Abstract
Tooth eruption is a complex and tightly regulated process that involves cells of the tooth organ and the surrounding alveolus. Mononuclear cells (osteoclast precursors) must be recruited into the dental follicle prior to the onset of eruption. These cells, in turn, fuse to form osteoclasts that resorb alveolar bone, forming an eruption pathway for the tooth to exit its bony crypt. Some of the molecules possibly involved in the signaling cascades of eruption have been proposed in studies from null mice, osteopetrotic rodents, injections of putative eruption molecules, and cultured dental follicle cells. In particular, recruitment of the mononuclear cells to the follicle may require colony-stimulating factor-one (CSF-1) and/or monocyte chemotactic protein-1 (MCP-1). Osteoclastogenesis is needed for the bone resorption and may involve inhibition of osteoprotegerin transcription and synthesis in the follicle, as well as enhancement of receptor activator of NFκB ligand (RANKL), in the adjacent alveolar bone and/or in the follicle. Paracrine signaling by parathyroid-hormone-related protein and interleukin -1α, produced in the stellate reticulum adjacent to the follicle, may also play a role in regulating eruption. Osteoblasts might also influence the process of eruption, the most important physiologic role likely being at the eruptive site, in the formation of osteoclasts through signaling via the RANKL/OPG pathway. Evidence thus far supports a role for an osteoblast-specific transcription factor, Cbfa1 (Runx2), in molecular events that regulate tooth eruption. Cbfa1 is also expressed at high levels by the dental follicle cells. This review concludes with a discussion of the several human conditions that result in a failure of or delay in tooth eruption.
(I) Introduction
The development of dentition is not complete when the mineralized tissues of the crown and root are formed, but continues through post-natal life until teeth emerging from within the alveolus reach the position of functional occlusion. For developmental biologists, tooth eruption is a fascinating post-natal event that does not lend itself easily to categorization. It is the only instance in which a developing organ (the tooth) must exit the confines of its bony crypt. Skeletal biologists view the process of tooth eruption as a valuable model to study bone remodeling, since the emergence of a tooth into the oral cavity involves both coupled and uncoupled bone turnover events. For cell and molecular biologists, tooth eruption provokes several questions concerning the tightly programmed series of signaling interactions between cells of the connective tissue sac surrounding the tooth (dental follicle) and the surrounding alveolus.
In recent years, knowledge of the biology of tooth eruption has greatly increased. What has emerged is the realization that interactions among osteoblasts, osteoclasts, and dental follicle involve a complex interplay of regulatory genes that encode various transcription factors, proto-oncogenes, and soluble factors. For the clinician faced with treating both simple and complex dental complications that arise from abnormal tooth eruption, as seen in numerous genetic and acquired disorders, knowledge about the basic molecular mechanisms involved is essential.
This review critically evaluates our current understanding of tooth eruption based on previous in vivo and in vitro studies. Gaps in our knowledge about its cellular, molecular, and genetic determinants are also highlighted. An effort has been made to include information about human conditions affecting tooth eruption. The discussion provided will point to future avenues for translational research and the development of new therapeutics that can effectively induce impacted teeth to erupt in a timely manner.
(II) Basic Biology of Tooth Eruption
(A) Bone resorption
Given that the unerupted tooth is encased in alveolar bone, an understanding of the biology of bone resorption to form an eruption pathway is a central theme in tooth eruption. To that end, Cahill (1969) dissociated tooth movement from eruption pathway formation by placing transmandibular wires over dog premolars prior to the onset of eruption. An eruption pathway formed in the alveolar bone above the temporarily impacted premolars, and when the wires were removed, the tooth rapidly erupted. In succedaneous dentition, this pathway follows the gubernacular canal above each tooth; i.e., bone resorption widens this canal to allow the crown to move through it and exit the alveolar bone (Cahill et al., 1988). Thus, there is a template (gubernacular canal) for the eruption pathway, and, at a given time, bone resorption by osteoclasts will occur in this template, even if the tooth is stationary. Consequently, formation of the tooth eruption pathway is a localized, genetically programmed event that does not require pressure from the erupting tooth.
The requirement of alveolar bone resorption for tooth eruption was first noted in osteopetrotic rodents. Osteopetrosis, a congenital bone disease marked by reduced bone resorption but not reduced bone formation (Marks, 1973), is often characterized by failure of teeth to erupt. For example, in the toothless rat (tl) first described by Cotton and Gaines (1974), the teeth are fully formed but do not erupt. Such animals have fewer osteoclasts that are probably non-functional, given their weak staining for tartrate-resistant acid phosphatase (Marks, 1977; Seifert et al., 1988). This was confirmed by scanning electron microscopy that showed the absence of bone resorption in the crypts of tl rats, in contrast to the scalloped crypt surfaces reflecting bone resorption in normal rats (e.g., see Marks et al., 1994). In another strain of osteopetrotic rats (ia) in which the teeth do not erupt, irradiation and injection of spleen cells from normal rats into the ia rats results in tooth eruption as a result of functional osteoclasts now being formed (Marks, 1981). Osteopetrotic (op/op) mutant animals have reduced numbers of osteoclasts compared with the normal mouse, and their teeth do not erupt (Marks and Lane, 1976).
A direct causal relationship between resorption of alveolar bone by osteoclasts and tooth eruption has been shown in studies in which pamidronate, a bisphosphonate that reduces bone resorption by osteoclasts, was injected into rats (Grier and Wise, 1998). In these animals, the eruption of the first mandibular molars and mandibular and maxillary incisors was delayed by 8, 1.6, and 2.5 days, respectively. The cytological effect of this treatment was to increase the size of the osteoclasts, including their number of nuclei, suggesting that the osteoclasts might be increasing in size to compensate for their reduced ability to resorb bone (Grier and Wise, 1998).
(B) Role of dental follicle
Originating from cranial neural crest mesenchyme, the dental follicle (DF) is a loose connective tissue sac surrounding the enamel organ of each tooth. Destined to develop into the periodontal ligament (PDL), the DF is required for eruption to occur, as not only a follicle but also perhaps as the PDL.
The necessity of the dental follicle for tooth eruption was demonstrated by elegant surgical studies in the dog showing that removal of the DF from premolars prior to the onset of eruption prevented the unerupted tooth from erupting (Cahill and Marks, 1980). In contrast, leaving the DF intact, but removing the tooth and inserting an artificial replica of the tooth such as dental amalgam, resulted in eruption of the artificial tooth (Marks and Cahill, 1984). This experiment eliminated many previous theories of eruption, because possible propulsive tissues such as the dental pulp and roots were absent. It has long been known that the roots of teeth are not required for eruption, because rootless teeth will erupt in children, just as they will when the roots are destroyed in irradiated monkeys (Gowgiel, 1961).
