. AlFx mimics a γ-phosphate at its transition state in a G α protein and is therefore able to inhibit its GTPase activity. AlFx also forms complexes with small GTP-binding proteins in the presence of their GTPase-activating proteins (GAP). As phosphate analogs, AlFx or BeFx affect the activity of a variety of phosphoryl transfer enzymes. Most of these enzymes are fundamentally important in cell signal transduction or energy metabolism. Al3+ and F- tend to form stable complexes in aqueous solution. The exact structure and concentration of AlFx depend on the pH and the amount of F- and Al3+ in the solution. Humans are exposed to both F and Al. It is possible that Al-F complexes may be formed in vivo, or formed in vitro prior to their intake by humans. Al-F complexes may play physiological or pathological roles in bone biology, fluorosis, neurotoxicity, and oral diseases such as dental caries and periodontal disease. The aim of this review is to discuss the basic chemical, biochemical, and toxicological properties of metallic fluoride, to explore its potential physiological and clinical implications.
Introduction
In the biochemical and cellular fields, aluminum fluoride (Al-F complexes, or AlFx) and beryllium fluoride (BeFx) are widely used to interfere with the activity of many enzymes. It is well-known that they can activate G proteins in eukaryotic cells (Gilman, 1987). AlFx and BeFx are small inorganic molecules that mimic the chemical structure of a phosphate (Bigay et al., 1987). As phosphate analogs, they affect the activity of phosphoryl transfer enzymes, such as GTPases, ATPases, phosphohydrolyases (Chabre, 1990), and phospholipase D (Li and Fleming, 1999a,b). Phosphoryl transfer is the fundamental mechanism underlying energy metabolism and signal transduction in cells (Knowles, 1980). As unique chemical tools in biological studies, AlFx and BeFx have been successfully used in the study of the structures and catalytic mechanisms of enzymes involved in phosphoryl transfer (Petsko, 2000; Thompson and Cole, 2001). A variety of such enzymes and their complexes with AlFx or BeFx have been crystallized. Some examples include: different classes of GTP-binding proteins (GTPases) (Sprang, 1997), myosin ATPases (Maruta et al., 1993), H+-translocating F1-ATPase (Braig et al., 2000), and phosphoserine phosphatase (Cho et al., 2001).
In addition to their role in basic biochemistry, Al-F complexes also have effects on human physiology. Chemical studies show that Al3+ binds F- more strongly than 60 other metal ions (Martin, 1988). Al is the most abundant metal on earth. It is ubiquitously present in all foodstuffs and drinking water (Macdonald and Martin, 1988). In contrast, only a μM level of Al is needed to form biologically effective Al-F complexes (Sternweis and Gilman, 1982). Fluoride is widely added to human drinking water (1 ppm) and in most toothpastes (500-1500 ppm) to prevent dental caries (Warren and Levy, 1999). In BC3H1 myocytes, AlFx was found to activate the MAP kinase pathway, which is the pivotally important pathway in cell proliferation (Anderson et al., 1991). It is well-known that fluoride affects bone physiology (Farley et al., 1983). It is prescribed as a drug to treat osteoporosis (Ringe and Rovati, 2001). Recent research strongly indicates that AlFx might be the active species that affects bone cell biology in vivo (Caverzasio et al., 1998; Susa, 1999). Exposure to fluoride induces asthmatic symptoms among workers in the aluminum industry, and recent studies indicate that this might also be due to AlFx (Refsnes et al., 1999). One study reported that chronic ingestion of 0.5 ppm AlFx in the drinking water induced neurohistological changes in experimental rats (Varner et al., 1998). Misra et al. (2002) recently reported that 1-5 nM beryllium fluoride and a μM level of AlFx induced peritoneal macrophage proliferation. The foregoing suggests that it is extremely important to understand the role of Al-F and Be-F complexes in human physiology, because of its potential effects on our health.
This review discusses the basic chemistry and biochemistry of Al-F complexes in relation to their potential physiological and toxicological implications. The first half of this paper is centered on the molecular mechanism of metallic fluoride on GTP-binding proteins. The second half of this paper is centered on the basic chemical and physiological processes of AlFx. Studies on the physiological relevance of AlFx in bone physiology, neurotoxicity, fluorosis, and oral diseases are discussed in detail. Questions are raised throughout this paper, particularly on the potential clinical implications of Al-F complexes and indications for future investigations.
Fluoride and the Discovery of G Protein
The concept of cell signaling dates back to the beginning of the last century, when Paul Ehrlich (Triggle, 2000) postulated a ’lock and key’ theory to explain the interaction between cell-surface receptors and their agonists. Five decades later, Sutherland’s lab first discovered that treatment of particulate fraction of dog liver homogenate with epinephrine and glucagon induced the formation of cyclic AMP (3′ 5′-adenosine monophosphate) from ATP (Rall et al., 1957). An enzyme, later named adenylate cyclase (AC), was proposed to catalyze this reaction (Rall and Sutherland, 1958). In these studies, 10 mM sodium fluoride (NaF), in itself, was found to stimulate cyclic AMP production, as well as to potentiate hormonal effects. Fluoride was thus identified as a stimulator of adenylate cyclase, but its mechanism of action was unknown. The early hypothesis was that fluoride directly stimulates the catalytic activity of AC (Sutherland et al., 1962). Rodbell made the critical discovery that GTP is required for the hormonal activation of AC in fat cells (Rodbell et al., 1971). These investigators were the first to speculate on the potential importance of a GTP-binding protein in a hormonal-sensitive AC system. The existence of such a protein was later suggested by the demonstration that a guanine nucleotide binding component could be partially resolved from the putative catalytic subunit of adenylate cyclase by affinity chromatography with GTP-sepharose (Pfeuffer, 1977). It was soon established that adenylate cyclase was composed of at least two separate components, a catalytic component and a regulatory component, referred to as G/F (an early name for Gs protein), because it contained the activation sites for both a guanine nucleotide and fluoride (Ross and Gilman, 1977). Meanwhile, a murine S49 lymphoma cell line which bears a somatic mutation cyc - and lacks AC activity was isolated (Bourne et al., 1975). These S49 cells were found to lack the regulatory component G/F, but they retain the catalytic component. By taking advantage of this genetically mutated cell line, Ross and Sternweis developed a reconstituted system, in which hormone-, guanine nucleotide-, and fluoride-sensitive AC activity could be restored to S49 cell membranes by the addition of detergent extracts of G/F from various sources (Ross et al., 1978; Sternweis and Gilman, 1979). Subsequently, fluoride was shown to act on the guanine nucleotide-binding regulatory component of AC (Howlett et al., 1979; Downs et al., 1980). The stimulatory component of AC (Gs) was first purified to homogeneity from rabbit liver, owing to its ability to reconstitute fluoride-stimulated AC activity in S49 cell membranes (Northup et al., 1980; Sternweis et al., 1981). Cassel and Selinger (1977, 1978) demonstrated, in the turkey erythrocyte membrane system, that adenylate cyclase activation after catecholamine receptor stimulation was due to GTP binding and hydrolysis (i.e., GTPase reaction) by Gs.
Fluoride Action on G Proteins is Associated with Al3+ Ions
The actual mechanism of fluoride action on guanine nucleotide-binding component of AC remained elusive. Experiments with detergent extracts of S49 plasma membranes suggested that activation of G/F by fluoride required Mg2+ and ATP, but the specificity of the requirement for ATP was not clear (Sternweis and Gilman, 1979). When these experiments were repeated with purified G/F from rabbit liver, the absolute requirement for ATP was shown to be somewhat erratic. The necessary ingredient donated by ATP did not appear to be the nucleotide (Sternweis et al., 1981). Both an extract from the glass test tubes or the use of tap water, rather than glass-distilled water, could substitute ATP and promote activation of G/F by fluoride. Sternweis persevered through these seemingly strange experimental phenomena and solved the puzzle (Sternweis and Gilman, 1982). He isolated the mysterious factor by passing ATP as well as the glass test tube rinses through a cation exchange resin column and eluted the factor with 3 M HCl. The factor purified by this procedure was subjected to elemental analysis by neutron activation, and it turned out to be Al3+ ions in both ATP and the glass tube rinses. Exogenous AlCl3 produced G/F activation in the presence of Mg2+ and fluoride. In addition, Al3+-free ATP failed to promote activation of G/F. In their assay system (which contained 10 mM Mg2+, 1 mM EDTA and 5 mM NaF), the Kact for Al3+ was about 4 μM. Martin (1986) later showed that 1 mM EDTA strongly chelates Al3+ ions. According to the calculation, this would leave 10-4 as much free Al3+ as the added total Al3+. However, Al3+ reacts with EDTA much more slowly than Al3+ and F-; therefore, effective amounts of Al-F complexes could still form (Martin, 1986). The specificity of the requirement for Al3+ was remarkable. Of a total of 28 metals tested, only beryllium was effective. Therefore, fluoride activation of G protein depends upon Al3+, which may also be substituted by Be2+ (for more information on their chemistry, see Fig. 1).
Aluminum is a minor structural component of glass. It usually exists in the form of alum or alumina in glass (Berry, 1979). mM fluoride etches Al3+ from glass into solution (Sternweis and Gilman, 1982). Aluminum is also a common contaminant of some commercial nucleotide products, such as ATP, which was shown to be ’necessary’ for fluoride action. Free Al3+ usually ’disappears’ into the sediment in the earth’s crust as a hydroxide, and is locked into minerals. However, with the advent of acid rain, aluminum escapes from mineral deposits, and a certain amount of free Al3+ is dissolved in fresh water (Martin, 1994). This is the most likely reason for the ’effectiveness’ of tap water vs. glass-distilled water in promoting the activation of G/F by fluoride.
