Abstract
The zebrafish, Danio rerio, is a small, freshwater teleost that only began to be used as a vertebrate genetic model by the late George Streisinger in the early 1980s. The strengths of the zebrafish complement genetic studies in mice and embryological studies in avians. Its advantages include high fecundity, externally fertilized eggs and transparent embryos that can be easily manipulated, inexpensive maintenance, and the fact that large-scale mutagenesis screens can be performed. Here we review studies that have used the zebrafish as a model for craniofacial development. Lineage studies in zebrafish have defined the origins of the cranial skeleton at the single-cell level and followed the morphogenetic behaviors of these cells in skeletal condensations. Furthermore, genes identified by random mutational screening have now revealed genetic pathways controlling patterning of the jaw and other pharyngeal arches, as well as the midline of the skull, that are conserved between fish and humans. We discuss the potential impact of specialized mutagenesis screens and the future applications of this versatile, vertebrate developmental model system in the molecular dissection of craniofacial development.
Introduction
The zebrafish has rapidly emerged as a model system for genetic studies of vertebrate development. Early pioneering studies in zebrafish demonstrated the wide range of experimental manipulations afforded by the transparent embryos of these small vertebrates, including cell labeling, lineage tracing, and cell transplantation (Ho and Kimmel, 1993; Kimmel et al., 1995). Their small size and fecundity also make zebrafish particularly well-suited for large-scale mutagenesis screens for the identification of genes essential for specific developmental processes (Culp et al., 1991; Grunwald and Streisinger, 1992a,b; Mullins et al., 1994; Solnica-Krezel and Driever, 1994; Driever et al., 1996; Gaiano et al., 1996a,b; Haffter et al., 1996; Amsterdam et al., 1999). One area of promise in this regard is the genetic analysis of craniofacial development (reviewed in Schilling, 1997; Kimmel et al., 2001a,b; Fig. 1). Zebrafish form essentially all of the same skeletal and muscle tissue types as their higher vertebrate counterparts, but in much more simple spatial patterns composed of smaller numbers of cells. Furthermore, their development has been studied with single-cell resolution. Examples of this are the lineage-tracing techniques used to characterize neural crest cell contributions to pharyngeal arch cartilages and mesodermal contributions to muscles (Schilling et al., 1994, 1996a). Mutagenesis screens have identified zebrafish mutants exhibiting both expected and surprisingly new types of craniofacial anomalies (Neuhauss et al., 1996; Piotrowski et al., 1996; Schilling et al., 1996b), and these promise to reveal many new essential functions for genes in the formation of the cranial skeleton and musculature.
Here we review recent work on zebrafish craniofacial development and its implications for other vertebrates, including humans. These studies address fundamental issues of skeletal patterning, such as the lineages of skeletal precursors, their patterns of gene expression, the re-arrangements and divisions of cells in skeletal condensations, and the coordinated development of skeletal, vascular, and nervous systems. With this as a background, we can begin to interpret the phenotypes of a variety of identified zebrafish craniofacial mutants, which we discuss in the context of a growing understanding of vertebrate craniofacial development, particularly with respect to the cell-cell interactions involved in skeletal organization and growth. The cumulative outcome of these studies suggests that many of the molecular mechanisms controlling early stages of development are highly conserved. Future studies in zebrafish, including specialized screens and the ongoing effort to sequence the genome, promise to provide many new insights into the molecular mechanisms regulating craniofacial development in vertebrates.
Fate Maps and Lineages of Skeletal Precursors in Zebrafish
How are cells specified to form different components of the craniofacial complex? Although zebrafish at first appear quite distinct from mammals, many of the principles and features that govern craniofacial development in higher vertebrates are conserved. Zebrafish contain craniofacial elements similar to those of higher vertebrates (Fig. 1).
Zebrafish have provided the opportunity for the examination of cell lineages during craniofacial development and the production of high-resolution fate maps, which suggest that many aspects of its development are very similar to those of mammals. Analysis of cells simply injected with fluorescently labeled dextrans and followed in living embryos has shown that craniofacial mesenchyme is derived from both migratory neural crest and paraxial mesoderm (Schilling and Kimmel, 1994), as have chick-quail chimeric studies in avians (Koentges and Lumsden, 1996). Migrating neural crest cells adopt cartilage, pigment, neuronal, and glial cell fates, while the surrounding mesoderm forms muscle and endothelia. The neurocranium derives from both cranial neural crest and mesoderm, while the pharyngeal skeleton, including the jaw and branchial arches, derives solely from cranial neural crest cells (Fig. 2A). This dual mesenchymal origin has made it complicated for us to understand the exact nature of tissue interactions and gene functions that create craniofacial structures.
The fact that cell lineages and morphogenesis can be followed in living zebrafish has helped to resolve these issues, in terms of the positions and movements of individual cells and their final craniofacial fates. Migratory cranial neural crest cells arise from dorsal and lateral regions of the neural ectoderm, and are identifiable in 12-hour post-fertilization (hpf) zebrafish embryos. At this time, after gastrulation but before neural crest cells have begun to migrate, restrictions in mixing between future segments are already evident, suggesting that anterior-posterior cell movements are already limited at this stage, as is also observed in the chick (Lumsden et al., 1991, 1994). The most anteriorly positioned cranial neural crest (CNC) cells begin to emigrate first, followed by the more caudally located cells. Based on their segmental origin along the AP axis, neural crest cells migrate in three streams to populate each of seven pharyngeal arches (the mandibular, hyoid, and 5 branchials), which eventually become separated by endodermal epithelial pouches.
The ultimate destination of the migrating CNC cells corresponds to their rostro-caudal and medial-lateral location prior to migration: The rostro-caudal position corresponds to segmental fate, while the medial-lateral position corresponds to the cell-type-specific fate—either cartilage, pigment, glial, or neuronal (Schilling and Kimmel, 1994). Lineage tracing of superficial neural crest cells has demonstrated that the most laterally located neural crest cells assume neuronal or Schwann cell identities, while more medial cells assume cartilage, pigment, and connective tissue cell identities (Schilling and Kimmel, 1994). Recent evidence suggests that determination of these different cell-type-specific fates of pre-migratory neural crest cells may be controlled, at least in part, by secreted proteins of the Wnt family expressed at the dorsal embryonic midline (Dorsky et al., 1998; Fig. 3B). While such positional differences in the fates of pre-migratory neural crest cells have not been described in other vertebrates, they may well reflect a conserved underlying organization that is more easily observed in the relatively small numbers of rapidly developing cells of transparent zebrafish embryos. In support of this idea, mammalian and chick neural crest cells exhibit some fate restrictions in vitro (for review, see Baroffio et al., 1990; Henion and Weston, 1997). Furthermore, loss-of-function approaches to disrupt certain Wnts in mice and Xenopus (Wnt1/Wnt3a) (Augustine et al., 1993; Saint-Jeannet et al., 1997) lead to neural crest defects.
Segment-specification in the Cranial Skeleton
How are the segmental fates of components of the serially reiterated pharyngeal arch skeleton specified? These analyses are particularly straightforward in zebrafish, given the relative simplicity of their arch patterning. The segmental origins of CNC cells align with the segments of the hindbrain, the rhombomeres (Fig. 2B). Segment-restricted neural crest cell (NCC) emigration thereby coordinates the segmentation of pharyngeal arch cartilages and the cranial nerves that innervate them (Raible and Kruse, 2000). The most anteriorly positioned CNC cells contribute to the first and second (mandibular and hyoid) arch structures, and align with rhombomeres 2/3 (r2/3) and 4/5 (r4/5), respectively. NCC emigrating from the r6 level and more posterior levels contribute to the branchial arches (Schilling and Kimmel, 1994). Surprisingly, lineage-tracing studies of mesoderm adjacent to the neural crest revealed a similar rhombomeric alignment, suggesting that the metameric organization of the head can be extended to include the paraxial mesoderm, which morphologically exhibits no overt segmentation (Fig. 2B; Schilling and Kimmel, 1994). Perhaps, as is thought to occur in other body regions, the mesoderm plays a role in organizing and maintaining craniofacial segmentation, as well as coordinating the formation of the craniofacial skeleton, musculature, and vasculature.