The derivative of the dental follicle, the PDL, appears to play a role in continuous eruption of rodent and lagomorph incisors. In mandibular incisors of rabbits (Berkovitz and Thomas, 1969) and rats (Moxham and Berkovitz, 1974), transection of roots, followed by insertion of an impermeable barrier, resulted in the distal portion continuing to erupt. As emphasized by Berkovitz and Thomas (1969), this experiment downplayed previous theories of tooth eruption mechanisms such as root elongation, pulp cell proliferation, bone deposition, and tissue fluid pressure. Thus, they concluded that the force of eruption might come from the PDL.
Two caveats pertain to these elegant studies of the PDL in continuously erupting incisors. First, these results apply to the supraosseous and supragingival phases of eruption and do not address the factors involved in the intraosseous phase of eruption. Second, these are continuously erupting teeth, not a limited eruption as in human dentition. In teeth of limited eruption, the fibers of the DF are not attached to the alveolar bone and are not oriented to move the tooth during the intraosseous phase of eruption (Cahill and Marks, 1982). Perhaps during the supraosseous phase of eruption, after the tooth has pierced the gingiva, the DF derivative (PDL) provides an eruption force.
The requirement for the presence of the DF, or its successor, is also seen clinically. A rare disease of multiple calcifying hyperplastic dental follicles (MCHDF) is characterized by unerupted teeth with atypical follicles containing hyperplastic dense fibrous connective tissue and numerous deposits of calcified tissue (Sandler et al., 1988; Gardner and Radden, 1995; Gomez et al., 1998). In a genetic disorder, mucopolysaccharidosis VI (Maroteaux-Lamy Syndrome), eruption of the permanent molar teeth is retarded, and the dental follicles of such teeth are abnormal in that they have excessive accumulations of dermatan sulfate (see review by Sauk, 1988). Thus, in both of these syndromes, abnormal DFs result in unerupted teeth.
(C) Cellular events
Early cytological studies of dog premolars showed that, prior to the onset of eruption, there is an influx of mononuclear cells into the coronal portion of the dental follicle, with a concurrent increase in the numbers of osteoclasts in the coronal third of the alveolar bony crypts (Marks et al., 1983). This is followed by a sharp decrease in both mononuclear cell and osteoclast numbers. Subsequent examination of the ultrastructure of the DF demonstrated that the mononuclear cells had features of osteoclast precursors, and such cells were often observed immediately adjacent to osteoclasts (Wise et al., 1985). Thus, it was postulated that the influx of mononuclear cells was required for formation of osteoclasts needed to resorb bone for the eruption pathway. The sequence of cellular events—influx of mononuclear cells to form osteoclasts followed by a decline in the number of both—was shown to occur in rats (Wise and Fan, 1989; Cielinski et al., 1994). In the rat first mandibular molar, the influx of mononuclear cells into the DF reaches its peak at day 3 post-natally, the time of maximal number of osteoclasts on the surface of the bony crypt. This is followed by a sharp decline in numbers for both cell types. Although eruption of the first mandibular molar does not occur until day 17 or 18 post-natally, it is apparent that the major cellular events must occur earlier to have time to resorb the alveolar bone.
Recently, the cellular events of eruption have been described in a mouse. As with the rat and dog, there is a peak of mononuclear cells and osteoclasts, but that peak is at day 5 post-natally in the mouse, followed by a decline and then a lesser peak at day 9 (Volejnikova et al., 1997).
These studies demonstrated that some of the functions of the DF in tooth eruption are to serve as a target tissue to attract the mononuclear cells, to serve as a repository for the cells, and to provide a milieu whereby these cells can fuse to form osteoclasts. Morphologically, because the DF is interposed between the alveolar bone and tooth, it is in an ideal location to regulate the cellular events of eruption. Not only can the DF “deliver” the resorptive cells to the alveolar bone, but it also is in a position to receive signals from the tooth. As discussed in a later section, such paracrine signaling may emanate from the stellate reticulum of the enamel organ of the tooth.
(III) Molecules Involved in Tooth Eruption
(A) Eruption molecules
Tooth eruption appears to be a programmed, localized event whereby a given tooth erupts at its appointed time. The molecules that initiate eruption, their localization, and their regulation of the cellular events of eruption—all must fit within the context that each tooth erupts independently. In other words, tooth eruption is a localized event rather than a systemic one.
Determination of the molecules that may be required for eruption began with the isolation of epidermal growth factor (EGF) by Cohen (1962) and his discovery that its injection into rodents accelerated incisor eruption. Subsequent studies have amply confirmed these results (e.g., see Hoath, 1986; Lin et al., 1992) and demonstrated that, in rats, injections of EGF in the early post-natal period of days 0-3 are critical for maximizing the effects of EGF (Hoath, 1986). This theme of an eruption molecule exerting its effect on rodent eruption early in post-natal development is a common one that appears to apply to all the putative eruption molecules, as will be discussed.
Incisor eruption in mice was also shown to be accelerated by another growth factor, transforming growth factor-alpha (TGF-α) (Tam, 1985). In view of the fact that TGF-α and EGF utilize the same receptor (Todaro et al., 1980; Brachmann et al., 1989; Wong et al., 1989), it is not surprising that both molecules would have the same effect upon eruption. Consequently, in null mice devoid of the TGF-α gene, the teeth still erupt on schedule (Mann et al., 1993), suggesting that EGF alone can initiate incisor eruption. This redundancy of function of potential tooth eruption molecules is a common theme that will be revisited later.
Osteopetrotic rodents have also provided information about the molecules that are required for eruption. In particular, osteopetrotic (op/op) mice have unerupted teeth and lack functional colony-stimulating factor-1 (CSF-1) activity (Felix et al., 1990a; Wiktor-Jedrzejczak et al., 1990; Yoshida et al., 1990). Injections of CSF-1 induce tooth eruption in these op/op mice (Kodama et al., 1991; Wiktor-Jedrzejczak et al., 1991; Niida et al., 1997). In toothless (tl/tl) rats, injection of CSF-1 restores bone resorption and tooth eruption (Iizuka et al., 1992). It is important to note that injections of CSF-1 must be done early post-natally for eruption to occur—i.e., no later than day 1 to induce incisor eruption and no later than day 3 for first mandibular molar eruption (Iizuka et al., 1992). Molar eruption in normal rats is also accelerated by 3 to 4 days following injections of CSF-1, an effect that is probably due to the CSF-1 increasing the number of TRAP-positive mononuclear cells present in the follicle at day 3, as well as an increase in the number of osteoclasts on the alveolar bone (Cielinski et al., 1994).