The use of a controlled concentration of Al3+ plus fluoride (here referred to as AlFx) in a reconstituted system enabled the experiments to be more sensitive and stable. Activation of G/F by GTPγS and AlFx was found to protect the protein from thermal denaturation and chemical inactivation. Such activation allowed for the resolution of the active 45-KDa α subunit and the 35-KDa β subunit from each other by high-performance gel filtration (Northup et al., 1983a,b). The β subunit was later found to be associated with a smaller peptide of 8 KDa, named as the γ subunit (Hildebrandt et al., 1984). Analysis of these data permitted a definitive statement to be made on the activation mechanism of adenylate cyclase by AlFx and guanine nucleotide analogs. Gs protein is a heterotrimer, composed of α, β, and γ subunits. The binding of GTPγS or AlFx to the α subunit dissolves α and βγ. The activated and resolved α subunit itself was able to reconstitute AC activity. βγ heterodimer inhibited AC activity by re-associating with the α subunit. Moreover, incorporation of purified β-adrenergic receptor and Gs into phospholipid vesicles reconstituted hormone-stimulated GDP-GTP exchange on the α subunit and its GTPase activity (Asano et al., 1984). Recombination of the purified adenylate cyclase catalytic unit with Gs and the receptor reconstituted hormone-stimulated synthesis of cAMP (May et al., 1985). In essence, Gs protein mediates hormone stimulation of the effector adenylate cyclase, which catalyzes the production of the second-messenger cAMP. Other members of the G protein family have also been identified and characterized (for a review, see Gilman, 1987). Such G proteins include: Gi (the G protein which transduces inhibitory hormone regulation of AC) and Gt (also called transducin; see next section).
γ-Phosphate Analog Model
Transducin is a G protein with α, β, and γ subunits. It functions to mediate visual transduction by coupling the photoexcitation of rhodopsin to the stimulation of a cGMP phosphodiesterase in retinal rod cells (Fung et al., 1981). Transducin is structurally and functionally analogous to Gs and Gi. For a long time, fluoride was known to influence the activity of the cGMP phosphodiesterase system (Sitaramayya et al., 1977). Fluoride acts on the α-subunit of transducin (Stein et al., 1985). AlCl3 synergistically enhanced the effect of fluoride by promoting dissociation of Gt α from Gt βγ and inhibiting GTPase activity (Kanaho et al., 1985).
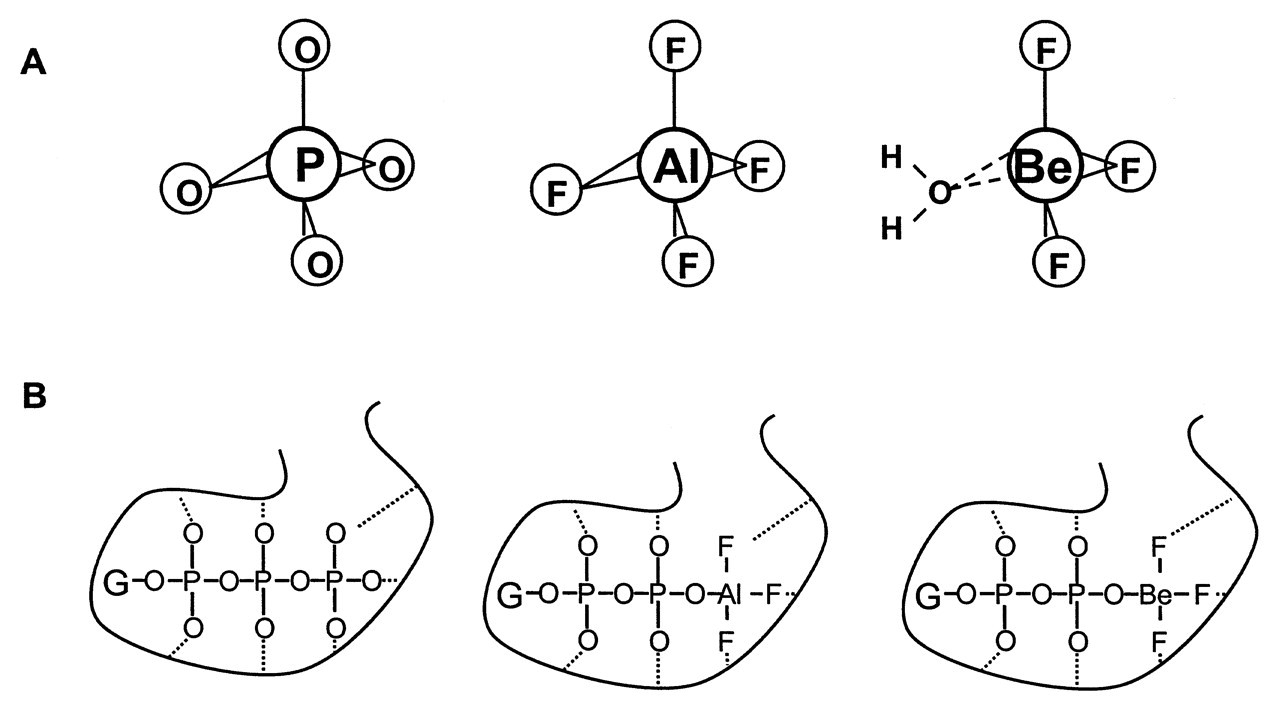
A breakthrough on the mechanism of AlFx on G proteins was achieved by Bigay and Chabre (Bigay et al., 1985, 1987). Their work on transducin focused on the point that, to allow Gt α activation by AlFx, a GDP is required to bind to the nucleotide site of Gt α. Analogs of GDP were accessible only if the terminal oxygen on the β-phosphate remains unsubstituted. AlFx was effective with GDPαS or GP-NH-P, but not with GDPβS, whose β-phosphate is chemically modified. This finding led them to conclude that AlFx interacts directly with the oxygen on the β-phosphate of GDP. This is the position where a γ-phosphate would bind if it were a GTP. They recognized in their experiments that maximum efficiency was achieved at a NaF concentration, where the predominant fluoroaluminate complex is AlF4 - in the solution (Goldstein, 1964). They also recognized the structural similarity between AlF4 - and PO4 -3 (Fig. 2A). Both complexes are tetrahedral. The Al-F bond length is very similar to that of the P-O bond in PO4 -3 (Bigay et al., 1985). This feature was further confirmed by the fact that BeF3 -, another complex of fluoride with a tetrahedral chemical structure similar to that of PO4 -3, fully mimicked the AlF4 - effect in this system (Bigay et al., 1987). Based on these recognitions, a γ-phosphate model was proposed to explain the AlF4 - effect on G proteins. Isolated G α protein normally keeps a GDP molecule permanently bound. AlF4 - interacts with G α protein through binding to the β-phosphate of GDP. The bound AlF4 - (or BeF3 -) simulates the presence of the bound γ-phosphate of GTP and therefore confers on the protein the structure of the active G α•GTP state. The high electro-negativity of F- allows F- to form strong hydrogen bonds with nearby amino acid side-chains. This tight bonding makes AlF4 - and BeF3 - non-hydrolyzable by the GTPase activity of G α, and thus maintains the G protein in its activated state (Fig. 2B).
Shortly after its proposal, the γ-phosphate analog model obtained immediate support from the structural studies on G α protein by tryptophan fluorescence and 19F and 31P NMR spectroscopy (Higashijima et al., 1987, 1991). Meanwhile, this model quickly expanded from the G protein system to other phosphoryl transfer enzymes, on which AlFx and BeFx were known to have an effect. AlFx and BeFx were thus recognized as a new group of phosphate analogs in the study of the enzymology of phosphoryl transfer enzymes (Chabre, 1990). Phosphoryl transfer reactions are catalyzed by enzymes such as ATPase, GTPase, protein kinases, phosphatases, nucleotidyl transferase, and phospholipase D (Knowles, 1980). The importance of AlFx or BeFx lies in that their complexes with these enzymes can be used to study the mechanistic aspects of phosphoryl transfer reactions (Lolis and Petsko, 1990). Existing experimental evidence indicates that the strictly tetrahedral BeF3 - complex is an analog of the phosphate in its ground state, whereas the trigonal bipyramidal or octahedral geometry of AlF3 or AlF4 - mimics a phosphate in its transition state (Wittinghofer, 1997; Thompson and Cole, 2001).
Al-F Complex Mimics a γ-phosphate at its Transitional State: Insight into the GTPase Mechanism
G protein is a superfamily of regulatory GTP hydrolyases. All members of this superfamily share a common structure core, which is exemplified by p21Ras. The G α subunits of the heterotrimeric G protein are also considered members of the Ras family of GTP hydrolyases. They are the enzymes that use the free energy of GTP hydrolysis to transduce signals from ligand-activated receptors to downstream effectors. The enzyme-product complex G α•GDP (the resting state) assumes an inactive signaling conformation, and continues to bind to Gβγ subunits. The enzyme-substrate complex G α•GTP (the ground state) assumes an active signaling conformation. It dissociates from Gβγ to bind to downstream effectors (Coleman and Sprang, 1999). The switch of these two functional states is regulated by the hydrolysis of GTP to GDP, which is catalyzed by the intrinsic GTPase activity of G α. Thus, GTPase catalyzed GTP hydrolysis is the central rate-controlling point of a G protein signaling pathway (Rodbell, 1997). AlFx activates a G protein signaling pathway by inhibiting the GTPase activity, thereby keeping the G α protein in its active signaling conformation. The popular statement that "AlFx activates G proteins" can sometimes be confusing. Its effects on other phosphoryl-transfer enzymes are also inhibitory. Early analysis of the stereochemical course of the P21Ras GTPase reaction showed that the hydrolysis of GTP occurs with inversion at the γ-phosphorus (Feuerstein et al., 1989). This indicates that the mechanism is most likely a single-step, in-line transfer, without forming a phosphoenzyme intermediate.
