Abstract
The synthesis of extracellular glucan is an integral component of the sucrose-dependent colonization of tooth surfaces by species of the mutans streptococci. In investigators’ attempts to understand the mechanisms of plaque biofilm development, several glucan-binding proteins (GBPs) have been discovered. Some of these, the glucosyltransferases, catalyze the synthesis of glucan, whereas others, designated only as glucan-binding proteins, have affinities for different forms of glucan and contribute to aspects of the biology of their host organisms. The functions of these latter glucan-binding proteins include dextran-dependent aggregation, dextranase inhibition, plaque cohesion, and perhaps cell wall synthesis. In some instances, their glucan-binding domains share common features, whereas in others the mechanism for glucan binding remains unknown. Recent studies indicate that at least some of the glucan-binding proteins modulate virulence and some can act as protective immunogens within animal models. Overall, the multiplicity of GBPs and their aforementioned properties are testimonies to their importance. Future studies will greatly advance the understanding of the distribution, function, and regulation of the GBPs and place into perspective the facets of their contributions to the biology of the oral streptococci.
Introduction
Many species of bacteria synthesize glucan polymers and glucan-binding proteins. Common forms of glucan include glycogen, a storage form of glucose (Preiss and Romeo, 1989), and the beta-linked-D-glucans that modulate osmolarity within the periplasm of Gram-negative bacteria (Kennedy, 1996; Bohin, 2000). Glucan-binding proteins may include the enzymes that catalyze the synthesis of the glucans, as well as enzymes capable of hydrolyzing glucans, including starch and cellulose, that can act as substrates for microbial growth (Warren, 1996).
For many oral streptococci, glucans comprise an extracellular slime layer produced in the presence of sucrose that promotes adhesion and the formation of a dental plaque biofilm (Gibbons and van Houte, 1973). These glucans are synthesized from sucrose by the enzymatic action of one or more glucosyltransferases (GTFs) and can be water-insoluble or -soluble. Research into dental plaque development and the etiology of dental caries established the central role of glucan in sucrose-dependent adhesion and the correlation between sucrose consumption and increased caries rates (Hamada and Slade, 1980; Loesche, 1986). For these reasons, many of the initial investigations into the virulence of the mutans streptococci (MS; including 3 serotypes of S. mutans, 2 serotypes of S. sobrinus, S. criceti, S. ratti, and S. downei) focused on the synthesis of glucan and its effects on promoting the adhesion, aggregation, and accumulation of MS onto smooth tooth surfaces.
However, glucan-binding proteins are also found among the commensal oral streptococci. Extracellular polysaccharides are synthesized by one or more GTF enzymes in S. salivarius, S. gordonii, S. sanguis, S. oralis, and S. cristatus (Whiley and Beighton, 1998). Less attention has been paid to the biological roles of glucans in commensal oral bacteria. The ability of oral streptococci to exploit glucan-binding properties of extracellular and cell-surface proteins to facilitate colonization and survival in the oral environment may have ecological implications for determining the bacterial composition of dental plaque. The composition of this microbial community could, in turn, affect the state of oral health or disease.
The GTF Enzymes
Enzymatic classification
The glucansucrase enzymes—which include the GTFs of oral streptococci, dextransucrases, and the alternansucrase of Leuconostoc mesenteroides, and GTFs of lactococci and lactobacilli—are extracellular enzymes of ca. 140 to 175 KDa that hydrolyze sucrose into its fructose and sucrose moieties; the same enzyme then polymerizes the resulting glucose moieties into glucans. In oral streptococci, the synthesized glucans contain various proportions and branches of alpha-1,3 and alpha-1,6 glucosidic linkages (Loesche, 1986). Although alpha-1,4 linkages have been reported in glucans synthesized by oral streptococci (Guggenheim and Schroeder, 1967), these linkages are more commonly synthesized by related glucansucrase enzymes from non-oral bacterial species [e.g., Leuconostoc mesenteroides (Monchois et al., 1999b) and lactobacilli (Kralj et al., 2002)]. The degree of branching and proportion of each type of glucosidic linkage determine the relative water-solubility of the glucan product. One classification system for GTFs relates to the glucans they produce. GTF-S enzymes synthesize water-soluble glucans, also termed dextrans, which have predominantly linear alpha-1,6 linkages. GTF-I and the S. mutans GTF-SI enzymes make water-insoluble glucans with various degrees of branching and predominantly alpha-1,3 linkages. This insoluble glucan, also termed mutan, is associated with more tenacious adhesion to the tooth surface and with dental plaque development (Loesche, 1986). Activity of some, but not all, GTF enzymes is dependent upon, or enhanced by, the presence of acceptor molecules (e.g., maltose, isomaltose, pre-existing dextrans) for the glucosyl moieties. The basis for primer dependence is not fully understood, and a generalized mechanism for glucan synthesis may not be applicable to all members of the glucansucrase enzyme family (Monchois et al., 1999b).