Further analysis of the effects of EGF and CSF-1 on tooth eruption suggest that they may have different actions on incisor vs. molar eruption. In a study comparing their direct effects on eruption, EGF accelerated incisor eruption but not molar eruption, whereas CSF-1 caused the opposite (Cielinski et al., 1995). In addition, EGF neither increased the numbers of mononuclear cells in the DF of the molars nor increased the numbers of osteoclasts on alveolar bone around the molars, in contrast to the increases induced by CSF-1 (Cielinski et al., 1995).
In recent years, experiments utilizing knockout mice have added to our knowledge of the molecules that are needed for eruption to occur. Thus, null mice devoid of either the transcription factor gene c-fos (Grigoriadis et al., 1994) or the transcription factor genes NFκB1 and NFκB2 (Franzoso et al., 1997; Iotsova et al., 1997) lack osteoclasts, and the teeth do not erupt. Mice lacking a functional parathyroid-hormone-related protein (PTHrP) gene die at birth due to premature ossification of the rib cage. However, genetic rescue experiments performed by the crossing of col II–PTHrP transgenics with PTHrP mutants survive and show a failure in tooth eruption (Philbrick et al., 1998). Recently, it has been shown that knockout mice devoid of the osteoclast differentiation factor (ODF) gene, a gene that is required for osteoclast formation and activation, have unerupted teeth (Kong et al., 1999).
We have also examined abnormalities in tooth eruption in knockout mice lacking a functional type I receptor for interleukin-1α (IL-1R). In such mice, eruption is delayed for 2 days in molars and 1 day in incisors (Huang and Wise, 2000). In rats, injection of IL-1α accelerated eruption by 2 days (Wise et al., 2000a). Thus, IL-1α may play a role in tooth eruption, but it is likely that another molecule may substitute for it, because eruption eventually occurs in the absence of its effect.
(B) Localization and chronology of eruption molecules
If we assume that eruption is a programmed, localized event, in what dental tissues are the eruption molecules localized? In vivo techniques, including in situ hybridization, reverse-transcription/polymerase chain-reaction (RT-PCR), and immunostaining, have been used to determine whether a given eruption gene is both transcribed and translated, as well as the time of its expression (see review by Wise, 1998a). Many of these techniques were also applied in vitro to cultured dental follicle cells (Wise et al., 1992a). These combined studies have demonstrated that the putative eruption genes and their products are localized primarily in either the dental follicle or stellate reticulum (SR), as seen in Table 1. The latter clearly reveals that the tissue required for eruption, the dental follicle, produces the majority of the potential eruption molecules. The remainder of the molecules reside in the stellate reticulum adjacent to the DF. Paracrine signaling from the molecules in the SR affects gene expression of the molecules in the DF (see reviews by Wise and Lin, 1995; Wise, 1998a). For example, the receptor for IL-1α (IL-1R) is localized in the DF, and its gene expression is enhanced either by a ligand present in the SR, TGF-β1, or by a molecule present in the DF, EGF (Wise et al., 1995b). The presence of this receptor in the DF enables IL-1α to act on the DF to enhance CSF-1 gene expression both in vitro (Wise and Lin, 1994) and in vivo (Wise, 1998b). IL-1α also enhances MCP-1 gene expression (Que and Wise, 1998), as well as MCP-1 synthesis and secretion (Wise et al., 1999a) in the DF. Recently, it has been shown that IL-1α enhances NFκB gene expression and translation to the nucleus (Que et al., 1999). Paracrine signaling between some of the molecules within the DF also can occur.
In the later 1980’s, Cahill et al. (1988) theorized that the enamel organ might contain the “biological clock” that regulates the timing of tooth eruption. Although still a hypothesis, the presence of IL-1α and PTHrP in the SR suggests that the SR portion of the enamel organ may play such a role. In the dog, a unique protein, DF-95, is present at the onset of eruption and then declines in amount (Gorski et al., 1988a,b). Surprisingly, DF-95 immunolocalizes to the SR (Gorski et al., 1994). Thus, if the enamel organ has a regulatory role in eruption, that regulation probably is coming from the SR.
The chronology of the localization of the potential eruption molecules parallels the timing of the cellular events of eruption. In the rat, the maximal influx of mononuclear cells into the DF of the first mandibular molar is at day 3 post-natally, the time at which almost all of the putative eruption molecules are maximally expressed (see review by Wise, 1998a). Following this peak of mononuclear cells and osteoclasts, there is a sharp decline in their numbers, just as there is a decline in the expression of the molecules after day 3. As an example, the peak expression of CSF-1 in the DF, as well as the maximal immunostaining, is at day 3, with a subsequent decline in expression and immunostaining, resulting in minimal expression and immunostaining by day 10 (Wise et al., 1995a).
Such correlation of the molecular expression and cellular events suggests that molecules such as CSF-1 play a role in eruption. This hypothesis is strengthened by recent studies on gene expression in the mouse DF, showing that CSF-1, c-fos, MCP-1, and NFκB are maximally expressed at day 5 post-natally (Wise et al., 1999b). In the mouse, the maximal influx of mononuclear cells into the DF, as well maximal osteoclast numbers on the alveolar bone, also occurs at day 5 post-natally (Volejnikova et al., 1997). Thus, in two different species, peak expression of putative eruption genes corresponds to peak mononuclear cells and osteoclast numbers.
(IV) Role of Osteoclasts in Tooth Eruption
(A) Recruitment of osteoclast precursors
The signaling cascades that initiate eruption must trigger the influx of mononuclear cells into the DF. Two molecules, CSF-1 and MCP-1, are prime candidates for recruiting the osteoclast precursors (mononuclear cells) into the DF. Although it is known that CSF-1 is required for the formation of osteoclasts from monocytes (mononuclear cells) (e.g., see Kodama et al., 1991; Takahashi et al., 1991; Tanaka et al., 1993; Felix et al., 1990b), it also is a chemoattractant for monocytes in vitro (Wang et al., 1988; Bober et al., 1995). Monocyte chemotactic protein-1 (MCP-1) is a well-known chemokine for monocytes (Rollins et al., 1988; Yoshimura et al., 1991) and is found in a variety of cell types, including fibroblasts (Yu and Graves, 1995).
