Abstract
Background:
Recurrent vulvovaginal candidiasis (RVVC) is defined as 3 or more acute symptomatic episodes in a year and affects nearly 138 million women globally. Oteseconazole selectively inhibits fungal cytochrome P51 and has potent activity against Candida albicans and resistant species, including C. glabrata and fluconazole-resistant strains. Over 90% of women randomized to receive oteseconazole in two recently completed global Phase 3 clinical trials (VIOLET trials) did not experience a RVVC episode during these 48-week clinical studies. This observational study was designed to extend these initial findings.
Patients and Methods:
A total of 70 eligible women previously randomized to receive oteseconazole were enrolled into an extension study and were followed an additional 48 weeks and monitored for recurrent infections. No additional investigational treatment was administered during the observational period.
Results:
Of the 70 eligible study participants who received oteseconazole, 85% completed the extension study, and 1/70 (1.4%) experienced a culture-verified recurrent infection during the observational period, weeks 48 to 96. Of the participants initially randomized to receive placebo, 76% completed the study and 3/17 (17.6%) experienced a recurrent infection during the extension study. The proportion of participants who prematurely discontinued the study was lower in the oteseconazole-treated group (16%) compared with the placebo group (29%).
Conclusions:
During the 48-week extension study, 1.4% of previously randomized subjects experienced a culture-positive recurrence of symptomatic vulvovaginal candidiasis, demonstrating a continued long-term protective effect of oteseconazole.
Keywords
Introduction
Recurrent vulvovaginal candidiasis (RVVC) is defined as 3 or more acute symptomatic vulvovaginal yeast infections within a 12-month period, with approximately 6–9% of women worldwide experiencing RVVC, presenting with a combination of vaginal signs (erythema, edema, and/or excoriation) and symptoms (itching, burning, and/or irritation).1–6 Approximately 6 million women in the United States and 138 million women globally develop RVVC, which is projected to increase to 158 million by 2030. 2 RVVC has a significant impact on the mental, emotional, and physical health of women who are experiencing frequent vulvovaginal candidiasis (VVC) episodes as well as an economic burden. 6 While the majority of all VVC infections are caused by Candida albicans species (approximately 90%) and are typically well managed with fluconazole, considered the standard of care for the past 30 years, reports indicate a significant increase in the prevalence of less common non-albicans species (NAC), such as C. glabrata, C. krusei, C. parapsilosis, and C. tropicalis, observed in 10–20% of women with RVVC. Conventional antimycotic therapies are not as effective against these NAC.7–9
While not approved by the U.S. Food and Drug Administration for the treatment of RVVC, maintenance oral fluconazole is recommended by the Infectious Disease Society of America, the Centers for Disease Control and Prevention, and the American College of Obstetricians and Gynecologist.10,11 These clinical guidelines suggest an induction antifungal regimen with either a topical azole agent or oral fluconazole at doses ranging from 100 to 200 mg for 7–14 days, followed by weekly fluconazole (100–200 mg) maintenance therapy for 6 months.12,13 In a controlled clinical study, approximately 10% of study participants experienced clinical VVC recurrence while receiving fluconazole and approximately 50–60% of patients experience a recurrent episode within 6 months of stopping the 6-month weekly fluconazole regimen. 9 However, fluconazole resistance is increasingly reported for C. albicans, particularly in women receiving maintenance fluconazole.14,15 While generally considered relatively safe and well tolerated, fluconazole use carries warnings of liver toxicity, allergic skin reactions, potential for fetal harm, drug-drug interactions, and QT prolongation.12,15
Oteseconazole is a novel, highly selective inhibitor of the fungal enzyme cytochrome p450 14 α-sterol demethylase (CYP51), required for fungal cell wall integrity. Oteseconazole was rationally designed to have a lower affinity for heme-iron and a greater affinity for the fungal CYP51 polypeptide than current azole drugs. As a result, oteseconazole more potently inhibits fungal CYP51 selectivity. 16 Limited binding to human cytochromes, typically responsible for drug metabolism and drug-drug interactions, together with a high human plasma binding profile of >99.5%, results in a compound with a median terminal half-life of approximately 138 days. These characteristics allow for in vivo retention of oteseconazole in the plasma and in target tissues that are relevant to RVVC and other fungal mucosal infections. These properties should translate to greater efficacy with an improved safety and tolerability profile.
