Abstract
Phospholipids have been identified in enamel and dentin. Before demineralization, a group of phospholipids extracted by lipid solvents was associated with cell membranes and is therefore closely related to cell growth and intracellular regulations. After demineralization, a second group of phospholipids, associated with the extracellular matrix, was extracted; this group is probably linked to the mineralized phase. Using imidazole-osmium tetroxide fixation of rat incisors, we stained cellular unsaturated fatty acids, so that we could visualize the membrane domains, coated pits, and endocytic inclusions. Filipin, a probe for cholesterol, varied in density along the plasma membrane of secretory ameloblasts, and allowed us to visualize membrane remnants inside the forming enamel. With respect to phospholipids located in the extracellular matrix, the malachite-green–glutaraldehyde (MGA) method or iodoplatinate (IP) reaction retains and visualizes enamel and dentin phospholipids. In predentin, aggregates appearing as granules and filaments, or liposome-like structures, were located in the spaces between collagen fibrils. In dentin, organic envelopes coating the crystals, also named “crystal-ghost” structures, outlined groups of collagen fibrils. Histochemical data provided evidence that phospholipids are co-distributed or interact with proteoglycans. Radioautography after IP reaction established that [3H] choline was detected in dentin as early as 30 min after the intravenous injection of the labeled precursor, before any labeling was seen in odontoblasts and predentin. This suggests that blood-serum-labeled phospholipids pass between odontoblasts, cross the distal permeable junctional complex, and diffuse in dentin prior to any cellular uptake and phospholipid synthesis. Pharmacologically and genetically induced pathology also supports the suggestion that phospholipids play an important role in the formation and mineralization of dental tissues.
(I) Concentration and Distribution of Lipids in Enamel and Dentin
Most of the molecules of the extracellular matrix (ECM) have been identified in both dentin and enamel. In addition to biochemical data, their specific temporo-spatial distribution sheds some light on their potential roles. However, the fundamental question cannot yet be answered: Which of these molecules triggers the initial nucleation of the mineral phase and stimulates or inhibits the successive growth of the crystallites? Are they only ECM components? There is now increasing evidence that they are also acting as signaling molecules and factors influencing the biological activities of the cells responsible for the production and spatial organization of mineralized tissues.
In this context, lipids, and especially phospholipids and lipoproteins as well, have been far less studied than phosphorylated and glycosylated proteins, although they may also be involved in such events. Historically, Krasnow (1934) was among the first to establish chemically the presence of lipids in dental tissues, by determining the phosphorus content of an alcohol-ester-soluble material. The amount of lipid phosphorus per 100 g was reported to range from 0.5 to 2.8 mg in enamel and from 0.1 to 1.7 mg in dentin. Pincus (1938) isolated a fatty substance that amounted to approximately 1% of dental tissue. Leopold et al. (1951) found 24 mg lipids/100 g of dentin. Soyenkoff et al. (1951) extracted 300 mg of a chloroform-soluble material from 300 g of mixed dental tissues. Dirksen (1963) established, by paper chromatography, the presence of cholesterol esters, cholesterol, fatty acids, triacylglycerols, diacylglycerols, monoacylglycerols, and various phospholipids. In addition, Dirksen and Ikels (1964) were the first to establish that certain lipid classes were not extracted from dentin samples unless the samples are demineralized by EDTA or by carious decay. This was confirmed by Shapiro et al. (1966) in fetal bovine dental tissues, and by Prout et al. (1973) in rat teeth. However, interpretation of extraction data must take into account that some components may not, in vivo, be in contact with the mineral phase. They may become associated with it during extraction. It is not clear to what extent these data are trustworthy or partly artifactual, but the abovementioned studies highlight the following findings:
First, a total of 330-350 mg lipid/100 g dentin and 540-570 mg lipid/100 g enamel were extracted from rat incisors and molars. In bovine fetal enamel, a total of 320 mg phospholipids were extracted/100 g, and 55 mg/100 g in dentin. These variations are probably related to the stage of maturation of the tissues, and to species differences. Second, 66% of dentin lipids and 57% of enamel lipids are extracted by lipid solvents prior to demineralization, and consequently, 33% and 43%, respectively, are tightly bound to the mineral phase, whatever the species might be. Third, as shown in Tables 1 and 2
, the composition of the extract obtain from enamel and dentin before demineralization differs from the composition of the mineral-associated phospholipids. The data obtained with bovine fetal tissue are very similar (Shapiro et al., 1966; Prout et al., 1973; Ellingson et al., 1977).
A large body of evidence has accumulated which indicates that the phospholipids extracted prior to demineralization are mostly associated with the plasma membrane of ameloblasts and odontoblast cell bodies and their processes. The group of phospholipids extracted only after demineralization is probably present in the extracellular matrix that is associated with the mineralized phase (Dirksen and Ikels, 1964; Shapiro et al., 1966; Prout et al., 1973). Consequently, the second group probably has the potential to play a role in the initiation and growth of the mineral phase in dental tissues. Since minute cell processes penetrate the depth of mineralized or mineralizing tissues, demineralizing agents facilitate access of the solvent and subsequent lipid membrane extraction. Therefore, the second extract is probably contaminated by a variable membrane-associated fraction.
Little is known about membrane-associated lipids and their specific role in the biology of enamel and dentin. It is highly probable that, as is the case for most cells, lipids in odontoblasts and ameloblasts are acting not only as major membrane constituents but also as molecules involved in signal transduction and membrane trafficking. They are believed to play a pivotal role in cell proliferation and apoptosis, cell recognition, and adhesion. Their heterogeneous distribution along the cell membrane is associated with the formation of microdomains, involved in secretion and endocytosis. These cellular aspects will be reviewed in the first part of this article. In the second part, we will focus on their visualization and distribution, their origin, and the potential roles of phospholipids associated with the extracellular matrix.
Since most of our histochemical data is derived from studies on the developing part of the rat incisor, a short description is provided for the benefit of those who are not aware of this biological model.
(II) The Forming Enamel and Dentin: Morphological Data
During the development of the enamel organ, the inner enamel epithelium is initially formed by pre-ameloblasts, which are proliferating cells with a fixed number of DNA replication cycles. Although at this stage a basement membrane (BM) separates the epithelial compartment from the mesenchyme, the future pulp and dentin, there is continuity between the extracellular matrix located in the mesenchyme, the BM, transmembrane proteins such as integrins and/or syndecans, and cytoskeletal proteins of epithelial cells. At that stage, most lipids are located in cell membranes (Dunglas et al., 1999). However, it should also be noted that the dental BM and early predentin has been shown to contain phospholipids (Goldberg et al., 1988, 1990).
Amelogenesis
After the last DNA replication, post-mitotic pre-secretory ameloblasts start to synthesize and secrete enamel proteins. This occurs at birth for the mouse molar but much earlier for the incisor. For a time, no enamel is formed, although intracellular synthesis of enamel proteins is already occurring. This is due to the fact that enamel proteins diffuse into the mineralizing dentin, probably acting, at that stage, as a signaling molecule for the odontoblasts. The cell bodies of secretory ameloblasts are closely connected laterally by terminal bars, i.e., a continuous junctional complex formed by tight and gap junctions. This junctional complex behaves as a permeability barrier, which isolates the intercellular compartment from the forming enamel, located distally (Fig. 1.). The transition from pre-secretory to secretory ameloblasts is accompanied by not only the formation of junctional complexes, but also by dramatic changes in the shape of the distal parts of ameloblasts, which are subsequently transformed into Tomes' processes. The presence of Tomes' process residues inside the forming enamel is the major reason for the high level of lipids found in the developing enamel compartment. In addition, it is now well-recognized that cell remnants are present during enamel formation and maturation, and also at later stages when enamel is fully mature, in the rat incisor as well as in unerupted and erupted human enamel (Goldberg et al., 1983, 1987, 1995, 1998). Therefore, forming enamel is loaded with membrane remnants, similar in some respects to matrix vesicles present in bone, mantle dentin, and cartilage, but, in contrast to matrix vesicles, Tomes' processes debris has no recognized role in initial enamel mineralization. In the course of this review, only the developing enamel will be considered; therefore, we are not going to discuss the changes that the enamel undergoes during maturation and post-eruptively, although there is a high probability that lipids of bacteria and saliva origin penetrate enamel, at least in the subsurface (Goldberg et al., 1987).