CSF-1 and MCP-1 are secreted molecules that act as chemokines to recruit mononuclear cells to the DF. Studies have shown that the dental follicle cells do secrete CSF-1 (Grier et al., 1998) and MCP-1 (Wise et al., 1999a). The CSF-1 and MCP-1 secreted by the DF cells are chemotactic for monocytes (Que and Wise, 1997).
Because the peak influx of mononuclear cells is at day 3 post-natally in the rat, the positive correlation of this cellular influx with the peak gene expression for MCP-1 (Que and Wise, 1997) and CSF-1 (Wise et al, 1995a) at day 3 post-natally is striking. Many of the eruption molecules, in turn, have been shown to enhance the gene expression of MCP-1 (Que and Wise, 1998) and the expression of CSF-1 (Wise and Lin, 1994; Wise, 1998b). Regarding CSF-1, in addition to IL-1α and PTHrP enhancing its gene expression, CSF-1 protein has an autocrine effect on its gene expression (Wise and Lin, 1994). Thus, once CSF-1 is induced to be expressed, the autocrine effect could lead to a burst of CSF-1 being produced to recruit the mononuclear cells. This positive feedback would eventually be inhibited, because CSF-1 at high concentrations reduces the gene expression of its receptor (Wise et al., 1997).
As seen in Fig. 1, there is a redundancy of function of different genes during recruitment of osteoclast precursors to the DF; i.e., both CSF-1 and MCP-1 could act as chemokines. In addition, different molecules can enhance the expression of either CSF-1 or MCP-1. This overlapping function of gene products, as well as overlapping signaling pathways, ensures that critical developmental events such as tooth eruption will occur. Eruption requires osteoclast progenitors and osteoclasts to form an eruption pathway. Thus, by having more than one chemokine capable of recruiting osteoclast precursors, the chances are better that the tooth will still erupt even if one chemokine is impaired. This seems to be a fundamental tenet of development, as seen in knockout mice studies in which embryonic development will still progress despite the lack of what would appear to be a molecule essential for development (e.g., see Shull et al., 1992).
(B) Signaling pathways in osteoclasts
After the mononuclear cells have been recruited to the DF, there must be a favorable milieu within the DF region to promote the fusion of these cells to form osteoclasts. Transcription factors (c-fos and NFκB) and osteoclast differentiation factor (ODF) may be required for this osteoclast formation. Regarding transcription factors, null mice devoid of either the c-fos or NFκB (p50 and p52) genes have no osteoclasts but do have macrophages and apparent osteoclast precursors (Grigoriadis et al., 1994; Franzoso et al., 1997; Iotsova et al., 1997). Thus, it is possible that genes activated by these factors are needed for the fusion of the mononuclear cells to form osteoclasts. As indicated earlier, c-fos immunolocalizes to the DF, and its gene expression is enhanced both in vitro and in vivo by either CSF-1 or EGF (Wise et al., 1996, 1998). NFκB appears to be stimulated only by IL-1α (Que et al., 1999).
The proliferation and differentiation of osteoclast progenitors require CSF-1 (e.g., see Felix et al., 1990b; Kodama et al., 1991; Takahashi et al., 1991; Tanaka et al., 1993). Once the progenitor cells are formed, CSF-1 appears to be indispensable for osteoclast formation (Yasuda et al., 1998a, 1999; Jimi et al., 1999). In vitro, the survival of osteoclast-like cells (OCL) is prolonged in the presence of CSF-1 and IL-1α (Jimi et al., 1995).
Current concepts of osteoclast formation assign a major role to three molecules: receptor activator of nuclear factor-6B ligand (RANKL), also known as TRANCE, ODF, or OPGL; osteoprotegerin (OPG), also known as osteoclastogenesis inhibitory factory (OCIF); and CSF-1 (see Fig. 2). RANKL is a membrane-bound protein that is a member of the TNF ligand family, is present on bone marrow stromal cells and osteoblasts, and induces osteoclast formation and activation from precursor cells (e.g., see Yasuda et al., 1998a). It is also found on dendritic cells (Anderson et al., 1997) and is identical to TRANCE (TNF-related activation cytokine) that is present on T-lymphocytes (Wong et al., 1997). RANKL recently has been designated as the preferred terminology of the three (ASBMR President’s Committee on Nomenclature, 2000).
Inhibiting the actions of RANKL is osteoprotegerin (OPG), a secreted glycoprotein that is a member of the TNF receptor superfamily (Tsuda et al., 1997; Simonet et al., 1997; Yasuda et al., 1998b). OPG is produced by a variety of cells and blocks osteoclast differentiation from precursor cells (Simonet et al., 1997). It appears that OPG is a receptor for RANKL and that its binding to RANKL inhibits cell-to-cell signaling between stromal cells and osteoclast precursors, such that osteoclasts are not formed (e.g., see Yasuda et al., 1998b, 1999). Regulation of RANKL action appears to be essential for tooth eruption, because, in null mice devoid of the RANKL (ODF) gene, the teeth are unerupted (Kong et al., 1999).
Current results indicate that OPG but not RANKL is expressed in the dental follicle of rat mandibular molars (Wise et al., 2000b). However, the gene expression of OPG is reduced in the first mandibular molar of the rat at day 3 post-natally, and, in vitro, its expression is reduced when the DF cells are incubated with either CSF-1 or PTHrP. Thus, it has been postulated that CSF-1 and PTHrP, seen in the follicle and stellate reticulum, respectively, may be responsible for the reduced gene expression of OPG (Wise et al., 2000b). Regardless, the inhibition of gene expression of OPG at that time would allow for osteoclast formation, resulting in the maximal number of osteoclasts seen. RANKL is present in the alveolar bone (Wise et al., 2000b) and thus, the cell-cell signaling could occur to promote osteoclast formation. Studies by Nakchbandi et al. (2000) suggest that RANKL might be expressed in the DF cells following incubation with PTHrP.
Fig. 2 depicts signaling cascades that could lead to osteoclast formation. As with the cascades leading to recruitment of osteoclast precursors into the DF, there is a redundancy of function. As examples, both EGF and CSF-1 enhance c-fos expression, and IL-1α enhances both NFκB and CSF-1 expression.
