Abstract
Introdution:
Hypertension (HTN), metabolic syndrome (MetS), and obesity are common in women with polycystic ovary syndrome (PCOS); these comorbidities may be more common in non-Hispanic Black (NHB) and Hispanic women with PCOS.
Methods:
We examined electronic health records in an academic health care system using International Classification of Diseases, Tenth Revision, diagnostic codes to identify PCOS cases (n = 4,479) and control women (n = 771). We examined associations between PCOS and blood pressure (BP) (elevated BP [Stage 0 HTN], Stage 1, Stage 2 HTN, and overall HTN [Stages 1 and 2]), with obesity (class 1 [body mass index or BMI, 30–<35 kg/m2], class 2 [BMI, 35–<40 kg/m2], and class 3 [BMI, ≥40 kg/m2]), and pre-metabolic syndrome (pre-MetS). Analyses were stratified by obese, BMI ≥30 kg/m2 versus nonobese, BMI <30 kg/m2. Age, race, and ethnicity were included as covariates for all adjusted analyses. Associations are presented as adjusted odds ratio (aOR) and 95% confidence interval (CI) for logistic regression and β coefficient (SEM) for linear regression analyses. PCOS had higher BMI (33 [27–40]) than controls (29 [23–33], p = 0.001), and obese PCOS had 35% greater HTN incidence (aOR 1.35, 95% CI 1.05–1.75).
Results:
Independent of PCOS, nonobese NHB were ∼60% more likely to be hypertensive (aOR 1.57, 95% CI 1.14–2.17). Obese Hispanic women were less likely to have Stage 1 (aOR 0.59, 95% CI 0.46–0.75), whereas nonobese Hispanic women were more likely to have Stage 1 (aOR 1.52, 95% CI 1.04–2.24) HTN. PCOS was six-fold more likely to have class 3 obesity versus controls (aOR 6.29, 95% CI 4.60–8.60). Adjusting for age, race, and ethnicity, PCOS (aOR 2.25, 95% CI 1.77–2.86) and NHB (aOR 1.83, 95% CI 1.53–2.20) had an increased likelihood for pre-MetS.
Conclusions:
Independent of PCOS, nonobese NHB women were more likely to be hypertensive and pre-MetS compared with other races. Our findings indicate roles for race and ethnicity on BP and metabolic regulation in PCOS.
Keywords
Introduction
Polycystic ovary syndrome (PCOS) is a common endocrine and metabolic disorder affecting 6–20% of women of reproductive age.1,2 Recognized as a heterogeneous disorder, PCOS is clinically defined by evidence of hyperandrogenism (clinical or biochemical), ovarian dysfunction (anovulatory cycles), and/or ultrasound evidence of polycystic ovarian morphology. 2 Beyond the burden related to PCOS symptomatology, the affected women are considered at risk for chronic disorders such as type II diabetes mellitus, obesity, and mental health disturbances.
Cardiovascular disease (CVD) is the leading cause of mortality in women in the United States. 3 Known risk factors and predictors of CVD, such as dyslipidemia, metabolic syndrome, endothelial dysfunction, inflammatory milieu, hypertension (HTN), and insulin resistance, are overrepresented in women with PCOS.4–12 However, large-scale studies examining the prevalence of clinical CVD events in women with PCOS compared with age-comparable women without this condition remain lacking. Given the interplay between obesity and hormonal dysfunction as mediators of cardiometabolic disease in women with PCOS, 13 there is a need to better characterize obesity and its independent role for CVD risk in women with PCOS. While women with PCOS have an increased prevalence of obesity in general and central obesity in particular compared with women without PCOS, 14 few studies have examined the prevalence of class 3 obesity in a large population of women with PCOS compared with controls. Despite a clustering of risks for CVD in the PCOS population, and evidence of subclinical vascular pathology being described and such findings correlated with blood pressure (BP) elevations and BP dysregulation, 15 data are limited and even conflicting regarding PCOS being an independent predictor of clinically elevated BP.16,17
In the United States, racial minorities have a greater burden of CVD and HTN compared with the White population.18,19 Based on the American Heart Association (AHA) data, among women 20 years and older, non-Hispanic Black (NHB) women had the greatest prevalence of CVD (58.8%) compared with Hispanic (42.7%) and non-Hispanic White women (NHW, 42.1%). In the same cohort of women, NHB women had the greatest prevalence of high BP (57.6%) compared with Hispanic (40.8%) and NHW (40.5%) women. Notably, the prevalence of CVD in NHB women 20 years and older (58.8%) exceeds that of NHW men (53.6%), emphasizing existing health disparities. 20
Data examining racial and ethnic differences in HTN in women with PCOS are limited. Black women with PCOS have higher body mass index (BMI) and are at an increased risk of CVD and HTN compared with White women with PCOS, despite having more favorable lipid profiles.21–24 Furthermore, the odds for HTN were higher in NHB women with PCOS (odds ratio [OR] 1.32, 1.19–1.48) but lower in women of Hispanic ethnicity (OR 0.68, 0.62–0.75) compared with NHW women with PCOS. 25 More data are needed to elucidate the BP trends in racial and ethnic minorities with PCOS because identifying such disparities is crucial for targeted screening and timely management of HTN toward minimizing cardiovascular morbidity and mortality.
With broadening adoption of electronic health records (EHRs) across health systems, EHR-based queries of clinical data allow a meaningful opportunity to examine clinically relevant queries. 26 Given the scarcity of epidemiological studies providing real-world data (versus lab setting) of CVD and metabolic disease in women with PCOS, we utilized the clinically captured data in our institutional EHR (EPIC) to examine the prevalence of elevated BP indices and meeting criteria for American College of Cardiology (ACC)/AHA defined stages of HTN in women diagnosed with PCOS versus age comparable controls without PCOS. We also examined the prevalence of meeting criteria for pre-metabolic syndrome (pre-MetS) (comprised 2/5 of the International Diabetes Federation [IDF] MetS criteria) in this population. To characterize the severity of obesity in reproductive-age women with PCOS, we examined the association between diagnosis of PCOS and meeting criteria for three World Health Organization (WHO) classes of obesity. We hypothesized that women with PCOS would have a greater incidence of BMI, HTN, and pre-MetS compared with women without PCOS. We further hypothesized greater disease burden in NHB and Hispanic women compared with NHW women with PCOS.
Materials and Methods
The study was approved by the Human Investigation Committee of Yale School of Medicine (HIC Protocol No. 2000031043). No minors were included in this investigation (Table 1). This was a case–control study based on retrospective analysis of cross-sectional data available in the EHR of a tertiary care health care system. The cases (women with a diagnosis of PCOS) and the controls (women diagnosed with tubal and/or male factor infertility and without a PCOS diagnosis) were identified by International Classification of Diseases, Tenth Revision (ICD-10), diagnostic codes for follow-up visits or treatment by reproductive endocrinologists: E28.2 for Polycystic Ovarian Syndrome, N97.1 for Tubal Infertility, and N46.9 for Male Factor Infertility. Criteria for identifying the eligible study population from patients seeking reproductive health care at a tertiary care subspecialty practice within the health care system included: (1) females of age ≥18 years, (2) ICD-10-based diagnoses of cases (PCOS) and controls, (3) inclusion criteria specified only tubal/male factor infertility without any ovulatory dysfunction (menstrual dysfunction) cases, and (4) receiving care between October 2011 and December 2021 and for whom information on BP and BMI was captured at a clinical encounter after the date of diagnosis (of PCOS or tubal/male factor infertility). When a patient had multiple clinical encounters, BP and BMI data were taken from the first clinical encounter after diagnosis (of PCOS or male/tubal factor infertility) to ensure temporality proximate to the time of diagnosis. Patients lacking information on the specified diagnoses, BP, and BMI were excluded. Clinical EHR data were provided by the Yale Joint Data Analytics Team (JDAT).