Dentinogenesis
Dentin, metadentin, and predentin form concentric layers, parallel to each other and to the dentino-enamel junction (DEJ). All three contain, unambiguously, three distinct types of extracellular matrix. Metadentin is the area where mineralization is initiated and which has its own specific characteristics (Goldberg and Septier, 1996). This terminology is different from the concept, as reported by paleohistologists, of metadentin, which is a specialized tissue that includes both ortho- and osteo-dentin (Ørvig, 1967). With respect to the cells, pre-odontoblasts originate from neural crests and migrate from the central part of the mesenchymal or embryonic pulp toward the periphery of the pulp. Like pre-ameloblasts, pre-odontoblasts are actively dividing cells, with a fixed number of mitoses. The final mitotic division is followed by the differentiation of two specific groups of cells. The outermost cells, which are in contact with the basement membrane, differentiate into post-mitotic polarizing odontoblasts, whereas the daughter cells, located some distance from the basement membrane, are included in the Höhl's layer and serve as replacement cells in case of odontoblast alterations. Dentin constitutes a mineralized layer which is secreted by odontoblasts (Figs. 2, 3 ) at a rate of approximately 4 μm per day. This increase in thickness occurs to the detriment of the pulp. The rate of dentin deposition decreases at the end of root formation. Predentin is an unmineralized layer, with a constant 15- to 20-μm thickness, where native fibrils of collagen are secreted together with proteoglycans and phospholipids, which collectively form more complex structures. At the junction between predentin and dentin, there is a transition zone 0.5 to 2.5 μm thick, termed metadentin (Goldberg and Septier, 1996) (Fig. 4). It is noteworthy that odontoblast cell bodies are located at the periphery of the pulp, and therefore easily accessible to lipid solvents. In contrast, the membrane lipids of odontoblast processes, and of the lateral branches located inside minute secondary dentin canaliculi, are extracted more easily by lipid solvents when the diameter of the dentin tubules is enlarged by demineralizing agents and when the permeability of the dentinal tissue is increased. Consequently, it must be borne in mind that the lipid material extracted from dentin after demineralization may be a mixture of cellular and extracellular components, even if most of it is obtained from the acellular part of the dentin. As noted earlier, this group of lipids may not be actually associated with the mineral in the in vivo situation but artifactually associated during the release of extracted species.
In addition, it is important to note that gap- and desmosome-like junctions form the junctional complex located in the distal parts of odontoblast cell bodies. Tight junctions, which are scarce, are associated with some stages of differentiation, and apparently detected only in some species. They do not contribute efficiently to the formation of a permeability barrier. A few molecules originating from the blood serum cross this intercellular barrier, and contribute to the extracellular matrix. This is the case for α2HS-glycoprotein and albumin, and probably for some other dentin proteins (Kinoshita, 1979; Takagi et al., 1990). In this context, the diffusion of fixative solutions, whether or not followed by lanthanum nitrate or other fluorescent tracers, was demonstrated to be transjunctional for odontoblasts; whereas, in contrast, intercellular tight junctions impede intercellular diffusion between secretory ameloblasts. Tannic acid passes through terminal junctions, diffuses, and stains glycoconjugates in predentin (Goldberg and Septier, 1989). Similar findings were reported when lanthanum nitrate was used as a tracer (Torrès-Quintana et al., 1999) (Fig. 4). Lucifer Yellow CH appeared to diffuse between odontoblasts (Ushiyama, 1989). This implies that all the molecules found in dentin are not derived from odontoblasts, but that some may originate in the blood serum and diffuse between the odontoblasts before being trapped in predentin and dentin. This is also suggested by some of our radioautographic data, at least for some choline-containing lipids, which apparently are not synthesized by the odontoblasts (Goldberg et al., 1999).
(III) Visualization of Lipids and Phospholipids in Dental Tissues
(1) Historical background
For years, Sudan black B has been considered as one of the most accurate methods used to detect lipids. Oil red and Sudan IV have also been used successfully. Wislocki et al. (1948) and Wislocki and Sognnaes (1950) reported that odontoblast processes and prism sheaths in the developing enamel stained with Sudan black B. Irving (1958a,b, 1973) and Irving and Wuthier (1961) reported later that after formaline-saline fixation, pyridine or ethanol-benzene lipid extraction, demineralization, gelatin embedding, sectioning, and Sudan black B staining, a sudanophilic material was associated with the border where mineralization was initiated in dental calcified tissues, and in bone and cartilage as well. These results are still quoted as a proof that lipids are actually involved in biomineralization. However, Fincham et al. (1972) pointed out that the total quantity of lipids is insufficient to explain the sudanophilic properties of forming enamel. They also stated that the lipophilic properties of some enamel proteins may be responsible for the staining. In addition, Beynon (1976) and Patterson et al. (1977) have shown that, after lipid extraction with acidified chloroform-methanol solvent, or phospholipases, or detergents, enamel matrix was still stainable by Sudan black.
(2) Ultrastructural cytochemistry: potential roles of membrane-associated lipids
For a long time, few ultrahistochemical studies were published. This was due to the limited availability of specific techniques. The characterization of lipids based on their osmiophilic properties is certainly not a sufficient criterion. Some of the available methods were usable for staining cellular lipids, but failed to identify phospholipids in the extracellular matrix.
(2a) Unsaturated fatty acids
After fixation by perfusion with 1.5% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.4, thick sections were placed in 0.2 M imidazole to which 4% aqueous osmium tetroxide was added, in the procedure of Angermüller and Fahimi (1982). OsO4 interacts with unsaturated fatty acids, and spot tests have shown that linolenic, linoleic, and oleic acids were densely stained by this method, whereas the saturated fatty acids palmitic and stearic acid remained unstained. By this technique, membrane microdomains were visualized along odontoblast processes, electron-lucent and electron-dense areas being visualized adjacent to each other, along the plasma membrane. Caveolae were densely stained. Such unsaturated fatty-acid-rich microdomains seemed to be associated with endocytosis. They are invaginated into coated pits, isolated in the form of coated vesicles, and driven back to the distal part of the Golgi dictyosomes, where presumably the discrimination occurs among what will be destroyed in the Golgi-Endoplasmic Reticulum-Lysosome (GERL), what will be conveyed to lysosomes, and what will be re-used (Goldberg and Septier, 1984). This is probably the reason why the GERL was also densely stained. In contrast, the method failed to reveal any lipidic component in the extracellular matrix. This localization seemed to be very similar to the staining obtained with tannic acid, known to interact with the choline component of phosphatidylcholine (Kalina and Pease, 1977). Again, coated pits and endocytic inclusions displayed enhanced electron-density (Goldberg and Septier, 1989). In addition, with tannic acid, some granules were stained in predentin as well as “crystal ghosts” in dentin.
(2b) Cholesterol
Another specific distribution was revealed by filipin, a probe for cholesterol. This polyene antibiotic forms complexes with membrane cholesterol and related β-hydroxysterols, producing distinct protuberances of the membrane, measuring 25-35 nm in diameter, as visualized in a freeze-fracture replica (Kinski, 1970; Elias et al., 1979). In odontoblast cell bodies, the plasma membrane displayed a density of 15.16 ± 0.91 filipin/cholesterol protuberances/μm2 (mean ± SD). In the membrane of lysosome-like vesicles, 451 ± 3.3 protuberances/μm2 were reported. When cell bodies are adjacent to other cell bodies, the cholesterol distribution was weak or undetectable with the technique used, cholesterol being below the level of detection. It is documented that maximum sensitivity of filipin as a probe to detect cholesterol lies close to 5 mol% cholesterol (Steer et al., 1984; Hayat, 1993). Along the plasma membrane of odontoblast processes, a density of 32.21 ± 0.71 protuberances/μm2 was scored in predentin, vs. 100.79 ± 2.49/μm2 in dentin (Figs. 5a, 5b) (Goldberg and Escaig, 1984b). A rather similar situation was seen in secretory ameloblasts, where 0-32 filipin/cholesterol protuberances/μm2 were scored along the plasma membrane in cell bodies, vs. 150-250/μm2 in Tomes' processes (Fig. 5c). Between 350 and 450 protuberances were scored/μm2 on membrane remnants found at some distance away from Tomes' processes inside the developing enamel (Fig. 5d). Such remnants were found at distances greater than 15 μm from the apparent ending of Tomes' processes, near the dentino-enamel junction. This method also allows for clear visualization of membrane remnants which otherwise were undetectable in the forming enamel (Goldberg and Escaig, 1984a).
Cholesterol in odontoblast processes is associated with a less fluid environment. The plasma membranes of processes need to become more rigid to resist damage. This may be the reason for the increasing gradient of filipin/cholesterol protuberances from the cell bodies to the processes in predentin and dentin, where the density reaches a maximal value. Similarly, Tomes' processes resist the pressure of the surrounding forming enamel, and vesicles isolated inside the forming enamel are isolated in a rigid environment. In many other cell types, it has also been reported that a high cholesterol/protein ratio makes the membrane especially rigid (Demel and de Kruijff, 1976). There is also a gradual reduction of motility of membrane components, which implies higher cholesterol content. This increase may be associated with a decrease in the membrane-associated proteins, or the latter may be unaffected (Feltkamp and Waerden, 1982; Hayat, 1993).