Two Phase 3 multicenter, multinational, double-blind, randomized, placebo-controlled clinical trials (NCT03562156, NCT03561701), referred to as the VIOLET trials, were conducted to evaluate the efficacy and safety of oteseconazole in the treatment of participants with RVVC. 17 Both trials consisted of two phases: an open-label induction phase and a maintenance phase. Participants with a history of RVVC (experiencing 3 or more acute episodes in the previous 12 months) and presenting with an acute episode received three sequential doses (every 72 hours) of fluconazole 150 mg.
To participate in the VIOLET trials, subjects were required to meet the following key inclusion criteria included: (1) Generally healthy, postmenarchal, nonpregnant females aged ≥12 years of age; (2) with a history of RVVC defined as ≥3 patient-reported and/or laboratory-confirmed episodes of acute VVC in the past 12 months, including the episode confirmed at screening, with ≥1 episode prior to screening documented by positive culture, PCR, Affirm test, KOH test, Gram stain, or a documented Pap test in the prior 12 months revealing yeast; and (3) a positive KOH wet mount preparation or Gram stain from a vaginal smear revealing filamentous hyphae/pseudohyphae and/or budding yeast cells, and an acute VVC episode at screening, defined as a total score of ≥3 for vulvovaginal signs and symptoms. The total score was established following the assessment of vulvovaginal signs (erythema, edema, and excoriation) and symptoms (itching, burning, and irritation) using the following scale: 0, none (complete absence of any signs or symptoms); 1, mild (slight); 2, moderate (present); and 3, severe (marked, intense). Individual scores were combined for a maximum score of 18.
In addition, subjects were excluded from the study if any of the following criteria were established: (1) Vaginal infection other than acute VVC; (2) use of systemic antifungal therapy within 7 days prior to screening; (3) Use of immunosuppressive medication or radiation therapy within 3 months prior to screening; (4) clinically significant major organ disease or current clinically significant infection or conditions (apart from acute VVC); (5) currently pregnant, lactating, or planning to become pregnant; (6) Absence of contraception for nonpregnancy women of childbearing potential; and (7) history of intolerance or hypersensitivity to azole antifungal drugs.
Participants returned 14 days after the first dose of fluconazole, and if the acute VVC episode was resolved, they transitioned to the maintenance phase and were randomized to receive either 150 mg of oteseconazole or placebo for 7 days, followed by 11 weekly doses of 150 mg oteseconazole or placebo. Participants were then monitored for an additional 36 weeks, for a total of 48 weeks. In these clinical trials oteseconazole was shown to be statistically superior (p < 0.001) to placebo in the prevention of culture-verified acute VVC infections (defined as a positive culture for Candida species and a clinical signs and symptoms score of ≥3) through 48 weeks. 17 Reduction of culture-confirmed episodes was evident with greater than 90% efficacy at the 48-week follow-up period. Treatment-emergent adverse events were similar between the oteseconazole and placebo groups. This extension study was undertaken to begin to answer the temporal question of disease prevention with oteseconazole.
Methods
Subjects
Eighty-seven (87) U.S.-based subjects who had completed one of two randomized, placebo-controlled Phase 3 trials (VIOLET trials) and had not experienced a recurrent VVC episode during the original 48-week study, and who remained blinded to their original treatment assignment cohort, were enrolled in the long-term extension (LTE) study. Subjects were followed for an additional 48 weeks, for a total of 96 weeks. This was an observational study, and no additional investigation agent was provided during the LTE. A summary of the overall study design is provided in Figure 1.