(V) The Role of Osteoblasts in Tooth Eruption
Similar to the bone resorption that results from compression of the periodontium during orthodontic tooth movement, alveolar growth and root elongation alter the microenvironment within the superior aspects of the crypt. Consequently, the activation of transcription factors, cytokines, and growth factors leads to the recruitment and activation of osteoclasts by the dental follicle. The latter can be viewed as a specialized periosteum that serves osteogenic functions during the formation of the periodontium—in particular, the deposition of bone lining the tooth socket as well as cementum and the ligament fibers. From the discussion presented above about the molecules involved in osteoclastogenesis, it is clear that the presence of osteoblasts is required for the activation of osteoclasts via the RANKL/OPG pathway.
Recently, scientists began an intense search for transcription factors that act as “master” regulators of osteoblast differentiation. In this quest, a cDNA encoding a transcription factor, Osf2 (
In the mouse, Cbfa1 expression has been reported in pre-osteogenic mesenchyme and sites of active osteogenesis as well as in developing tooth organs (Ducy et al., 1997; Komori et al., 1997; Otto et al., 1997; D’Souza et al., 1999). Mice lacking Cbfa1 [Cbfa1 (-/-) mice] show a complete absence of osteoblasts and bone and succumb at birth due to respiratory distress caused by a defective rib cage. Phenotypic studies of Cbfa1 (+/-) pups that survive birth revealed the presence of a skeletal phenotype involving open fontanelles, delayed development of membranous bones, multiple Wormian bones, defective clavicles, and other defects in the axial skeleton. This skeletal phenotype demonstrates that inactivation of one allele in mice is sufficient to produce defects in bone that closely resemble those found in the human autosomal-dominant condition of
Linkage studies and mutational analyses have recently identified loss-of-function mutations in one allele of CBFA1 as responsible for the defects seen in human CCD (Lee et al., 1997; Mundlos et al., 1997). Other individuals affected with CCD are heterozygous for the deletion, missense mutation, and substitution mutation in the DNA binding region of Cbfa1. Thus, the lack of expression of one allele of Cbfa1 is the basis for the cleidocranial dysplasia syndrome (CCD) in mice and humans. Taken together, results from in vivo and in vitro studies indicate that Cbfa1 is a master gene of osteoblast differentiation whose function is not redundant with that of other genes in vivo.
The defects seen in the dentitions of patients affected with CCD have been thought to arise from a disruption in the bone remodeling process. In addition to an increased density in the maxilla and mandible, multiple supernumerary teeth are present that show a marked delay or arrest in eruption (Rushton, 1937; Jensen and Kreiborg, 1990, 1993). Other dental defects include: irregular and compressed permanent tooth crowns, hypoplastic and hypomineralization defects in enamel and dentin, an excess of epithelial root remnants, the absence of cellular cementum, and abnormally shaped roots (Rushton, 1937, 1956; Smith, 1968; Hitchin, 1975; Jensen and Kreiborg, 1990, 1993; Lukinmaa et al., 1995). Intriguingly, initial studies of dentition in Cbfa1 (+/-) mice indicate a marked delay and/or failure in eruption (Fig. 3). This phenotypic change is not evident in every adult Cbfa1 (+/-) mouse studied and suggests an effect of incomplete penetrance. Cbfa1 is expressed by osteoblasts as well as in the dental follicle and its derivatives, but not in osteoclasts (D’Souza et al., 1999; Bronckers et al., 2001). Therefore, the delay or failure of tooth eruption in the Cbfa1(+/-) young adult mouse offers an exciting means for the study of the role of these cells in dental-follicle-mediated signaling of osteoclasts during tooth eruption. While defects in bone deposition by osteoblasts may contribute to the eruption anomaly, it is also plausible that Cbfa1 haploinsufficiency compromises osteoblast-osteoclast signaling during tooth eruption. Clearly, more research in this area is warranted to shed further light on the molecular basis for the eruption defect seen in Cbfa1 (+/-) mice.
(VI) Human Conditions Affecting Tooth Eruption
Disturbances in tooth eruption are most commonly attributed to mechanical interferences caused by supernumerary teeth, crowding, and soft-tissue impaction as well as by odontogenic tumors and cysts. Ankylosis typically occurs after partial eruption of the tooth into the oral cavity and is defined as fusion of cementum or dentin to alveolar bone due to cellular changes in the periodontal ligament caused by trauma and other pathologies. When the tooth becomes ankylosed, it appears to submerge in relation to adjacent teeth that continue to erupt. Eruption failure and delayed eruption are conditions that do not naturally involve ankylosis and are associated with craniofacial dysostosis, hypothyroidism, hypopituitarism, and several genetic and medical syndromes (Sauk, 1988; Gorlin et al., 1990; Jones, 1997).
To date, of the 25 known human syndromic conditions that involve disruptions in the eruption process, approximately half have led to the identification of a causative genetic mutation (Table 2). Clearly, there is a considerable amount of work remaining before the genetic etiology of the remaining eruption defects can be completely determined. In most of these human conditions, however, the mode of inheritance has already been determined (divided nearly equally between autosomal-recessive and -dominant or between X-linked recessive and dominant). But, while most eruption defects are part of a genetic syndrome, they can also be non-familial (caused by sporadic mutation), as is thought to be the case in
The eruption phenotype can be further characterized by whether the process is merely delayed or fails completely. It appears that “delayed eruption” occurs more frequently than complete “failure of eruption” in the majority of conditions reported thus far. We can now appreciate that the process of tooth eruption may be perturbed at many different time points and may also vary in severity. Both allelic heterogeniety and phenotypic variability are seen in syndromes such as Hunter’s, Aarskog, and Apert’s, where delays in eruption have been associated with different mutations in the same gene. Hence, further characterization of distinct human tooth eruption phenotypes with the corresponding causative mutations should provide several clues about how single genes interact in a common pathway.
Classified as the most intriguing among conditions affecting tooth eruption is
The complexity of PFE is illustrated in Fig. 4, which describes a case that was diagnosed and treated in the Orthodontic Clinics (University of Texas Health Science Center, Houston). Despite comprehensive orthodontic treatment and third molar extractions, orthodontic force was not sufficient to bring the teeth into the desired plane of occlusion. The genetic and molecular basis of PFE is not yet known. However, unique features of PFE offer exciting possibilities for unraveling the molecular basis of eruption failure. Since PFE exclusively affects posterior teeth without the involvement of any systemic disorder, we can deduce that candidate genes for PFE would be molecules that function solely in the pre-eruptive phase and that are expressed in cells of the dental follicle and surrounding structures. Hence, it is likely that genes like CSF-1, NFκB, and c-fos are prime and equally likely candidate genes responsible for the eruption defect in human PFE.