In conclusion, from these investigations it was possible to visualize two different groups of lipids. First, a group of membrane lipids associated with microdomains, rich in unsaturated fatty acids, is apparently involved in the motility of some clusters, fragments, or segments of the plasma membrane. These domains were seen to be preferentially implicated in endocytosis and subsequently in membrane transport from the plasma membrane to the Golgi apparatus. Such lipid domains, called rafts (Verkade and Simons, 1997), may serve as a platform for the performance of important functions in membrane trafficking and signaling.
Second, these specific histochemical techniques allowed for clear visualization of an increasing gradient of membrane-associated cholesterol, which provides rigidity to the plasma membrane system both in developing enamel and in dentin. However, it should be noted that even cholesterol-rich membranes cannot resist crystal growth. For example, in cartilage, matrix vesicles are cholesterol-rich (Wuthier, 1988). This is also the case in the initially formed mantle dentin (Katchburian, 1973). In both cases, growing crystals easily rupture the limiting membrane.
(2c) Essential fatty acid deficiency
Experimental approaches have been used to obtain a better understanding of the properties and role(s) that lipids may play in dental tissues, or at least in cells such as odontoblasts and ameloblasts. In this context, essential fatty acid deficiency is one of the best-documented models. Essential fatty acids are major constituents of structural lipids. As cellular components, membrane phospholipids are now recognized to be involved in membrane trafficking, signal transduction, and cytoskeletal regulation, including cell-cell contact and cell growth (Martin, 1998), while ceramides are implicated as messengers in signal transduction for catabolic cytokines as well as in apoptosis. Essential fatty acids (EFA) are specific polyunsaturated fatty acids, which are not synthesized by animals. Vertebrate animals are dependent on dietary plant material that contains two major precursors, i.e., linoleic acid and linolenic acid. In animal tissues, these acyl chains can be converted to other fatty acids, and elongation reactions can produce longer chains. Deficiency in EFA (EFAD) induces changes in water permeability of membrane or cell layers and epithelial hyperplasia. EFAD rats lose weight, and heart enlargement, decreased capillary resistance, and enlargement of the kidneys have been reported as consequences of EFAD. Alteration of the lungs, of adrenal and thyroid glands, and altered fatty acid composition of many tissues lead ultimately to death (Gurr and Harwood, 1991). Little is known with respect to the effects of EFAD on dental-forming cells. Cell alterations have been reported; however, alterations in enamel and dentin were relatively minor (Prout and Odutuga, 1974a,b,c). This was confirmed by our group and by others (Vermelin et al., 1995; Goldberg and Boskey, 1996; Goldberg et al., 1996).
One interesting and puzzling finding was that pulp cells were three-fold as numerous as in the normal pulp when newborn rats received a EFAD diet at weaning for 5 wks. They were twice as numerous when, during pregnancy, the mother was fed an EFAD diet and the offspring were normally fed after being weaned. It is hard to explain this increase in pulp cell number, since a reduction might have been expected as with any dietary deficiency. As a working hypothesis, we assume that the number of cells is normally regulated by apoptosis. Since enhanced numbers of mitoses were not observed, we hypothesized that normal apoptosis might be impaired, consequently extending the lifespan of many cells. In support of this hypothesis, chemical analysis of the developing part of rat incisors revealed that amounts of linoleic and arachidonic acid were reduced to half their normal levels (Goldberg and Boskey, 1996). Similar results have been obtained for bone (Alm et al., 1994). In addition, since these fatty acids are also precursors for prostaglandins and leukotrienes, it may also be that EFA have a role in the synthesis of these mediators. EFAD exerts an inhibiting effect on the synthesis of inflammatory mediators. The relationship between EFAD and the synthesis of prostaglandin and leukotriene is well-documented. The effects of EFAD on ceramide synthesis are less clear. There are considerable data that have established the effects of ceramide on apoptosis, at least in articular cartilage (Sabatini et al., 2000). Such data are missing for odontoblasts and ameloblasts. The results reported here concern pulp cells and may not apply to enamel- and dentin-forming cells. However, there are some data showing that although odontoblasts display severe morphological alterations and delayed incorporation of radiolabeled precursors, they still survive after 21 days (Vermelin et al., 1995; Goldberg and Boskey, 1996). Because they are post-mitotic cells, their number stays constant, but there is an increase in the number of pulp cells in EFAD sections. In dentin, choline-containing lipid uptake was reduced and delayed compared with that of rats fed normally. In contrast, no change was detected in the secretory ameloblasts and forming enamel (Goldberg and Boskey, 1996).
(IV) Ultrastructural Histochemistry
(1) Visualization of the phospholipids located in enamel and dentin extracellular matrix
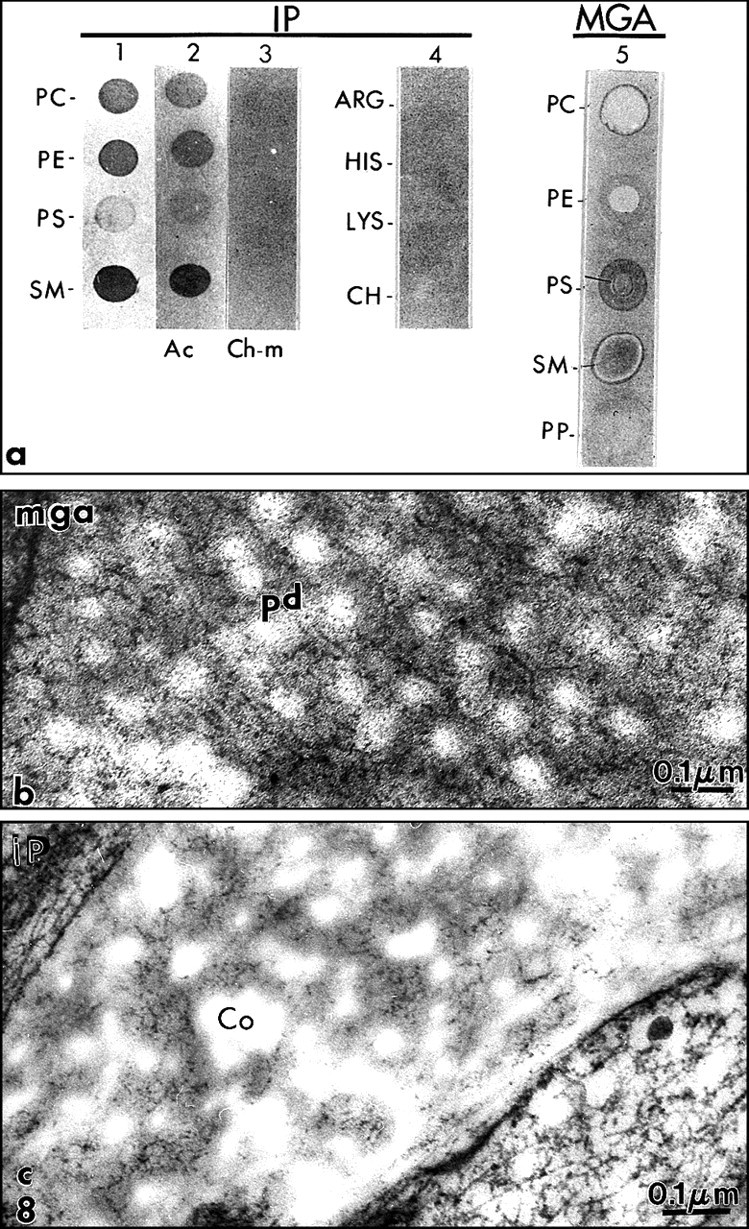
Two staining methods were used successfully to visualize and identify phospholipids in the dental extracellular mineralized tissues: the malachite green aldehyde (Goldberg and Septier, 1985) and iodoplatinate methods (Vermelin et al., 1994).