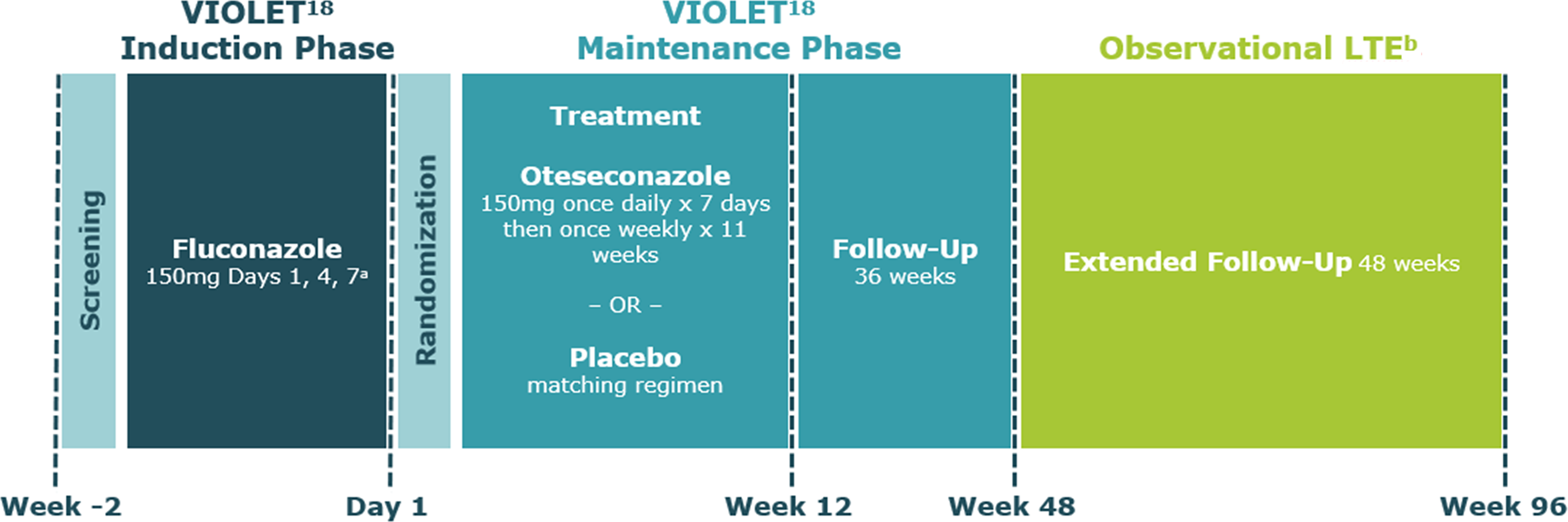
Clinical Study Design. Design and duration of the clinical study are schematically depicted, including the VIOLET phase 3 trials which comprised of the first 48 weeks (included randomization, treatment and the original 36 week follow-up, followed be the extension study for an additional 48 weeks for eligible U.S. participants. Abbreviations: LTE, Long term extension; VVC, vulvovaginal candidiasis. aBased on Infectious Disease Society Guidelines, bU.S. clinical study participants with no VVC recurrence during maintenance phase. 18Sobel JD, Donders G, Degenhardt et al. Efficacy and Safety of Oteseconazole in Recurrent Vulvovaginal Candidiasis. N End J Med Evid. 2022;1 (8).
Seventy (70) subjects had originally been randomized to receive oteseconazole, and 17 subjects had been randomized to receive placebo. While this study was not designed to be a comparative study, due to the nature of participant blinding, study participants who had originally been randomized to receive placebo and remained VVC attack free at week 48 were also enrolled and followed. No statistical comparisons were made given the small sample size.
Methods
Participants were contacted approximately every 6 weeks to monitor for VVC recurrence. Participants experiencing a suspected recurrent VVC episode were required to return to the investigational site for evaluation. Culture-confirmed recurrent VVC episodes were recorded during the 48-week LTE study. Exclusion criteria included: topical products applied to the vulva or vagina (e.g., antibiotic, antifungal, antitrichomonal, corticosteroids, or anti-inflammatory agents); oral antibacterial, antiviral, or antitrichomonal agents for the treatment of bacterial vaginosis, trichomonas, or other concomitant (urogenital) infection; and vaginal douches, boric acid, or benzalkonium chloride disinfectant.