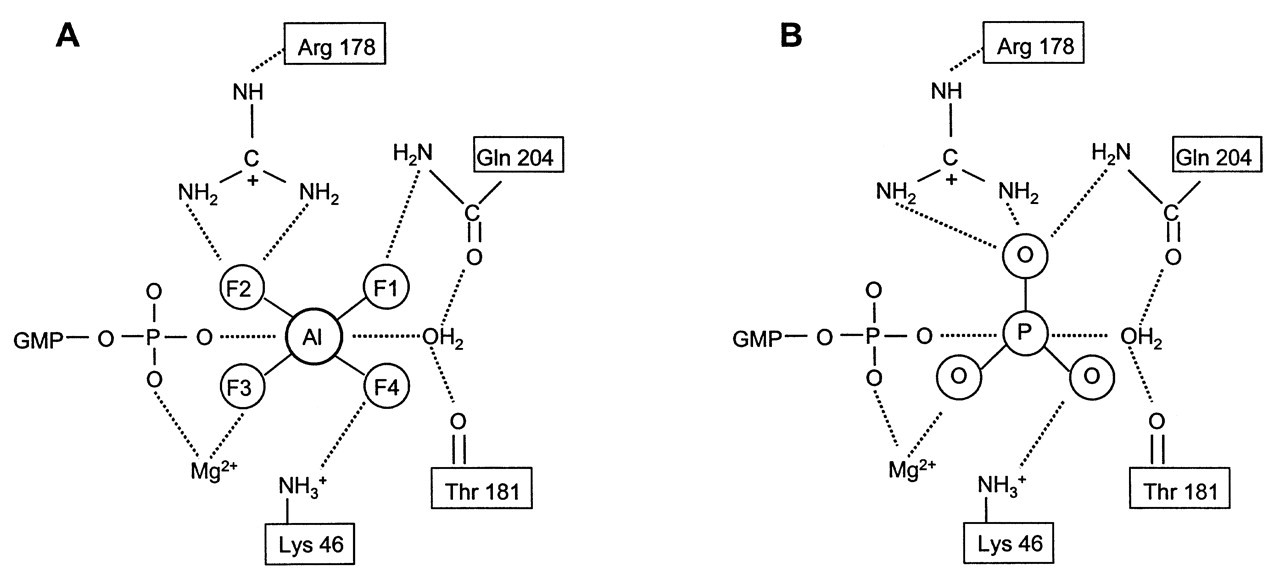
The breakthrough in the GTPase catalytic mechanism comes from the determination of the x-ray crystallographic structures of G α proteins. Giα and Gtα, coupled with GTPγS or GDP•AlF4 -, were among the first to be crystallized (Noel et al., 1993; Coleman et al., 1994; Sondek et al., 1994). The molecular structure of Giα•GTPγS•Mg2+ showed that this protein contains two general domains: one helical domain, which is unique to all trimeric G α proteins; and one Ras-like domain, which is typified by P21Ras. It contains switches I, II, and III. Switches I and II interact with Mg2+ and γ-phosphate, respectively, and switch III is unique to all trimeric G α subunits. A guanine nucleotide binds with the pocket formed by these two domains. Nucleotide makes direct contact only with the Ras-like domain, but is shielded from solvent by the helical domain. Two important residues within the active site—Gln204 and Arg178—are present in all G α family members. They are crucial for enzyme catalysis, since point mutation of either residue abolishes enzyme activity (Kleuss et al., 1994). It is therefore surprising that, in the Giα•GTPγS•Mg2+ ground state complex, none of these residues is in contact with either the nucleotide or the hydrolytic H2O. It appears that Gln204 and Arg178 are not involved in the binding of GTP and H2O to Giα. The Giα•GTPγS•Mg2+ complex provides a model of the ground state enzyme-substrate complex in its active signaling conformation. However, it provides little insight into the catalytic mechanism. It did show that the active site contains a well-coordinated H2O molecule that appears to be positioned for an in-line nucleophilic attack on the γ-phosphate group (Coleman et al., 1994).
The most revealing structure concerning enzyme catalysis was provided by the crystal structure of Giα•GDP•AlF4 -•Mg2+ (Coleman et al., 1994). This crystal structure is essentially identical to Giα•GTPγS•Mg2+, except near the region of the γ-phosphate-binding site. AlF4 - is located at the γ-phosphate-binding site and covalently linked to the β-phosphate. This confirmed the early γ-phosphate analog model (Fig. 3A). However, the central Al atom exhibits an octahedral, hexacoordinated binding geometry, rather than the earlier proposed tetrahedral orientation. Four fluorine atoms bind around aluminum in an equatorial plane (90° between each other). A GDP β-phosphate oxygen and a hydrolytic H2O molecule occupy the axial positions (perpendicular to the fluorine plane) (Fig. 3A). This structure confirmed an earlier proposal by Martin (1988) that, in aqueous solution, AlF4 - is not tetrahedral, but occurs as the hexacoordinate (H2O)2AlF4 -. The most important finding of this crystal complex is the change in positions of the catalytically important residues Gln 204 and Arg 178. The side-chains of both residues interact directly with GDP•AlF4 -•H2O through hydrogen bonds (Fig. 3A). An almost identical interaction was observed in GDP•AlF4 -•H2O complexed with transducin (Gtα) (Sondek et al., 1994). Their counterparts Gln200 and Arg174 assumed the same roles. The remarkable complementary nature of Giα and Gtα active sites to GDP•AlF4 -•H2O and the close resemblance of their structure with the described bipyramidal transition state of phosphate indicate that AlF4 - is a transitional state analog in both situations (Fig. 3B). Based on the crystallized analog structure, an approximate picture of the G α transition state can be reconstructed by replacing the AlF4 - moiety with a γ-phosphate (Fig. 3B). The detailed mechanism of Gln204 and Arg178 (Gln200 and Arg174 in Gtα) in stabilizing the phosphate transition state can be addressed (for details, see Sprang, 1997). In essence, the positions of the key residues re-orient during the catalysis to stabilize the reaction transition state, thus enabling the attacking nucleophile (H2O) to replace the leaving group (the oxygen linking to the β-phosphate) and resulting in GTP hydrolysis to GDP.
Al-F Complex and Small GTP-binding Proteins
In contrast to the trimeric G protein, another group of G proteins behaves differently toward AlFx. Small GTP-binding proteins are a superfamily of monomeric G proteins that are Ras-like. Major families include Ras, Rho, Rab, Rac, Arf, and Ral (Wittinghofer, 1998). These proteins play pivotal roles in the signaling of cell growth and differentiation. Their molecular weights are all in the range of 20-30 kDa (for this reason, they are also called small-molecular-weight G proteins). They are structurally similar to the a subunits of trimeric G proteins. Members of this family process consensus amino acid sequences responsible for GDP/GTP-binding and GTPase activities (called Ras-like domain). Kahn (1991) found that, in contrast to their trimeric counterparts, under the same conditions, Ras-like G proteins do not bind to AlFx, and their biochemical activities were not affected by AlFx. This was surprising, since the GTP-binding and hydrolysis mechanism is conserved among all G protein members, and the AlFx effect has been attributed to the guanine nucleotide hydrolysis. Since many cellular processes are regulated by both trimeric and monomeric G proteins, the response to AlFx has been used as an indication of trimeric G protein and not of monomeric G protein involvement. In contrast, guanine nucleotide analogs, such as Gpp-NH-p and GTPγS, stimulate both families of G proteins.