Overall Population Characteristics
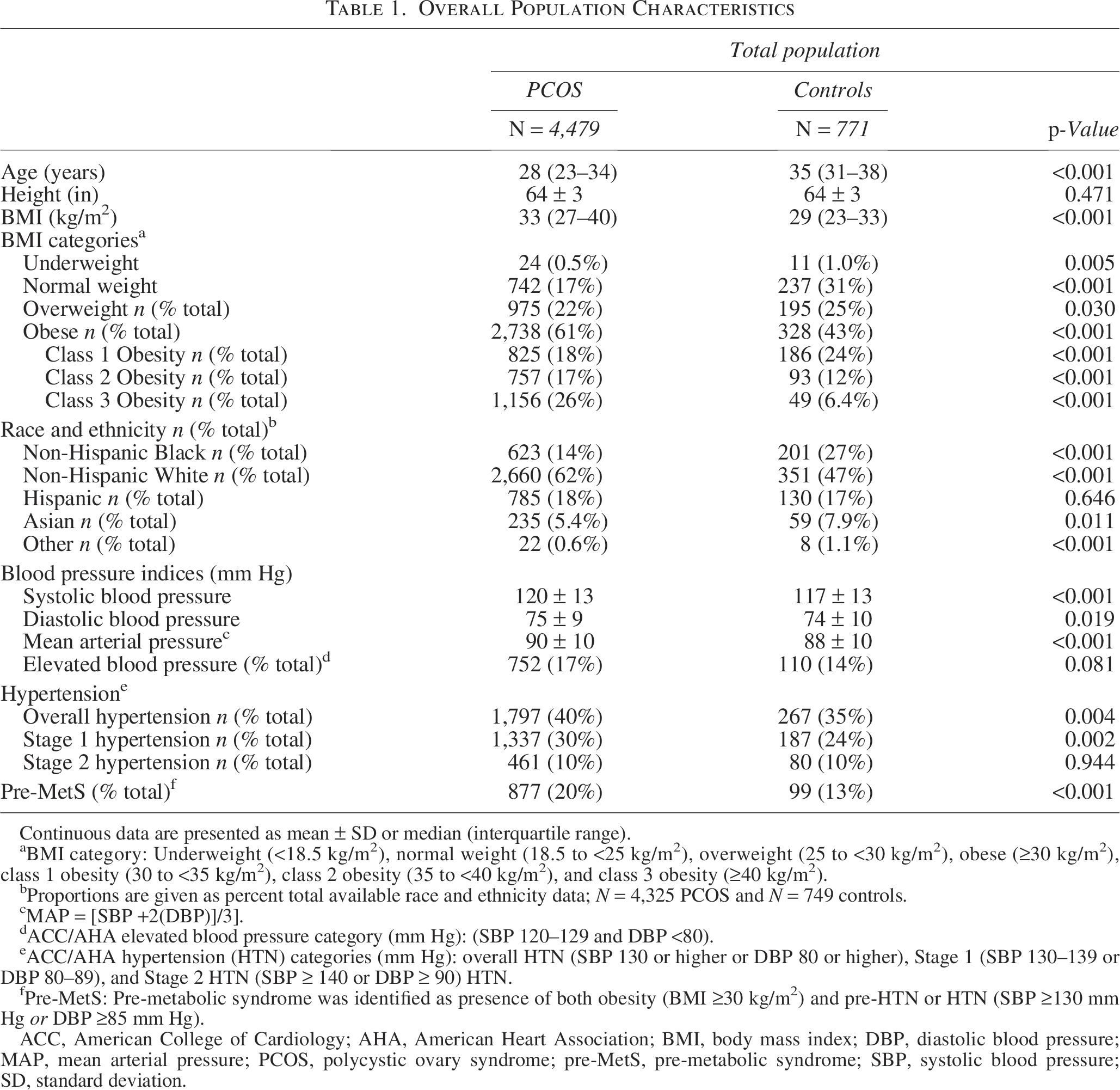
Continuous data are presented as mean ± SD or median (interquartile range).
BMI category: Underweight (<18.5 kg/m2), normal weight (18.5 to <25 kg/m2), overweight (25 to <30 kg/m2), obese (≥30 kg/m2), class 1 obesity (30 to <35 kg/m2), class 2 obesity (35 to <40 kg/m2), and class 3 obesity (≥40 kg/m2).
Proportions are given as percent total available race and ethnicity data; N = 4,325 PCOS and N = 749 controls.
MAP = [SBP +2(DBP)]/3].
ACC/AHA elevated blood pressure category (mm Hg): (SBP 120–129 and DBP <80).
ACC/AHA hypertension (HTN) categories (mm Hg): overall HTN (SBP 130 or higher or DBP 80 or higher), Stage 1 (SBP 130–139 or DBP 80–89), and Stage 2 HTN (SBP ≥ 140 or DBP ≥ 90) HTN.
Pre-MetS: Pre-metabolic syndrome was identified as presence of both obesity (BMI ≥30 kg/m2) and pre-HTN or HTN (SBP ≥130 mm Hg or DBP ≥85 mm Hg).
ACC, American College of Cardiology; AHA, American Heart Association; BMI, body mass index; DBP, diastolic blood pressure; MAP, mean arterial pressure; PCOS, polycystic ovary syndrome; pre-MetS, pre-metabolic syndrome; SBP, systolic blood pressure; SD, standard deviation.
Information collected for the final study population included demographics (patient age, race, and ethnicity as available in the EHR [White, Black, Hispanic, Asian, Other race]), clinical data including height (meters) and weight (kilograms), and BP indices (millimeters of mercury) including systolic BP (SBP) and diastolic BP (DBP). Based on patient self-identity, race and ethnicity were categorized as NHB, NHW, and Hispanic. BMI was derived using an established clinical formula; BMI = (weight, kilograms)/(height, square meters). 27 Mean arterial pressure (MAP) was derived from SBP and DBP measurements using an established clinical formula; MAP = [SBP +2(DBP)]/3]. 28
Based on documented indices for the clinical visits, BP (millimeters of mercury) was categorized based on the ACC/AHA’s current guidelines for elevated BP (Stage 0 HTN) (SBP 120–129 and DBP <80), Stage 1 HTN (SBP 130–139 or DBP 80–89), Stage 2 HTN (SBP ≥ 140 or DBP ≥ 90), 29 and overall HTN (Stages 1 and 2). 30
Pre-MetS was identified as the presence of both obesity (BMI ≥ 30 kg/m2) and pre-HTN or HTN (SBP ≥130 mm Hg or DBP ≥85 mm Hg), which represent two of the five specified criteria by the IDF. 31 Data for the remaining IDF MetS criteria (hypertriglyceridemia, reduced high density lipoprotein, elevated fasting plasma glucose) were unavailable, and BMI ≥30 kg/m2 was used as a proxy for excess waist circumference as per IDF guidelines. 31 Based on BMI, patients were assigned to WHO-defined categories as nonobese (<30 kg/m2) and obese (≥30 kg/m2), and patients with obesity were further subcategorized by classes of obesity; class 1 obesity (BMI, 30 to <35 kg/m2), class 2 obesity (BMI, 35 to <40 kg/m2), and class 3 obesity (BMI, ≥40 kg/m2). 32 Nonobese patients were also further classified as underweight (<18.5 kg/m2), normal weight (18.5 to <25 kg/m2), or overweight (25 to <30 kg/m2) for descriptive studies.
Data distribution was visually assessed using histogram plots and Q-Q graphs. Relationship of PCOS diagnosis (independent variable of interest) with specified outcomes is as follows: (1) BP indices taken as a continuous variable and categorized as elevated BP (Stage 0), Stage 1 HTN, Stage 2 HTN, and overall HTN (Stage 1 and Stage 2 HTN); (2) pre-MetS; and (3) WHO classes of obesity were examined. Continuous data were compared between groups (cases and controls) utilizing Student’s t test (parametric) or Mann–Whitney U (nonparametric) as appropriate, whereas categorical variables were compared utilizing Chi-square test. Interaction between PCOS diagnosis and BMI (using a product term) for specified outcomes was examined, and interaction was identified; therefore, subsequent analyses (multivariable linear and logistic, as appropriate) examining the relationship between PCOS diagnosis and BP indices after adjusting for age, race, and ethnicity were stratified by BMI.
Continuous data are presented as mean ± standard deviation (SD) or median ± interquartile range and categorical data as percentages (%). Magnitude of associations are presented as β coefficient ± standard error (±SEM) for linear regression analyses and OR with 95% confidence interval (adjusted OR [aOR], 95% CI) for logistic regression analyses. Multivariable regression models were constructed with stepwise inclusion of race, ethnicity, and age using forward model building. Statistical significance of p-values was set at <0.05. All p-values are reported to the third decimal place. Analyses were performed using SPSS software version 28.0.0.0 (IBM Corp. Released 2021. IBM SPSS Statistics for Macintosh, Version 28.0. Armonk, NY: IBM Corp).