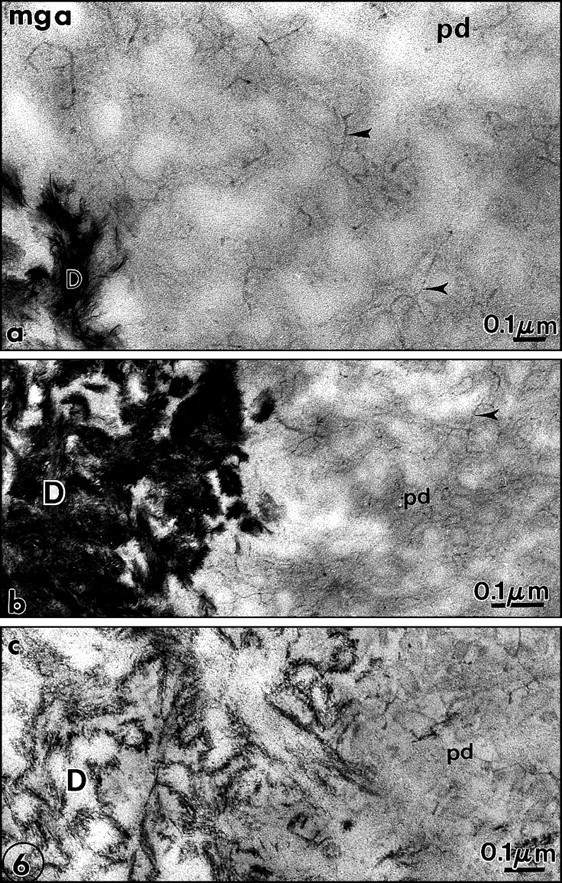
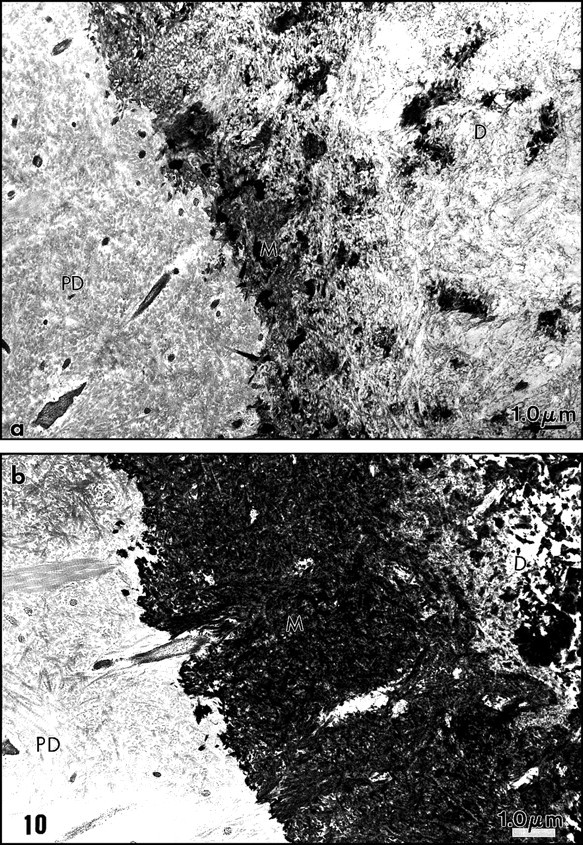
The malachite green aldehyde (MGA) fixation was devised by Teichman et al. (1972, 1974). The reaction involves the formation of stable complexes between malachite green and phospholipids in the presence of glutaraldehyde, which is a cross-linking agent. During fixation, lipids which would otherwise have been extracted or translocated during dehydration are retained with this technique. After SDS-PAGE, malachite green has been used successfully as a staining agent to visualize phosphoproteins in the gel. Spot-test experiments demonstrated that phosphatidylethanolamine (PE) was faintly stained by MGA. Phosphatidylcholine (PC) staining was weak, however. In contrast, phosphatidylserine (PS) and sphingomyelin (SM) were intensely stained by MGA. Dentin phosphoprotein was unstained (Vermelin et al., 1994). This method was applied to dentin and enamel, and, for the first time, phospholipids were identified in the extracellular matrix and not only as membrane components. Aggregates of phospholipids appear as granules (7-10 nm in diameter) or filaments (25-35 nm in length). They were located in the spaces between collagen fibrils in predentin (Figs. 6a, 6b). Ramified structures displayed increasing diameter from the proximal to the distal predentin. In dentin, without demineralization, needle-like structures appeared in the mineralized zone, along and in the spaces located between the collagen fibrils. In sections floated on EDTA or acetic acid solutions, crystal-like structures were still present despite the demineralization procedure. They are reminiscent of the “crystal ghosts” described by Bonucci (1987) in cartilage and bone, that outlined individual or groups of collagen fibrils (Fig. 6c). This suggests that some organic material is closely associated with the surfaces of the crystallites, or, after being processed for electron microscopy, is precipitated at the surfaces of crystals. The organic envelope coating crystallites persisted after demineralization. Histochemically, this material is positively stained as a protein-rich structure associated with glycosaminoglycans and phospholipids, as has been previously reported in bone (Bonucci, 1987). No specific staining for phospholids was detected at the junction between predentin and dentin, the so-called mineralization front or metadentin, in contrast to what was reported by Irving with Sudan black (Irving and Wuthier, 1961; Irving, 1973).
We modified the method to improve the retention of extracellular lipids. Rapidly frozen samples of rat incisor dentin and predentin were fixed by freeze substitution in a mixture consisting of 0.02 g malachite green and 0.4 cc undiluted acrolein in 20 mL acetone, and placed in a Cryocool CC-100 for 3 days. They were freeze-substituted for 3 days in 100 mL 2.5% osmium tetroxide in acetone, and then embedded in Epon (Goldberg and Escaig, 1987). The thickness of the electron-dense intercollagen network of branched fibrils appeared to be increased with this anhydrous technique, and in many areas, it was clear that in predentin the network was formed by connected vesicle-like or by liposome-like structures. Collagen appeared electronlucent in the sections, and although some contacts were detected between MGA-phospholipid aggregates and collagen fibrils, these aggregates were mostly located in intercollagenic spaces (Figs. 7a, 8b ). To control the specificity of the reaction, investigators treated some samples by methanol, a phospholipid solvent, prior to freeze-substitution. After such treatment, the electron-dense structures forming network in predentin were abolished (Fig. 7b). Chloroform could not be used at such a low temperature. In dentin, crystal-like (or crystal ghosts) structures were seen along the mineralized collagen fibrils, and the staining persisted even after demineralization with EDTA or acetic acid solutions (Fig. 6c). Spot tests established that MGA stains PS and SM and, to a lesser degree, PC (Vermelin et al., 1994). Dentin phosphoprotein (DPP), which is stained in SDS-PAGE MGA-fixed gels, remains unstained with this experimental spot-test approach (Fig. 8a).
The second method used successfully for electron-ultrahistochemistry was the iodoplatinate reaction (IP), according to Kao and Lichtenberger (1987) (for details of the technique, see Vermelin et al., 1994). Spot tests indicated that PC, PE, and SM were densely stained. By contrast, PS remains unstained (Fig. 8a). Therefore, the staining obtained with MGA and IP did not totally overlap, because it was specific for different types of phospholipids. Treating the spot tests by acetone, which is not a phospholipid solvent, did not alter the staining pattern. Chloroform-methanol pre-treatment abolished the staining. Since IP interacts with quaternary ammonium compounds, some basic compounds (arginine, histidine, lysine, and choline) were included in the spot tests. They remained unstained (Fig. 8a).
Again, electron-dense granules (3-12 nm in diameter), linked by filaments, were observed after such treatment. They formed a network in the intercollagen spaces, increasing in size and number in the distal predentin (Fig. 8c). In dentin, crystal-like structures were stained. They resisted treatments with acid or chelating solutions. Treatment of the sections with acetone, which extracts some lipids but not phospholipids, did not modify the staining pattern. Chloroform-methanol pre-treatment largely abolished the staining, as did phospholipase C. This was also the case for the needle-like structures in dentin.
Taken together, the staining suggests that phospholipids are present in predentin as extracellular matrix components. They are organized as a network of liposomes or vesicles, observed at various stages of aggregation, and located in spaces between collagen fibrils. In contrast, in dentin, needle-like structures or “crystal ghosts” are closely associated with the surfaces of mineralized collagen fibrils. These are likely the extracellular components that were obtained only after demineralization and believed to be associated with the mineral phase. We have already commented on the extraction procedures and possible artifactual re-association that should be taken into account in the interpretation of the results. Such re-associations could occur during fixation and demineralization. Some electron-dense amorphous spots were also stained in dentin, unrelated to any identified structures. They disappeared after treatment with either chloroform/methanol or phospholipase, whereas they resisted acetone treatment, and therefore are probably phospholipids.
These data are in agreement with the results which were published by Ellingson and Smith (1975) in unerupted bovine pulp, by Ellingson et al. (1977) in predentin, and by Shapiro et al. (1966) in embryonic bovine dentin. Collectively, these results provide a clearer idea of which phospholipids are related to cell membranes and which phospholipids belong to the mineralized extracellular matrix, respectively (Table 3). Again, liberation of the extracted species from another location and artifactual re-association should also be considered.
Chemical analyses and histochemical studies were carried out on phospholipid distribution in mouse embryonic tooth germs (day 18 and newborn) and in the post-natal first molar (day 7 and day 21, with and without demineralization). The coronal part of the tooth is already formed at day 18 of gestation. The root starts to be formed at birth (day 0). Lengthening of the root and eruption occur at day 7, whereas the tooth becomes functional and fully mineralized at day 21. Analysis of these data indicates the same tendencies to incorporate in the extracellular matrix lipids taking origin from membranes during development of dental tissues and their subsequent mineralization (Dunglas et al., 1999). Table 4 summarizes the findings.
It is clear that the high PC, PE, and SM content of the undemineralized fraction reflects more or less a plasma membrane composition. In contrast, the presence of PS, PI, and PA in the demineralized dentin extract, a higher PE and SM percentage, and lower PC content characterize the mineral-associated phospholipid fraction.