Distribution of GTF enzymes among cariogenic and commensal streptococci
The number of GTF enzymes within species of streptococci varies. The MS have multiple GTFs which encode both water-soluble and water-insoluble glucans within one organism. In S. mutans, the tandemly arranged gtfB and gtfC encode GTF-I and GTF-SI enzymes, respectively, and the distally located gtfD encodes a GTF-S. One GTF-I and two tandemly arranged GTF-S enzymes have been isolated and characterized in S. criceti (Inoue et al., 2000). S. downei and S. sobrinus both have three GTF-S and one GTF-I enzymes (Colby et al., 1999; Hanada et al., 2002). In contrast, members of the oralis group of streptococci (S. sanguis, S. oralis, and S. gordonii) have a single GTF enzyme which appears to make glucans with various proportions of alpha-1,3 and alpha-1,6 linkages (Grahame and Mayer, 1984; Kopec et al., 2001). The structural genes for S. gordonii gtfG (Vickerman et al., 1997b) and S. oralis gtfR (Fujiwara et al., 2000) have been sequenced and characterized. A partial sequence of the S. sanguis gene thought to encode the single GTF has been designated gtfP in GenBank (Kawaguchi, M., et al. Direct submission; Accession number AB056712.1). However, not all non-MS streptococci have only one GTF. The commensal species S. salivarius has two GTF-I (encoded by gtfJ and gtfL) and two GTF-S (encoded by gtfK and gtfM) enzymes (Simpson et al., 1995). In species with multiple GTF enzymes within one organism, the alpha-1,6 linked dextrans synthesized by the GTF-S enzymes may function as acceptors for the GTF-I enzymes in the same species. In this manner, one species may make a variety of glucan products.
Structure
The primary amino acid sequences of the streptococcal GTF enzymes are highly homologous. Although the large size of the GTF molecules has precluded complete functional domain mapping, three-dimensional models have been proposed (Devulapalle et al., 1997). Secondary structural predictions suggest that the GTFs are members of the alpha-amylase superfamily and contain a circularly permuted (alpha/beta)8-barrel motif (MacGregor et al., 1996). After a conserved signal sequence, followed by a ca. 200-amino-acid non-conserved region, the catalytic domain of ca. 1000 amino acids contains conserved amino acids necessary for sucrose hydrolysis (Mooser et al., 1991; Funane et al., 1993; MacGregor et al., 2001). The carboxyl-terminal one-third consists of a series of direct repeats that function in glucan binding (Ferretti et al., 1987; Mooser and Wong, 1988; Abo et al., 1991). The conserved amino acids in the carboxyl-terminus repeats have been defined in various lengths by different investigators, although these repeat lengths have not been related to any functional activities. In the GTF and GBPs, the two main methods of classification have been: (1) 33-aa "A" , 48-aa "B", 20-aa "C" [reviewed by Russell (1990)], and 14-aa "D" repeats (Giffard et al., 1991); and (2) the 21-aa YG repeats (Giffard and Jacques, 1994). The "A" repeats are shared by all the streptococcal GTF enzymes as well as by the first sequenced MS glucan-binding protein, GbpA (Banas et al., 1990).
The glucan-binding domain, which makes up approximately one-third of the streptococcal GTF protein, shares sequence similarity with ligand-binding domains found near the NH2 terminal (Garcia et al., 1999) or carboxyl-terminal regions of proteins with various functions in several Gram-positive bacteria (Wren, 1991). These proteins include the Clostridium difficile toxins A and B (von Eichel-Streiber et al., 1992), and lytic enzymes from S. pneumoniae and its bacteriophages (Garcia et al., 1990), where the glucan-binding domain is thought to interact with carbohydrate components in target cells. Similarly, repeats on the carboxyl terminus of pneumococcal surface protein A (PspA) are thought to facilitate cell-surface anchoring through interactions with choline in the S. pneumoniae cell wall (Yother and White, 1994). Such interactions would not be expected to occur in oral streptococci, whose cell walls do not contain choline. Nevertheless, it is possible that the carboxyl-terminal repeats can affect cell association of GTFs. Nested carboxyl-terminal deletions of recombinant enzymes expressed in GTF-negative streptococcal species suggested that the repeat units are necessary for sucrose-dependent cell association (Kato and Kuramitsu, 1991). Since a minimum number of repeats is necessary for optimum GTF activity (Ferretti et al., 1987; Abo et al., 1991), this effect may be due to their role in glucan synthesis as well as their ability to bind to developing glucan polymers.
Carboxyl terminal repeats and glucan binding
The role of the carboxyl-terminal repeats in determining the structure of the glucans synthesized by the GTF enzymes has not yet been fully determined. Although decreases in the number of repeats can decrease or abrogate GTF activity and influence the structure of the glucan products, usually by increasing the water solubility of the glucan product, no consistent domain patterns have yet been identified that predict the linkages in the glucans synthesized by the enzymes (Monchois et al., 1999b). In general, these glucan-binding domains of the GTFs are thought to bind alpha-1,6-linked glucans. However, there is one report that the recombinant GtfJ of S. salivarius can bind to alpha-1,3-linked mutan (Kingston et al., 2002). Similarly, four mutan-binding peptides were recovered from a trypsin digest of the related enzyme, dextransucrase, from Leuconsotoc mesenteroides NRRL B-512; only one of these peptides was from the carboxyl terminus repeat region (Funane et al., 1998).