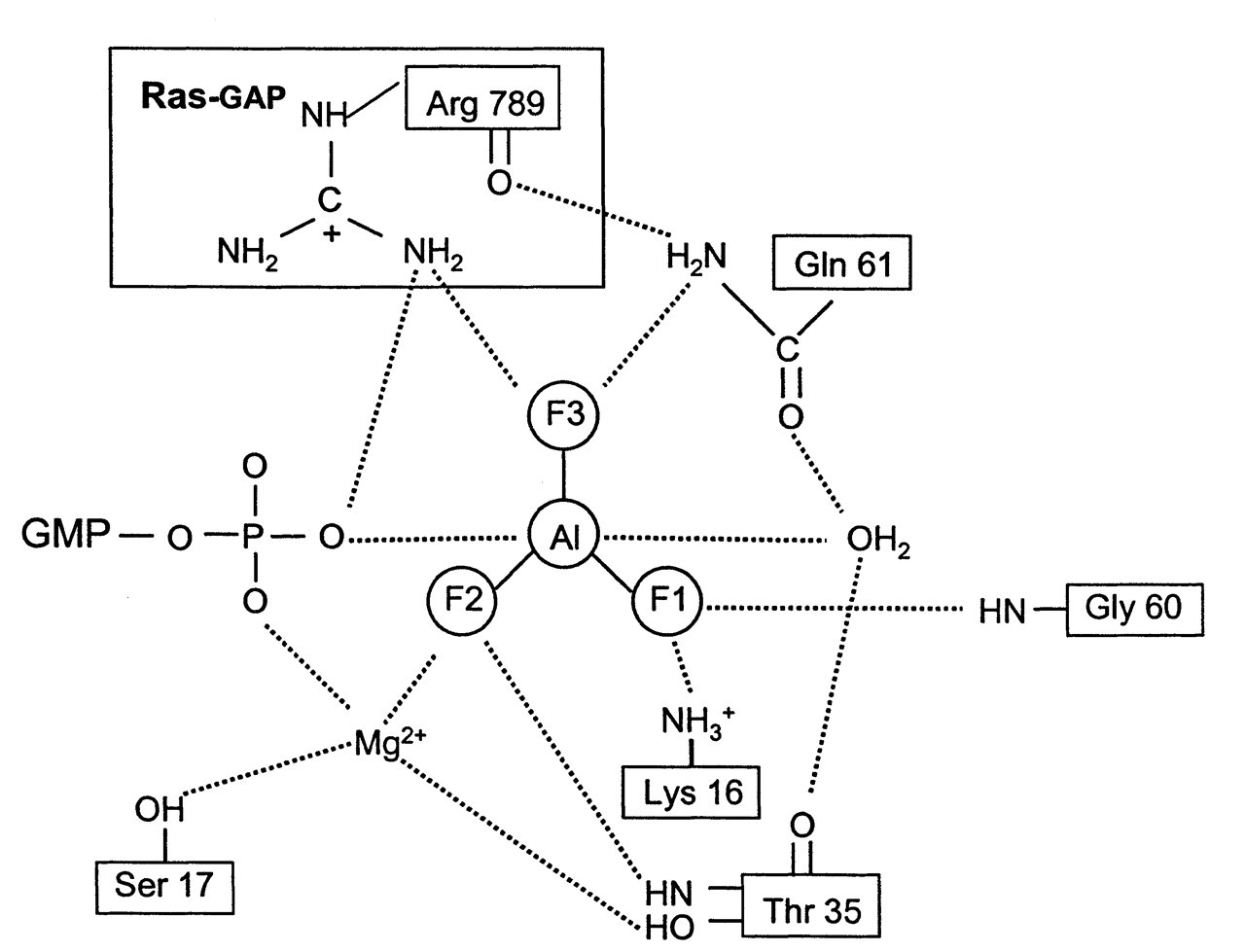
The Ras superfamily of monomeric G proteins has low intrinsic GTPase activity. Its hydrolysis rate is about 1/100 of that of an average G α protein (2-5 min-1) (Wittinghofer et al., 1997). Thus, Ras proteins require their GTPase activating proteins (GAP) to accelerate the GTP hydrolysis process. Two Ras-specific GAPs, the P120GAP and neurofibromin proteins1 (NF1), increase the hydrolysis rate by 105 (Scheffzek et al., 1998). Oncogenic Ras mutants have impaired GTPase activity, and, more importantly, they are insensitive to their GAPs; thus, oncogenic Ras remains bound with GTP. This overactive signaling conformation results in tumorigenesis. Point mutations in Ras genes have been found in 30% of all human tumors (Scheffzek et al., 1997). Trimeric G α protein and Ras proteins both contain the conserved Ras-like domain, which is responsible for GTP hydrolysis. This seems to imply a similar mechanism of GTPase catalysis for both families of G proteins. However, Kahn (1991) found that members of the Ras superfamily of small GTP-binding proteins do not bind to AlF4 -, indicating a significant difference in their active site structures. As reviewed in the last section, AlF4 - mimics the transition state of γ-phosphate in the G α protein. Prive et al. (1992) proposed that the GAP may induce the transition state formation by introducing an amino acid side-chain structure during GTP hydrolysis. Using a fluorescence emission spectrum, Mittal et al. (1996) first detected that, in the presence of a stoichiometric amount (1:1) of GAP (P120GAP or NF1) and Ras•GDP, Ras interacts with AlFx. The ternary complex Ras•GDP•AlFx•GAP was subsequently isolated by gel filtration (Ahmadian et al., 1997). Other members of the Ras superfamily, such as Cdc42, Rap, and Ran proteins, also form ternary complexes with AlFx in the presence of their respective GAP proteins. The crystal structure of the complex between Ras•GDP and GAP-334 (the catalytic fragment of P120GAP) with AlFx was resolved (Scheffzek et al., 1997) (Fig. 4). An arginine side-chain, Arg789 (the equivalent of Arg178 in Giα), in GAP-334 is positioned at the active site of Ras to neutralize the developing negative charges on the γ-phosphate during the transition state. This allows glutanine-61 (the equivalent of Gln204 in Giα) to participate in the catalysis. AlFx again mimics the γ-phosphate at its transitional state, which is stabilized by Ras and GAP interaction through the above mechanism (Fig. 4). In trimeric G α protein, an arginine of similar function is provided from the same molecule (Arg178 in Giα), rather than from a different molecule (Arg789 in P120GAP). Another difference observed in Scheffzek’s study (1997) was that an AlF3, instead of an AlF4 -, was bound to the active site (compare Fig. 3A with Fig. 4). The Al-F bonds in both situations are planar. The two axial bonds with β-phosphate oxygen and nucleophilic H2O were very similar. Rather than attaining an octahedral geometry in the case of Giα and Gtα, aluminum fluoride assumes a trigonal bipyramidal structure in Ras•GAP. This conformation more closely resembles the true transitional state of γ-phosphate (compare Fig. 3B with Fig. 4). The reason for these two different conformations was recently found to be caused by experimental conditions rather than to be due to any basic structural differences. By changing the pH value in UMP/CMP-kinase crystallization buffer, Schlichting and Reinstein (1999) discovered that the bonding configuration of AlFx can switch from AlF4 - in acid pH to AlF3 in alkaline pH. A brief survey of the pH conditions in other phosphoryl-transfer enzymes crystallized with bound AlFx supported the same principle. Factors other than pH, such as F concentration, may also have effects on AlFx configuration.
Basic Chemistry of Al- and Be-F Complexes
Aluminum is a group 3B light metal (Fig. 1). It is an extremely rich but somewhat ’hidden’ element. Comprising 8% of the earth’s crust, Al is the most abundant metal and the third most abundant of all elements (Liptrot, 1974). It is concealed in minerals such as bauxite (Al2O3•2H2O) and cryolite (Na3AlF6). The Al3+ level is usually very low in natural water, due to the fact that free Al3+ instantly precipitates as hydroxides (Martin, 1986). With the advent of acid rain, metal ions, including Al3+, escape from minerals, dissolve in fresh water, and thus become available to man (Martin, 1994).
Al3+ is the only accessible oxidation state for aluminum in biological systems (Macdonald and Martin, 1988). In aqueous solution, Al3+ forms different species with water components at different pH. Free Al3+ [it is, in fact, Al(H2O)6 3+] exists mainly in acidic solutions (pH < 5.0) According to Martin’s calculation (Martin, 1986), upon addition of 1 mmol per liter of an Al3+ salt to a solution at pH 7.4, the free Al3+ concentration is not 1 mM, but only about 3 x 10-12 mol/L. Most of the Al3+ ions form insoluble Al(OH)3. The predominant Al3+ ions are in the form of Al(OH)4 - at around 8 μmol/L. However, when there are ligands available, free Al3+, rather than Al(OH)4 -, would bind to them, and this would shift the equilibrium to more free Al3+ ions (Martin, 1986).
Fluorine is a group 7B halogen (Fig. 1). It is the most chemically reactive non-metal. It is also the most electro-negative element (Liptrot, 1974). Of more than 60 metal ion species, Al3+ binds F- most strongly (Martin, 1988). The Al-F complex exists in nature in cryolite (Na3AlF6). Another metal ion which binds F- with high affinity is beryllium (Be). Be is a group 2A alkaline earth metal (Fig. 1). It is a relatively rare element. Coal combustion is the chief reason for its presence in the environment. Fresh water contains less than 0.001 ppm Be (Fishbein, 1981). The chemistry of Be differs considerably from that of the other members of this group (such as Mg and Ca), but resembles that of Al due to similar electro-positivity (Liptrot, 1974). Be-F complexes are strictly tetrahedral due to the sp 3 orbital hybridization (Fig. 1), whereas Al-F complexes have different bonding configurations (Chabre, 1990). F- is very electro-negative and has the greatest capacity to form hydrogen bonds. A metallic fluoride complex can bind to a protein molecule through hydrogen bonds formed between F atoms and the nearby amino acid side-chains. Be-F, Al-F, and P-O bonds are very similar in length (around 1.55 Å). However, the Al-F bond is ionic, whereas the P-O bond is covalent, and the Be-F bond is somewhere in between, but closer to being covalent (Emsley and Hall, 1976).