The primary efficacy outcome measure was to determine the proportion of participants who experienced one or more culture-verified acute VVC episodes during the observational extension study (week 48 through week 96). Pharmacokinetic samples were only collected if a study participant was experiencing a potential recurrent VVC episode during the LTE and came into the clinic for testing.
Safety assessments were not planned in this study. For any participant that became pregnant, the outcome of their pregnancy (birth or termination) was documented in the subject’s medical record.
The LTE study was conducted in accordance with applicable International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use Guidelines, Good Clinical Practice, and the guiding principles of the current version of the Declaration of Helsinki. The study was sponsored by Mycovia Pharmaceuticals Inc. (Durham, NC) and approved by a central institutional review board (WIRB-Copernicus Group). Written informed consent was obtained from all participants prior to study inclusion.
Results
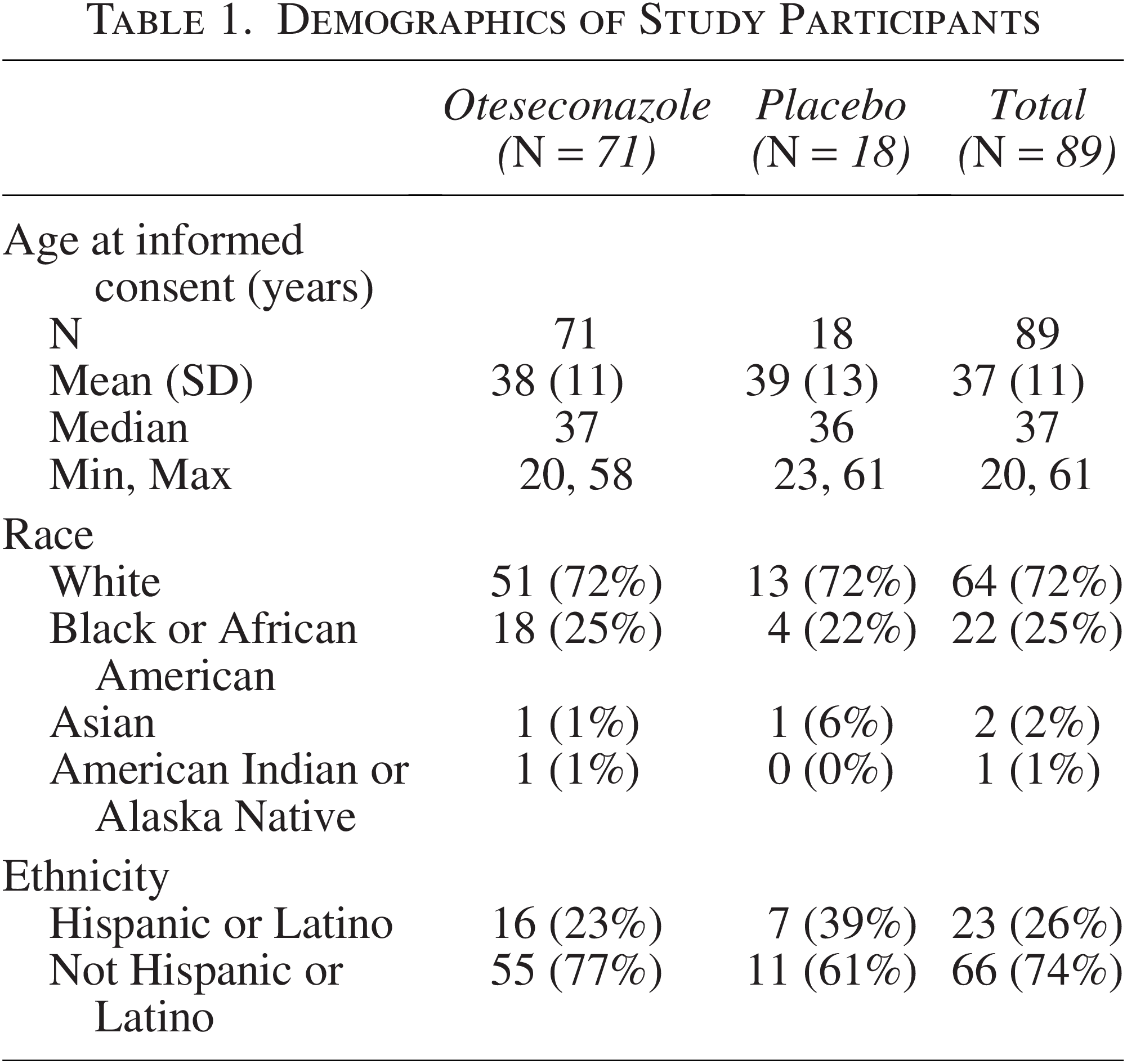
There were no notable differences in demographics and baseline characteristics across the treatment groups (Table 1).