Discovery of Glucan-binding Proteins
During the investigations of MS aggregation properties, it was noted that a certain proportion of extracellular GTFs could become cell-associated in the presence of glucan or commercially prepared dextran (Gibbons and Fitzgerald, 1969; Gibbons and van Houte, 1973; Mukasa and Slade, 1974). This cell-association of GTFs was determined to be due to binding by the GTFs to glucan or dextran that coated the bacteria (Mukasa and Slade, 1974; Kuramitsu and Ingersoll, 1978) in conjunction with a proposed proteinaceous receptor for glucan (Scherp, 1971; Kelstrup and Funder-Neilsen, 1974; Kuramitsu, 1974; Olson et al., 1974; McCabe and Smith, 1975, 1976; McCabe, 1976; Slade, 1977; Kuramitsu and Ingersoll, 1978; Montville et al., 1978; Olson, 1978; Wu-Yuan et al., 1978). Several groups examined Streptococcus mutans proteins, most often via affinity chromatography, using glucan or dextran, in attempts to isolate and identify the putative cell-surface receptor or protein(s) that might contribute to overall plaque development (McCabe et al., 1977; McCabe and Smith, 1977; McCabe and Hamelik, 1978; Russell, 1979; Smith et al., 1979, 1994, 1998; Landale and McCabe, 1987; Wu-Yuan and Gill, 1992; Sun et al., 1994; Ma et al., 1996; Sato et al., 1997). These studies confirmed the glucan-binding properties of the GTFs and led to the isolation of several non-GTF glucan-binding proteins (GBPs). Some of these GBPs are indeed surface proteins and may fit the role of a cell-surface receptor for glucan. Others represent secreted proteins that can become cell-associated when glucan coats the bacterial cells. The GBPs are grouped together on the basis of their glucan-binding properties, but it is clear that they represent a heterogeneous group of proteins with variations in size, glucan-binding domain, glucan-binding affinity, distribution among the MS, and, most importantly, function.
Distribution of Glucan-binding Proteins
The ability of some MS to aggregate in the presence of exogenously added glucan or dextran precipitated a search for a GBP that might be responsible for this property. Many of the early investigations utilized strains of S. mutans that later became classified as S. sobrinus. After the taxonomic split, it was apparent that S. sobrinus strains more readily aggregated than did strains of S. mutans, and a major GBP from S. sobrinus was designated the glucan-binding lectin (GBL) (Drake et al., 1988). Subsequently, Ma et al. (1996) defined GBLs as GBPs that confer upon bacteria the property of aggregation in the presence of exogenous alpha-1,6 glucan. According to this definition, all GTFs and GBLs are glucan-binding proteins, but not all glucan-binding proteins are GTFs or GBLs. This review will focus on the properties of the GBPs relevant to glucan binding. [For a recent review of the chemistry and structure of the GTFs, see Monchois et al. (1999b).] Non-GTF GBPs have been identified and characterized only among the MS, although both commensal and cariogenic mutans oral streptococci possess GTFs. The distribution of the GBPs among different oral bacteria may provide clues to their function. Perhaps the GBPs serve to aid adhesion of the MS to the plaque biofilm, whereas the non-MS oral streptococci possess efficient means for colonization independent of glucan binding. As the genome sequences of additional bacteria become available, the identity and extent of GBPs should become more obvious. It is conceivable that even non-streptococcal plaque species could possess GBPs to promote their colonization. The Table lists the known GBPs.
S. mutans GBPs
The GBPs of S. mutans were designated in the order of their discovery. GbpA was originally isolated by Russell (1979) and designated GBP (Douglas and Russell, 1982; Russell et al., 1985). Several years later, Smith et al. (1994) reported the isolation of GbpB. Unlike GbpA and GbpB, which were isolated primarily by affinity chromatography, GbpC was identified by Sato et al. (1997) following mutagenesis to pinpoint the genetic locus responsible for conferring dextran-dependent aggregation when the bacteria were grown under certain defined stress conditions. Shah and Russell (2002) analyzed the S. mutans genome sequence for amino acid repeats common to the carboxyl terminus regions of GTFs and GbpA to locate gbpD.
GBPs in Other Mutans Streptococci
Several GBPs have also been found in other MS, particularly S. sobrinus (Table). Wu-Yuan and Gill (1992) reported weak cross-reactivity with an 87-kDa S. sobrinus B13 GBP (GBP-2) using antisera directed against the S. mutans GbpA. However, the gbpA gene did not hybridize with S. sobrinus chromosomal DNA (Wu-Yuan and Gill, 1992), and Western immunoblots with sera against S. sobrinus 6715 GBPs 2, 3, or 5 did not cross-react with any S. mutans GBPs (Smith et al., 1998). Since most of the genes encoding S. sobrinus GBPs have not been cloned, it is difficult to evaluate the extent of similarity between GBPs among the MS, as well as intraspecies relationships. For example, GBP-5 and GBP-3 may be similar proteins, or GBP-5 might be a degradation product of GBP-3 (Smith et al., 1998).