(VII) Future Challenges and Directions
It is clear from the discussion above that tooth eruption is a complex process that involves the timely action and interaction of cells of the dental (enamel) organ, follicle, and alveolus (osteoclasts and osteoblasts). Despite the wealth of knowledge established about the basic biology of the eruptive process, the precise relationship of molecules involved in signaling cascades remains to be determined. As has been demonstrated for the developing skeleton and tooth organ, it is highly likely that a high level of redundancy is shared by growth factors and cytokines that influence tooth eruption. In contrast, transcription factors that are produced in small amounts within the nucleus are likely to play critical, non-redundant roles in cell-signaling functions during tooth eruption. Elucidating the physiologic role(s) and relationship of known key transcription factors, growth factors, and cytokines as well as the discovery of novel molecules will require the use of multifaceted approaches that involve both mouse and human genetic studies.
When the eruption process is disturbed, we are presented with a clinical situation that is challenging to diagnose and treat. The prospect of better control over the eruptive process and improving the overall health of dentition is an obvious reason to advance in this area of oral biology. Limited therapeutic interventions available today offer very little promise of completely correcting the inevitable orthodontic and prosthodontic complications that result from a delay or failure of eruption. As research advances are made, it is now foreseeable that future treatment modalities could include therapeutics that re-create eruption events. Critical to the success of such treatment would be the diagnostic tools that can positively distinguish between the various types of tooth eruption defects and ultimately target the appropriate treatment regimen. Clearly, the development of stringent inclusion and exclusion criteria for delineation of the variety of conditions that lead to delayed or failed eruption is needed.
The availability of rodent models as well as recent progress in the Human Genome Project offer an optimistic outlook for the identification of the genes and underlying mutations associated with PFE and syndromes involving eruption failure. Clearly, it will prove advantageous if clinicians can avail themselves of new and enabling technologies, such as laser capture microdissection, DNA chips, proteomic arrays, and functional assays with reliable cell lines. Such knowledge will not only provide us with a better understanding of the tooth eruption process itself, but will also lead to the development of effective therapeutic interventions for these otherwise clinically challenging eruption defects. When “molecular orthodontic” procedures are available for delivering the appropriate molecules or factors necessary to “erupt” individual teeth, the clinician can select the appropriate treatment for the patient based on the underlying cause of the eruption problem.
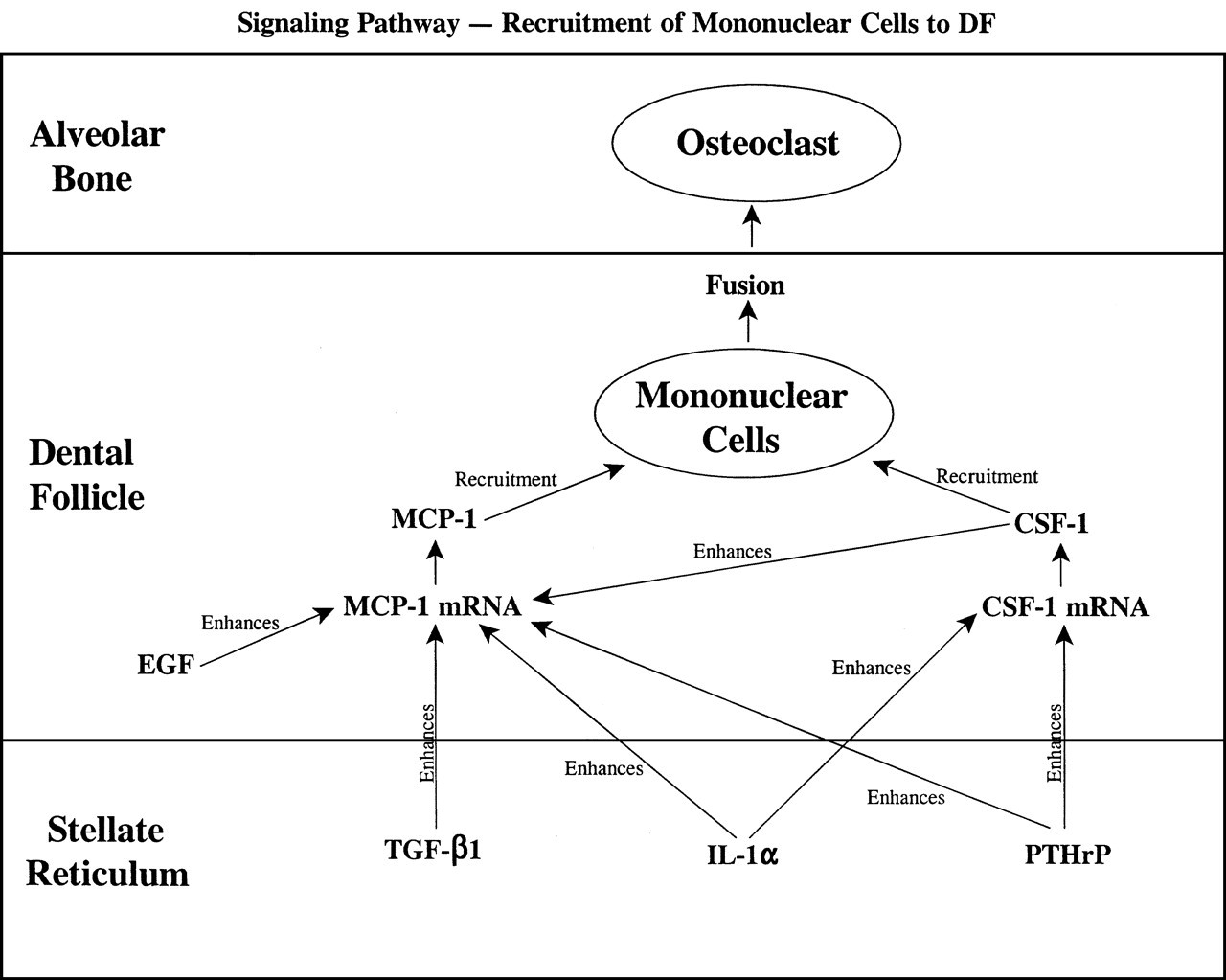
Paracrine signaling between the SR and DF, as well as within the DF only, ultimately results in the synthesis and secretion of the chemotactic molecules MCP-1 and CSF-1 for the recruitment of mononuclear cells into the DF. Note that a given molecule can often enhance the expression of more than one gene, and that two chemokines (MCP-1 and CSF-1) with redundant functions are produced. “Enhances” refers to up-regulation of a given gene, and “recruitment” is the chemotactic effect of CSF-1 and MCP-1. Modified from Wise (1998a).