Demographics of Study Participants
The percentage of participants who completed the additional 48-week observational study was high in both the oteseconazole group (60/70 participants [85%]) and the placebo group (13/17 participants [76%]). A total of 16 (18%) subjects prematurely discontinued study participation; one each from the oteseconazole- and placebo-treated cohorts were deemed ineligible. The proportion of participants who prematurely discontinued the study was lower in the oteseconazole group (11 participants [16%]) compared with the placebo group (5 participants [29%]). Reasons participants prematurely discontinued included: withdrawal of consent, met the primary endpoint of confirmed RVVC, unconfirmed RVVC site closure, and for use of an excluded medication. Study enrollment and discontinuation are shown schematically in Figure 2. Adverse events were not collected during the study. One participant (previously randomized to receive oteseconazole) became pregnant and delivered a healthy baby approximately 2.5 months following enrollment.
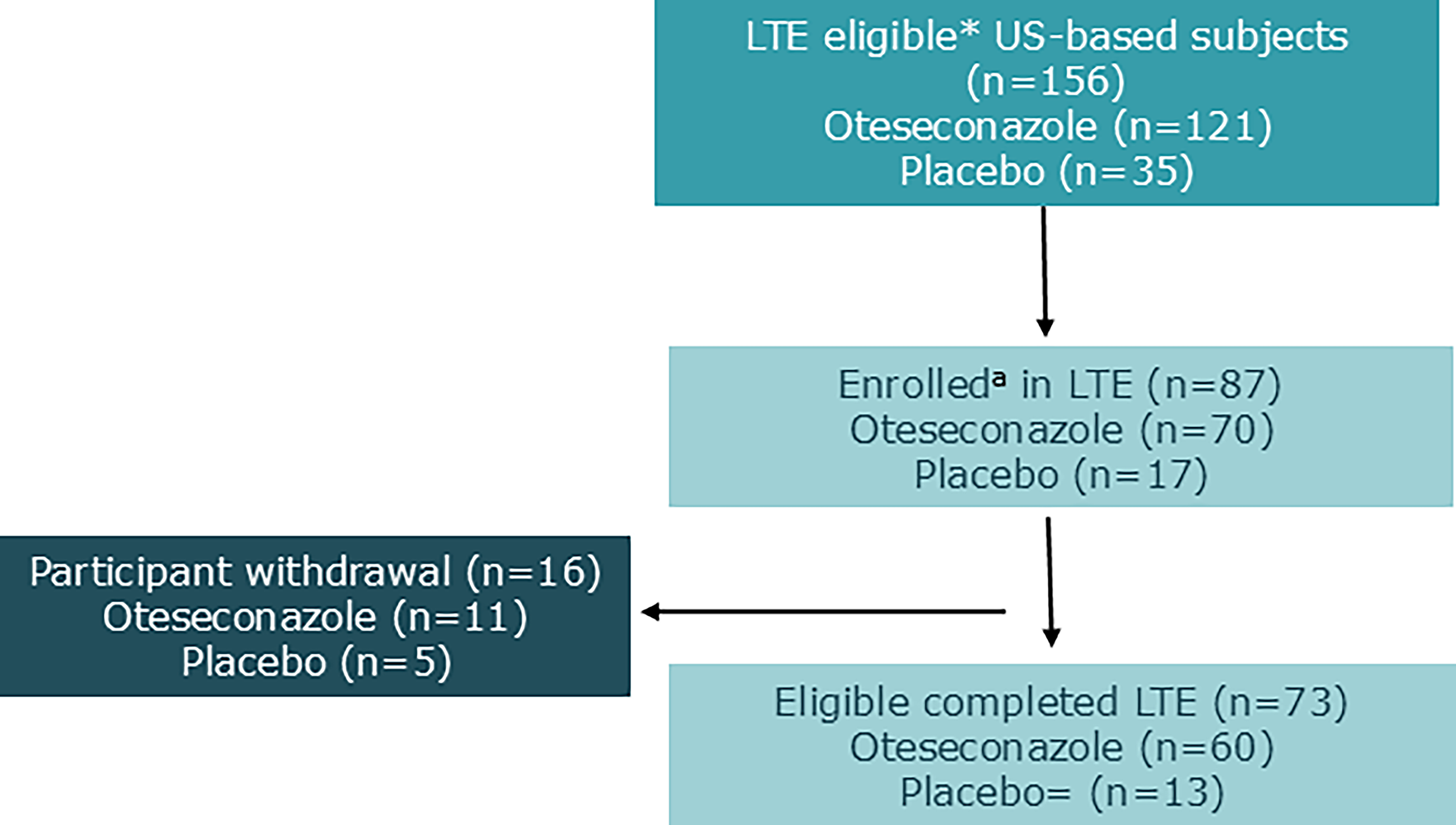
Study Enrollment and Discontinuation. *eligibility corresponds to U.S.-based participating subjects who had not experienced a recurrent episode whereas participating in the Phase 3 RWC studies. Two participants (1 in the oteseconazole group and 1 in the placebo group) were enrolled in the LTE but were not eligible and not induded in the final analysis. LTE-long-term trial extension.
Eighteen participants were seen in clinic for a self-suspected recurrent VVC episode during the extension study (weeks 48–96); 6 from the placebo group and 12 participants from the oteseconazole group. Two subjects in the oteseconazole-treated group and one in the placebo group returned on more than one occasion. A total of 4 subjects who returned to the clinical (1 from the otesaconazole-treated group and 3 from the placebo treated group) met the criteria for a recurrent episode. Of the 70 eligible extension study participants previously randomized to receive oteseconazole, only 1 (1.4%) experienced a recurrent episode between weeks 48 and 96, compared to 3 of 17 participants who originally received placebo. Figure 3 summarizes the disease recurrence rates for participants randomized to receive either oteseconazole or placebo and participated in the VIOLET and LTE study.