Studies in the chick have suggested that segmental organization in the arches is imposed by the migrating neural crest, since heterotopically transplanted crest can re-organize an arch’s skeleton and musculature in some cases (Nodén, 1983). Support for this idea comes from genetic analysis of the chinless (chn) mutation in zebrafish, in which muscle defects accompany mutant cartilage defects and can be rescued by the restoration of wild-type cartilage in a chn mutant by transplantation (Schilling et al., 1996a). New evidence suggests that this is not the entire story, however, since surgical removal of the entire hindbrain in chick embryos prior to neural crest cell emigration does not alter arch specification, as assessed by the expression of arch epithelial markers (Veitch et al., 1999). Furthermore, recent transplantation studies in mouse and zebrafish embryos have demonstrated that neural crest cells are plastic with respect to their segment-specific patterns of Hox gene expression, and again, these implicate the cranial mesoderm as a source of environmental signals that may mediate such plasticity (Trainor and Krumlauf, 2000; Schilling et al., 2001). In the zebrafish, such plasticity is seen in the lineal descendents of single transplanted neural crest cells, both in Hox gene expression and in their final skeletal fates, but not in transplants of larger cohorts of cells. These results suggest that segmental identities among crest cells are maintained through community effects.
In zebrafish, each arch forms a unique set of neural-crest-derived cartilages and bones that resemble those of other vertebrates (Langille and Hall, 1989; Hall and Miyake, 1995; Schilling and Kimmel, 1997). First-arch derivatives include a ventral Meckel’s cartilage and dorsal palatoquadrate (pq); second-arch structures include ventral ceratohyal (ch), basihyal (bh), and interhyal cartilages, and the dorsal hyosymplectic (hs) cartilages; and third- through seventh-arch structures include, from dorsal to ventral, pharyngobranchial, epibranchial, bilateral ceratobranchial cartilages, and corresponding midline hypobranchial and basibranchial cartilages (Fig. 2a). Cartilage development is particularly rapid, and by five days most of the cartilages of the pharyngeal skeleton are well-formed, and larvae begin to feed (Schilling and Kimmel, 1994).
There are six different types of cartilage present in the zebrafish cranium, cell-rich hyaline cartilage being the most abundant (Benjamin, 1990), and as in other vertebrates, two types of bone, dermal and cartilage-replacement (Cubbage and Mabee, 1996; Schilling, in press). In teleosts, the bone is sometimes referred to as “acellular”, since the osteocytes tend to lie peripherally. Dermal bone (bones that develop directly within a connective tissue) and cartilage-replacement bone (bones that develop indirectly through a cartilage intermediate) first develop during larval stages. The majority of bones of the zebrafish skull are cartilage bones (43/74) which undergo either endochondral (ossify within cartilage) or perichondral (ossify around cartilage) bone formation. Because bones do not appear until 5 to 6 days of development, beyond the stages examined in lineage studies or in mutational screens (most of the existing mutants are lethal by 5 days), little is known about their early development, and we do not discuss it here.
Using Zebrafish to Study Skeletal Organogenesis: Development of Pharyngeal Teeth
One area in which the fine cellular resolution possible in zebrafish shows great promise is in the study of how cells organize and divide to define the complex sizes and shapes of skeletal organs, including teeth. As in other cyprinids, zebrafish lack teeth on the oral jaws, but have well-developed pharyngeal teeth that form in a bilaterally symmetric pattern posterior and medial to the fifth ceratobranchial (cb5) arches, or pharyngeal jaws. A five-day-old zebrafish larva has two or three pharyngeal teeth that form on each cb5 arch cartilage, juvenile zebrafish carry up to 20 teeth on each arch, and 11 teeth are present on each arch in adults (Van der Heyden et al., 2000, 2001; Van der Heyden and Huysseune, 2000). Zebrafish pharyngeal teeth exhibit all of the characteristics of true teeth in other vertebrates. Descriptions of early stages of zebrafish pharyngeal tooth development were included in reports on zebrafish skeletal morphogenesis (Cubbage and Mabee, 1996; Schilling and Kimmel, 1994, 1997), and in the analysis of zebrafish pharyngeal arch mutants (Neuhauss et al., 1996; Piotrowski et al., 1996; Schilling et al., 1996b). More recently, detailed morphological and histological studies have described the pattern of tooth formation from the first appearance of tooth buds until 14 dpf (Van der Heyden and Huysseune, 2000), and the odontogenic waves regulating adult replacement tooth formation (Van der Heyden et al., 2001). Zebrafish teeth arise in an ordered and synchronized fashion, and development can be divided into three stages: (1) initiation and morphogenesis; (2) cytodifferentiaton, including enameloid and dentin matrix deposition; and (3) attachment and functional stages, followed by tooth resorption. Zebrafish tooth germs arise directly from the pharyngeal epithelium and independently of one another, and pharyngeal tooth formation closely resembles that of higher vertebrates, beginning with the formation of an epithelial organ that subsequently acquires a bell shape (for review, see Thesleff, 1995; Huysseune et al., 1998; Peters and Balling, 1999). Tooth buds first appear as cartilaginous anlage at hatching (approximately 48 hpf), as a pair of germs on each side. Even at this early stage, the tip of the more medial and probably more mature of the two tooth germs already contains enamel and dentin, demonstrating the extremely rapid rate of development. One day later, this tooth has fully differentiated and is connected by attachment bone to the perichondral bone of the 5th ceratobranchial. Subsequent teeth arise more medially and dorsally to the initial ventral ones, and eventually form three transverse rows from anterior to posterior. Tooth development is reproducible and bilaterally symmetric, with some variation. Pharyngeal teeth contain a pulp cavity, dentin, and enameloid components, including an enamel organ, at the time of attachment. Thus, they exhibit many of the same developmental stages and structures as their mammalian counterparts and, like their associated cartilages, arise in a rapid and simple, segment-specific pattern. Some characteristics and organization of adult zebrafish pharyngeal teeth are shown in Fig. 3.
Based on these similarities, we expect that similar epithelial/mesenchymal cell interactions and gene expression patterns govern both zebrafish and mammalian tooth development. Over the past two years, enormous progress has been made toward the identification of genes involved in mammalian tooth development and specification. Position-dependent control of tooth germ initiation and morphogenesis involves restricted epithelial signals that elicit the differential expression of transcription factors, particularly homeobox genes, in the adjacent dental mesenchyme (see Maas and Bei, 1997; Thesleff and Sharpe, 1997; Peters and Balling, 1999; Tucker and Sharpe, 1999; and references therein). The original hypothesis that regionalization of the pharyngeal mesenchyme specifies different tooth types has evolved into what is now termed the odontogenic homeobox code of tooth specification (Sharpe, 1995), reflecting the expansion of the well-documented Hox gene patterning of the axial skeleton to include patterning of the teeth.
Major players in mammalian tooth specification, as revealed in gene expression patterns in tooth primordia, include four growth factor families—Bone Morphogenetic Proteins (BMPs), Fibroblast Growth Factors (FGFs), Wnts, and Hedge Hog growth factors (Hhs)—and the transcription factors they induce (Jernvall and Thesleff, 2000). It is the reiterative expression of many of these genes during various stages of tooth development that results in the formation of teeth of specific size and shape (Thesleff and Jernvall, 1997; Jernvall and Thesleff, 2000).
In mammals, tooth development begins with the expression of Pitx2 and Shh in the thickened epithelial stripe marking the dental lamina (Thomas et al., 1998; Zhang et al., 1999; St Amand et al., 2000). Subsequently, epithelial expression of FGF and BMP family members and the induction of Pax9 and Msx1 in the adjacent dental mesenchyme, and a shift of the inductive potential from the epithelium to the mesenchyme, are required for tooth morphogenesis to proceed (Peters et al., 1998a,b). Recent studies indicate that the four types of mammalian teeth—incisor, canine, premolar, and molar—are specified by early epithelial signals and not by neural-crest-derived signals, as was previously thought (Tucker et al., 1998). This was dramatically demonstrated by the transformation of an incisor to a molar through the inhibition of epithelial bmp4 signaling with the Bmp antagonist, noggin (Tucker et al., 1998). In addition, analysis of loss-of-function mutations in homeobox genes Dlx1 and 2 and compensatory activities of Dlx 5 and 6 reveals requirements for these homeobox genes in tooth morphogenesis (Thomas et al., 1997; Weiss et al., 1998a,b).