The dextranase inhibitor, Dei, is another GBP whose range is limited to only certain members of the MS. Dei activity can be found in S. sobrinus, S. criceti, and S. downei (Sun et al., 1994). The Dei glucan-binding domain shares homology with those found in the GTFs and GbpA.
Functions of GBPs
Functionally, the S. mutans GbpC and the GBL/GBP-4 from S. sobrinus or S. criceti are involved in dextran-dependent aggregation (Ma et al., 1996; Sato et al., 1997), though the conditions for aggregation of S. mutans are more limited than for S. sobrinus or S. criceti. Sato et al. (1997) report that the high alanine and proline content of GbpC is similar to that reported for GBL/GBP-4 (Ma et al., 1996; Sato et al., 1997). Accordingly, the GbpC and GBL/GBP-4 may represent the cell-surface glucan receptors originally predicted to explain dextran-dependent aggregation and the sucrose-dependent cell association of GTFs and GBPs. It should be noted, though, that spontaneous and chemically induced aggregation-deficient mutants of S. sobrinus have been found to be missing either a band corresponding to the 87-kDa GBP-2 (Wu-Yuan and Gill, 1992) or the 60kDa GBL/GBP-4 (Ma et al., 1996). Final resolution of the protein(s) responsible for dextran-dependent aggregation in S. sobrinus will likely be achieved following genomic sequencing and the development of an efficient method for the genetic inactivation of individual genes in this species. Consistent with cell-surface expression, GbpC contains a putative membrane anchor and cell-wall-binding sites (Sato et al., 1997).
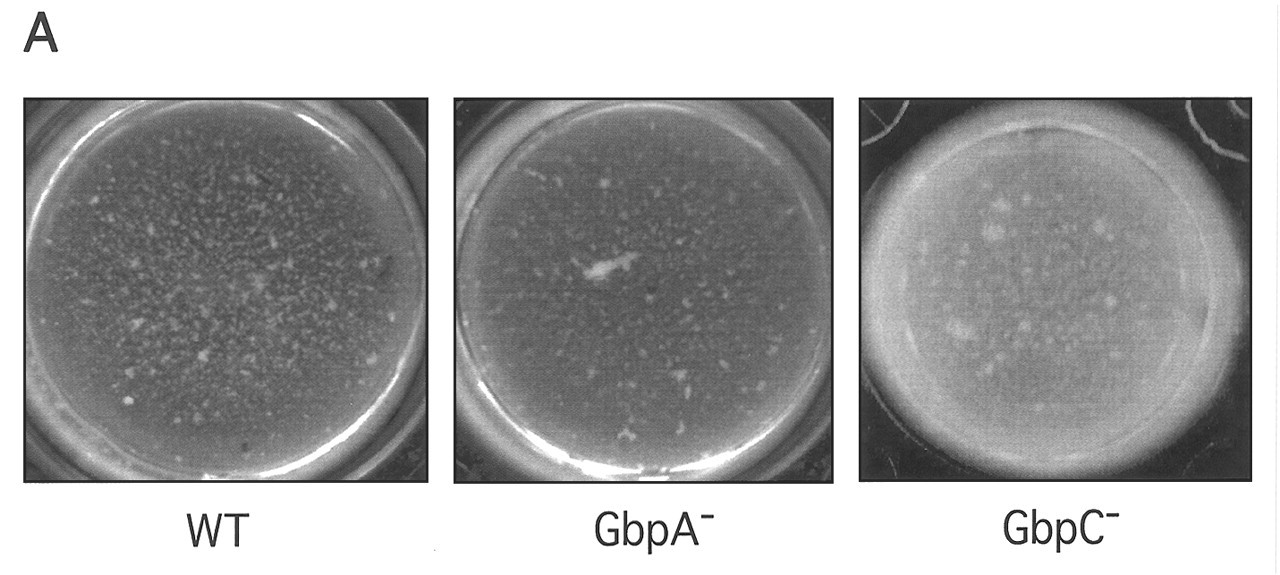
Since glucan and mutan are exo-polysaccharides that promote the formation of oral biofilms, there is considerable interest in whether some or all of the GBPs contribute to plaque biofilm development or whether glucan is used as an alternative form of cell anchorage. As a first step in answering this question, null mutants for each of the S. mutans GBPs have been engineered, and the nature of the biofilms formed by pure cultures in vitro has been examined. The loss of any of the GBPs has an impact on adhesion or biofilm structure. The loss of GbpA changes the architecture of sucrose-dependent biofilm deposited within microtiter dishes (Hazlett et al., 1999) (Fig.). The null mutant of GbpB is unstable (Mattos-Graner et al., 2002), precluding in vitro investigation. However, the amount of GbpB produced by clinical isolates correlates with their biofilm-forming abilities (Mattos-Graner et al., 2001). The loss of GbpC in the same model system as used with GbpA results in a biofilm about twice as thick as normal (Fig.). It is still unclear whether the thickness of the GbpC biofilm is due to the deposition of a greater number of organisms, an enhanced synthesis of glucan, or occurs via a mechanism that has not yet been described. The loss of GbpD is associated with the formation of an extremely fragile biofilm on wires suspended in sucrose-containing broth cultures (Russell, personal communication).