Results
Figure 1 provides an overview of the study population screening and enrollment. Table 1 identifies the overall population demographics by cases and controls. Women with PCOS were younger and were of significantly higher BMI than the controls (p < 0.001). Racial distribution of NHB and NHW women differed between women with PCOS versus controls (p < 0.001).
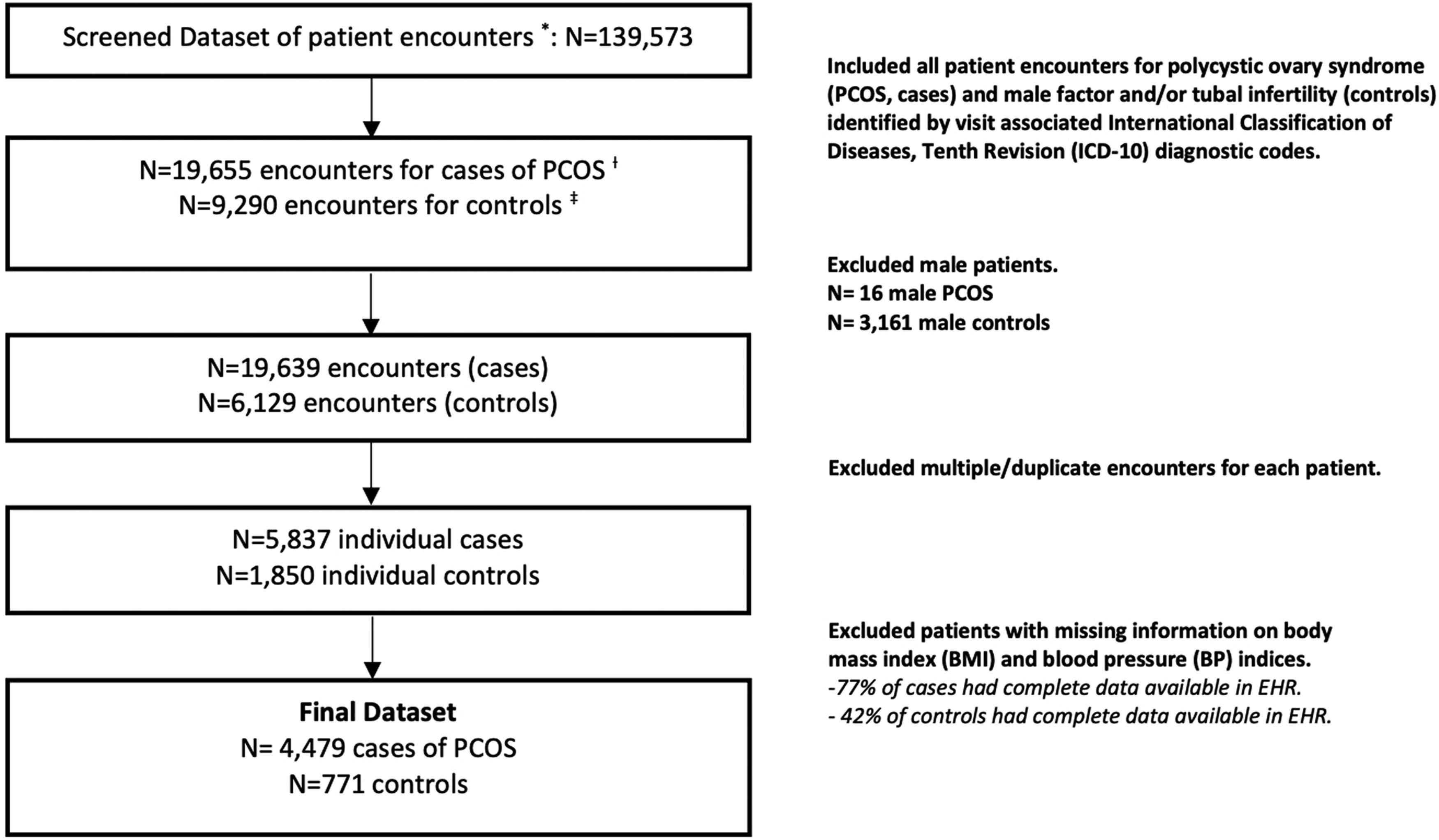
Study population. *Electronic health record (EHR) data. (†) Cases identified by ICD-10 diagnostic code for PCOS (E28.2). (‡) Controls identified by ICD-10 diagnostic codes for male factor infertility (N46.9) and tubal infertility (N97.1). ICD-10, International Classification of Diseases, Tenth Revision; PCOS, polycystic ovary syndrome.
Association between PCOS and BP indices and HTN
On univariate simple linear regression analysis, PCOS diagnosis was associated with significantly higher BP indices compared with the controls (Table 1). On univariate binary logistic regression analysis, PCOS diagnosis (compared with controls) was associated with an increased odds of meeting ACC/AHA criterion for Stage 1 HTN in the population with obesity alone (OR 1.37, 95% CI 1.07–1.76). On multivariable linear regression analyses adjusting for age, race, and ethnicity, PCOS diagnosis was associated with greater BP indices (SBP and MAP) compared with controls in the population of women with BMI ≥30 kg/m2, whereas there was no association between PCOS and the continuous BP indices in the population with BMI <30 kg/m2 (Tables 2 and 3, respectively). On multivariable binary logistic regression analyses adjusting for age, NHB race, and Hispanic ethnicity, PCOS diagnosis (compared with controls) was associated with an increased likelihood of meeting ACC/AHA defined criteria for overall HTN (aOR 1.36, 95% CI 1.05–1.75) and Stage 1 HTN (aOR 1.41, 95% CI 1.07–1.85) in the population with BMI ≥30 kg/m2 but not in the nonobese (Tables 4 and 5). PCOS diagnosis was not associated with an increased likelihood of meeting the criterion for ACC/AHA-defined elevated BP category in either population.
Results of BMI Stratified Stepwise Multivariable Linear Regression Analyses Examining the Relationship Between PCOS Diagnosis (Versus Controls) and Blood Pressure Indices
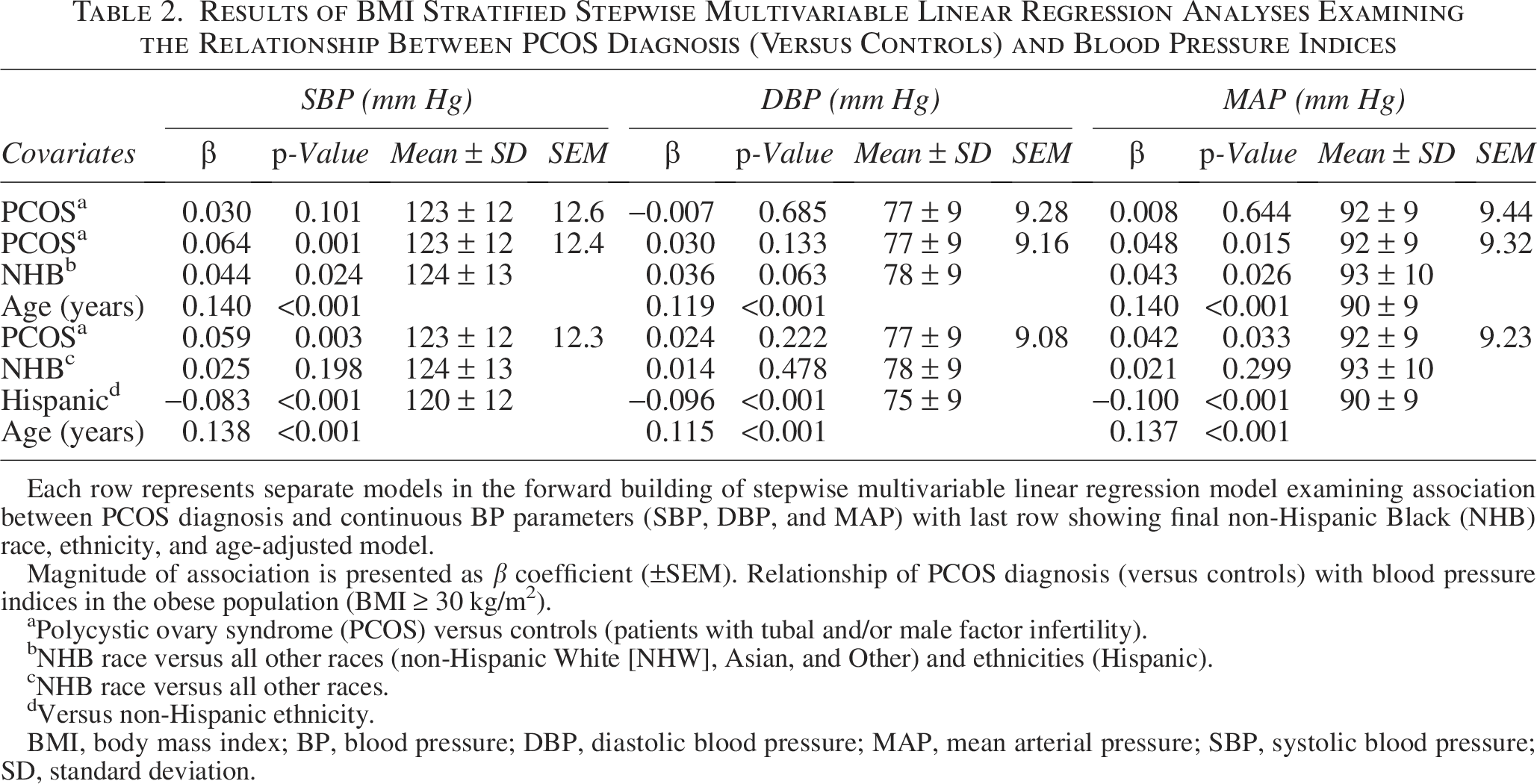
Each row represents separate models in the forward building of stepwise multivariable linear regression model examining association between PCOS diagnosis and continuous BP parameters (SBP, DBP, and MAP) with last row showing final non-Hispanic Black (NHB) race, ethnicity, and age-adjusted model.