From Table 4, it appears that, during tooth development, PE increased in percentage from E18 to D7, reached a maximum, and then decreased at D21. PC peaked at D1, but remained at a constant level afterward. In contrast, SM and PS gradually increased and, together with PI, culminated at D21 in the demineralized fraction. From the weight of the extracts per germ or tooth, PC and PI increased 2.3-fold between E7 and D21, whereas PE, SM, and PS increased by 4.8-, 5.4-, and 5.9-fold, respectively.
From the above discussion, the data reported in the literature and our own findings fit well together and provide evidence that some phospholipids are closely related to the mineralization processes, whereas some others are related to cell growth and intracellular regulations.
(2) Phospholipid and proteoglycan interactions
It is also interesting to note that, in both predentin and dentin, phospholipids share the same locations as proteoglycans. Ultrastructural histochemical studies have shown that glycosaminoglycans (GAGs) are present in predentin and dentin and are closely associated with phospholipid-stainable material.
In predentin, GAGs appear as aggregates or as granules when cationic dyes such as alcian blue, ruthenium red, or ruthenium hexamine trichloride were used. They appear as stellated structures with cuprolinic blue, and as forks or boomerang-like structures with cationic detergent such as cetylpyridinium chloride (for review, see Goldberg and Takagi, 1993; Embery et al., 2001). GAGs are not stained by MGA. Depending on the method used, aggregates are located either in close contact with the collagen fibrils and/or in intercollagenous spaces. However, it has also been shown that, after rapid-freezing and freeze-substitution, which minimizes loss of proteoglycans and artefactual aggregation of the components, GAGs appear as an amorphous expanded gel, filling all the available space between the collagen fibrils (Goldberg and Escaig, 1984c). Taken together, in predentin, the staining for GAGs appears as quite uniform. More recently, immunogold detection revealed that chondroitin sulphate/dermatan sulphate (CS/DS) displays a decreasing gradient from the proximal to the distal predentin, and the reverse has been observed for keratan sulphate (KS) (Goldberg et al., 2000; Hall et al., 2000). The same applies to the CS/DS- and KS-related leucine-rich proteoglycans decorin and lumican, respectively, which are not uniformly distributed and increase in the distal predentin. Apparently, from our electron-histochemical observations, phospholipids also display such a gradient, with increasing size and number of aggregates produced by the staining method. Therefore, it can be concluded that, in predentin, some phospholipids and some proteoglycans share the same territories in intercollagen spaces. They may be involved in the transport of collagen fibrils from the proximal predentin, the place where they are secreted, to the distal part, where mineralization occurs. It is possible that PGs together with lipoproteins play roles in the fibrillation process of collagen, as is the case in vitro (Pentikäinen et al., 1997). Liposomes, on the other hand, as observed in many areas in our predentin sections, may contribute to calcium transport toward the mineralization front. It should be stated, to avoid any confusion, that these liposomes are distinct from extracellular matrix vesicles.
Liposomes form a network of extracellular phospholipids in the predentin at all stages of dentinogenesis. Interconnected filaments and granules, 3–12 nm thick, form the mesh of this irregular network, about 0.1-0.2 mm in diameter. This network is totally unrelated to matrix vesicles (MV), seen only during the formation of the mantle dentin, and never observed in the developing enamel. MV originate from cell budding and shedding. The proteins and phospholipids of the limiting membrane of MV have a composition that differs from that of the plasma membrane, at least for chondrocytes. Proteoglycans are located in the extracellular matrix and at the membrane surface. Enzymes and proteins known to be associated with the mineralization processes are also present in MV. In this context, a high level of alkaline phosphatase, an enzyme that generates inorganic phosphate from organic phosphate compound, has been detected. Calcium-binding proteins of the annexin family (i.e., annexin II, V, and VI), matrix metalloproteases such as MMP-2, -3, -9, -13, and growth factors such as transforming growth factor-β have been identified in MV (Wuthier, 1988; Nefussi et al., 1989; Dean et al., 1992; D'Angelo et al., 2001). Many studies have been carried out on MV and provide information on the mechanisms of cartilage mineralization. Interactions between MV and collagen (Wu et al., 1991), description of the nucleational core complex (Wu et al., 1993), and identification of annexin (Wuthier, 1988) shed light on the phenomenon and how it is triggered. In the absence of MV, which is the case with circumpulpal dentin and enamel, mineralization is also occurring, but in a different way. However, some events are similar and suggest some common features. Although there are many differences between the cell-derived MV and the liposome network visualized in predentin, they are both formed by lipids involved in biomineralization.
The nature of the interaction with PGs is not yet established, but it can be argued that either they are acting in synergy in the mineralization processes, or PG molecules exclude calcium ions at their periphery, thereby favoring calcium concentration within extracellular phospholipids. In doing so, the inhibition of calcium phosphate precipitation and/or the increased concentration of calcium ions inside phospholipid-rich liposomes may contribute to events which promote initial mineral nucleation. In vitro, it has been shown that when aggrecan—a cartilage proteoglycan which is not apparently present in predentin—is added to the suspending medium, it has no effect on intra-liposomal precipitation, but it severely reduced its subsequent spread into the medium by ∼ 70% (Eanes et al., 1992). This is not the first time that PGs have been shown to interact with lipids and contribute to the formation of mineralized structures. Such a phenomenon has been reported in atherosclerotic plaque, and the same may apply to the dentin situation (Jarrold et al., 1999; Kaplan and Aviram, 2000). Decorin links low-density lipoproteins (LDL) to collagen, and this interaction seems to contribute to the formation of atherosclerotic plaques which subsequently underwent mineralization (Kovanen and Pentikäinen, 1999). Biglycan, found in cartilage, bone, skin, dentin, and many other tissues, is also a vascular proteoglycan. It binds differently to the high-density lipoproteins 2 and 3 (HDL2 and HDL3) (Olin et al., 2001). This has been established in atherosclerotic plaque but cannot be generalized. In bone, dentin, and cartilage, initial mineralization is provided by matrix vesicles: the phospholipid/proteinaceous surrounding membrane is protected or covered by PGs, and the latter are either closely associated with MV membrane or located in the surrounding extracellular matrix (for review, see Wuthier, 1988; and Nefussi et al., 1989). Mineralization of matrix vesicles occurs only after degradation of PGs by specific MV-associated metalloproteinases such as MMP-3 or stromelysin-1 (Dean et al., 1992). It has been reported that MMP-2, -9, and –13 are also implicated in these events in growth plate cartilage (D'Angelo et al., 2001). In 1976, Vogel and Boyan-Salyers (1976) proposed that acidic phospholipids may be implicated in the following cascade of events: (1) the liposomes or plasma membranes provide an environment in which mineralization may occur; (2) after desolvation and ion concentration; (3) in addition to Ca++ binding, the formation of CaHPO4 dimers and their condensation into a (Ca9PO4)6 unit bound to the membrane are achieved by crystal growth, which, in turn, destroys the hydrophobic domain; and (4) once mineralization is initiated, the crystals grow along the surfaces of collagen fibrils, and this is the reason for the presence of needle-like structures, also termed “crystal ghosts” (Bonucci, 1987), which are composed of a mixture of proteins, proteoglycans, and phospholipids. This co-distribution is also observed in dentin.
In this context of interactions between proteoglycans and low-density lipoproteins, some data suggest that low-density lipoproteins (LDL) interact with the GAG chains of decorin and biglycan, which themselves are subject to receptor-mediated endocytosis through lipoprotein lipase-mediated interactions (Schönherr et al., 2000). It is also noteworthy that LDL-related scavenger receptor and thrombospondin 2 (Yang et al., 2001) regulate extracellular matrix metalloproteinase-2 (MMP-2) levels.
(3) Origin of predentin and dentin phospholipids
The incorporation and fate of phospholipid precursors have been studied mainly by radioautography. For years, the results were impaired by the fact that extraction and diffusion of the labeled precursor(s) could occur during fixation, dehydration, and embedding (in Epon). Therefore, all the data obtained with conventional methods were unreliable. Our attempts to label precursors with palmitic acid (Goldberg and Escaig, 1984d) or oleic acid (Goldberg et al., 1990) failed to provide accurate information. The background labeling was so high that interpretation of the results was hampered. In contrast, when [3H] choline is used in conjunction with MGA or iodoplatinate fixation and staining, aggregates are formed which prevent translocation of the labeled compound. Background labeling in this instance was moderate and therefore acceptable.