The instability of the gbpB mutant indicates that it may be an essential protein; its homology to peptidoglycan hydrolase may mean that GbpB is essential for cell-wall cycling and synthesis (Mattos-Graner et al., 2001, 2002). Recently, it became apparent that GbpB was identical to the immunodominant glycoprotein IDG-60 and the general stress protein GSP-781 (Chia et al., 2001a,b; Mattos-Graner et al., 2001). Loss of IDG-60 did indeed affect the stability of the cell wall, resulting in a pleomorphic shape and retarded growth rate (Chia et al., 2001b). It is unclear why the insertionally inactivated idg60 strain was apparently more stable than the insertionally inactivated gbpB strain, though different growth conditions or strain backgrounds might provide an explanation. Thus, GbpB may be an enzyme whose glucan-binding properties help keep it cell-associated in the absence of a cell-wall anchor. Alternatively, its glucan binding may be an artifact, its primary ligand perhaps residing within the cell wall.
GbpD may also function as an enzyme, since it displays amino acid homology to the alpha/beta hydrolase family of enzymes (Shah and Russell, 2002). Unlike the GbpB, however, GbpD shares homology in its glucan-binding domain with those in GbpA and the GTFs. Therefore, the fragile nature of the biofilm formed by the GbpD knockout mutant could be due to the loss of the physical presence of this GBP, the loss of its putative enzymatic activity, or a combination of the two.
The GbpA and GbpC may be GBPs whose physical presence contributes to optimal aggregation and plaque biofilm architecture. When GbpA and GbpC knockouts are tested for their abilities to form a biofilm in a microtiter dish model (Banas et al., 2001) with sucrose-containing medium, they both behave quite distinctly compared with the wild-type. The GbpA knockout forms a smoother biofilm that coats the substratum more evenly (Hazlett et al., 1999), while the GbpC knockout produces a biofilm at least twice as thick as that produced by the wild-type (Fig.; unpublished data). Within the biofilm formed by the GbpA knockout, there is an accumulation of organisms that have undergone a genetic re-arrangement between the contiguous and highly homologous gtfB and gtfC genes (Hazlett et al., 1998). Clones with this recombinant, hybrid GTF have previously been shown to produce less total glucan, and, within the lower total, the ratio of water-soluble to -insoluble glucan is increased (Ueda and Kuramitsu, 1988). The spontaneous occurrence of this recombination has been known for some time (Perry et al., 1983; Ueda and Kuramitsu, 1988; Yamashita et al., 1992). Whether or not an increase in the frequency of recombination, or ecological pressures, can explain the large number of recombinants in the GbpA knockout biofilm is uncertain, but their accumulation may be an indication that the change in biofilm structure that accompanies the loss of GbpA leads to a more stressful environment. The precise mechanisms for how GbpA and GbpC contribute to biofilm architecture are not known and will require further research.
The significance of the dextranase inhibitor, Dei, may be to affect the ratio of GTF to dextranase (Sun et al., 1994), which, in turn, influences the synthesis of water-insoluble glucan (Walker et al., 1981; Tanzer et al., 1985). Shaw et al. (1997) have determined that, at low growth rates, the activity of free Dei is high, and predominantly water-soluble glucan is synthesized. At high growth rates, S. mutans synthesizes predominantly water-insoluble glucan that binds Dei and suppresses its inhibitor activity. Any free Dei is bound to dextranase in enzyme-inhibitor complexes, but dextranase remains in excess (Sun et al., 1995; Shaw et al., 1997). While the pathogenic mechanisms of the MS for humans or animals are generally thought to be similar, the various numbers and types of GTFs and GBPs highlight the unique biology of each species.
Mechanisms of Glucan Binding
As noted above, the glucan-binding domains of the GTFs generally share considerable levels of amino acid homology. Certain of the GBPs, such as GbpA, GbpD, and Dei, also share homology with GTF glucan-binding domains. Circular dichroism analyses of the C-terminal glucan-binding domains of GbpA (Haas et al., 1998) and the S. downei GTF-I indicate a high percentage of beta-sheet (Monchois et al., 1999a). The beta-sheets may be capable of folding to form a hydrophobic core with an outer domain that interacts with glucan. The flexibility of the beta sheets may facilitate binding to both water-soluble and -insoluble glucan, but the specific sequence of any particular domain may be critical for optimal tertiary folding. The basis for glucan binding by GbpB, GbpC, or the S. sobrinus GBPs has not been determined. GbpC shares homology with the AgI/II family of proteins that at one time were reported to possess glucan-binding properties. However, this claim could be traced only to a personal communication, and experimental verification could not be found (Curtiss, 1985).