Magnitude of association is presented as β coefficient (±SEM). Relationship of PCOS diagnosis (versus controls) with blood pressure indices in the obese population (BMI ≥ 30 kg/m2).
Polycystic ovary syndrome (PCOS) versus controls (patients with tubal and/or male factor infertility).
NHB race versus all other races (non-Hispanic White [NHW], Asian, and Other) and ethnicities (Hispanic).
NHB race versus all other races.
Versus non-Hispanic ethnicity.
BMI, body mass index; BP, blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure; SBP, systolic blood pressure; SD, standard deviation.
Relationship of PCOS Diagnosis (Versus Controls) with Blood Pressure Indices in the Nonobese Population (BMI <30 kg/m2)
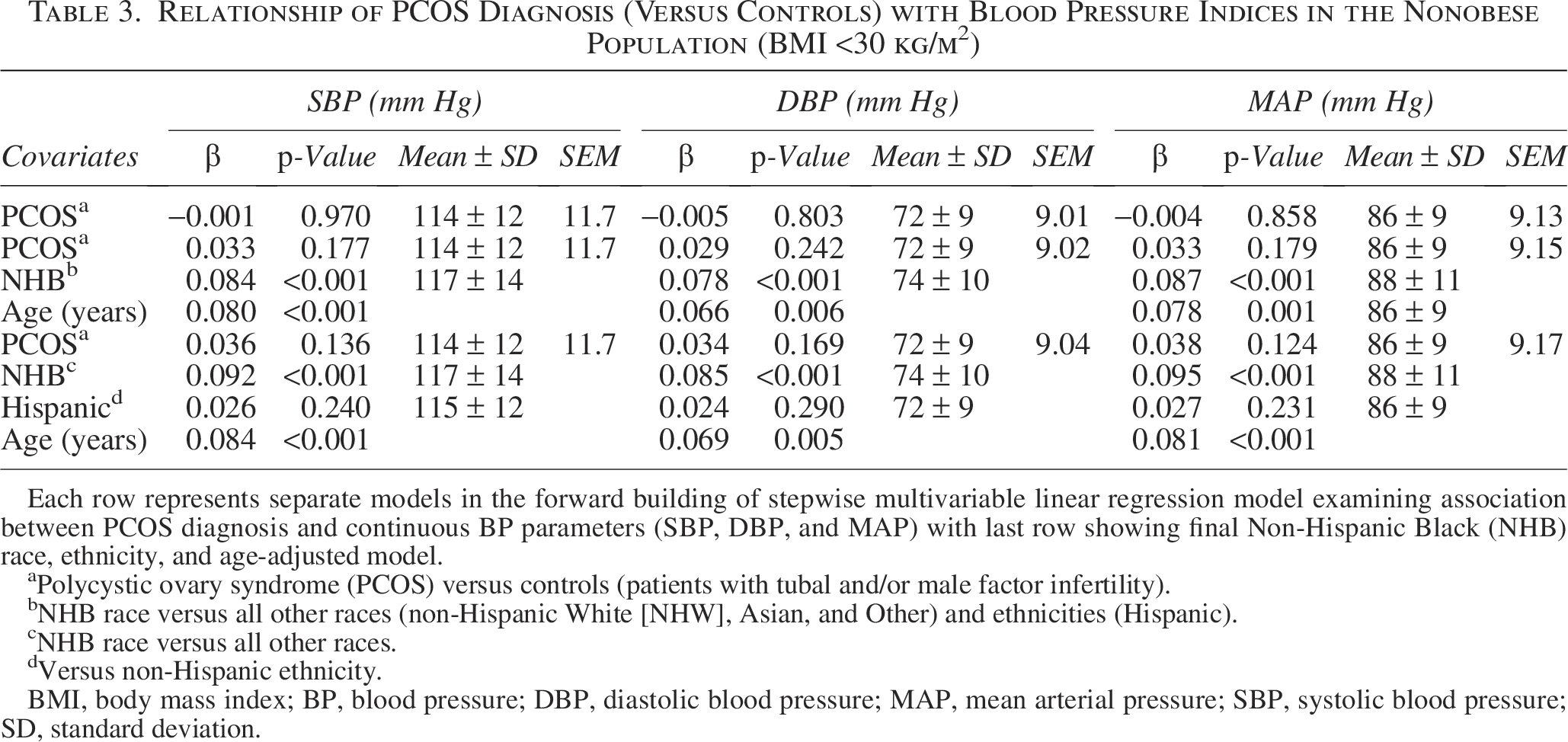
Each row represents separate models in the forward building of stepwise multivariable linear regression model examining association between PCOS diagnosis and continuous BP parameters (SBP, DBP, and MAP) with last row showing final Non-Hispanic Black (NHB) race, ethnicity, and age-adjusted model.
Polycystic ovary syndrome (PCOS) versus controls (patients with tubal and/or male factor infertility).
NHB race versus all other races (non-Hispanic White [NHW], Asian, and Other) and ethnicities (Hispanic).
NHB race versus all other races.
Versus non-Hispanic ethnicity.
BMI, body mass index; BP, blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure; SBP, systolic blood pressure; SD, standard deviation.