A great many zebrafish orthologues of genes involved in mammalian tooth development have now been identified. Examples of these include members of all four growth factor families, including BMPs (refs), FGFs (Hauptmann and Gerster, 2000a,b), Wnts (refs), and Hhs (refs), as well as homeobox genes such as Hoxb-2 and –5 (Thomas and Sharpe, 1998; Yan et al., 1998; Suzuki et al., 2000), and dlx1 and 2 (Zerucha et al., 1997), among others (Table; Fig. 5). Representatives of all of these have been shown to be expressed in localized regions of the early craniofacial mesenchyme in zebrafish, like their counterparts in mammals, though most have not been examined at later stages in the 5th branchial arch during the formation of the tooth primordia (Fig. 4).
To date, the expression of only two genes, the homeodomain transcription factor engrailed and the novel type I TGFβ family member receptor alk8 (Hatta et al., 1991; Yelick et al., 1998), has been described in developing zebrafish teeth. Alk8 has also been demonstrated to be required for normal tooth development, by microinjection studies of dominant-negative alk8 mRNAs, and through analysis of the alk8 mutant lost-a-fin (Bauer et al., 2001; Mintzer et al., 2001; Payne et al., 2001a). The majority of gene expression studies in zebrafish have focused on early developmental events up to 24 hpf, at least a day before tooth formation begins. Further examination of the expression patterns of the abovementioned genes in later-staged embryos, and in identified zebrafish mutants when possible, provides an exciting prospect for the future and is likely to reveal roles in tooth and arch specification and differentiation.
It is possible, though, to examine tooth development in zebrafish harboring mutations in genes with demonstrated mutations in mammalian and/or zebrafish tooth development, where tooth phenotypes are expected. In fact, tooth phenotypes are readily observed in snh/bmp7 (Dick et al., 2000; Schmid et al., 2000) and laf/alk8 (Bauer et al., 2001; Mintzer et al., 2001) mutants (Fig. 5).
Zebrafish Skeletal Mutants
The zebrafish complements genetic studies in mammals by providing a tool for “forward genetics” to be performed, with random screening for mutations in processes of interest such as craniofacial development. This ability, combined with the ease of manipulation of zebrafish embryos and increasing tools for genome analysis (linkage and physical mapping, DNA libraries, and expressed sequence tags), is quickly bringing the fish up to speed as a genetic system. Zebrafish embryos are particularly well-suited for mutagenesis screens based simply on visual inspection via a dissecting microscope, allowing for powerful in vivo analysis of gene function. Such screens have resulted in the identification of mutants encompassing a wide range of developmental processes, although only a small fraction of the genes underlying these mutations has been identified to date (Knapik, 2000).
Most of the zebrafish jaw and branchial arch mutants were identified in two large-scale chemical mutagenesis screens in the early 1990s: one at the Max-Planck Institute in Tübingen, Germany, and one at the Harvard Medical School in Boston, USA (Fig. 6). Both used similar methods. The mutagen, N-ethyl N-nitrosourea (ENU), was titrated to induce single base-pair mutations and F3 embryos screened for craniofacial morphology (Mullins et al., 1994; Solnica-Krezel et al., 1994; Driever et al., 1996; Haffter et al., 1996). The Tübingen screen alone resulted in the isolation of over 100 skeletal mutants, including at least four Classes with defects in: (1) the ventral skeleton of each pharyngeal arch, particularly the mandibular and hyoid arches; (2) the posterior five gill-bearing arches, including the tooth-bearing seventh arch; (3) cartilage differentiation and growth; and (4) the midline of the braincase (Brand et al., 1996; Piotrowski et al., 1996; Schilling et al., 1996b). The Boston ENU mutagenesis screen produced 48 skeletal mutations in 34 genetic loci that exhibited many similarities to those identified in Tübingen, and which were classified into three Groups having defects in: (1) the overall pattern of the craniofacial skeleton; (2) cartilage differentiation and morphogenesis; and (3) spatial arrangement of otherwise normal skeletal elements (Neuhauss et al., 1996). A few additional unique mutations that did not fit into any of these categories were also identified. Perhaps most intriguing were whole classes of genes—for example, the anterior arch mutants described in more detail in the following section—the loss of which produced nearly identical phenotypes that differed in severity, suggesting that they may lead to the discovery of entire genetic pathways required for craniofacial development. Testing this hypothesis requires an understanding of both the cellular basis for the observed defects in these mutants and the cloning of the underlying genes, and this is currently the focus of major efforts in several laboratories.
Organization and Division Patterns in Cartilage Condensations
Like teeth, cartilages must form and maintain precise shapes and sizes during development to serve its function. The morphogenetic events that regulate cartilage organogenesis and the genes that control these events are poorly understood. Cartilages are exceptional in their relatively simple make-up, having only two major cell types: chondrocytes and the perichondrial cells that surround them. In zebrafish, these cells secrete relatively little extracellular matrix, making it easy for investigators to observe individual cells and follow their re-arrangements. An understanding of this wild-type pattern of chondrification facilitates important predictions for the types of mutant phenotypes that may occur.
Cartilage patterning appears to reflect a progressive subdivision in which individual skeletal elements arise out of common primordia (Bertmar, 1959), and early work in zebrafish, where Nomarski optics was used for the visualization of cartilage precursors in the pharyngeal arches, has supported this notion (Schilling and Kimmel, 1997). The first cranial chondrification begins at 2 days post-fertilization (dpf). From an initial condensation, which is similar in each arch, protrusions develop that pre-figure individual skeletal elements. Kimmel et al. (1998) have taken this a step further to describe two major stages in cartilage formation in the hyoid (second pharyngeal) arch: an early morphogenetic phase of re-organization within the condensation, followed by a growth phase. In the hyoid, two sites of chondrification—a dorsal hyosymplectic and ventral ceratohyal—appear within the larger condensation. Simple movements of cartilage precursors in these sites correlate with the future shapes of the cartilages, forming “stacks” that are polarized and aligned with the long axis of the element. Overlapping in time with these re-arrangements, the cells begin to divide and the cartilage grows isometrically, generally preserving its initial shape. Interestingly, growth is concentrated near articulations or joints between cartilages, and in many ways future joint regions appear to be regions where chondrogenesis patterns are organized.
The usefulness of such descriptive studies was immediately obvious in the analysis of one group of zebrafish mutants, those that affect the ventral pharyngeal cartilages. Four mutants—sucker (suc), schmerle (she), hoover (hvr), and sturgeon (stu)—show reductions in the ventral cartilages and loss of joints between these cartilages and the dorsal elements in each arch. Particularly in the most severe member of this class, suc, differences are apparent at the earliest stages of cartilage condensation in the hyoid arch, for example. In contrast to wild-type embryos, only a tiny ventral set of precursors of the ceratohyal cartilage appears to condense in the mutant, and these have a reversed polarity with respect to the dorsal hyosymplectic. Furthermore, this remnant of the ventral cartilage is fused to its dorsal neighbor, beginning even at these earliest stages of condensation, presumably due to the absence of joint cartilage. Thus, suc—and probably she, hvr, and stu as well, which show similar but less severe phenotypes—is required to orient the way in which cells stack together during cartilage condensation and to form a joint between these regions of chondrogenesis. Similarities in the phenotypes of all four members of this class of mutants suggest the exciting possibility that all may lie in a common genetic pathway.