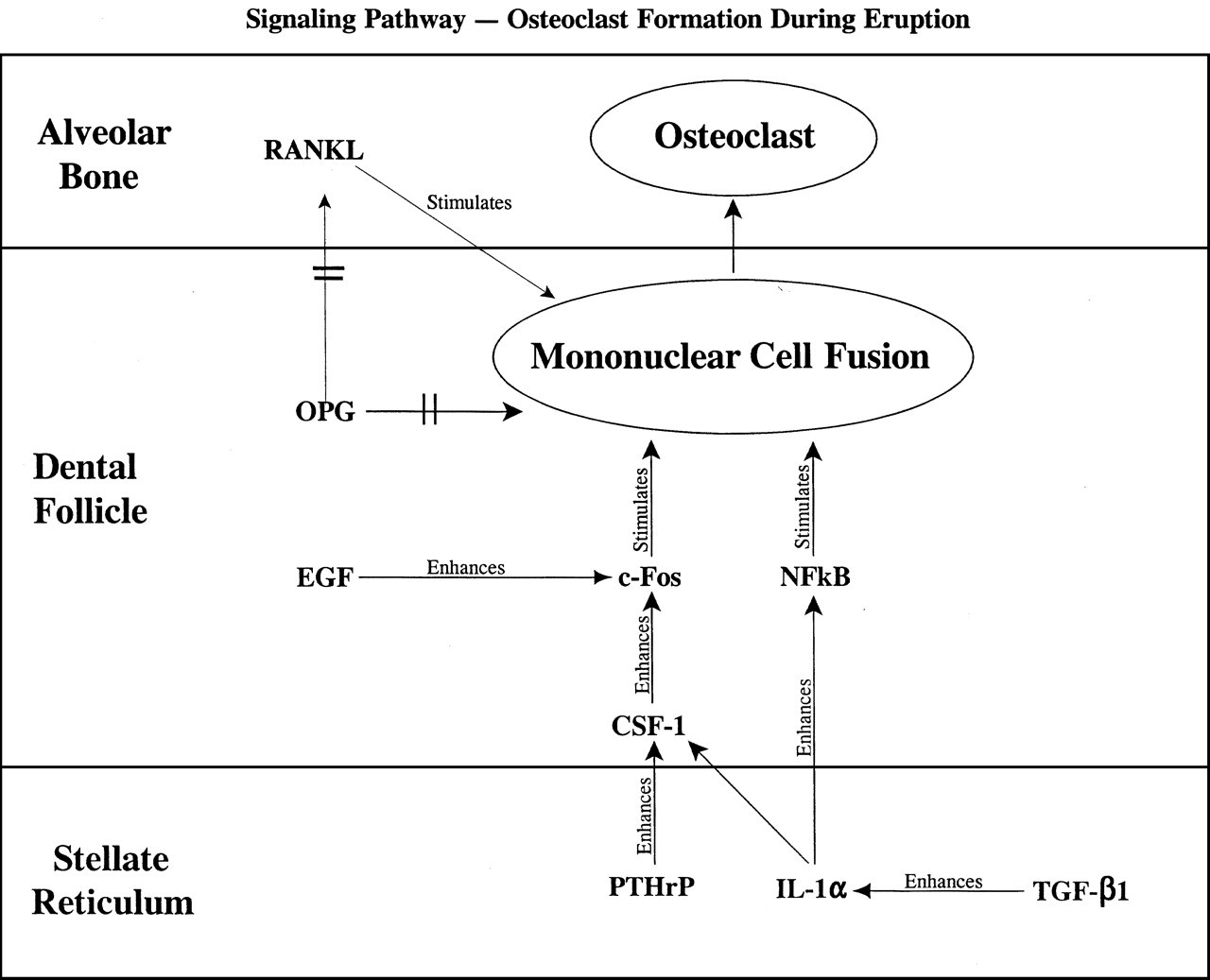
Possible signaling cascades that may promote the fusion of the mononuclear cells recruited to the DF. Both CSF-1 and ODF are known to promote osteoclast formation, whereas OPG inhibits this by preventing cell-to-cell signaling. The downstream products of the transcription factors c-fos and NFκB that promote osteoclast formation are not yet known. Arrows with crosses ( → ) reflect inhibition of gene expression, whereas other arrows reflect enhancement of gene expression or stimulation of cell fusion.