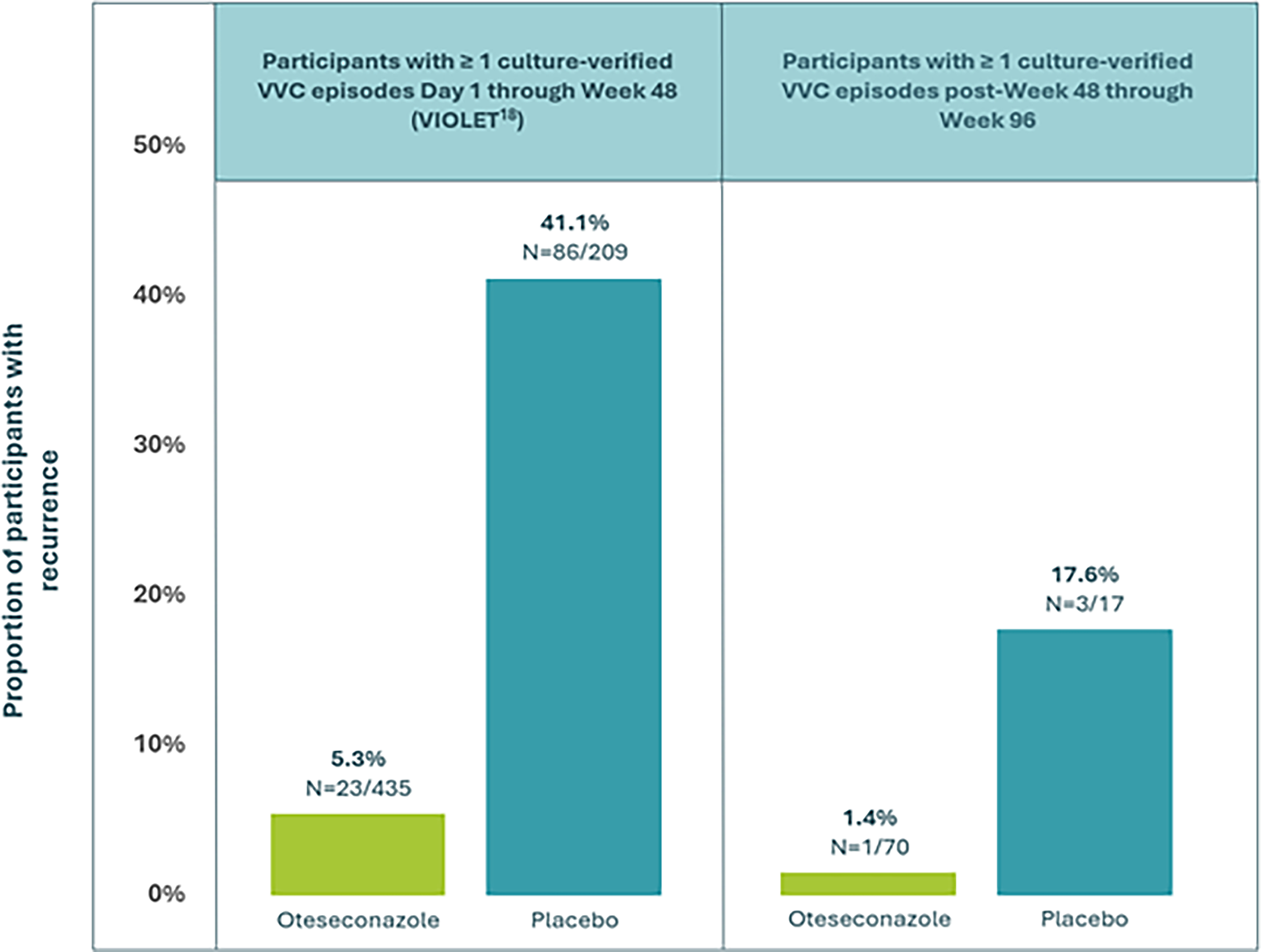
Primary Efficacy Endpoint for VIOLET Studies and the Extension Study. VVC, vulvovaginal candidiasis.
During these visits, plasma samples were also collected to determine oteseconazole concentration, with the goal to correlate drug concentrations to efficacy outcomes. Six samples were from participants randomized to receive placebo, and hence no further testing was performed. Fourteen samples were collected from participants who had been randomized to receive oteseconazole; 3 samples had drug concentrations of 365 ng/mL, 1000 ng/mL, and 1140 ng/mL, and 3 were below the level of assay quantitation (including the subject with a culture-verified recurrent VVC infection). Of note, based on in vitro minimum inhibitory concentration testing data, 1,000 ng/mL is at the lower range of the effective drug concentration deemed necessary to exert an antifungal effect. Concentrations were not determined for 5 samples due to site collection error, and 3 samples were omitted from testing.