Zebrafish Mutants Reveal Cell Interactions in the Developing Craniofacial Skeleton
Recent identification of the genetic lesion underlying the suc mutation now provides a molecular entry point into the significance of the ventral arch mutant class and the descriptive studies of zebrafish chondrogenesis. By a candidate gene approach, based on its map position on linkage group 19 and synteny between this position and a similar region of human chromosome 6, suc was shown to encode a zebrafish endothelin1 (et1) gene (hereafter referred to as suc/et1; Miller et al., 2000). As in the mouse, suc/et1 in zebrafish is expressed in pharyngeal arch epithelia and in the paraxial mesoderm-derived core of each pharyngeal arch (see Fig. 2), but not in the neural crest. suc/et1 mutants lack expression of several transcription factors (e.g., dHAND, gsc) in the ventral arch neural crest. Similarly, et1 knockout mice exhibit numerous craniofacial defects, including the lower jaw defects and defects in transcription factor expression characteristic of suc/et1 mutants, suggesting that the role of et1 is conserved throughout vertebrates. Thus, like its mammalian counterparts, zebrafish suc/et1 is thought to be an epithelial and paraxial mesoderm-derived arch core signal that regulates gene expression in adjacent arch skeletogenic cells (Clouthier et al., 2000). This idea is supported by mosaic studies in the fish demonstrating that suc/et1 mutant neural crest cells are able to express ventral ectomesenchymal markers when transplanted into wild-type arches. Based on descriptive studies of chondrogenesis in suc/et1 mutants, we can now interpret how skeletogenic cells respond to suc/et1, both in the orientations of their movements and in division patterns with respect to the developing joint region. It remains to be determined how such a signal expressed broadly in the arch environment results in a specific joint defect. It will be interesting to examine how suc/et1 affects the expression of other secreted growth and differentiation factors, including ligands of the TGFâ superfamily, that are required for joint formation in mice, and are expressed in developing joint regions (Kingsley, 1994; Storm and Kingsley, 1999). Other zebrafish mutants with phenotypes similar to that of suc/et1 may help resolve these issues, and are prime candidates for additional genes in the suc/et1 signaling pathway.
Recent analysis of another mutant, van gogh (vgo), for which the underlying gene is still unknown, supports the idea of a signaling role for the pharyngeal endoderm in chondrogenesis (Piotrowski and Nusslein-Volhard, 2000). Mutation of vgo affects many of the same structures disrupted in suc/et1 mutants but with very different consequences. In vgo mutants, Meckel’s cartilage is well-developed but occasionally points posteriorly, where it often fuses with ventral elements of the second pharyngeal arch. Ceratohyal cartilages appear to be abnormal and fuse with posterior pharyngeal arch cartilages. Unlike the ventral arch mutants, vgo mutants have severe segmentation defects in the skeleton, such that cartilages of adjacent arches often fuse. In some cases, the ventral elements of arches 2-5 coalesce into a common mass. Although segmental patterning of the hindbrain is not disrupted in vgo mutants, the endoderm of the foregut does not form pharyngeal pouches, and the surrounding mesoderm is mis-patterned. This results in the fusion of initially well-patterned migrating neural crest cells once they reach the arches. Defects in the separation of migrating groups of neural crest cells are therefore thought to result in a failure to separate cartilage condensations, presumably because there is no pharyngeal pouch epithelium keeping them apart. These possibilities can now be examined through more detailed cellular analysis of chondrification in vgo, as described above for the ventral arch mutants. In addition, a direct patterning role for the endoderm in inductive interactions with cartilage precursors can now be tested with mosaic analysis. In addition, several mutations in zebrafish genes such as casanova (a novel Sox-related protein [Dickmeis et al., 2001; Kikuchi et al., 2001], bonnie-and-clyde, a Mix homeoprotein [Kikuchi et al., 2000], and faust/gata5 (Reiter et al., 1999) that are required for early endodermal development also show defects in craniofacial chondrogenesis, and these provide further tools for the study of endodermal requirements in skeletal development.
Another intriguing class of zebrafish mutants with clear human correlates includes those that disrupt the midline, resulting in fusions of the ventral neurocranium (Brand et al., 1996; Kimmel et al., 2001a). Such phenotypes were not observed among the jaw or general chondrogenesis mutants from either the Tübingen or Boston screen. Recent cloning of one of these mutants, you too (yot), has shown that it encodes gli2, a transcription factor in the Sonic hedgehog (Shh) signaling pathway (Karlstrom et al., 1999). This implicates Shh in midfacial cartilage patterning and provides a zebrafish model for the human syndrome holoprosencephaly, in which midline fusions have been linked to human mutations in Shh (see below). Surprisingly, mutations in shh itself in zebrafish (known as sonic you, syu) do not exhibit such neurocranial fusions. However, recent evidence inhibiting translation of both syu and its close relative tiggy-winkle hedgehog (twhh), with simultaneous use of the new technique of gene “knockdown” with morpholino oligonucleotides (Nasevicius and Ekker, 2000), has demonstrated their requirements in specification of the neurocranial midline. This suggests some redundancy between hedgehog family members in zebrafish, but reinforces the notion that the mechanisms of midline craniofacial development have been conserved between mammals and zebrafish.
Correlating Zebrafish Mutants with Human Syndromes
In humans, genes underlying craniofacial anomalies can broadly be divided into three types, affecting: (1) the specification, survival, and/or migration of neural crest cells; (2) mesenchymal cell condensation and differentiation; and (3) cell proliferation or maturation, including those affecting extracellular matrix (ECM) components and ECM homeostasis (Mundlos and Olsen, 1997a,b). A major goal of craniofacial screens in zebrafish is to identify genes whose counterparts control craniofacial development in humans. The recent results described here provide the first glimpses of progress toward this goal. For example, both the ventral arch defects in suc/et1 mutants and cartilage fusions in vgo mutants, as well as the phenotypes of et1(-/-) mice, fall within the range of phenotypes observed in humans afflicted with DiGeorge and velocardiofacial syndromes (Kurihara et al., 1995; Lindsay, 2001; see below).
In the making of comparisons between zebrafish and human craniofacial phenotypes, several caveats must be considered. One is the predominance of dominant alleles underlying most human syndromes, while, to date, most of the identified zebrafish craniofacial mutants are homozygous recessive lethal alleles. Thus, corresponding zebrafish phenotypes may have very different presentations and may be much more severe than heterozygous mutations in human orthologues. Another consideration is that independent duplications of genes within teleosts or mammals may have led to distinct and/or partially redundant gene functions. In several cases, the functions of a single mammalian gene appear to be distributed among multiple zebrafish genes, probably due to an ancient genome duplication in an ancestor of the ray-finned fishes (Postlethwait et al., 1998, 1999; Taylor et al., 2001), with the result that a gene mutation in zebrafish may not be as severe as that of its human homologue. This can be an advantage for genetic studies in that single gene mutations in zebrafish may result in less severe and—more importantly—non-lethal phenotypes than would result in other vertebrates, allowing for the characterization of distinct gene functions. Interestingly, however, gene order or synteny is surprisingly similar between zebrafish and human genomes relative to that of mice and humans for example, which facilitates direct comparisons. Given these caveats, in the paragraphs that follow, we will attempt to correlate some of the many identified zebrafish mutants with possible corresponding human craniofacial anomalies.
Class 1: Genes affecting the specification, survival, and /or migration of neural crest cells
Of the hundreds of mutants identified in the large-scale zebrafish mutant screens, surprisingly few display obvious defects in early neural crest specification or migration, although these may become more apparent as the phenotypes are further analyzed. As discussed above, if neural crest cells are specified by progressive fate restrictions, we might predict the existence of genes that regulate lineally related subsets of neural crest cell fates. Interestingly, several zebrafish mutants have phenotypes that appear to be restricted to specific neural crest sublineages while not overtly affecting others, and in some cases these appear to have direct mammalian correlates. For example, the pigmentation mutant colourless (cls), which encodes a zebrafish orthologue of the HMG-box-containing transcription factor, sox10 (Dutton et al., 2001), exhibits defects in NCC-derived pigment cells, neurons and glia, while the craniofacial skeleton appears normal (Kelsh and Eisen, 2000). The combination of pigment and enteric neuronal defects in these mutants is reminiscent of mutations in 3 genes in mice—piebald (s), lethal spotting (ls), and Dominant megacolon (Dom). Molecular characterization has shown that s and ls in mice encode an endothelin receptor (a B-type; Hosoda et al., 1994) and its ligand, endothelin 3 (Baynash et al., 1994), respectively, while Dom encodes Sox10, the functional homologue of cls (Herbarth et al., 1998; Southard-Smith et al., 1998). These phenotypes all bear a striking resemblance to the human Waardenburg-Shah syndrome and Hirschsprung’s disease, which are characterized by both otic and pigmentation defects (Hassinger et al., 1980). Similarities in the pigmentation and peripheral neural defects between these human syndromes and cls/sox10 mutants in zebrafish suggest that the mechanisms of specification of neural-crest-derived melanocytes and enteric neurons are conserved between fish and humans, and validate the use of zebrafish as an appropriate model for the molecular/genetic characterization of these syndromes. The experimental advantages available in zebrafish have allowed researchers to demonstrate that pigment cell precursors, and possibly other crest cells, fail to migrate in cls/sox10 mutants and die instead, as may also occur in Waardenburg-Shah and Hirschsprung’s patients. Similarly, recent studies suggest that apoptosis is the underlying reason behind the autosomal-dominant human disorder Treacher Collins syndrome (Dixon et al., 2000), which predominantly affects first- and second-arch structures, and may underlie additional mouse and human syndromes as well. From a developmental perspective, the demonstrated requirement for cls/sox10 in all non-skeletogenic neural crest derivatives is one of the first indications that skeletogenic and non-skeletogenic populations of CNC may be under separate genetic control at early developmental stages (Kelsh and Eisen, 2000).