To investigate how labeled precursors are incorporated into dental tissues, and the fate and turnover of phospholipids, we injected rats intravenously through the jugular vein with [3H] choline. The rats were killed 30 min, 1 hr, 2 hrs, 4 hrs, 24 hrs, 48 hrs, and 4 days later by intracardiac perfusion of a MGA solution for 10 min. The developing parts of the mandibular incisors were dissected out, and slices were immersed in the fixative overnight. After several rinses in the buffer alone, samples were dehydrated and embedded in Epon. Semi-thin sections were processed for radioautography. Using quantitative methods, we scored silver grain density automatically by compartment and for each period of time (for technical details, see Goldberg et al., 1999). This was carried out in odontoblast cell bodies, predentin, dentin, forming enamel, and ameloblasts. Analysis of variance (ANOVA) and statistical significance of the differences between two mean values, assessed by Fischer's PLSD test, Scheffé's F test, and Dunnett's t test, were calculated by Statview (Statview II, Abacus Concepts, Calabasas, CA, USA). After 30 min, in all the compartments of the sections, incorporation of [3H] choline-labeled precursor was below or equal to the background labeling, except in dentin, where labeling was significantly above background. The same occurred 1 hr after the precursor injection. Dentin labeling was even increased in that period of time. After 2 hrs, labeling began to be significantly above background in odontoblasts, predentin, developing enamel, and secretory ameloblasts. Dentin was densely labeled. After 4 hrs, labeling was still increasing in odontoblasts and ameloblasts, reaching maxima at 24 hrs. At 48 hrs, silver grains were redistributed and appeared to be concentrated in the part of dentin corresponding to what we have termed metadentin (Goldberg and Septier 1996). Labeling remained rather high in enamel and dentin at 4 days, although a slight decrease was apparent. The main finding of this investigation was that dentin was labeled before odontoblasts, the cells producing this tissue. We had expected to find an eventual incorporation into odontoblasts at 30 min-1 hr, followed by a subsequent secretion or release of phospholipids in predentin. This was not the case. The fact that labeling appeared in dentin prior to any visible uptake into odontoblast and predentin was totally unexpected. Our working hypothesis is that isotopic exchanges occur between the membranes of blood cells, which incorporate and synthesize labeled phospholipids, and lipoproteins (very low, intermediate, and high-density lipoproteins). These lipoproteins may be transported by a lipid carrier and rapidly penetrate the dentin. The time needed for these events to occur is within the limits of 30 min, and therefore fits in well with our observation. Odontoblast incorporation of the precursor and subsequent secretion would exceed this period of time. It is actually known that labeled albumin, a serum protein, passes between the distal junctional complex located between odontoblasts, and diffuses throughout predentin. Subsequently, albumin is incorporated into dentin (Kinoshita, 1979). In this context, albumin is one of the well-recognized lipid-binding proteins, or lipid carriers (Veerkamp and Maatman, 1995). The presence of labeled (3H) choline-containing phospholipids in predentin and dentin as early as 30 min-1 hr supports diffusion rather than incorporation and secretion by odontoblasts. This hypothesis sheds lights on the possible origin of phospholipids implicated in dentin biomineralization. This cannot be applied to secretory ameloblasts, which are connected at their distal parts by several strands of tight junctions and which act as a permeability barrier between cells of the enamel organ and the forming enamel. In this regard, it has been shown by Wortsman and Traycoff (1980) that a tightly bound albumin-calcium fraction is present in both human and rat sera. This calcium is complexed to albumin via fatty acids, and this circulating tightly bound calcium fraction or albumin-fatty acid complex may be taken up by living cells. For the predentin/dentin system, we would like to suggest that the albumin-fatty acid complex will not be immediately incorporated into cells but passes between odontoblasts, crosses the distal permeable junctional complex, and diffuses into dentin prior to any cellular uptake and phospholipid synthesis. We cannot preclude that the early accumulation of label in dentin may be due to physico-chemical interactions among the precursor, collagen, and the mineral and therefore does not reflect a controlled biological event. However, the redistribution of silver grains in the metadentin area that occurred between 2 and 4 hrs after injection suggests that this event cannot be a simple binding of the phospholipid to the mineral, but rather is an active process.
Whatever the origin of these phospholipids may be, there is still some conflict between histochemical data concerning the potential role of phospholipids in predentin, in calcium transport toward the mineralizing metadentin, and the results obtained by radioautography which suggest direct interaction between lipids of serum origin and the lipid fraction in dentin via albumin. The two groups of phospholipids might be totally different in composition and have complementary functions. Further investigations are needed to clarify this issue.
(4) Enamel lipids
In enamel, histochemical investigations failed to visualize specifically MGA- or iodoplatinate-stained structures in the extracellular matrix, except for a few crystallite-like or “crystal ghost” structures located in the inner forming enamel, near the dentino-enamel junction. However, by the histochemical techniques reported above, and in the context of a pharmacological lipidosis induced by the use of chloroquine, membrane remnants were visualized in spaces left more or less empty by Tomes' processes. Usually, these spaces are gradually filled by rod material. These membrane remnants, also visualized with the filipin/cholesterol method, may have biological role(s). However, up to now, we do not know why such membrane debris is released by secretory ameloblasts and what role they might have. They are certainly not involved directly in biomineralization, because we have no evidence of such nucleating functions, even by routine electron microscopy. Apparently, there are no matrix metalloproteases associated with these remnants, at least those (MMP-2, -3, -9) which have been studied by our group, as well as enamelysin (MMP-20), which seems to be specific for enamel protein degradation. Many other enzymes, such as alkaline phosphatase, are candidates for potential roles in mineralization processes. Immunohistochemical observations revealed that MMP-2, -3, and -9 are actually located in the forming enamel matrix, but do not seem to be associated with membrane remnants. However, in some locations, there is evidence which supports a possible relationship between IP-stained disintegrating membrane remnants and enamel initial mineralization. Membrane debris may be degraded by phospholipases, a phenomenon which might contribute to the potential release of some phospholipids from these membranes. This material could be transferred to the phospholipid-containing crystal ghosts, structures which are detected in the forming enamel, exactly at places where enamel mineralization is initiated. This assumption still needs to be reinforced and awaits stronger arguments than morphological data alone.
(V) Pharmacological and Genetically Induced Pathology
To evaluate the potential role of lipids in the mineralization of dental tissues, we used several experimental approaches. Pharmacological approaches provide some interesting data. Rats injected with a single injection of suramin develop a mucopolysaccharidosis combined with a lipidosis. Suramin, a polysulfonated naphtylurea with polyanionic character, used as an anti-trypanosomal drug, is taken into cells by endocytosis, accumulates in lysosomes, and inhibits phagosome-lysosome fusion (Hart and Young, 1975). Following intravenous administration, suramin induces an accumulation of GAGs and sphingolipids (Constantopoulos et al., 1980) due to inhibition of many lysosomal enzymes. The function of various ion pumps is inhibited (Moriyama and Nelson, 1988). Ultrastructurally, secretory ameloblasts and odontoblasts were loaded with lysosomes. In dentin, MGA stained amorphous electron-dense patches that were not seen in control sections (Figs. 9a, 9b). These patches are eliminated by chloroform/methanol treatment and are therefore identified as lipids. Although a clear relationship was found between intracellular events—namely, that suramin induced accumulation of lysosome-like structures, concomitantly with an alteration in the histochemical distribution of lipids in dentin—there is no clear-cut evidence on the effects of suramin, and consequently of some lipids, on biomineralization (Gritli et al., 1993). This may be due to the fact that suramin induced not only mucopolysaccharidosis and lipidosis, but also interfered with growth factors such as transforming growth factor-β, epidermal growth factor, and fibroblast growth factor-2, by blocking binding to their respective cell-surface receptors (Coffey et al., 1987).