Evidence for the functionality of the glucan-binding domains has been obtained by a variety of glucan-binding methods, often following proteolytic cleavage or the generation of deletion clones and fusion proteins (Ferretti et al., 1987; Mooser and Wong, 1988; Kato and Kuramitsu, 1990; Wong et al., 1990; Abo et al., 1991; Nakano and Kuramitsu, 1992; Lis et al., 1995; Vickerman et al., 1996; Haas and Banas, 2000). Despite the similarity in amino acid sequence and composition, glucan-binding domains cannot necessarily be interchanged among GTFs without affecting the nature of the glucan product (Nakano and Kuramitsu, 1992). It might be expected that the glucan-binding domain for each GTF would have highest affinity for the product it synthesizes. However, this might not be true. Haas and Banas (2000) estimated the dissociation constant for the S. mutans Gtf-S (gtfD) glucan-binding domain and dextran as being identical to that for the S. mutans Gtf-I (gtfB), 2.5 x 10-7 M and 2.4 x 10-7 M, respectively. These values are in the range of the affinity of GBP-1 (3 x 107 M-1) (Landale and McCabe, 1987) for dextran, but all are significantly lower than the dissociation constant measured for the glucan-binding domain from GbpA, 8.3 x 10-8 M (Haas and Banas, 2000). The lower affinity for dextran of these GTFs, relative to that of GbpA, may be related to the energetics of glucan synthesis; this may involve repeated association and dissociation of the GTF enzyme from the polymer during glucan synthesis (Kaseda et al., 2000). The unit of a single dextran chain that interacts with the glucan-binding domain of S. sobrinus GTF-I is about 15 glucosyl residues and has a relatively low affinity of 2 x 105 M-1 (Kaseda et al., 2000). However, the large number of glucosyl moieties in dextran available to interact with the glucan-binding domain strengthens the overall interaction and can be exploited for the purification of recombinant proteins (Kaseda et al., 2001).
Most of the studies examining glucan-binding domains have been done with commercial preparations of dextran. These proteins can also bind both water-soluble and -insoluble glucan produced by the MS. As was previously noted, the Dei has a preference for water-insoluble glucan, and its affinity is reportedly stronger than that for GTFs or GbpA (Sun et al., 1994). This is consistent with observations for GbpA where 1000-fold greater quantities of mutan than dextran were necessary to bring about comparable retardation of migration of GbpA by affinity electrophoresis (Haas and Banas, 2000). The significance of a stronger affinity for either dextran or mutan will likely depend on the contribution of each GBP.
Regulation of Expression
Expression and activity of the GTF enzymes can be influenced by several environmental conditions, including pH, ion concentration, availability of acceptor molecules, and oxidation-reduction potential (reviewed by Monchois et al., 1999b). The presence of multiple enzymes involved in carbohydrate metabolism and transport, glucan synthesis and secretion, and degradation (e.g., hydrolases and dextranases) in the oral streptococci, as well as factors that affect post-translational modifications of the GTF enzymes, have traditionally complicated the interpretation of regulatory studies. Although expression of gtf genes was originally thought to be constitutive, molecular analysis with transcriptional reporter gene fusions (Wexler et al., 1993; Goodman and Gao, 2000; Li and Burne, 2001) or direct measurements of specific mRNA transcripts (Fujiwara et al., 2002) has resulted in the identification and characterization of environmental signals, including pH, carbohydrate availability, and growth phase, that have different influences on expression of gtfB, gtfC, and gtfD genes in S. mutans. However, specific regulatory determinants have not been identified for gtf genes in the MS. In contrast, a regulatory determinant for gtf structural genes has been identified in commensal streptococci, which appear to have only a single GTF enzyme. The first-identified was S. gordonii rgg, which positively regulates transcription of gtfG (Sulavik et al., 1992). Similarly, disruption of the rgg gene in S. oralis also resulted in decreased GTF expression (Fujiwara et al., 2000). A phase variation between high and low levels of GTF activity (Tardif et al., 1989), as well as genetic or nucleotide sequence analysis of mutant strains (Haisman and Jenkinson, 1991; Vickerman et al., 1997a), indicates that additional genes also affect GTF expression in S. gordonii.
Little is known about the regulation of GBPs. Yoshida et al. (2002) have recently reported that the disruption of the luxS gene in S. mutans disrupts biofilm formation, perhaps due to enhanced auto-aggregation. The authors speculate that the expression of GTFs and GBPs may be regulated by autoinducer-2 (AI-2), the signaling molecule for a quorum-sensing system that is conserved among many Gram-positive and Gram-negative bacterial species (Schauder et al., 2001). Similarly, there is also a recent report that differential display PCR indicated that inactivation of luxS in S. gordonii resulted in a down-regulation of several genes, including gtfG (McNab et al., 2002). Additional studies will be needed to confirm that the varied expression of GTFs and/or GBPs seen in streptococcal luxS mutants is not due to indirect effects resulting from the role of LuxS in S-adenosyl-homocysteine metabolism (Winzer et al., 2002). Sato et al. (2000) have provided evidence for negative regulation of gbpC from a locus with homology to a family of response regulators that are part of two-component signaling systems. Indirect evidence hints at regulation for gbpA, but the genetic basis for this, if indeed it exists, is not known. Measurement of transcription from the gbpA promoter with use of a cat reporter gene provided evidence for constitutive expression in the presence and absence of sucrose (Banas et al., 1997). An S. mutans strain with streptococcal GTP-binding protein (SGP) synthesis suppressed by antisense RNA displayed significantly greater levels of GbpA as detected by Western immunoblotting (Baev et al., 1999). It is likely that, in the near future, several microarray analyses will be undertaken and the basis for the regulation of GTF and GBP expression will be known in much greater detail, both at the level of the individual gene and at the level of signaling networks.