Results of BMI Stratified Stepwise Multivariable Logistic Regression Analyses Examining the Relationship Between PCOS Diagnosis (Versus Controls) with Hypertension
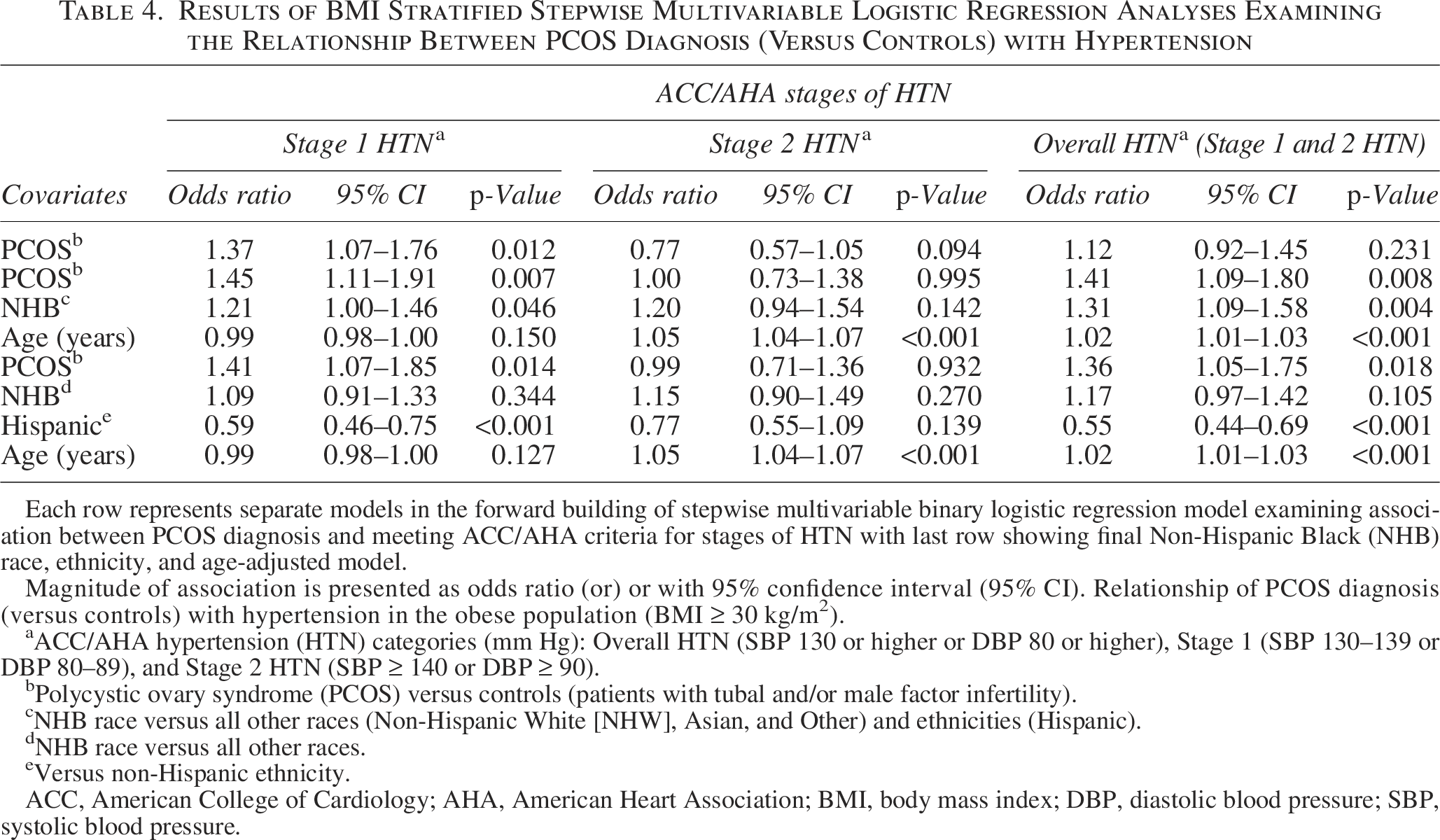
Each row represents separate models in the forward building of stepwise multivariable binary logistic regression model examining association between PCOS diagnosis and meeting ACC/AHA criteria for stages of HTN with last row showing final Non-Hispanic Black (NHB) race, ethnicity, and age-adjusted model.
Magnitude of association is presented as odds ratio (or) or with 95% confidence interval (95% CI). Relationship of PCOS diagnosis (versus controls) with hypertension in the obese population (BMI ≥ 30 kg/m2).
ACC/AHA hypertension (HTN) categories (mm Hg): Overall HTN (SBP 130 or higher or DBP 80 or higher), Stage 1 (SBP 130–139 or DBP 80–89), and Stage 2 HTN (SBP ≥ 140 or DBP ≥ 90).
Polycystic ovary syndrome (PCOS) versus controls (patients with tubal and/or male factor infertility).
NHB race versus all other races (Non-Hispanic White [NHW], Asian, and Other) and ethnicities (Hispanic).
NHB race versus all other races.
Versus non-Hispanic ethnicity.
ACC, American College of Cardiology; AHA, American Heart Association; BMI, body mass index; DBP, diastolic blood pressure; SBP, systolic blood pressure.
Relationship of PCOS Diagnosis (Versus Controls) with Hypertension in the Nonobese Population (BMI <30 kg/m2)
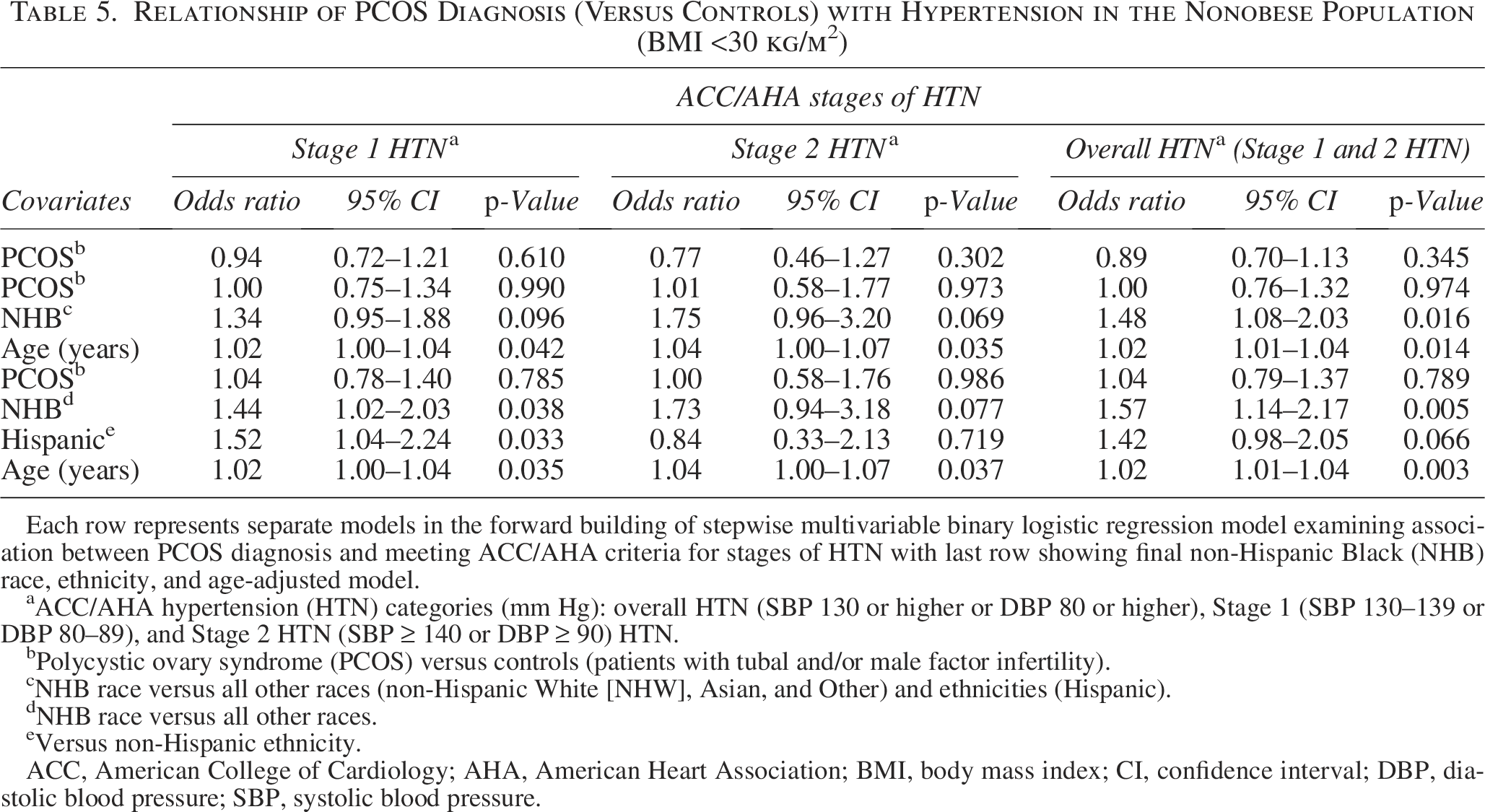
Each row represents separate models in the forward building of stepwise multivariable binary logistic regression model examining association between PCOS diagnosis and meeting ACC/AHA criteria for stages of HTN with last row showing final non-Hispanic Black (NHB) race, ethnicity, and age-adjusted model.
ACC/AHA hypertension (HTN) categories (mm Hg): overall HTN (SBP 130 or higher or DBP 80 or higher), Stage 1 (SBP 130–139 or DBP 80–89), and Stage 2 HTN (SBP ≥ 140 or DBP ≥ 90) HTN.
Polycystic ovary syndrome (PCOS) versus controls (patients with tubal and/or male factor infertility).
NHB race versus all other races (non-Hispanic White [NHW], Asian, and Other) and ethnicities (Hispanic).