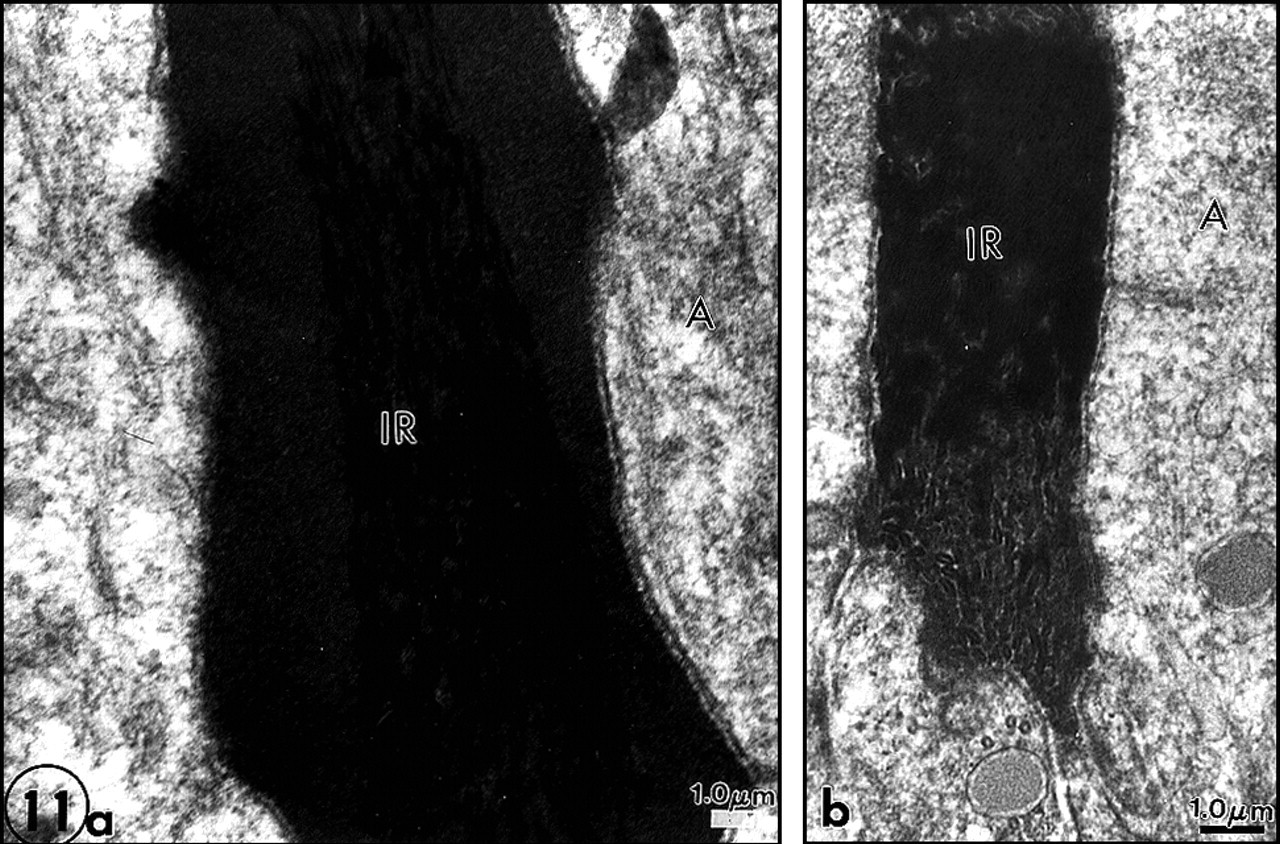
A more simple pharmacological approach was undertaken to assess more specific effects of lipidosis. Chloroquine given for 6 wks in the drinking water induces lysosomal accumulation of sphingolipids. Again, odontoblasts, pulp cells, and cells of the enamel organ, but not secretory ameloblasts, were loaded with lysosomes containing myelin-like structures. Two striking effects were observed with respect to dentinogenesis and amelogenesis:
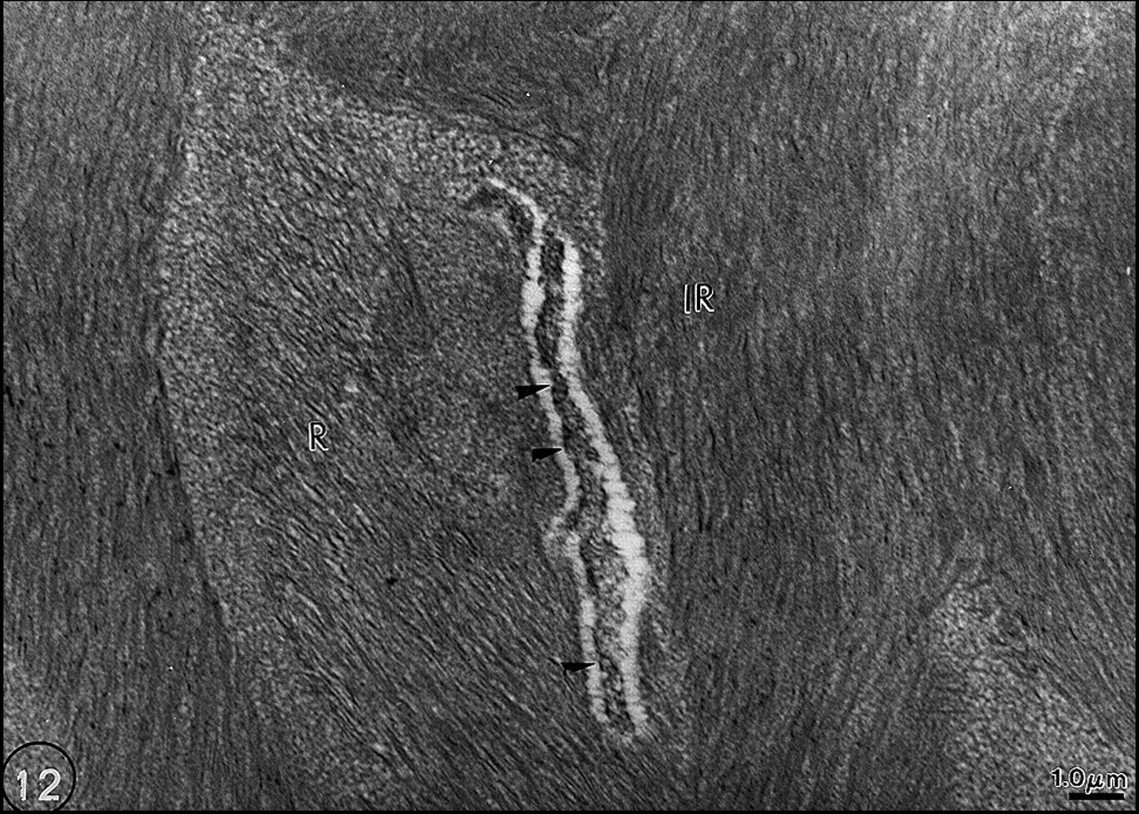
First, the metadentin zone was wider and more dense (Figs. 10a, 10b). This evidences that lipid alterations influence the area where the mineralization process is initiated. If metadentin is widened, this would suggest that the process is delayed. Second, many defects of enamel formation were seen in the interrod domains, at the growing site where apposition of newly formed enamel occurred (Figs. 11a, 11b). Either the central part of interrod was normal and covered by amorphous material, or aberrations in the normal structure were seen, with discontinuous sequences of normal and altered enamel formation. In addition, accumulation of membrane remnants occurred inside rods (Goldberg et al., 1998) (Fig. 12).
Since genetic diseases may provide some insights into the effects of lipid alterations on the formation and mineralization of dental tissues, a genetic pathology, Krabbe disease, was studied (Bloch-Zupan et al., 1994). SEM analysis revealed defects in enamel and dentin, including enamel hypoplasia, inclusions of proteins forming bundles at right angles with the DEJ, and inclusions of amorphous material in the mantle dentin of the deciduous teeth. With TEM, pulp cell alterations were detected. In addition, the odontoclast-like cells were extremely prominent, which probably contributed to the precocious shedding of the teeth. The complexity of the disease added to the confusion, and clearly there is a need for a more simple model.
In the Fabry's mice engineered by Kulkarni and co-workers (Functional Genomic Unit, NIDCR, NIH, Bethesda, MD, USA) (Oshima et al., 1995), phospholipid (namely, sphingolipid) alteration induces several structural defects. In enamel, tiny holes which may contain Tomes' process remnants were seen at the junction between rods and interrod, giving the prismatic enamel a riddled appearance (Dunglas et al., 2001). In dentin, as was the case with chloroquine treatment, metadentin was substantially widened. Disturbances of the collagen network are still under investigation.
Although each model has its own specificity and complexity, and may exhibit a convergence of plural effects, we conclude that phospholipids play a role in amelogenesis and dentinogenesis.
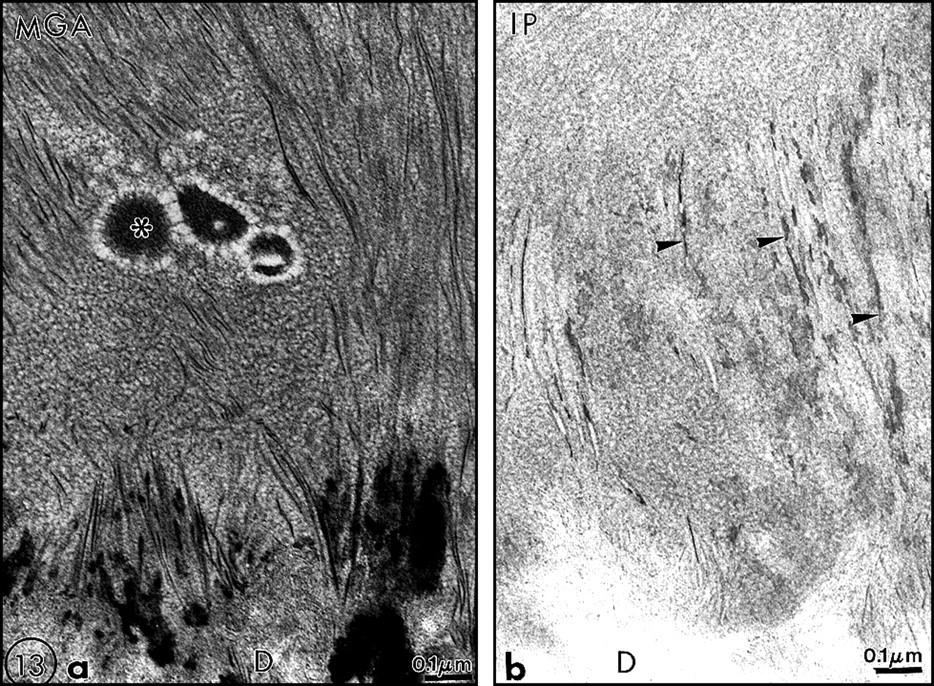
At the moment, we have provided evidence which indicates that lipids, namely, phospholipids, play a role in the formation and mineralization of dental tissues. Experimental pharmacological approaches and studies on genetic disease, and now the use of transgenic mice, support this assumption. Therefore, phospholipids have to be considered to be as important as any molecule or group of molecules belonging to the dentin non–collagenous proteins. This is probably also true for enamel extracellular matrix components, although there is still a debate on the cellular or extracellular nature of enamel lipids (Figs. 13a, 13b). There is still a need to investigate further the nature of the molecules implicated in these processes. It is also important to study the regulation of events by enzymes involved in the synthesis of lipids, such as the fatty acid synthetase complex and many others, and those involved in their degradations, such as phospholipases and others which might be less specific. The physico-chemical interactions among proteoglycans, collagen, lipids, LDL or HDL, and/or other yet-to-be-identified molecules in the predentin/dentin compartments are worthy of further research, as are also the interactions between enamel proteins and phospholipids.