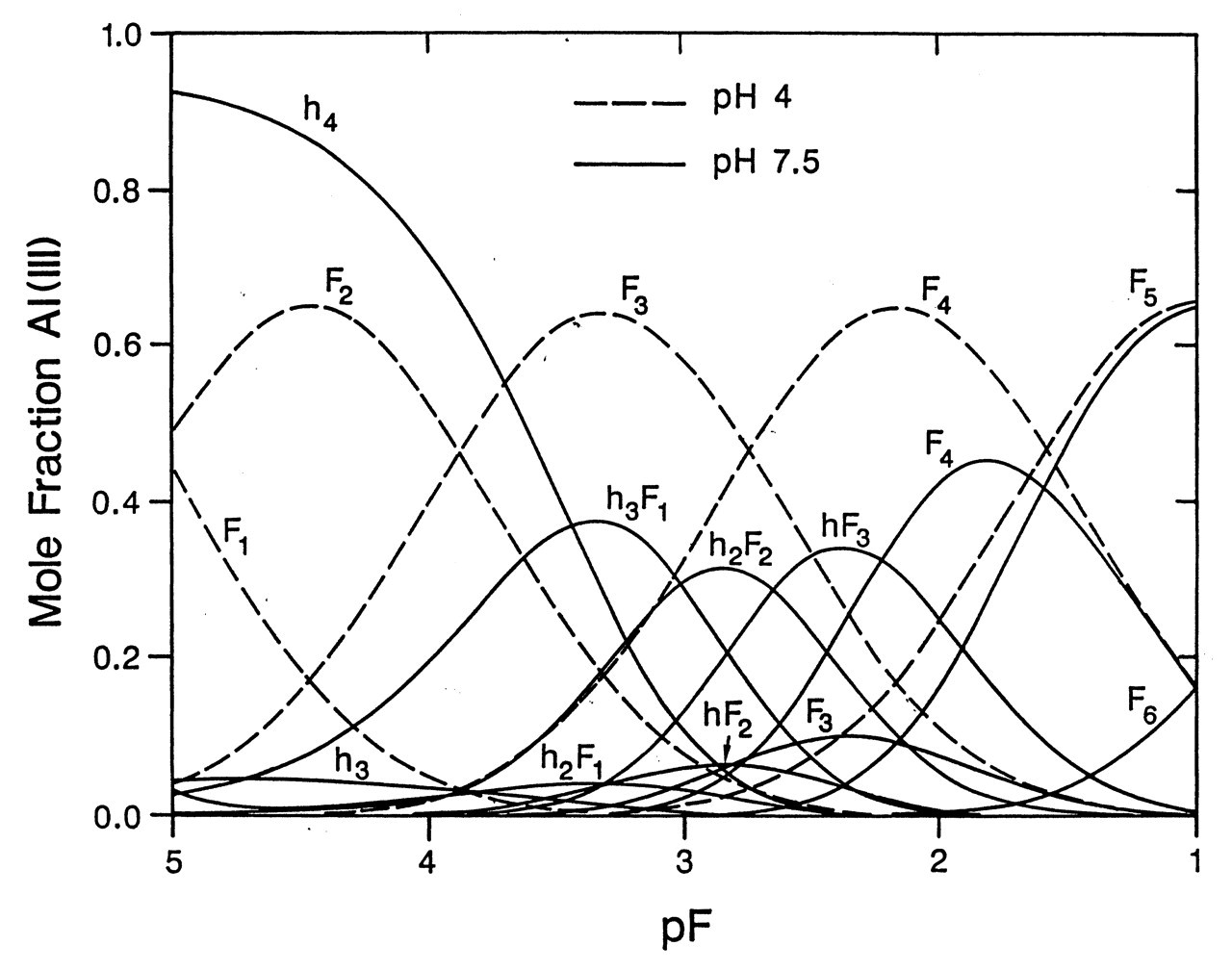
The structure and concentration of Al-F complexes in a solution depend on both F- concentration and pH (Martin, 1994). According to Martin’s calculation, in drinking water with 1 ppm F- (pF = 4.3), at pH 7.5, the predominant Al-containing species is Al(OH)4 -; at pH 4, the main species are AlF2 + and AlF3 (Fig. 5). Free Al3+ concentration in an aqueous solution changes dramatically with pH. The more acidic the solution, the more free Al3+ is available, the less the OH- group competes with Al3+ in binding to F-, and the more Al-F complexes are formed (Martin, 1996). The distribution graph shifts to the left as pH goes down. When pH reaches 2, there are more AlF3 complexes. This low pH is physiologically relevant, since the pHs of gastric juice, dental plaque fluid, and some popular beverages (such as cola) are around this level. At neutral pH, when F- is at the 5-mM level, the main species are a mixture of AlF3 and AlF4 - (Fig. 5). This is the condition in most biochemical and cellular studies. [For more detailed information on the structural dynamics of Al-F complexes, readers can refer to the two recent NMR studies (Bodor et al., 2000; Yu et al., 2001).]
Questions of Physiological Relevance
In spite of the detailed knowledge of its chemistry, the physiological relevance of Al-F complexes remains elusive. Many issues need to be considered in attempts to ascertain how metallic fluoride might potentially affect the host. The first question is how to relate its known biochemical effects in physiological situations. Known biochemical actions of Al-F complexes were performed in cell-free conditions with purified enzymes or extracted membranes where enzymes are directly accessible to AlFx binding. Would the same effect hold true when AlFx is applied to intact cells? In many intact cell models, G protein-regulated signaling pathways can be activated by AlFx at concentrations similar to those used in cell-free studies (Gilman, 1987). However, some evidence indicates that the effects are not always the same. For example, Inoue et al. (1990) concluded that AlFx activated both Gs and Gi proteins in membranes, but activated Gi only in cells. The difficulty in explaining this discrepancy lies in the lack of accurate knowledge on the transmembrane migration of AlFx. In physiological situations, for AlFx to activate an enzyme, which resides on the inner membrane of the lipid bilayer or in the cytoplasm, AlFx needs to cross the cell membrane. This has been clearly demonstrated by the clamp-patch technique, by which AlFx was directly delivered inside cells and activated G proteins (Chen and Penington, 2000). One early study estimated the intracellular F level to be around 0.35 mM after cells were incubated with 1 mM F for 10 min. When a 10-mM quantity of F is used, the intracellular F is around 2.6 mM. A decrease in the pH of the medium facilitated F influx into cells (Kawase and Suzuki, 1989). With certain concentrations of Al3+ and F- applied in the extracellular solution, the intracellular concentration of AlFx and their actual structures are not known. Nevertheless, the complexing between Al3+ and F- would inevitablely change the permeability of both Al3+ and F-.
The second question is related to the availability of Al-F complexes in human tissues. Are these AlFx complexes formed in vivo? Or are they formed in vitro and then taken up into the human body? Brudevold and colleagues (1972) studied the fluoride complexes in the tap water from 26 communities in three states in the United States. The total fluoride concentration in these sources of water ranged from 0.2-5 ppm. They found that Al was the principal complexing element for F in the drinking water. Higher Al concentration and marked complexing with F were found to be associated with treatment of water with alum. Boiling of the drinking water (1 ppm F) in an aluminum pot increased the water Al content from 0.03 ppm to 0.20 ppm, and a concomitant increase of complexed F from non-detectable to 50% (Brudevold et al., 1972). If the pH of the boiling water was adjusted to 3.5, 76% of the F was in a complex form. Many foods are prepared in acidic conditions in aluminum pots. Some popular beverages, such as cola, contained in aluminum cans, have pH values around 2. In the stomach, where most of the fluoride is absorbed, pH is around 1.2. When fluoride level is between 1 and 5 ppm, a significant protion of Al-F complexes in the stomach is AlF3 (see Fig. 5). AlF3 is an electrically neutral species. It is much more likely to be absorbed in the GI tract than other charged complexes of Al and F.
Bioavailablity and the in vivo State of Al and F
Daily Al intake in the typical UK/USA diet is about 10-20 mg. Most of the Al intake comes from Al-containing food additives, which are very common in the developed world (for a review, see Priest, 1993). Some beverages, such as Al-canned cola and tea, contained high content of aluminum. Certain medications, such as Al-containing antacids and buffered aspirin, as well as baking powder also contain very high levels of Al. For example, regular antacid users can consume 1 g Al per day. Drinking water contributes only a small fraction of the daily total Al intake. Al concentration in drinking water is rarely over 0.4 mg/L (Priest, 1993). There are two recognized sources of Al in drinking water (for a review, see Flaten, 2001). First, acid rain enhances the leaching of Al from minerals into natural water. Second, Al is widely used as a coagulant in water treatment to improve the water color. This often results in an increased Al concentration in drinking water (Flaten, 2001). Another potential source of Al is related to water fluoridation. Sodium fluoride, used in water fluoridation, usually comes as a by-product from Na3AlF6 in the aluminum smelting industry. It is not unlikely that a small amount of Al is present in sodium fluoride and may even exist in the form of Al-F complexes. Because of the chemistry, completely eliminating Al from NaF in this process is quite difficult. Nonetheless, it is certainly not likely to be a main source of Al in drinking water.
Al is very poorly absorbed in the GI tract. Only about 0.1% of the dietary intake of Al is absorbed, which amounts to 10 μg/day (Priest, 1993). In the extreme acidic conditions in the stomach, most of the ingested Al is in a soluble form. When the stomach contents reach the duodenum, it is rapidly neutralized; soluble Al would then precipitate out as hydroxides, and become unavailable for absorption. It is thought that a small amount of Al is absorbed through the gastric mucosa and proximal small intestine immediately before precipitation (Powell and Thompson, 1993). In human plasma, most Al is bound to transferin and citrates. Research indicates that citrates in the intestine maintain Al in a soluble form at neutral pH, and may promote absorption of the metal (Ohman and Martin, 1994). Serum Al concentration is about 6-7 μg/L (Powell and Thompson, 1993). Endogenous Al accumulates on the surface of bone. As the bone thickness increases, Al is buried in the mature bone matrix (Priest, 1993).
Sources of fluoride include natural fluoride in foodstuffs and water: fluoridated water (usually at 1.0 ppm, i.e., 1 mg/L), fluoride supplements (such as fluoride tablets), fluoride dentifrices (containing on average 1000 ppm F), and professionally applied fluoride gel (containing on average 5000 ppm F). Fluoride is also prescribed at a dose between 10 and 50 mg/day by some physicians to treat osteoporosis, although its therapeutic effect is controversial (Ringe and Rovati, 2001).
F- is very electro-negative. In an aqueous solution of F-, HF (pKa = 3.4) is formed. F- transport through biological membranes occurs primarily by non-ionic diffusion of HF. Classic studies with artificial lipid bilayers and pH electrodes indicated that HF is a highly permeant solute and has a permeability coefficient similar to that of water. Membrane permeability to HF is 5 to 7 orders of magnitude above that of F- (Gutknecht and Walter, 1981). Animal studies indicated that F- absorption from the stomach and oral mucosa is pH-gradient-dependent. HF is in diffusion equilibrium across the cell membrane (for a review, see Whitford, 1990). Recent studies showed that F- absorption from the intestine is less sensitive to pH, and may occur via a carrier-mediated process (i.e., facilitated diffusion) (He et al., 1998). It is not known whether such a carrier protein is also present in the membranes of other cells. The reported values for fluoride in human plasma range from 0.7 to 2.4 μmol/L. Ninety-nine percent of the endogenous F- accumulates in bone and other calcified tissues, such as enamel and dentin (Whitford, 1990).