Discussion
Current treatment guidelines from the Infections Disease Society of America, the Centers of Disease Control and Prevention, and the American College of Obstetricians and Gynecologists and professional societies worldwide recommend the off-label use of fluconazole as an induction therapy followed by 6-month maintenance fluconazole to treat RVVC. Although efficacious in preventing recurrent infections during weekly maintenance therapy for most patients, upon cessation of this regimen, recurrent infections are common. Moreover, reports of fluconazole resistance in C. albicans continue to increase and is not limited to women receiving maintenance fluconazole. 15 Frequent recurrence of VVC often requires long-term use of weekly prophylactic fluconazole administration. 15 Until recently, health care providers and patients suffering with RVVC had limited treatment options available. Oteseconazole was approved in the United States by the FDA in 2022 to treat women with a history of RVVC who are not of reproductive potential. This contraindication is based on ocular abnormalities reported in the offspring of some rats at doses 3.5 times the recommended human dose; these findings and additional prescribing information are further highlighted in the published prescribing information. 18 In two Phase 3 clinical studies, oral oteseconazole had previously been shown to be effective in preventing recurrent VVC episodes for nearly a year in the global VIOLET clinical trials. In these clinical studies the recorded safety outcomes were similar between subjects randomized to receive either oteseconazole or placebo. Clinical data generated in the study described herein suggests that oteseconazole can prevent disease recurrence well beyond the initial 48-week study endpoint, as only one participant of 70 enrolled in the LTE study experienced a recurrent episode during the 48-week or second-year observational period. Improved efficacy outcomes together with a significantly shorter, well-defined dosing regimen compared to the current standard-of-care should benefit patients in need of a new treatment option. Due to practical considerations, the study was limited to enrolling U.S.-based participants only. Descriptive statistics were employed due to the small sample size. An additional study limitation was the less extensive subject monitoring throughout study, when compared to the original VIOLET studies. A larger randomized controlled study is warranted to confirm these encouraging findings.
Conclusions
This observational extension study demonstrated that over 98% of recurrent VVC study participants receiving 12 weeks of oteseconazole once weekly following induction remained infection-free for an additional 48 weeks. These results are superior to previous prophylaxis studies and suggest that oteseconazole can provide improved clinical outcomes.
Footnotes
Acknowledgments
The authors acknowledge participating principal investigators and clinical sites of the VIOLET Long-Term Extension Study including: Mark D, Akin MD, Austin Area OBGYN, Austin, TX; Samual Alexander MD, Southern Clinical Research Associates, LLC, Metairie, LA; Steven Chavoustie MD, Segal Trials, Miami, FL; Harold Green MD, Columbus OB/GYN Inc./ClinOhio Research, Columbus, OH, Mark Jacobs MD, TMC Life Research, Inc., Houston, TX; Robert E Littleton MD, Unified Women’s Clinical Research, Raleigh, NC, Rex G. Mabey, Jr., MD., Rex Garn Mabey, Jr., M.D. Chartered, Las Vegas, NV; Paul Nyirjesy MD, Department of Obstetrics and Gynecology, Sidney Kimmel Medical College at Thomas Jefferson University, Philadelphia, PA, Ruth Palma, MD Corvallis Clinic, Corvallis, OR; Lamar Parker MD, Unified Women’s Clinical Research-Lyndhurst Clinical Research, Winston-Salem, NC, Ramana K Puppala MD; NuDirections Clinical Research, Marietta, GA; Alison Rodriguez MD, Clinical Trials Management, LLC, Metairie, LA; Steven Sussman MD, Capital Health Ob-Gyn, Lawrenceville, NJ; Denis G Tarakjian MD; Wake Research Medical Center for Clinical Research, San Diego, CA. The authors also thank the participants and clinical site staff for their contributions to this study.
Author Disclosure Statement
J.D.S. has been a consultant to Mycovia and received speaker’s fees from Mycovia Pharmaceuticals. S.B.C. and S.R.B. are employees of Mycovia Pharmaceuticals. All other authors: no potential conflicts of interest. The authors alone are responsible for the content and writing of the article.
Funding Information
This research was fully funded by Mycovia Pharmaceuticals, Inc. and did not receive any other specific grants from any funding agencies (public, commercial, or not-for-profit sectors).