NHB race versus all other races.
Versus non-Hispanic ethnicity.
ACC, American College of Cardiology; AHA, American Heart Association; BMI, body mass index; CI, confidence interval; DBP, diastolic blood pressure; SBP, systolic blood pressure.
Association between PCOS and WHO classes of obesity
On univariate analysis, PCOS diagnosis was associated with an increased likelihood of meeting WHO criteria for severe obesity; obesity (BMI ≥30 kg/m2, OR 2.12, 95% CI 1.82–2.48), class 1 obesity (BMI, 30 to <35 kg/m2, OR 0.71, 95% CI 0.59–0.85), class 2 obesity (BMI, 35 to <40 kg/m2, OR 1.48, 95% CI 1.18–1.87), and class 3 obesity (BMI ≥40 kg/m2, OR 5.13, 95% CI 3.81–6.90). The prevalence of WHO classes of obesity differed significantly between the cases and the controls (Fig. 2); the prevalence of class 3 obesity was significantly higher in the PCOS group compared to the controls (OR 4.16, 95% CI 3.04–5.69, p < 0.001). On multivariable binary logistic analysis adjusting for age, NHB race, and ethnicity, PCOS diagnosis was associated with an increased likelihood of meeting criteria for more severe classes of obesity, with a near 2-fold greater likelihood for class 2 obesity (aOR 1.72, 95% CI 1.34–2.21) and 6-fold greater likelihood for class 3 obesity (aOR 6.29, 95% CI 4.60–8.61) compared with the controls (Table 6). Women with PCOS were less likely to meet criteria for less severe (class 1) obesity compared with controls.
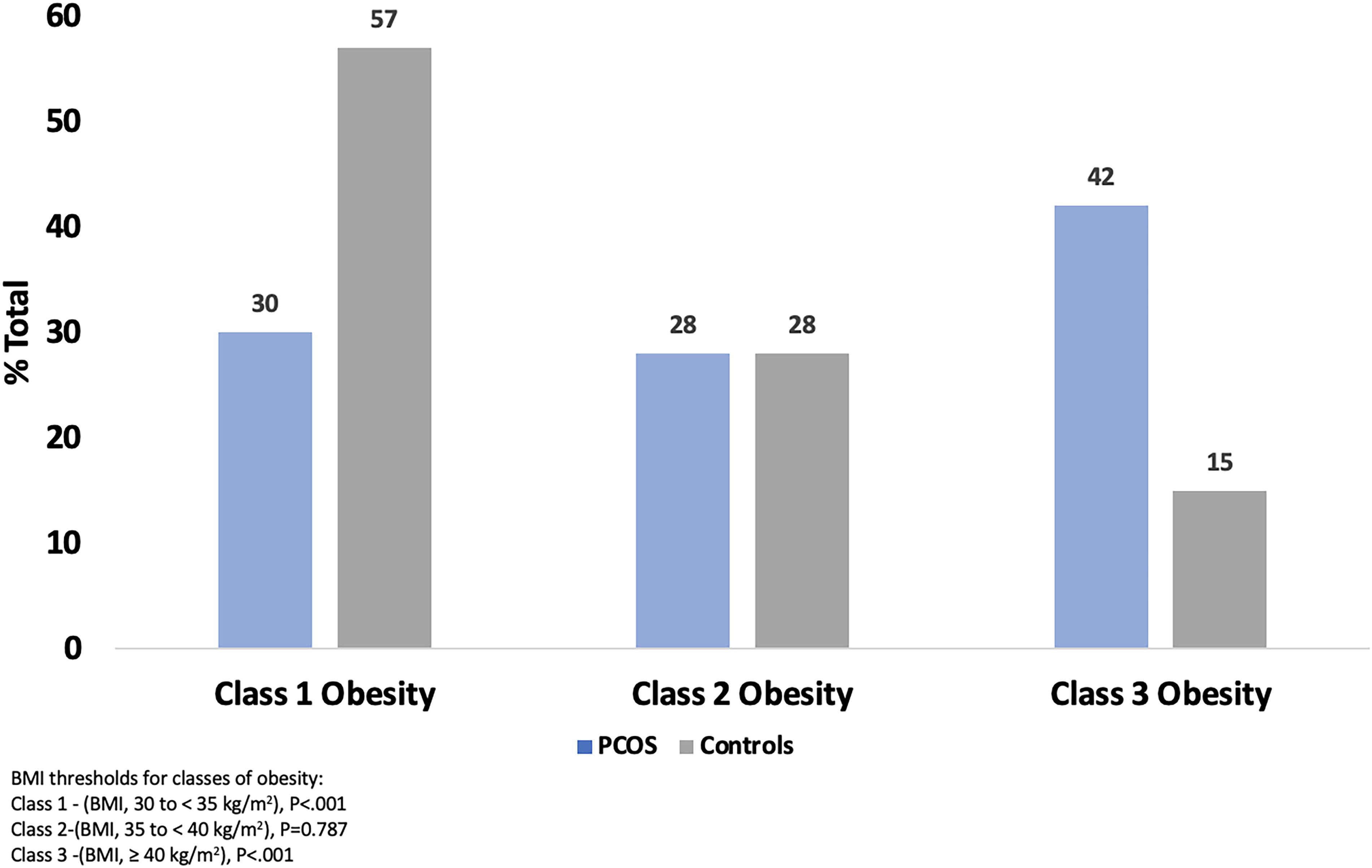
Categorizing obesity severity among the population with obesity—prevalence (%) of severe obesity is higher in cases of PCOS compared with controls. BMI thresholds for classes of obesity: Class 1—(BMI, 35 to <40 kg/m2), p < 0.001; Class 2—(BMI, 30 to <35 kg/m2), p = 0.787; and Class 3—(BMI, 30 to ≥35 kg/m2), p < 0.001. BMI, body mass index; PCOS, polycystic ovary syndrome.
Association Between PCOS Diagnosis (Versus Controls) and WHO Classes of Obesity—Results of Multivariable Binary Logistic Model Adjusting for Race, Ethnicity, and Age Presented as Odds Ratio or with 95% Confidence Interval
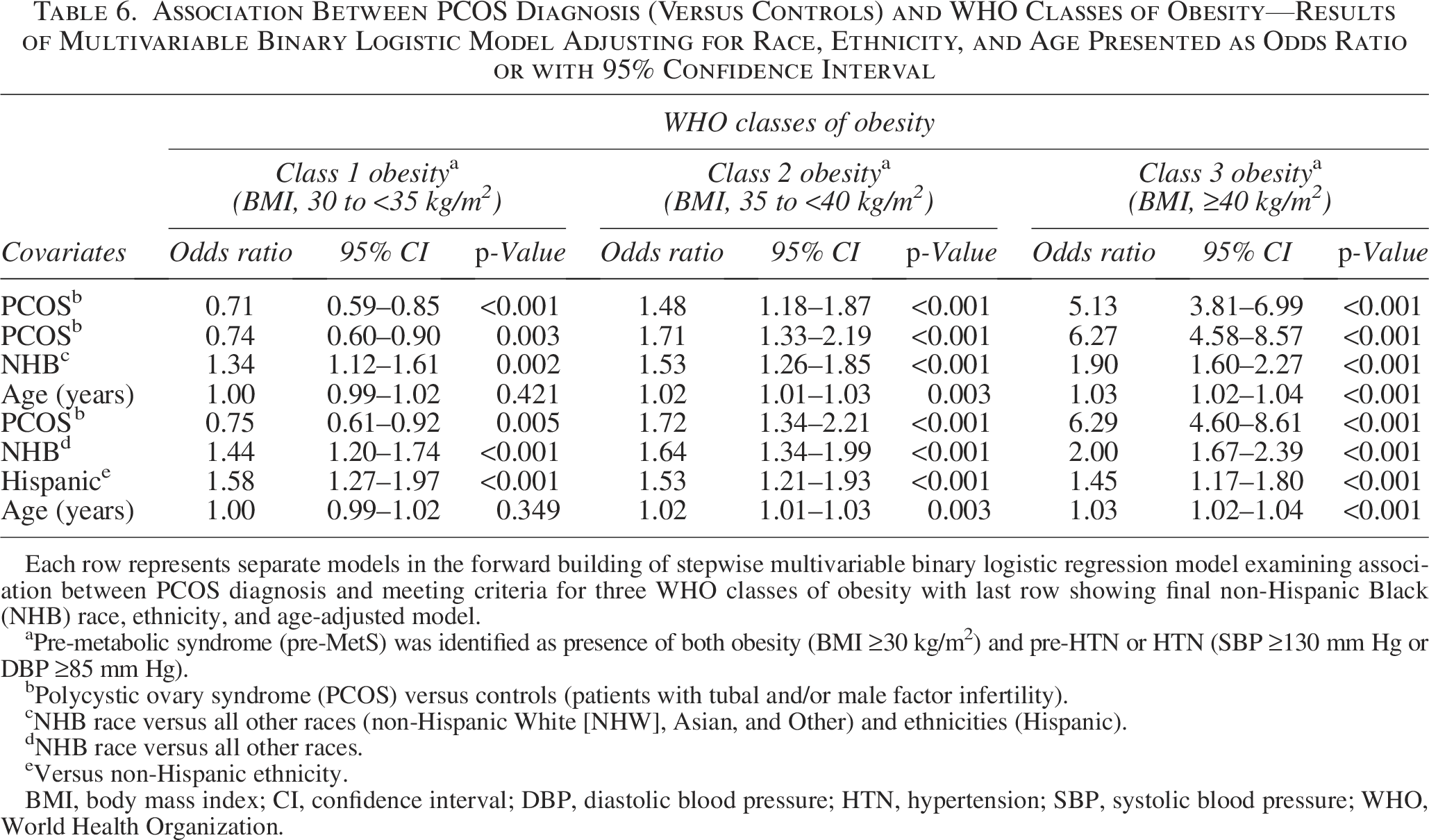
Each row represents separate models in the forward building of stepwise multivariable binary logistic regression model examining association between PCOS diagnosis and meeting criteria for three WHO classes of obesity with last row showing final non-Hispanic Black (NHB) race, ethnicity, and age-adjusted model.