Al-F Complexes and Al Neurotoxicity
Aluminum is generally considered a neurotoxin. Al is strongly associated with Alzheimer’s disease (AD). Many epidemiological studies seem to suggest a correlation between Alzheimer’s disease and Al in drinking water. It is possible that Al in drinking water is more physiologically available in man (for a recent review, see Flaten, 2001). An epidemiological study by Still and Kelley (1980) looked at the effect of water fluoride level on the incidence of AD. They compared the hospital-admitted cases of AD between one county with a very high water fluoride level (4.2 ppm) with those of two counties with low water fluoride levels (0.49-0.61 ppm). Interestingly, they found that the incidence rate of AD in the high-water-fluoride county was only one-fifth of that in the two low-water-fluoride counties (Still and Kelley, 1980). This study suggests the possibility that fluoride may have a protective effect against AD. This point of view was further elaborated by Forbes (Kraus and Forbes, 1992; Forbes and Agwani, 1994). Could the observed protective effect of F- on AD relate to the actions of AlFx on intracellular signaling pathways? Recent studies suggested that post-receptor signaling pathways, in particular that mediated by the G-protein-regulated phosphoinositide hydrolysis and adenylate cyclase pathways, are disrupted in certain areas of the brain in AD patients (Cowburn et al., 2001).
One research group (Isaacson et al., 1997; Varner et al., 1998) tested the neurotoxicity of Al-F complexes in rats. They administered either plain double-distilled water (ddw), or ddw containing 0.5 ppm, 5.0 ppm, 50 ppm AlFx, or 2.1 ppm NaF (containing an amount of F equivalent to that in 0.5 ppm AlF3) to a group of rats for 52 weeks. Both the NaF and the AlFx groups showed increased brain Al levels as compared with controls (2 times more Al in the NaF group, and 2.5 times more in the 0.5 ppm AlFx group). This indicates that the physiological level of fluoride (2.1 ppm) can increase Al absorption and deposition in the brain. The AlFx animals showed a reduction of neuronal density in the neocortex of the left hemisphere. Cellular histological changes include chromatin clumping, pyknosis, and vacuolation, and the presence of ghost-like cells was observed in both the AlFx group and the NaF groups, although they are more common and obvious in the former group. The AlFx-fed animals presented an unusual appearance, with sparse hair and discoloration of underlying skin (Varner et al., 1998). In summary, this study seems to show that chronic administration of a fairly low level of AlFx (0.5 ppm) in drinking water results in distinct morphological changes in the brain. One possible reason for these seemingly striking results could be related to the preparation of AlFx complexes in this study. Instead of adding F and Al salts to the diets of rats, the investigators carefully prepared the Al-F complex solution in double-distilled water by following a mole ratio of 1:6 (Al:F). Their stoichiometric calculation indicated that the predominant species formed in this condition was a mixture of AlF3 and AlF4 -. It is likely that the experimental condition in this study might have provided the optimal condition for the formation of the electrically neutral species AlF3. AlF3 may readily cross both the blood-brain barrier and neuronal cell membranes. It is known that AlF3 and AlF4 - are the active species in activating GTP-binding proteins (Coleman et al., 1994; Scheffzek et al., 1997). The potential actions of Al-F complexes on intracellular signaling pathways could be the underlying mechanism(s) for the morphological changes. It would appear that more studies need to be done to confirm these findings. Recently, an AlF3 salt became commercially available. It would be interesting to repeat the above experiments with different doses of AlF3 salt.
Al-F Complexes and Bone Physiology
Fluoride is a well-known bone-forming agent. Since it was shown to enhance calcium retention in osteoporotic patients, it has been used as a bone anabolic agent to treat osteoporosis (Rich and Ensink, 1961). Clinical studies have shown that fluoride treatment resulted in increased bone mass and bone density in spinal bone. However, a high concentration of fluoride can accumulate in bone. This can have an inhibitory effect on bone mineralization and may reduce the mechanical quality of bone crystals (Caverzasio et al., 1998). Due to the above facts, the therapeutic use of fluoride in osteoporosis is highly controversial. Nonetheless, fluoride is well-recognized as one of the strongest bone anabolic agents. Its clinical use in different conditions of osteoporosis is actively being tested (Ringe and Rovati, 2001).
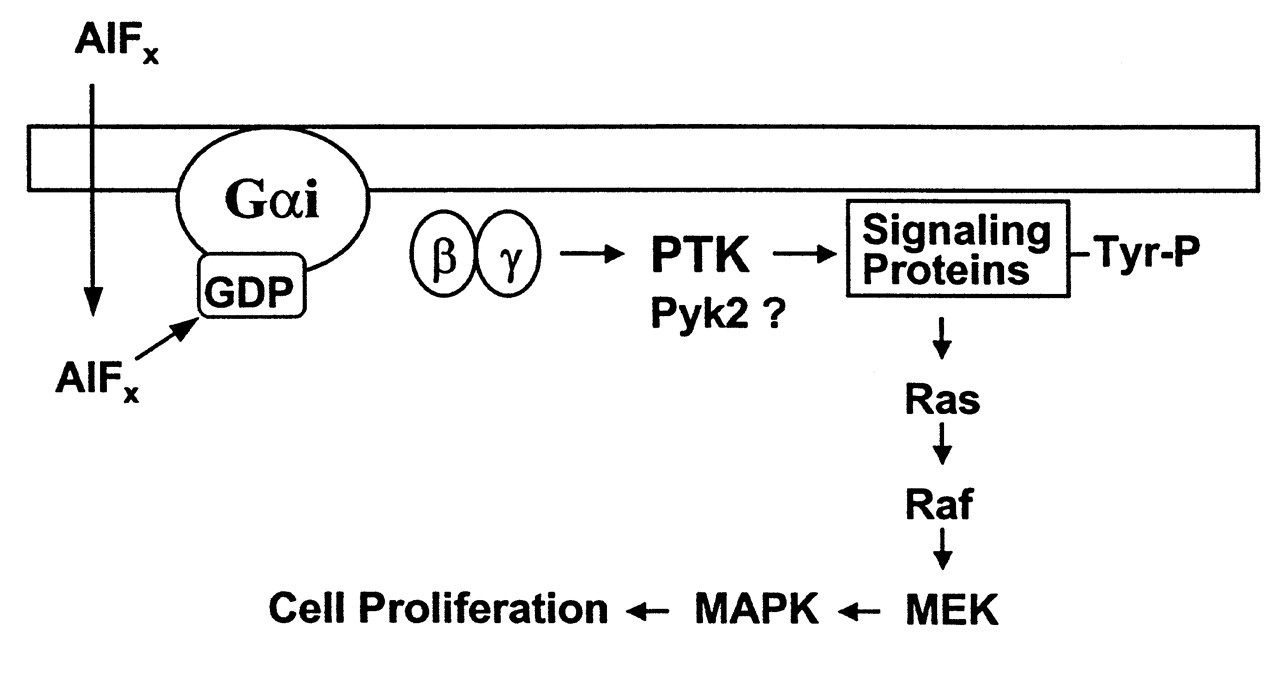
Farley et al. (1983) made the first observation that fluoride directly increases the proliferation and alkaline phosphatase activity of avian osteoblastic cells in culture. The optimal fluoride concentration for this action is about 10 μmol/L, which is within the plasma fluoride level in osteoporosis patients receiving fluoride treatment (5-30 μmol/L). Some phenomena indicate the possible involvement of Al in the effects of fluoride on bone. Fluorosis is an occupational disease among workers in the aluminum smelting industry. A high rate of osteosclerosis occurs in the miners of cryolite (Na3AlF6) (Caverzasio et al., 1996). Caverzasio and colleagues found that, in MC3T3-E1 cells (non-transformed osteoblast-like cells derived from the mouse), fluoride alone had no effect on cell proliferation. In the presence of 5 μM Al, fluoride (50-750 μM) stimulated tyrosine phosphorylation and cell proliferation (Caverzasio et al., 1996). A μM level of Al also potentiated the increase in P(i) transport across cell membranes induced by F in a dose- and time-dependent manner (Imai et al., 1996). Several key proteins in the MAP kinase pathway are tyrosine-phosphorylated. The mitogenic effect of fluoride on MC3T3-E1 cells can be blocked by genistein (a protein tyrosine kinase inhibitor) (Caverzasio et al., 1997; Susa et al., 1997), as well as by pertussis toxin (a specific inhibitor of heterotrimeric Gi/Go protein). These findings led these investigators to conclude that AlFx activates a specific protein tyrosine kinase (PTK) through a Gi protein. This PTK phosphorylates the downstream key enzymes in the MAP kinase pathway, and thereby triggers the proliferation response (Fig. 6) (Caverzasio et al., 1998; Susa, 1999). A novel cytoplasmic tyrosine kinase, Pyk2, has been identified in MC3T3-E1 cells and has been shown to be activated by AlFx through a G protein (Jeschke et al., 1998). Recently, two important signaling proteins, p130 Cas and Fak, have been found to be tyrosine-phosphorylated by PTK after AlFx stimulation. AlFx also increases the attachment and spreading of MC3T3-E1 cells (Freitas et al., 2002). Taken together, these studies strongly suggested that an Al-F complex is likely to be the active species that stimulates bone cell proliferation. Most recently, it was shown that AlF4 - and AlF3, as well as NaF, were all able to stimulate the proliferation of human TE85 osteosarcoma cells (Lau et al., 2002). Interestingly, 25 μM of AlF3, the lowest dose they tested, produced the highest effect on bone cell proliferation as compared with other treatments.
A study with rabbits showed that Al levels in tibia were significantly increased by the addition of F to the drinking water, even in animals receiving no additional Al in their drinking water (Ahn et al., 1995). Bone crystals are actively resorbed by resorbing osteocytes and osteoclasts. It seems that both F and Al tend to be concentrated in a surface layer of mineral, where active bone growth and remodeling occur (Smith, 1985; Priest, 1993). It is reasonable to suspect that high concentrations of labile F and Al and their complexes may exist in the extracellular fluid surrounding these cells. This unique microenvironment is the likely site where Al-F complexes exert their effects on bone cells.