Class 2: Genes regulating mesenchymal cell condensation and differentiation
Zebrafish mutations affecting patterning of the arches, such as suc/et1 and vgo, as well as mutants that disrupt the midline of the neurocranium are examples of a second general class that resembles human mutations defective in chondrocyte condensation and differentiation. Phenotypes of both suc/et1 and Et1(-/-) mice, as well as vgo mutants, resemble the DiGeorge syndrome (DGS) and velocardiofacial syndrome in humans, which are associated with translocations and deletions in chromosome 22q11, and are characterized by cleft palate, conotruncal heart anomalies, and thymus and thyroid defects (Kurihara et al., 1995; Hong, 1998). Several genes within this chromosomal region, including a ubiquitin fusion degradation gene (UFD; Lindsay, 2001), are candidates both for the human disorders and for the genetic defect underlying the vgo phenotype in zebrafish, as are several other genes that give DGS-like phenotypes when mutated in mice, such as the A-type endothelin receptor, the transcription factor dHAND, and the Tbox transcription factor, Tbx1 (Jerome and Papaioannou, 2001; Lindsay et al., 2001; Merscher et al., 2001). The midline phenotypes of zebrafish yot/gli2 mutants resemble holoprosencephaly in humans associated with mutations in Shh signaling, since both are characterized by ventral brain and midfacial skeletal defects (Muenke and Cohen, 2000). Therefore, the zebrafish provide a model system both for DGS/velocardiofacial syndromes and for holoprosencephaly, in that chondrocyte condensation and differentiation can be observed directly in response to suc/et1 or Shh signaling, respectively.
A much larger group of zebrafish mutants defective in skeletal condensations, for which the underlying genes have not yet been cloned, are the “flatheads”, which disrupt the posterior branchial arches, including those that bear teeth (Schilling et al., 1996b). These are caused by mutations in at least 8 genes with at least another 30 or so mutations for which complementation analysis is currently unresolved. We might speculate that human counterparts of such gill arch mutants in zebrafish would likely correspond to defects in throat structures such as the larynx, which derive from homologous arch segments. The flathead mutants have been classified based on their segment-specific branchial arch defects: (1) all pharyngeal arches, (2) arches 3-7, and (3) arches 4-6. Surprisingly, pharyngeal teeth, which form only on arch 7, are generally present in mutants in classes 1 and 2, suggesting that teeth develop somewhat independently of their underlying cartilages (see below). In addition, the disruption of whole contiguous sets of branchial arches in many flathead mutants appears to reflect a normal process by which these posterior arches all subdivide from a common mesenchymal primordium (Schilling and Kimmel, 1994). As described above for individual cartilages of the hyoid arch, progressive condensation from a common primordium may thus be a feature of many different aspects of craniofacial development.
Classes 3 & 4: Proliferation, differentiation, and possible ECM mutants
The other large group of zebrafish craniofacial mutants for which the underlying genetic causes are still unknown are the “hammerheads”. Hammerheads result from mutations in at least 7 different genes, with over 20 additional mutants still unresolved (Piotrowski et al., 1996). Hammerhead mutants lack tissue rostral to the eyes, causing the eyes to bulge laterally. In many of these mutants, individual chondrocytes appear small and misaligned, and the overall body length is reduced up to 20%, similar to many human cartilage differentiation and ECM defects. Hammerhead mutants fall into classes based on their chondrocyte morphologies: (1) kinked cartilages and shortened bodies, (2) severely reduced numbers or absence of chondrocytes, (3) reduced Alcian staining, (4) reduced or malformed cartilages, and (5) chondrocytes with tumor-like growths.
The jellyfish (jef) mutant is a particularly notable and severe example of the second class, in that it lacks all cartilages, including cartilaginous elements of the pectoral girdle. Here we can draw a parallel with mammalian mutants in genes such as sox9 (campomelic dysplasia), in which cartilage condensations form but fail to differentiate (Bi et al., 2001). As in flathead mutants, teeth are present in jef mutants, despite the absence of cb5 arch cartilages that normally support these teeth. This observation and the fact that tooth development appears largely unaffected in mutants hammerhead (ham) and head on (hen), both of which exhibit reduced numbers of chondrocytes, reinforce the notion that differentiation of teeth can occur independent of overtly differentiated cartilage or bone of attachment.
One clear set of candidates for genes, mutations in which might cause phenotypes like those observed in ham and hen, includes fibroblast growth factor receptors fgfr1, -2, and -3, dominant mutations of which, in humans, result in defects in cell proliferation. In humans, defects are evident in the growth plates of cartilages that undergo endochondral bone formation, and generally result in reduced bone length but not necessarily in altered bone shape. In addition, defects are often observed in cranial sutures, which often exhibit premature fusion. Syndromes included in this category are: Pfeiffer syndrome (FGFR1); Crouzon, Apert, Jackson-Weiss, and Saethre Chotzen syndromes, all of which can result from mutations in FGFR2; and achondroplasia, hypochondroplasia, and thanatophoric dysplasias, many of which are due to mutations in FGFR3.
Finally, a third class of mutants, including geist (get), whitebread (wib), or jekyll (jek), was characterized as having reduced Alcian blue staining, which specifically labels proteoglycans of the ECM. Clear candidates for genes underlying these phenotypes include ECM components such as aggrecan, a cartilage-specific proteoglycan core protein, whose mutation results in the nanomelia phenotype in chick and cartilage matrix deficiencies in mice (Vertel et al., 1994), as well as hyaluronan synthase, a regulator of ECM deposition (Spicer and Nguyen, 1999). Collagens are also likely candidates, since many human defects in ECM components and matrix homeostasis are due to mutations in collagen genes. This is not surprising, since collagens are the most abundant ECM proteins, with type I collagen constituting about 90% of total protein mass in bone. Human syndromes for which these fish mutants could serve as models include a long list for which the underlying genetic basis is mutations in a variety of collagen genes (So et al., 2001, review). Although zebrafish type II collagen has been identified (Yan et al., 1995; Lele and Krone, 1997), a mutant has not yet been identified, and most zebrafish collagen genes have yet to be characterized.
Using Retroviral Insertional Mutagenesis in Zebrafish
So far, we have discussed only the large spectrum of ENU-induced mutations available in zebrafish. However, extremely productive insertional mutagenesis screens in zebrafish have also been performed with the use of pseudotyped, mouse retroviral vectors (Gaiano et al., 1996a,b; Amsterdam et al., 1999; Amsterdam and Hopkins, 1999). Insertional approaches to mutagenesis screens offer distinct advantages over chemical mutagenesis. Although less efficient (approximately nine-fold at present, one embryonic lethal mutation in nearly 100 proviral insertions), an insertion provides a tag with which flanking gene sequences can be easily identified and the mutated gene cloned. In an early pilot screen of over 200 proviral insertions, three insertional mutants were found, for which two of the genes were identified (Gaiano et al., 1996b). One of these, no arches, resembles mutants of the flathead class that are required for normal branchial arch development, and encodes a zebrafish relative of the Drosophila zinc-finger-containing ribonuclease gene, clipper. A large-scale retroviral insertional mutagenesis screen is currently under way with the goal of screening over a hundred thousand insertions and isolating approximately a thousand embryonic mutations, nearly half of the genes that some estimates suggest are required for normal development (Amsterdam et al., 1999). Out of approximately 150 mutants, two-thirds have been linked to phenotypes, and nearly one-third to candidate genes (Talbot and Hopkins, 2000). Among these, the three craniofacial mutants no knack, hi37, and hi63 all lack jaws, among other defects. An additional insertional mutant alyron (aln), generated by microinjection of plasmid DNA, has also been described as having craniofacial, pigmentation, and circulation defects (Cretekos and Grunwald, 1999). These examples highlight the speed with which such insertional mutagenesis screens promise to uncover genes required for craniofacial development, though, so far, the techniques available have proved less efficient than traditional screening methods in generating mutations.