Clinical Significance
Role in dental plaque biology
The significance of glucan-binding proteins to the biology of the oral streptococci is often evaluated in the context of virulence, particularly dental caries. This is understandable given the integral role of glucan in the cariogenicity of the MS, and the fact that non-GTF GBPs have so far been characterized only in the MS. Nevertheless, as genome sequences are determined for more commensal streptococci, proteins with sequence similarities to the GBPs continue to be detected. At present, a BLAST search (Altschul et al., 1990) of the NCBI database as well as unfinished oral streptococcal genomes (e.g., www.tigr.org and www.sanguis.mic.vcu.edu) suggests that proteins with amino acid sequences significantly similar to gbpA, gbpB, and gbpC are present among a variety of streptococci. Although results of searching sequence homology databases must be interpreted cautiously, functional analysis of these similar proteins may provide insights into additional biological roles for GBPs and their homologues.
The evaluation of MS proteins in caries development is most often based on experiments in rodent models that allow for the comparison of isogenic strains. The most popular model is the rat, though hamsters, mice, and primates have also been used. When gnotobiotic rats are used, they are typically fed a diet with 5% sucrose. This amount was found to be sufficient for maximal caries development (Michalek et al., 1977). In specific-pathogen-free rodents, a higher level of sucrose, 56%, is required to establish infection (Krasse, 1965). Disruption of genes encoding GTFs reduces the amount of glucan synthesized, and the efficiency of sucrose-dependent colonization and/or the cariogenicity usually decreases (Tanzer et al., 1974, 1985; Munro et al., 1991, 1995; Yamashita et al., 1992, 1993; Kuramitsu, 1993), though the extent of caries reduction can vary and depend on the model system. The extent to which disruption of GTFs would reduce caries in humans is uncertain. However, a positive correlation between water-insoluble glucan synthesis and caries rates has been reported (Mattos-Graner et al., 2000). No information is available on the in vivo effects of knocking out GTFs in the non-MS oral streptococci. It is likely that a full understanding of the significance of GTFs and GBPs will require a broader consideration than their contribution to caries, including comparisons with proteins that share homology within non-MS hosts.
Among GBPs, only a GbpA knockout mutant has been tested in an animal model (Hazlett et al., 1998). Unexpectedly, it proved to be more cariogenic than the wild-type. Hazlett et al. (1999) proposed that the increase in cariogenicity was related to a change in the plaque biofilm structure to one that coats the tooth surface more completely. In this model, the smaller microcolonies characteristic of the GbpA knockout biofilm harbor a greater percentage of organisms directly exposed to available carbohydrates and thereby produce more acid in direct proximity to the tooth enamel. The flatter biofilm formed by the knockout may be due to the loss of structural strength normally provided by GbpA. The ratio of glucan to GbpA may also be critical. Overexpression of GbpA results in larger than normal aggregates of S. mutans (unpublished observations) consistent with GbpA making a contribution to biofilm cohesiveness.
Role in potential caries vaccines
A substantial body of literature supports the importance of GTFs and GBPs in dental caries based on experiments in which immunization with these proteins (or portions of them) can reduce caries rates upon challenge with the MS [see recent reviews by Koga et al. (2002) and Michalek et al. (2001)]. The GTFs have been an obvious target for immunization, given their integral contribution to sucrose-dependent colonization and accumulation. Immunogens corresponding to the glucan-binding domain shared by the GTFs, GbpA, and GbpD have been shown to be protective in rats (Yu et al., 1997; Jespersgaard et al., 1999a,b; Taubman et al., 2000), though immunization with the entire GbpA protein did not elicit a protective immune response (Smith et al., 1997). Another prominent immunogen has been AgI/II (also designated as SpaP, AgB, 1F, P1, SR, MSL-1, or PAc), which coats the cell surface and promotes sucrose-independent adhesion. GbpC was found to have homology with the AgI/II family of proteins (Sato et al., 1997). GbpC and GbpD have not been tested in immunization studies, but it is conceivable that immunization with GTFs or AgI/II elicits antibody that cross-reacts with these GBPs, and that interference with the GBPs could contribute in some measure to the protective effects afforded by the immunization. Previously, Smith et al. (1997) demonstrated that antibody raised against GbpA reacted with MS GTFs, and antibody raised against GTF had reactivity with GbpA. Immunization with GbpB is protective against cariogenic challenge in rats (Smith and Taubman, 1996), as is passive antibody to GbpB (Smith et al., 2001). The protective effects of the GBPs from other MS have not been tested.
Role in infective endocarditis
Recently, Nakano et al. (2002) reported the isolation of S. mutans strains from the blood of endocarditis patients. One of the strains was negative for GbpA, based on Southern hybridization. A second strain had multiple defects, including a lack of GbpA, a lack of the serotype-specific polysaccharide antigen, and an altered GbpC; the alteration in GbpC consisted of a deletion in the GbpC cell-wall anchoring region. This latter strain displayed significantly reduced sucrose-independent adhesion and dextran binding in vitro, and resulted in decreased caries scores in rats compared with those resulting from colonization with an S. mutans MT8148 reference strain. The reductions in adhesion and dextran binding matched those seen with a genetically engineered MT8148 strain interrupted in the gbpC gene, but caries rates for the isogenic mutant were not reported.