Al-F Complexes and Fluorosis
Historically, because Al and F are known to form stable complexes, there have been studies that have looked at the interaction between Al and F and its effect on fluorosis. A few studies showed that Al ameliorates fluorosis in animals by interfering with F absorption (Becker et al., 1950; Kessabi et al., 1986). This was also the case in man. Spencer et al. (1980, 1985a,b) reported that ingestion of a relatively small dose of Al hydroxide (1.8 mg/day), regardless of the level of F intake (from 4 to 50 mg/day), was associated with a significant increase in fecal F excretion and a decrease in net F absorption by 57%. The plasma F level decreased by 41% (Spencer et al., 1985). Consistent with these early results, a more recent study on rabbits showed that F accumulation in plasma, urine, incisors, and tibia decreased as Al concentration increased in drinking water (Ahn et al., 1995). If Al indeed antagonizes F absorption, could Al salts be used in treating acute fluoride toxicity (Whitford, 1987)? Could Al be used to counteract F in areas where water fluoride levels are too high? Such attempts would have to consider the potential toxicities of Al-F complexes to the host.
Fluoride is incorporated into enamel during the tooth-forming stage, when ameloblasts are functionally active (Bawden et al., 1995). Enamel is first secreted as a protein matrix by ameloblasts (secretory stage). These same cells subsequently secrete proteinases that degrade matrix proteins almost completely (maturational stage) (Limeback, 1994). There are two mechanisms by which excess fluoride may negatively influence enamel formation. The first is by interfering with cell activities, i.e., ameloblasts. The second is by interfering with the dynamics of extracellular matrices (Aoba and Fejerskov, 2002). This review will focus on the action of fluoride on ameloblasts.
Ameloblasts can be affected by an increased level of fluoride during both secretory and maturational stages. A study by Matsuo et al. (1996) clearly showed the influence of fluoride on the secretory pathway of ameloblasts in enamel fluorosis. For four days, 20 mg/kg body weight of NaF was subcutaneously injected into rats with developing tooth germs. Morphological and cytochemical studies revealed the accumulation of small vesicles in the secretory pathway between rER and the Golgi apparatus. Golgi stacks were disorganized. Abnormally large granules appeared at the distal cytoplasm in the secretory ameloblasts (Matsuo et al., 1996). Apparently, acute exposure to a high concentration of fluoride disturbs the synthetic and secretory pathways in ameloblasts. Ameloblasts synthesize and secrete large amounts of proteinases during the post-secretory stage. Thus, some of the effects of fluoride on secretory ameloblasts may also hold true in the maturational stage. It does not seem likely that fluoride would directly affect the proteolytic activity of enamel matrix proteinases (Gerlach et al., 2000). Fluoride may influence the maturational stage of ameloblasts by two means. It may reduce the quantity of the proteinases by interfering with the synthetic or the secretory pathway of ameloblasts, or it may reduce the quality of the proteinases by affecting gene expression or the protein synthesis process (DenBesten and Heffernan, 1989).
The site where fluoride comes into contact with ameloblasts is at the extracellular fluid surrounding the cells. Through the ameloblast cell layer, F- ions are transported from the plasma in the nearby capillaries to the enamel matrix. A substantial fraction of F- ions exists in labile form in the enamel fluid during the secretory stage (25-30% of the total fluoride) (Aoba et al., 1989). Due to technical difficulties, the actual concentration of free F- in enamel fluid has not been widely measured. Aoba and Moreno (1987) reported a value of 5 μM in the enamel fluid from the soft, ’cheese-like’ enamel of porcine teeth during the secretory stage. Is it reasonable to suspect that, during the maturational stage, when mineralization occurs, the free fluoride concentration would be higher than in the secretory stage? Furthermore, the free F- concentration in the enamel fluid of animals with a high intake of F, i.e., sufficient to cause fluorosis, is not known. Osteoblasts are sensitive to the μM level of fluoride. It is not clear, at present, whether fluoride at this concentration range has any effect on ameloblasts.
Another basic question is how fluoride might trigger intracellular events in ameloblasts. A study by Matsuo and colleagues (1998) indicated, for the first time, that G proteins might be involved in the development of enamel fluorosis. They applied immunoblotting and pertussis-toxin-induced ADP ribosylation to the intracellular fractions of the ameloblasts from the enamel fluorosis model rats (Matsuo et al., 1996). It was found that Gi3 and Go proteins are present in rER and Golgi apparatus, and that NaF treatment decreased the amounts of these G proteins bound to both membranes. It was assumed that, in their study, NaF was transformed into AlFx in vivo (Matsuo et al., 1998). Their study suggested that G proteins may participate in the disturbance of the ameloblastic secretory pathway in enamel fluorosis. It is well-known that AlFx and GTPγS blocked in vitro vesicle formation and transport in a cell-free system, and that trimeric G proteins are involved in secretory vesicle formation (Melancon et al., 1987; Leyte et al., 1992). The findings in the present animal study by Matsuo et al. (1998) are consistent with these early in vitro data.
Matsuo’s study did not look at whether fluoride would interfere with the intracellular signal transduction pathways by activating G proteins on the plasma membranes of ameloblasts. Such studies are hampered by the lack of a suitable cell-culture system for ameloblasts. Nevertheless, a few G-protein-regulated signal transduction pathways have recently been delineated in ameloblasts by immmunohistochemical techniques (Moran et al., 2000). With increasing understanding of enamel cell biology (Hubbard, 1998), as well as the recent advances in the cell-culture system for primary enamel organ epithelial cells and the development of ameloblast-like cell lines (DenBensten et al., 1998 DenBensten et al., 1999), progress in this field of research is likely to occur in the near future.
Al-F Complexes, Bacterial Physiology, and Oral Diseases
It is well-established that fluoride reduces acid production by inhibiting the carbohydrate metabolism of the acidogenic plaque flora (Hamilton, 1990). H+-translocating ATPase is an important component of the anti-microbial action of fluoride. H+-translocating ATPase is the key enzyme for generating the pH gradient (▵pH) across the cell membrane and helps cells to survive in the acidic environment (Bowden and Hamilton, 1998). With the action of this enzyme, acidogenic bacteria extrude H+ at the expense of ATP, and are thereby able to maintain the cytoplasm in a relatively neutral pH for metabolism. Fluoride, in the form of HF, diffuses into and accumulates in cells. HF acts to short-circuit the outflow of H+, and, as a consequence, the cytoplasm becomes acidic and the ▵pH is dissipated (Marquis, 1990). Sutton et al. (1987) were the first to report that H+-translocating ATPases isolated from membranes of oral bacteria were inhibited by fluoride. Shortly after this study, Lunardi et al. (1988) demonstrated that inhibition of H+-translocating ATPases by mM level of fluoride was dependent on the presence of trace amounts of Al3+. Such a mechanism was quickly confirmed in oral bacteria. Sturr and Marquis (1990) found that isolated H+-translocating ATPases from Streptococcus mutans and Lactobacillas casei can be protected from fluoride inhibition by an Al-chelator, deferoxamine. This protection was lost when a μM level of Al3+ or Be2+ was added. Their study strongly suggested that F inhibits H+-translocating ATPases by forming Al-F complexes with trace amounts of Al3+, and that Al-F complexes work by mimicking a phosphate analog (same as BeF3 -). Recently, the structure of a H+-translocating F1-ATPase complexed with MgATP and AlF3 has been determined by x-ray crystallography (Braig et al., 2000). Al- and Be-F complexes have lately been used in determining the three-dimensional structures and the catalytic mechanisms of several important bacterial enzymes such as nitrogenase (Schindelin et al., 1997), and sensor kinases-bacterial response regulators such as NtrC and CheY (Yan et al., 1999; Cho et al., 2000; Lee et al., 2001).
The clinical relevance of the above findings depends on the bioavailablity of Al-F complexes in the oral cavity. Al is known to have anti-caries activity of its own (for review, see Kleber and Putt, 1984). Early studies found that human saliva contains 10 ppm Al (Dreizen et al., 1970), and human whole dental plaque contains 35 ppm Al (Swift, 1967). Dietary Al tends to accumulate on the enamel surface (Kleber and Putt, 1994). A level of 100-700 ppm Al is present in the surface layer of enamel in teeth collected from different regions (Cutress, 1972). This study found that teeth in the high-water-F region (5 ppm) contain six- to seven-fold higher levels of Al than teeth in the low-water-F regions. This suggests that Al and F may interact chemically during the mineralization process. It is long known that dental plaque can accumulate F (Dawes et al., 1965). A steady F concentration in the whole plaque from people who use NaF-containing toothpaste (1000-1500 ppm F) was found to be around 4 ppm (Duckworth et al., 1994). Thirty min after the use of a mouthrinse containing 228 ppm F, the F concentration in plaque fluid was found to be 148 μmol/L (Vogel et al., 2000). According to Martin’s model, in an acidic aqueous solution (such as plaque fluid), under these concentrations of F (pF 3.6-3.8), the dominant species would be the electro-neutral AlF3 complexes (Fig. 5). Another potential source of Al-F complexes is toothpaste. Over 95% of the population in developed countries use fluoride-containing toothpaste, which contains, on average, 1000 ppm F (Warren and Levy, 1999). A 1000-ppm quantity of F in an aqueous solution would generate 53 mM F. In contrast, many toothpastes use Al compounds as the abrasive base, such as alumina (Al2O3•2H2O) and ordinary alum [KAl(SO4)2•12 H2O] (Hanachowicz, 1984; Wulknitz, 1997; Heidmann and Poulsen, 1997). Many brands of toothpaste are packed in Al tubes, especially in developing countries (Rajwanshi et al., 1997). One recent study reported that when bacterial Salmonella typhimurium was incubated with various concentrations of AlCl3 (1.5-9.0 ppm) for 1 hr at a neutral pH of 7.4, intracellular Al accumulated in a concentration-dependent manner from 0.5 to 4.5 ppm (Ahn and Jeffery, 1994). They also found that, at neutral pH, F requires the presence of Al to be transported into these cells. It is known that, in the acidic pH environment of dental plaque, F can diffuse into bacterial cells in the form of HF, and subsequently, dissociate into free F- in the more alkaline cytoplasm. There would also be more free extracellular Al3+ available in an acid pH and hence more intracellular Al3+. Based on our current knowledge of basic chemistry, the probability that intracellular F- and Al3+ would form complexes cannot be ignored. If Al-F complexes could be formed intracellularly or transported into bacterial cells from dental plaque or toothpaste, in what concentration would they exist in the cytoplasm? Would that concentration of Al-F complexes have an inhibitory effect on H+-translocating ATPases or other enzymes? What implications does this have for caries prevention and periodontal disease? These questions certainly warrant investigation.