Conclusions and Caveats
The combination of genetics and embryology afforded by the zebrafish embryo has led to many insights into the mechanisms that pattern the early craniofacial skeleton. Here we have tried to review briefly the huge range of skeletal mutants now available. Though the cloning of the underlying genetic basis for most of these mutants is still in progress, many have already given considerable insights into the mechanisms by which neural crest lineages are established (e.g., cls/sox10) and the signals and tissue interactions that control chondrocyte condensation and differentiation (e.g., suc/et1, vgo, etc.). In addition, the detailed lineage studies that are uniquely possible in this small, transparent vertebrate embryo have led to new hypotheses and provide great advantages for the future analysis of craniofacial mutant phenotypes. Such detailed embryological data, together with further genetic studies in which mutants are combined and ordered into pathways, promise to lead to a much more comprehensive understanding of the developmental basis for many human skeletal syndromes. We have tried to speculate on possible future avenues where zebrafish may prove to be important models in this regard, such as in velocardiofacial syndrome and Hirschsprung’s disease, for which the underlying defects in neural crest development have been difficult to pinpoint in other model systems such as mice.
However, there is clearly a long way to go. The zebrafish screens have not approached saturation, making the absence of certain expected phenotypes, such as the homeotic phenotypes observed in Hoxa2 mutant mice, uninformative. Recent efforts to use morpholino oligonucleotides to knockdown Hox gene functions are beginning to clarify these discrepancies and reveal unforeseen redundant functions among closely related Hox genes (McClintock et al., 2001). Allele number is a good indicator of extent of saturation; for example, among the pharyngeal cartilage mutants identified in the Boston screen, only one set of multiple alleles was isolated among 34 mutated loci. The use of more specialized and perhaps more arduous screens—through antibody staining, in situ hybridization, or Alcian blue staining of developing cartilages, for example—may result in the identification of mutants that would otherwise be overlooked in screens based solely on morphological criteria.
An additional complication is that early lethal phenotypes thwart the analysis of later gene function. Defects in relatively late events such as chondrification or ossification will not be apparent if early lethality prevents the embryos from developing to the appropriate stage. This problem can be overcome only by more sophisticated methods of conditional gene inactivation, techniques which are being explored in zebrafish, such as temperature-sensitive mutant alleles (Rawls and Johnson, 2001), or caged forms of morpholino oligonucleotides (Ando et al., 2001). Specialized screens, perhaps for dominant alleles more like those typically found in many human syndromes, will be necessary to overcome this obstacle, and will result in the isolation of mutants that were missed in previous large-scale chemical and insertional mutagenesis screens.
Future Prospects
The clarity and rapid development of the zebrafish embryo make it an ideal system for the design of marker-based screening strategies that should allow researchers to target classes of craniofacial mutants overlooked in previous screens, such as those affecting tooth development. Mutants in tooth initiation, adult replacement tooth formation, and the craniofacial skeleton can be identified in screens by traditional x-ray techniques for visualization (Fisher and Halpern, 1999), which historically have been used in the analysis of developing mineralized tissues, and the use of more recently developed fluorescent compounds that are selectively taken up and retained by mineralized tissues, making them easily visualized under fluorescent illumination of live zebrafish.
The use of fluorescently labeled transgenes is one attractive approach that has already shown promise and allows for high throughput screening of living embryos. Zebrafish lines can be made to express transgenes constructed from tissue- and/or stage-specific promoters driving fluorescent reporter genes such as green fluorescent protein (GFP) in the developing craniofacial skeleton (e.g., type I collagen) or musculature (e.g., actin or myosin), and in the latter case one such alpha-actin-GFP line is already available (Higashijima et al., 1997). In addition, researchers can use zebrafish mutants already available in the community to “re-mutagenize” and assay for phenotypic suppressor and/or enhancer activities, allowing for unprecedented detailed characterization of molecular regulatory mechanisms.
Large-scale efforts currently under way to sequence the zebrafish genome are expected to be completed by the end of 2002 (Nasevicius and Ekker, 2000), allowing for the rapid identification of every mapped mutant zebrafish allele. This accomplishment will facilitate the identification of syntenic regions between zebrafish, human, and other vertebrate and invertebrate species for which entire genome sequences are available, providing insights into genome evolution, the significance of genome duplications (Postlethwait et al., 1998), and the significance of synteny in the maintenance of developmental processes. With the entire genome sequence, sophisticated microarray techniques, and available zebrafish mutants in hand, experiments can be designed to associate functional pathways with entire developmental gene expression profiles. Furthermore, the availability of morpholino oligonucleotides for the efficient inhibition of the translation of targeted genes (Nasevicius and Ekker, 2000) allows for the rapid identification of essential functions of new candidate genes as soon as they are identified.
New techniques continue to emerge that are facilitating the molecular dissection of craniofacial developmental processes in zebrafish. As reviewed here, gene discovery in zebrafish through mutational analysis, and soon through functional genomic approaches, promises to have a huge impact on the elucidation of gene function, and the in vivo characterization of functional interactions of gene products. This knowledge will have a particularly significant impact on the field of tissue engineering and on efforts to regenerate diseased or missing craniofacial cartilages, bones, and teeth. These available and rapidly emerging tools ensure that the zebrafish will continue to be a leading model for the molecular dissection and analysis of genes regulating craniofacial development, and for functional studies of craniofacial disease and repair.
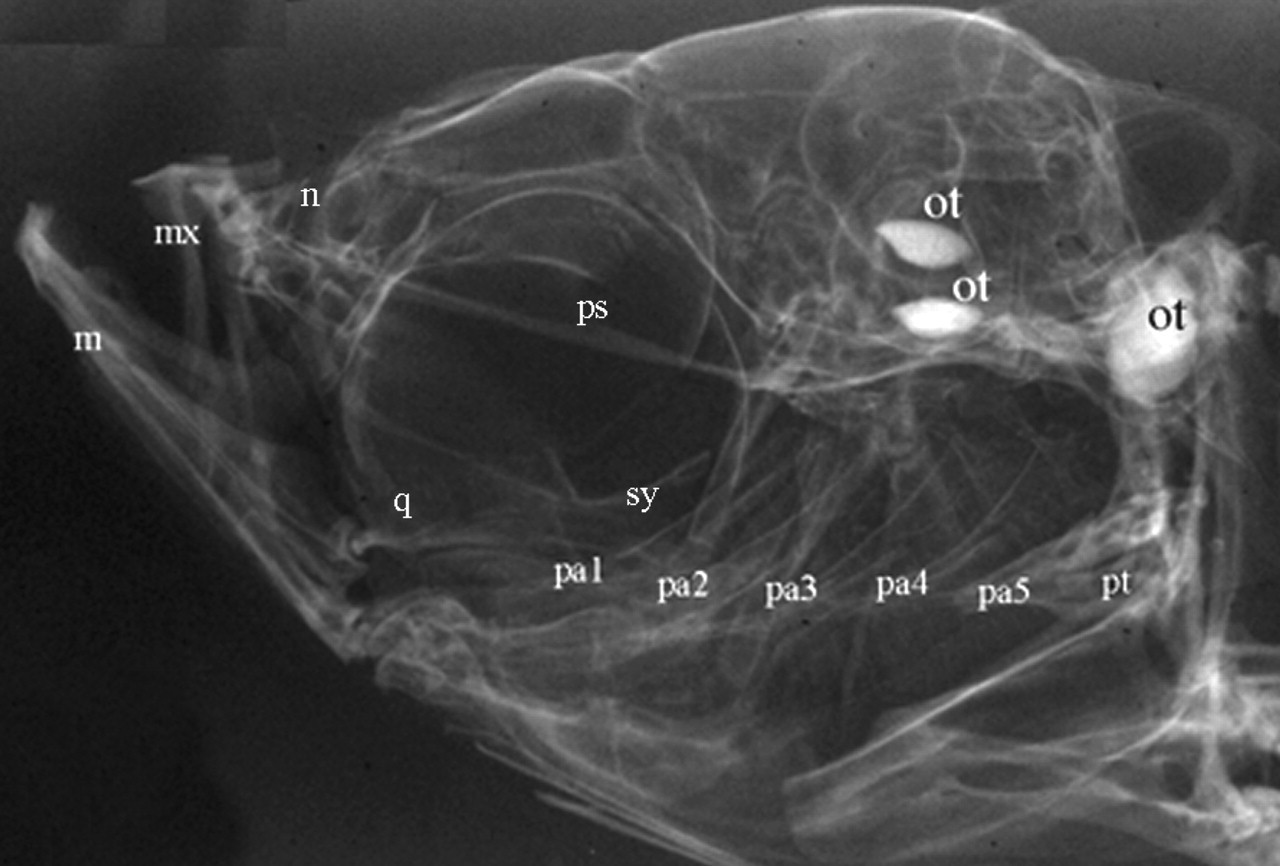
Craniofacial skeleton of an adult zebrafish. An x-ray of an adult zebrafish reveals segmented pharyngeal arch skeletal elements that closely resemble those of higher vertebrates. Abbreviations: m, mandible; mx, maxilla; n, nasal; ot, otoliths; pa1-5, pharyngeal arches 1-5; pt, pharyngeal teeth; q, quadrate; and sy, symplectic.