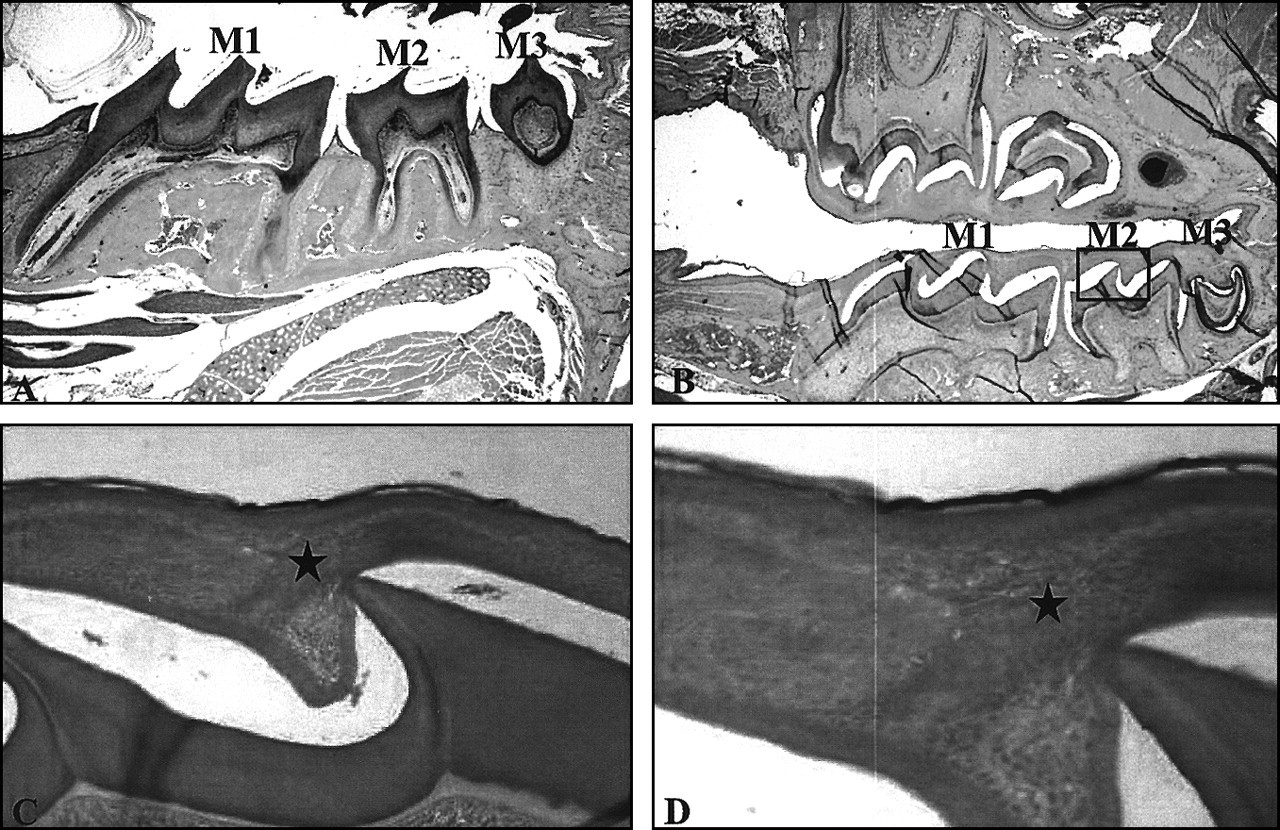
Hematoxylin-and-eosin-stained sagittal sections through demineralized mouse heads at Day 33 of post-natal life.
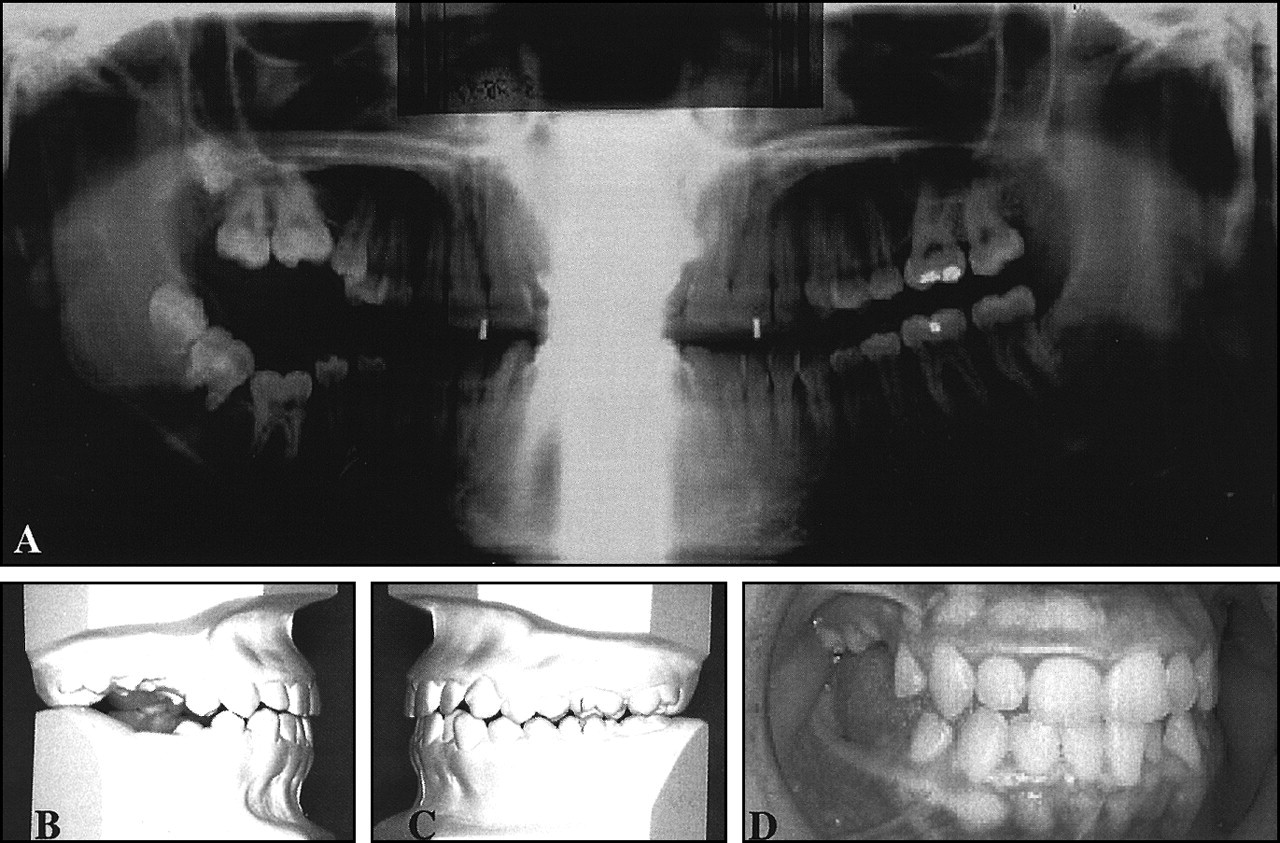
Dental records of a 15-y/o white female who first presented with missing maxillary and mandibular left third molars, small upper right and left second premolars, partially erupted right maxillary and mandibular second molars, and impacted right maxillary and mandibular first and third molars. The patient was in treatment for approximately 18 mos and has been in retention approximately 24 mos, now at age 18+. Note that the maxillary and mandibular teeth on the right side of the patient were affected from the second premolar distally
Footnotes
Acknowledgements
The authors are grateful for support from the National Institute of Dental and Craniofacial Research (Grants DE 08911 to GW; DE 13368 and DE 11663 to RDS). The advice and input received from Dr. Marion Messersmith (University of Texas Health Science Center, Houston) and Dr. Robert Gorlin (University of Minnesota Dental School), as well as the assistance provided by Ms. Cindy Daigle and Ms. Adriana Cavender in the preparation of the manuscript, are appreciated.