The recovery of GbpA- and GbpC-deficient strains of S. mutans from endocarditis patients may be of little significance or may be an indication that the GBPs do not contribute to virulence within the blood. Many species of oral streptococci are associated with infective endocarditis, and it has been proposed that the glucans synthesized by GTFs play a role in virulence. Several studies have presented data showing that glucan-deficient strains of S. mutans or S. sanguis do not establish endocarditis as efficiently as parental strains in rabbit models (Pelletier et al., 1978; Scheld et al., 1978; Meddens et al., 1984; Munro and Macrina, 1993), and are less adhesive to endothelial cells or heart tissue (Ramiriz-Ronda, 1978, 1980; Vacca-Smith et al., 1994). Dall and Herndon (1990) found a correlation between the amount of glycocalyx produced by clinical isolates of viridans streptococci and the size of infected cardiac vegetation. However, other studies could not corroborate a role for GTFs and glucan, at least for certain strains of S. sanguis or S. gordonii (Herzberg et al., 1983, 1990; Crawford and Russell, 1986; Wells et al., 1993). The variable results obtained with different oral streptococcal species may reflect the differences in model systems used in these studies. Thus, the role of glucans and GBPs in the pathogenesis of infective endocarditis is not clear. Since blood does not contain sucrose, one potential role of GTFs and glucan would be to coat the bacteria prior to their introduction into the bloodstream. It is possible that the exo-polysaccharide might then act in a manner similar to that of other streptococcal capsular polysaccharides to protect the bacteria from phagocytosis (Claverys et al., 2000). In addition, it is possible that glucans and/or the glucan-binding domains on cell-associated GBPs (including GTFs) could enhance the initial colonization of cardiac tissues, either directly or indirectly, by allowing formation of an optimally sized bacterial-glucan aggregate.
Summary
The GBPs of the oral streptococci represent a heterogeneous group of proteins. It is clear that their designation as glucan-binding proteins is based on a common property that does not necessarily define a single function. It has been well-established that the glucans synthesized by GTFs make a significant contribution to the extracellular polysaccharide matrix that forms an integral component of the dental plaque biofilm. These glucans are exploited by the MS, which do not have the range of ligands for the acquired enamel pellicle that are typical of primary plaque colonizers, to colonize smooth tooth surfaces. The well-characterized GBPs of MS are primarily thought to play this role. Nonetheless, the large complement of GTFs within Streptococcus salivarius, a species that is preferentially found on the soft tissues of the oral cavity (Hamada and Slade, 1980), suggests that biology of GTFs may be more sophisticated than just a contribution to colonization of dental plaque. Glucans have been postulated to act alone, or via interactions with other amphipathic molecules, to protect bacteria from potentially toxic components (Gibbons, 1983), e.g., by modulation of local acid and/or nutrient concentrations, as well as to affect diffusion of molecules within the plaque matrix (McNee et al., 1982; Dibdin and Shellis, 1988; van Houte et al., 1989). The ability to interact with and maintain this glucan-rich biofilm via GTF activity may provide significant ecological advantages to oral bacterial species.
GBPs such as GbpA and GbpD may have evolved from GTFs, retaining the glucan-binding domain, but with greater flexibility for evolving higher affinities for glucan. This may elevate adhesion to glucans synthesized not only by the MS but also by pioneer commensal plaque colonizers, providing a colonization advantage in the presence of sucrose. The lectin-like activity of GBPs like the S. mutans GbpC and the S. sobrinus GBP-4/GBL might contribute to colonization by promoting the optimal aggregate size for subsequent attachment to the plaque surface. The various types of binding domains found among the GBPs may be an example of convergent evolution that takes advantage of the properties of the extracellular milieu. Thus, the glucan binding by GbpB may represent a means of cell-surface association independent of common amino acid sequence motifs for Gram-positive cell membrane anchoring and cell wall attachment (Navarre and Schneewind, 1999).
The number of GBPs, the conservation of certain types of GBP properties, the role of some GBPs in modulating virulence, and the immunogenic properties of certain GBPs are testimonies to their importance. Future studies, including genome sequencing of both pathogenic and commensal oral species, should greatly advance the understanding of the distribution, function, and regulation of the GBPs in the biology of the oral streptococci. Comparing the genomes of oral bacteria occupying the same ecological niche may also provide insights into the specificity of these proteins. There is still much to be learned about roles that GBPs play in maintaining a stable, commensal microbial population in the oral cavity. Biological balance in this system is essential for protection against pathogenic states such as caries or periodontal disease that may result from a disruption of the ecological balance among a mixed-species biofilm community.
Footnotes
3
present address, Department of Periodontics and Endodontics, 240 Squire Hall, SUNY at Buffalo, Buffalo, NY 14214;
Acknowledgements
We gratefully acknowledge the critical input of Roy R.B. Russell and the support from NIDCR grants DE10058 (J.A.B.) and DE11090 (M.M.V.).