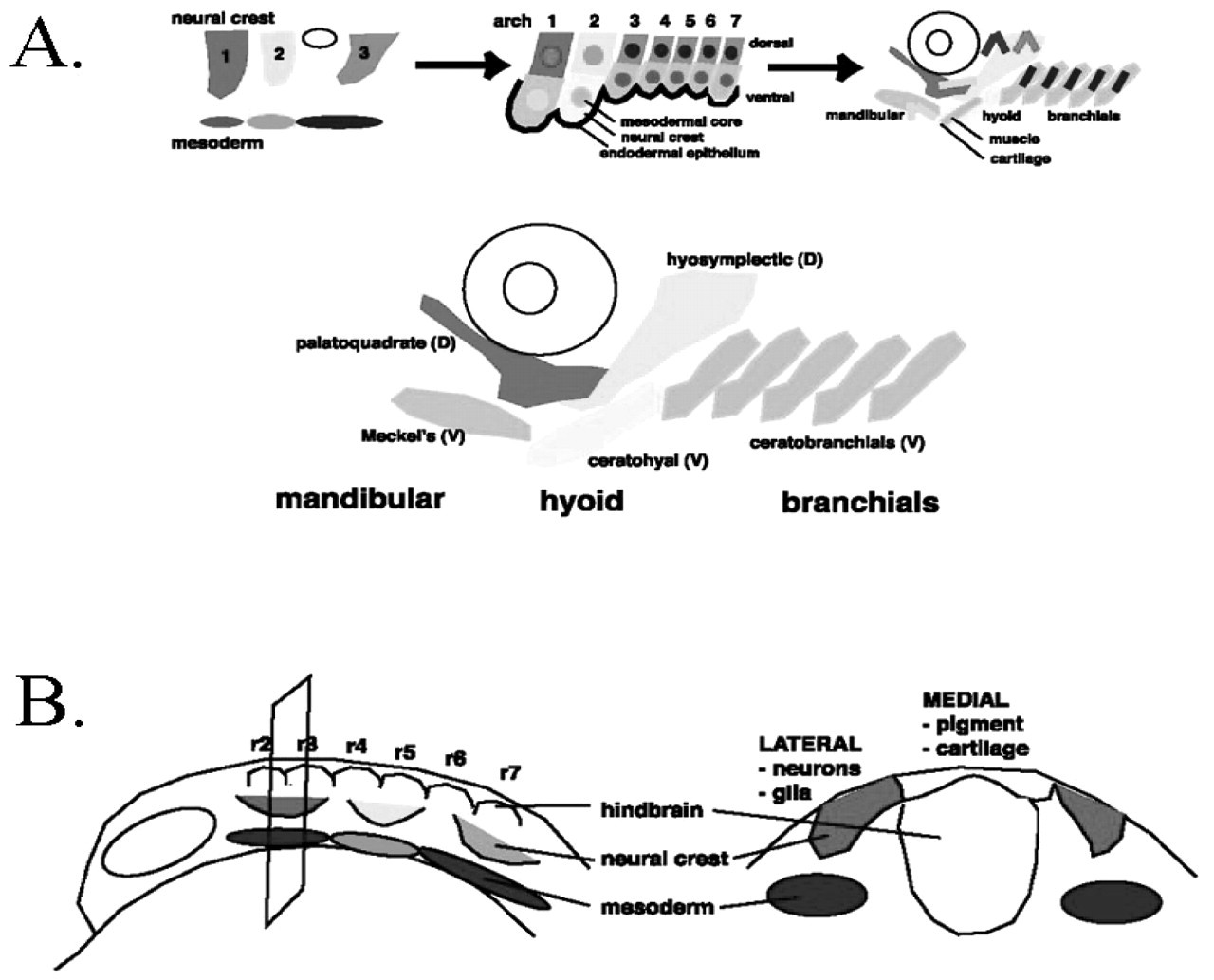
Components and patterning of the zebrafish pharyngeal skeleton.
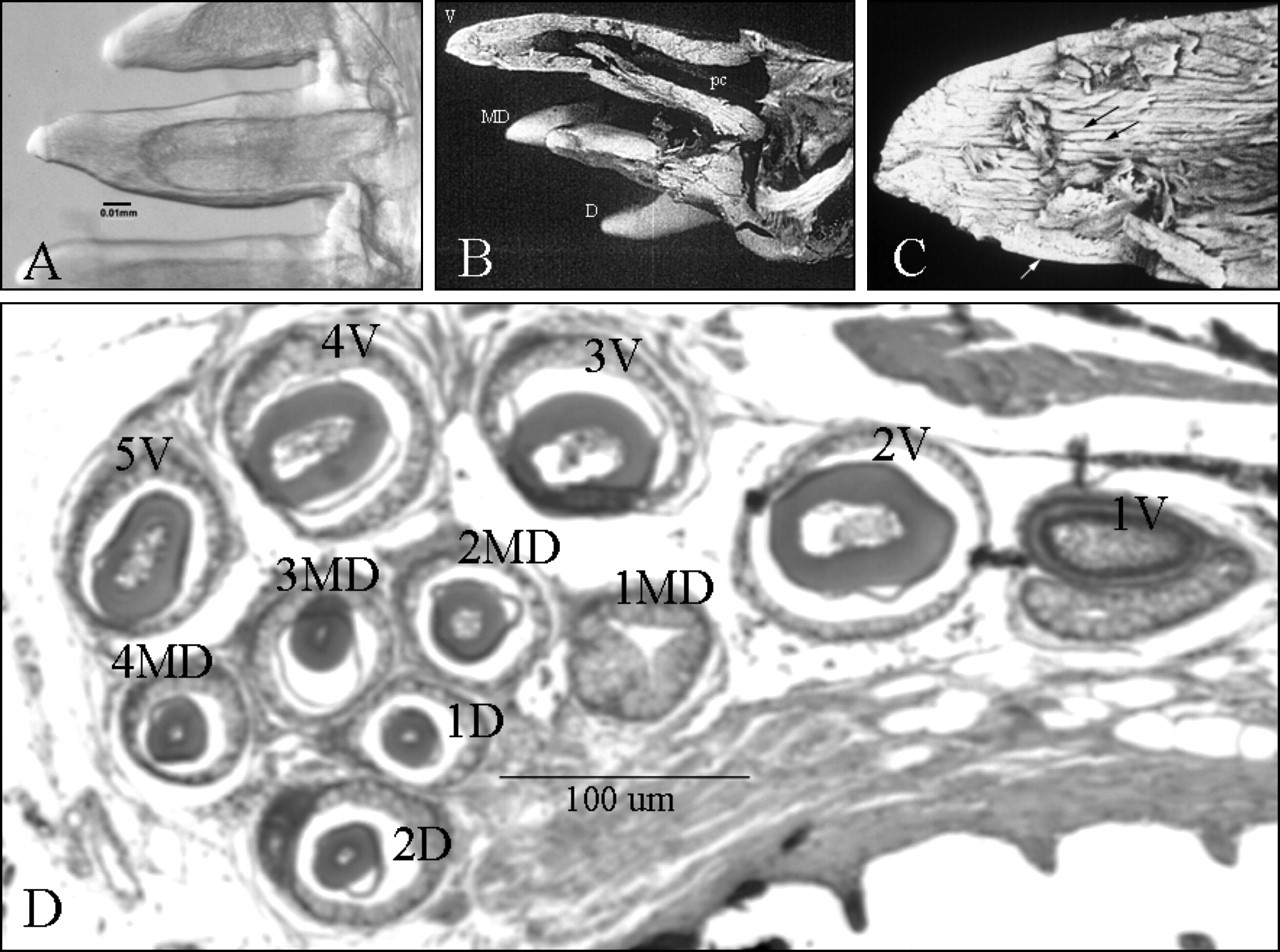
Adult zebrafish pharyngeal teeth.