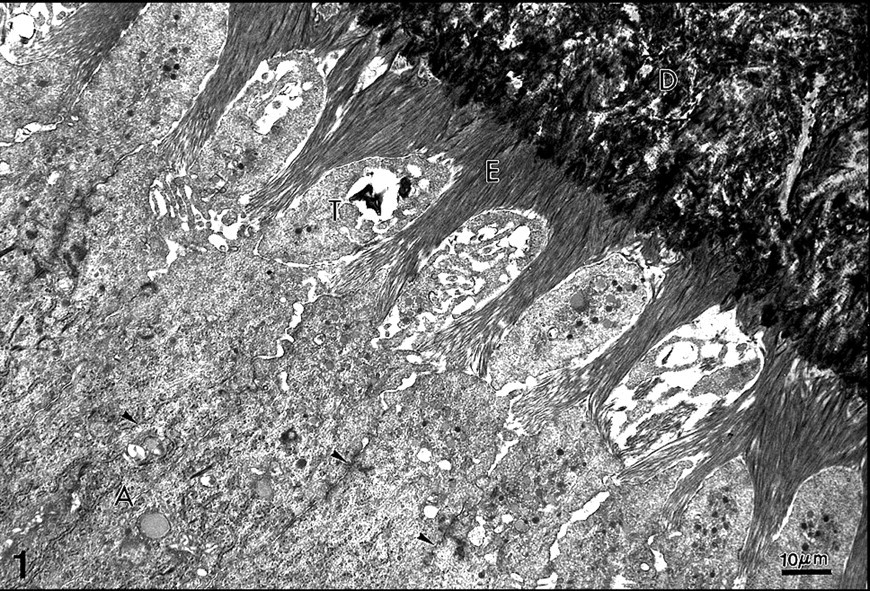
Rat incisor. Initial stage of enamel (E) formation. Terminal junctional complexes (arrowheads) are present between the cell bodies of secretory ameloblasts
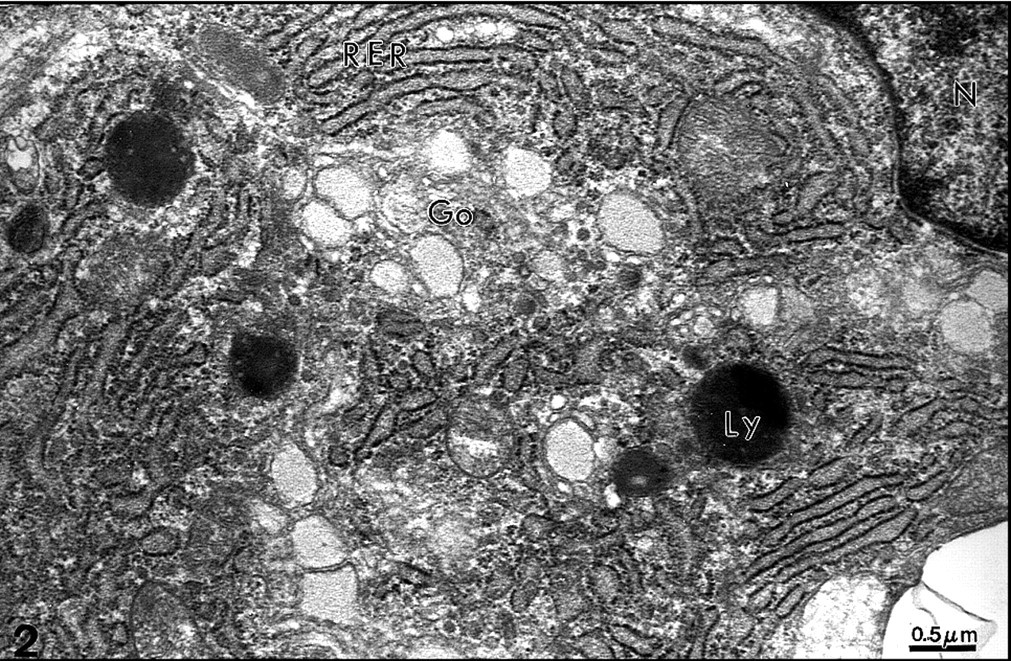
Rat incisor. Odontoblast cell body. Near the nucleus, the rough endoplasmic reticulum (RER), Golgi apparatus (Go), and lysosomes (Ly) are involved in synthesis and secretion of extracellular matrix components. Uranyl acetate, lead citrate. x27,000.
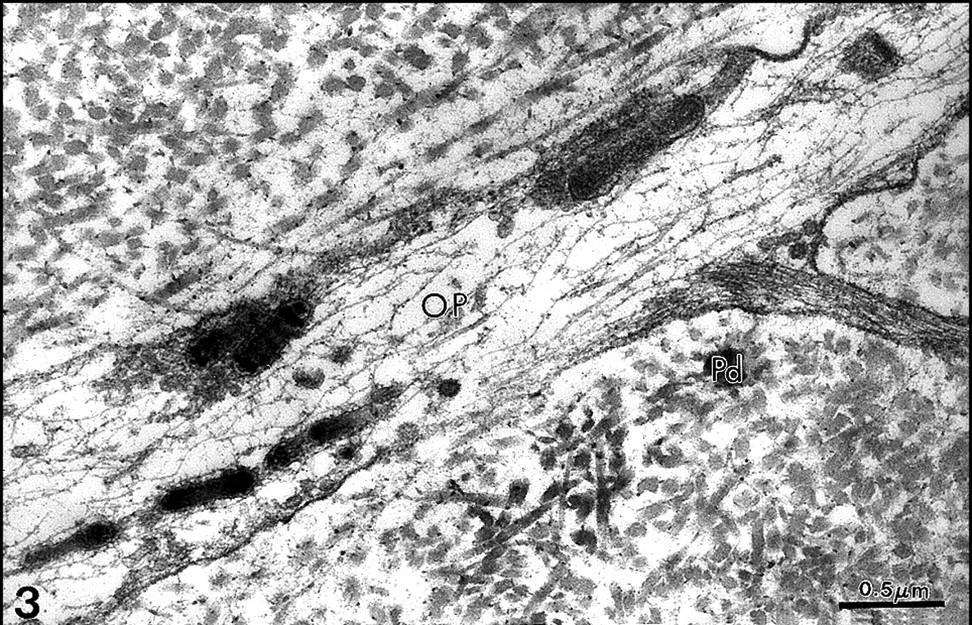
Odontoblast process (OP) and its lateral branch. Electron-dense vesicles contain a material with an appearance of periodicity. A dense cytoskeleton, formed by intermediate and micro-filaments, is seen in the process. Predentin (Pd) surrounds the process. Uranyl acetate, lead citrate. x42,000.

Lanthanum nitrate passes the terminal junctional complex located between the distal parts of odontoblast cell bodies, stains some matrix components in the predentin (Pd), and diffuses in metadentin (M). By contrast, dentin (D) is poorly stained. x54,000.

Filipin-cholesterol complexes are seen along odontoblast cell processes in predentin


Control
Control
Chloroquine induces defects during interrod enamel formation: lateral defects along a central core apparently normal
MGA staining. Chloroquine-treated rat. Remnants of Tomes' processes are visualized (arrowheads) far away from the cell bodies inside rods (R) in the forming enamel. Interrod enamel (IR). x13,500.
Footnotes
Acknowledgements
We thank Prof. Steinar Risnes, Oslo, Norway, for his advice and helpful discussions during the preparation of the manuscript. These investigations were supported by grants from the Ministry of Education and the University René Descartes, Paris V (Axes d'Excellence)..