Summary and Future Prospects
Fluoride has been found to inhibit a variety of enzymes for more than half a century. Since the discovery of G protein activation by AlFx, many enzymes involving phosphoryl transfer reactions are found to be affected by AlFx or BeFx. AlFx tends to mimic the terminal phosphate group in the transition state of the enzyme-catalyzed phosphoryl transfer reactions. Its biochemical interactions with enzymes such as GTPases, ATPase, and phosphatases have been proven by x-ray crystallography.
Al is the most abundant metal on earth. A mM level of fluoride etches Al3+ ions from laboratory glassware. Of all metal ions, F- binds to Al3+ most strongly. Only a trace amount of Al is needed to form biologically active fluoride complexes. Such Al-F complexes are usually formed in routine laboratory solutions, cell culture media, and body fluid (e.g., saliva), when F concentration approaches the 5 mM level. This AlFx, rather than F itself, is most likely the biologically active species. The concentration of AlFx is limited by the Al3+ concentration in a solution. Although a mM level of F is required to form AlFx at neutral pH, the AlFx concentration is no more than a μM, since only a μM level of total Al is needed. AlFx is a multiple-edged sword. Various phosphoryl-transfer enzymes may have different sensitivities toward different forms and concentrations of AlFx. This is due to different binding strengths between AlFx or AlFx-nucleotide complexes and the enzyme-active sites. Therefore, one needs to be cautious in always attributing an AlFx effect to a G protein, especially in intact cells. Nevertheless, since AlFx activation of heterotrimeric GTP-binding proteins seems to be the predominant action in many systems, this effect of AlFx always needs to be excluded before other mechanisms are considered.
The potential physiological implications of AlFx are revealed by many studies cited in this paper. In most of the situations, the investigators have not proved that Al-F complexes are present in vivo. Are they formed in the GI tract before being absorbed? In what concentrations and in what forms are they present in plasma? Are they bound to nucleotides in plasma? How are they delivered to the extracellular fluid? An understanding of these questions is fundamental to the comprehension of its physiology. At acidic pH (e.g., pH 1-4), the concentration requirement for F to form effective Al-F complexes is greatly reduced. At pH 4, a significant amount of AlF3 is formed in the presence of 50-100 μM F. Regular fluoridated drinking water (1 ppm F) contains 53 μM F, and gastric juice usually falls between pH 1 and 2. A central issue concerning the absorption and distribution of AlFx seems to be how AlFx crosses a biological membrane. An artificial lipid bilayer system can be used for studying the transmembrane migration of AlFx. It is suspected that the electro-neutral complex, AlF3, would be the most deliverable species in the in vivo system.
Fluoride is particularly related to the physiology of mineralized tissues, such as bone and enamel. There has been convincing evidence that AlFx affects bone cell physiology. Dental plaque and periodontal pockets also provide a unique acidic environment for the formation of Al-F complexes. AlFx may affect the survival of oral bacteria, especially the acidogenic and aciduric microbes, by inhibiting the H+-translocating ATPases. This effect may be of significance in dental caries and periodontal disease. The key issue is to test whether Al-F complexes are indeed present in dental plaque and periodontal pockets as well as in toothpaste or are formed inside the bacteria. It is important to remember that Al-F complexes have clinical implications only if they are available to the biological tissues. The study of Al-F complexes may lead to the optimal use of fluoride, which is both safer and more effective. In the future, It may also promote metallic fluoride drugs for specific health purposes.
A Special Note
Glass-ionomer cement (GIC) is manufactured from compounds containing both Al and F. Different GIC products have dramatically different Al- and F-releasing profiles. Studies have shown a definite positive correlation between Al and F release in different GICs. Al-f complexes may contribute to the biological effects of GICs in areas such as caries prevention, reparative dentin formation and dentin remineralization, and apical bone repair after root canal treatment, as well as new bone matrix formation after GIC implantation in orthopedic bone reconstruction. Bioactive GIC formulations capable of releasing optimal concentrations of Al-F complexes in an effective timeframe could be developed in the future. These may induce favorable biological responses on cells such as osteoblasts and odontoblasts.
A section of the Periodic Table. For the elements related to phosphate or its analogs, their symbols, atomic numbers, oxidation states, and electron configurations are highlighted.
Schematic illustration based on the crystal structure of Ras•GDP•AlF3•GAP. The important elements of catalysis and their interaction with AlF3 (γ-phosphate) at the transition state are shown. Aluminum fluoride forms a trigonal bipyramidal complex. RasGAP stabilizes the transition state by supplying the critical arginine (Arg 789) in trans to neutralize the developing charges. In heterotrimeric G protein, such a critical arginine (Arg 178 in Giα1) is supplied in cis from the Giα1 itself (see Fig. 3A) (adapted from Scheffzek et al., 1997). Reprinted with permission from the journal Science and The American Association for the Advancement of Science. Speciation of Al-F complexes in aqueous solution. Mole fraction of total Al(lll) vs. pF.pF = - log[F-], where [F-] is the ambient fluoride molar concentration. F complexes with Al were measured at two pH values, dashed curves for pH 4 and solid curves for pH 7.5. Symbols on curves designate the number of fluoride groups (F) or hydroxy groups (h) bound to Al(lll). Thus, h4 represents Al(OH)4
-; F4 represents AlF4
-; and hF3 represents (OH)AlF3
- (adapted from Martin, 1994). Reprinted with permission from Elsevier Science. G protein hypothesis in bone cells. In osteoblast-like cells, fluoride forms a complex with aluminum (AlFx), which interacts with GDP to form a GTP-like molecule. Activation of the Gi protein stimulates the tyrosine phosphorylation of signaling proteins by an unknown protein tyrosine kinase (PTK), such as the recently identified Pyk2. Activation of the MAPK pathway through the Ras pathway leads to enhanced cell proliferation (adapted from Caverzasio et al., 1998). Reprinted with permission from Elsevier Science.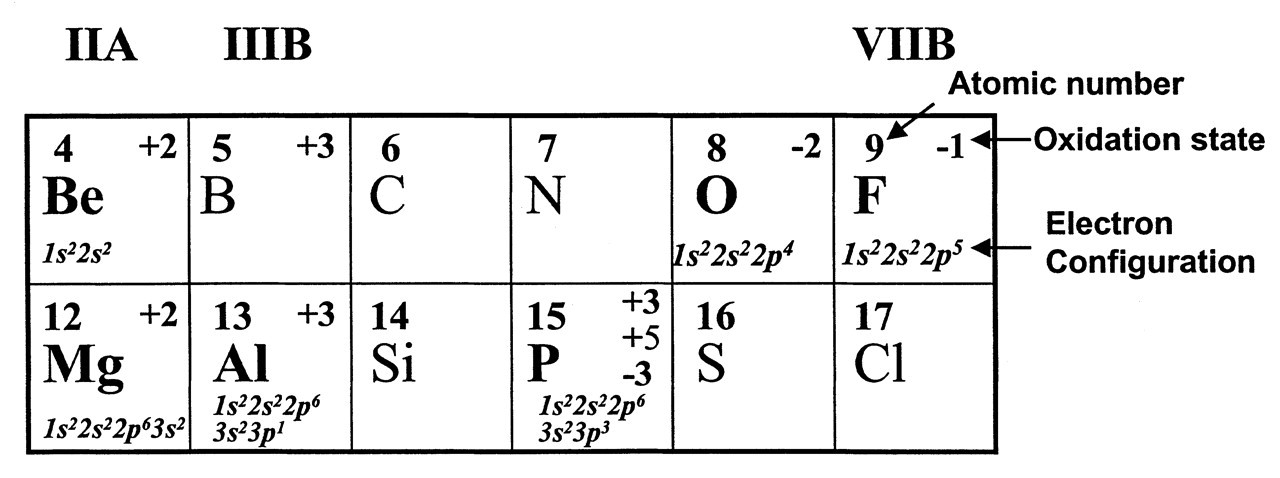
Footnotes
Acknowledgements
I thank Drs. Marc Chabre, Stephen Sprang, Alfred Wittinghoger, Bruce Martin, and Joseph Caverzasio for allowing me to adapt the illustrations from their original publications. I also thank Drs. Bruce Martin and Robert Marquis for communicating and discussing unpublished studies. I am particularly thankful to Dr. Hardy Limeback for reviewing the manuscript and providing valuable suggestions. I am most grateful to my former PhD mentor, Dr. Norman Fleming, for his steadfast encouragement and support. I am also thankful for the encouragement of three other teachers of mine in the Department of Oral Biology, University of Manitoba—Drs. George Bowden, Ian Hamilton, and Colin Dawes.