Pre-metabolic syndrome (pre-MetS) was identified as presence of both obesity (BMI ≥30 kg/m2) and pre-HTN or HTN (SBP ≥130 mm Hg or DBP ≥85 mm Hg).
Polycystic ovary syndrome (PCOS) versus controls (patients with tubal and/or male factor infertility).
NHB race versus all other races (non-Hispanic White [NHW], Asian, and Other) and ethnicities (Hispanic).
NHB race versus all other races.
Versus non-Hispanic ethnicity.
BMI, body mass index; CI, confidence interval; DBP, diastolic blood pressure; HTN, hypertension; SBP, systolic blood pressure; WHO, World Health Organization.
Association between PCOS and pre-MetS
In a univariate model, PCOS diagnosis was associated with an increased odds of meeting the criterion for pre-MetS (OR 1.65, 95% CI 1.32–2.07). On multivariable binary logistic regression analysis adjusting for age, NHB race, and Hispanic ethnicity, cases of PCOS (compared with controls) were two-fold more likely to meet the specified criterion for pre-MetS (aOR 2.25, 95% CI 1.77–2.86) (Table 7).
Association Between PCOS Diagnosis (Versus Controls) and Meeting Pre-MetS Criteria—Results of Multivariable Binary Logistic Model Adjusting for Race, Ethnicity, and Age Presented as Odds Ratio or with 95% Confidence Interval
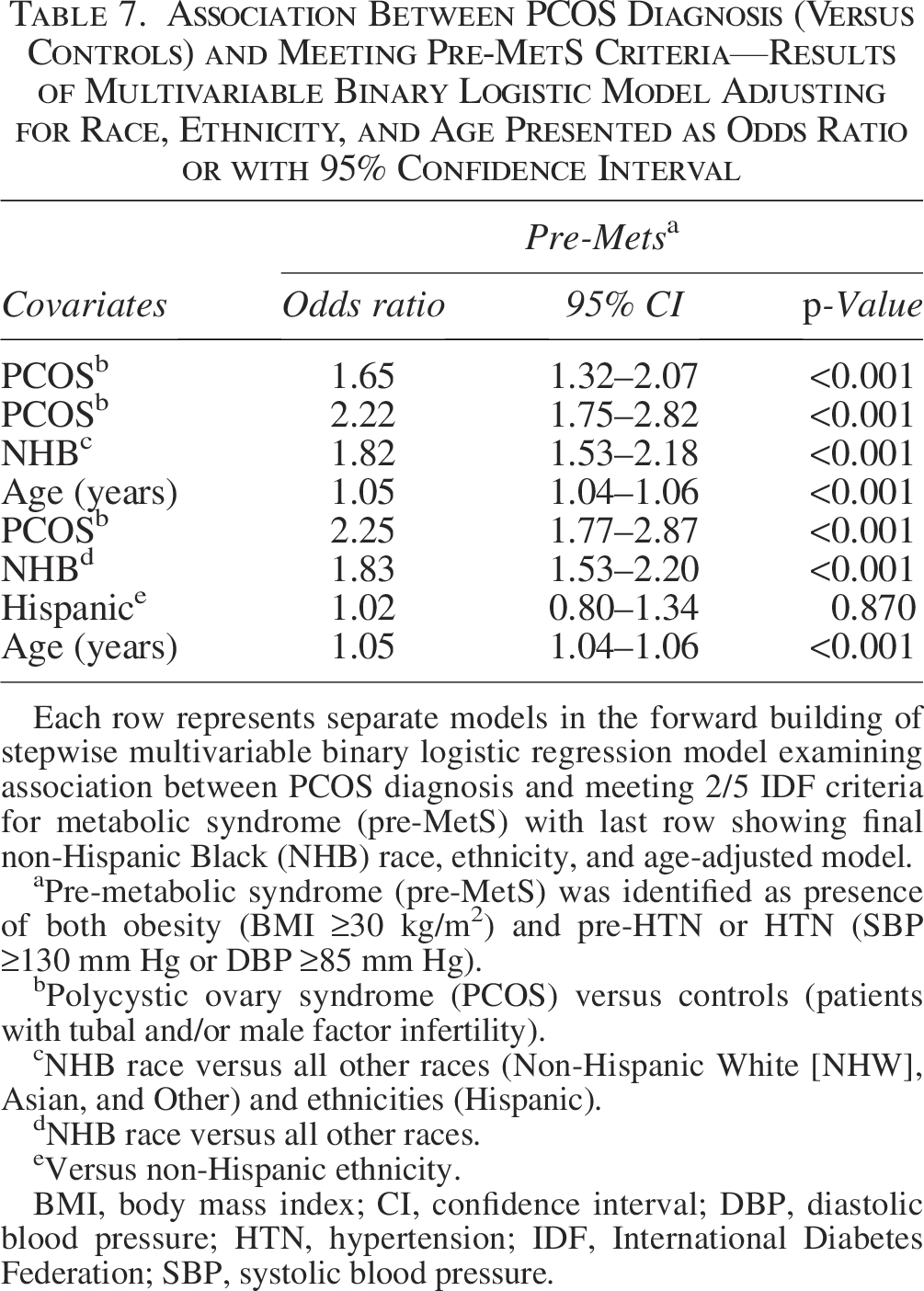
Each row represents separate models in the forward building of stepwise multivariable binary logistic regression model examining association between PCOS diagnosis and meeting 2/5 IDF criteria for metabolic syndrome (pre-MetS) with last row showing final non-Hispanic Black (NHB) race, ethnicity, and age-adjusted model.
Pre-metabolic syndrome (pre-MetS) was identified as presence of both obesity (BMI ≥30 kg/m2) and pre-HTN or HTN (SBP ≥130 mm Hg or DBP ≥85 mm Hg).
Polycystic ovary syndrome (PCOS) versus controls (patients with tubal and/or male factor infertility).
NHB race versus all other races (Non-Hispanic White [NHW], Asian, and Other) and ethnicities (Hispanic).
NHB race versus all other races.
Versus non-Hispanic ethnicity.
BMI, body mass index; CI, confidence interval; DBP, diastolic blood pressure; HTN, hypertension; IDF, International Diabetes Federation; SBP, systolic blood pressure.