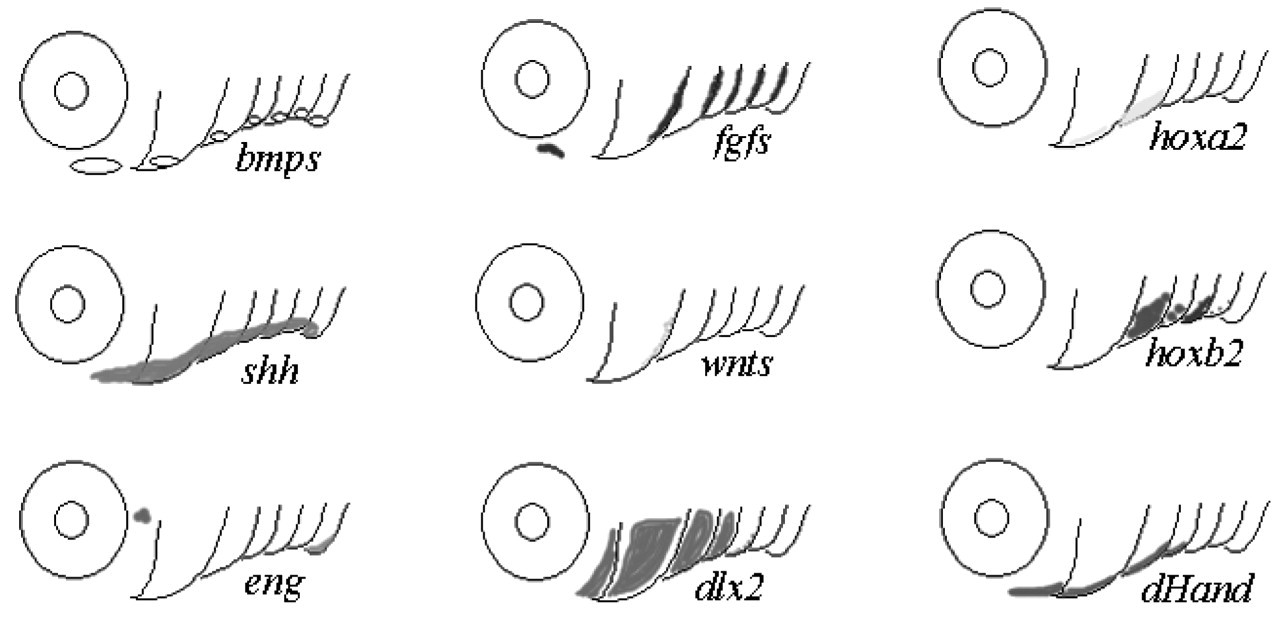
Conserved gene expression in mammalian teeth and zebrafish pharyngeal arches. The expression patterns of genes expressed in developing zebrafish pharyngeal arches are shown.
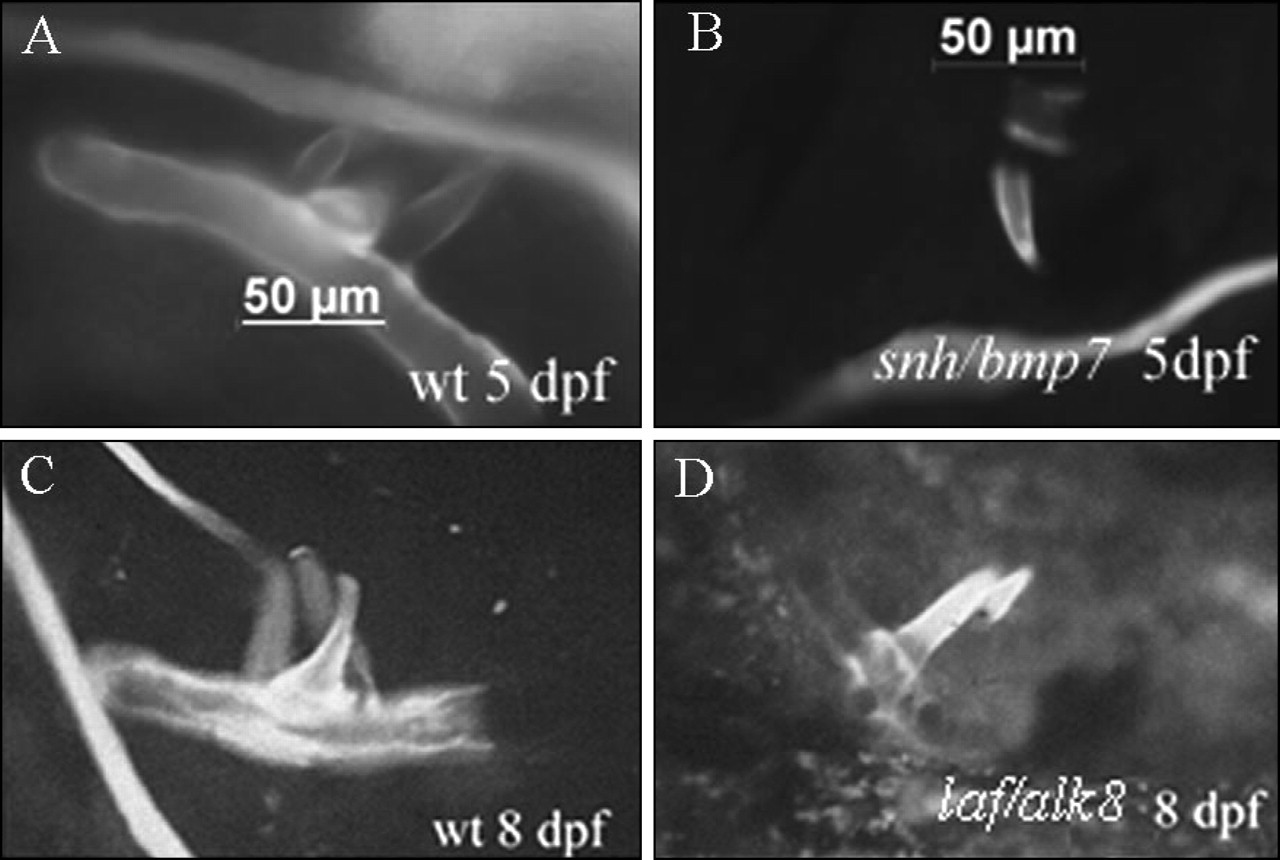
Tooth phenotypes in laf/alk8 and snh/bmp7 zebrafish mutants. Wild-type and mutant embryos at 5 and 8 days of age were stained with Quercetin (100 μg/mL, Sigma) for 3 hrs so that developing pharyngeal teeth could be visualized. At 5 days post-fertilization (dpf), age-matched wild-type siblings exhibit 3 teeth on each cb5 cartilage

Zebrafish craniofacial mutants. Alcian blue staining of 5-day post-fertilization wild-type and Boston mutants m526 and m533 embryos reveals specific distinct pharyngeal arch phenotypes. Ventral views