Racial differences in relationship of PCOS diagnosis with HTN, obesity, and pre-MetS
Women of NHB race and ethnicity (versus all other races and Hispanic ethnicity) were at a significantly higher risk of meeting ACC/AHA defined criteria for HTN in the population with BMI <30 kg/m2 ([overall HTN, aOR 1.57, 95% CI 1.14–2.17]) (Stage 1 HTN, aOR 1.44, 95% CI 1.02–2.03). Hispanic ethnicity itself was independently associated with HTN, although the directionality of this relationship differed by BMI status. Hispanic women meeting BMI-specified obesity criterion were significantly less likely to meet ACC/AHA-defined Stage 1 HTN (aOR 0.59, 95% CI 0.46–0.75), whereas an opposite directionality was noted in nonobese Hispanic women who had a significantly greater likelihood of meeting ACC/AHA Stage 1 HTN (aOR 1.52, 95% CI 1.04–2.24). Racial differences were apparent in relationship with obesity; NHB race (versus all other races) and Hispanic ethnicity (versus non-Hispanic ethnicity) were associated with an increased odds of all classes of obesity (p < 0.001). Furthermore, NHB race was associated with an increased likelihood of meeting criteria for pre-MetS independent of PCOS diagnosis (aOR 1.83, 95% CI 1.53–2.20). Hispanic ethnicity was not associated with pre-MetS (aOR 1.02, 95% CI 0.89–1.34).
Discussion
Utilizing a clinically characterized patient population of reproductive-age women, we demonstrate that diagnosis of PCOS is associated with a predisposition to CVD risk as reflected in elevated BP indices and increased odds of meeting criteria for ACC/AHA-defined stages of HTN, 2/5 of the IDF specified criteria for MetS (pre-MetS, BMI ≥30 kg/m2 and SBP ≥130 mm Hg or DBP ≥85 mm Hg), and WHO-classified severe obesity (Class 3). Based on clinical data, our findings offer a meaningful addition to existing literature that delineates increased cardiometabolic risk in women with PCOS.33–35 PCOS diagnosis was associated with increased odds of HTN independent of age, race, and ethnicity in the population with obesity but not the population without obesity. Importantly, we also show increased risk of mild HTN in obese NHB women with PCOS relative to NHW and Hispanic women with PCOS. An association between PCOS and HTN has been described,25,36 although the data are limited and variable. The interaction observed between obesity and PCOS diagnosis allowed us to conduct multivariable analyses stratified on BMI to gain clarity on the relevance of PCOS diagnosis itself for BP indices. Women with the obese/overweight PCOS phenotype represent 75% of women with this syndrome, and we would expect BP regulation to differ between women with PCOS with and without obesity.37,38 Indeed, we observed elevated BP and a greater likelihood of HTN only in the obese PCOS population.
Androgen excess in women with PCOS has been associated with increased prevalence of cardiometabolic disease, HTN, and endothelial dysfunction,11,12 and hyperandrogenemia may play a role in monocyte adhesion to endothelial cells in the development of atherosclerosis in this population. 39 A limitation of this study is that biochemical androgen indices data were not available. More data are needed to elucidate the interplay between obesity and hyperandrogenemia, as our findings suggest that BMI is clearly relevant as a CVD risk factor in women with PCOS.
In addition to the observed association with HTN, PCOS diagnosis, as well as NHB race, was associated with an increased likelihood for meeting the two specified criteria for MetS (classified as pre-MetS); these observations are concordant with existing data that support a higher prevalence of dysmetabolism in these specific populations. 23 Furthermore, the magnitude of prevalent obesity was significantly higher in women diagnosed with PCOS compared with age-comparable controls; women with PCOS were significantly more likely to meet BMI criteria for class 3 obesity compared with the controls. A large meta-analysis of 20 prospective studies estimating sex-and age-adjusted total and cause-specific mortality rates in adults with class 3 obesity compared with normal weight controls found that class 3 obesity was associated with substantially elevated rates of total mortality, with most excess deaths due to heart disease, cancer, and diabetes. 40 There is a need for prospective studies characterizing the relative risk of CVD and HTN in women with PCOS with more severe obesity (class 2 and class 3) compared with populations with normal weight or less severe obesity (class 1), as this subgroup may have substantially greater disease burden.
Racial differences for CVD risk were apparent in our relatively young patient cohort. NHB race was associated with an increased likelihood of meeting ACC/AHA-defined criteria for Stage 1 HTN in the population with BMI ≥30 kg/m2. However, the association between Hispanic ethnicity and meeting ACC/AHA-defined criteria for Stage 1 HTN diverged in the context of obesity status. Hispanic women had comparatively lower BP indices in the population with BMI ≥30 kg/m2, consistent with one large study of 11,035 reproductive-age women with PCOS, which found that NHB women were more likely and Hispanic women were less likely to be hypertensive in multivariable logistic regression analyses that adjusted for BMI category and age. 25 However, our findings suggest that BP in minority women with PCOS may vary with obesity status. Our findings support existing data suggesting that PCOS diagnosis and race are important considerations when evaluating CVD risk in general, or specific risk for hypertensive disorders in reproductive-age women. These results are also consistent with findings from a large cross-sectional analysis of data from the Dallas Heart Study, which demonstrates a greater prevalence of CV risk factors in Black compared to White women with PCOS. 24 Thus, our findings underscore a need for vigilance and further examination of the underpinnings of observed predisposition of reproductive-age NHB women to CVD and HTN.
A strength of this study is that the stratification of HTN by the ACC/AHA categories of elevated BP (Stage 0), Stage 1 HTN, Stage 2 HTN, and overall HTN allows for a more nuanced examination of the role of PCOS in HTN and greater clinical applicability. Although our study may not have been powered to fully examine the association between PCOS and Stage 2 HTN, the significance of PCOS as a predictor of our overall HTN criteria suggests that the association between PCOS diagnosis and an increased likelihood of meeting criteria for Stage 2 HTN might reach significance in a population with a greater prevalence of Stage 2 HTN. Similarly, stratification of obesity by WHO classes of obesity allows for greater clarity on the severity of obesity in women with PCOS.
Limitations
Our study comprised a unique population of women seeking care at a reproductive endocrinology clinic versus a standard community-based population. A strength of the study is that the patients were under care of reproductive endocrine specialists and ICD-10 diagnostic codes were for follow-up and/or treatment of PCOS and thus we assume a high level of diagnostic accuracy; we however are unable to differentiate the diagnostic criteria (National Institutes of Health [NIH] versus Rotterdam versus AES) among the cases given the nature of the available “macro-level” data provided by ICD-10 diagnostic codes and lack of androgen indices. We were limited in that data regarding comorbidities, medications, and socioeconomic factors were not available in this dataset. Also, women meeting BP criteria for HTN at a clinical visit may not have a known diagnosis of HTN, and given the cross-sectional design of the study, we are unable to determine chronicity and persistence of abnormal indices. Furthermore, self-reported race and ethnicity data from EHR are subject to individual variations in perception of race. Clinical measures of BP were not taken in controlled environments and may be confounded by White Coat HTN, a phenomenon suggested to be more prevalent in the NHB population. 41 Additionally, we do not have data on the biochemical metabolic indices needed for the full IDF criteria for MetS (obesity, pre-HTN or HTN, hypertriglyceridemia, reduced high density lipoprotein elevated fasting plasma glucose), 31 which limited our ability to examine the prevalence of meeting criteria for MetS in this population. However, we were able to demonstrate an association between PCOS and NHB race and meeting criteria for pre-MetS, which allowed for examination of metabolic disturbances in this population.
Utilizing EHR data on a sample of reproductive age women seeking reproductive health-related care at a tertiary care institution, higher BP indices were identified in obese women with PCOS compared to the specified age and BMI comparable controls. We also supported earlier data indicating greater HTN in NHB women with PCOS. Our findings underscore a need for appropriately designed future studies that aim at teasing out the interplay of PCOS and obesity in the causation of HTN that includes a focus on race and ethnicity. Importantly, our study highlights the inconsistencies in monitoring and documentation of vital signs and anthropometric data in outpatient clinical settings: data on BMI and BP were documented in 77% of women with PCOS and only 42% of controls. With the increasing application of data in clinical research, the collaborative role of healthcare teams and their responsibility toward ensuring valid and complete data documentation is key to success.
Authors’ Contributions
F.O. collected the data, analyzed the data, and wrote the article. M.M. assisted with data analysis. L.P. assisted with data collection, analysis, and writing the article. N.S. assisted with data analysis and writing the article.
Footnotes
Acknowledgment
The authors would like to thank the Yale JDAT for providing the EHR data used in this study.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was supported by NIH R01 HL135089. F.O. was supported by NIH T35 HL 7649-35.
Data Availability
All datasets on which the conclusions of the article rely are available to readers upon request.
