Abstract
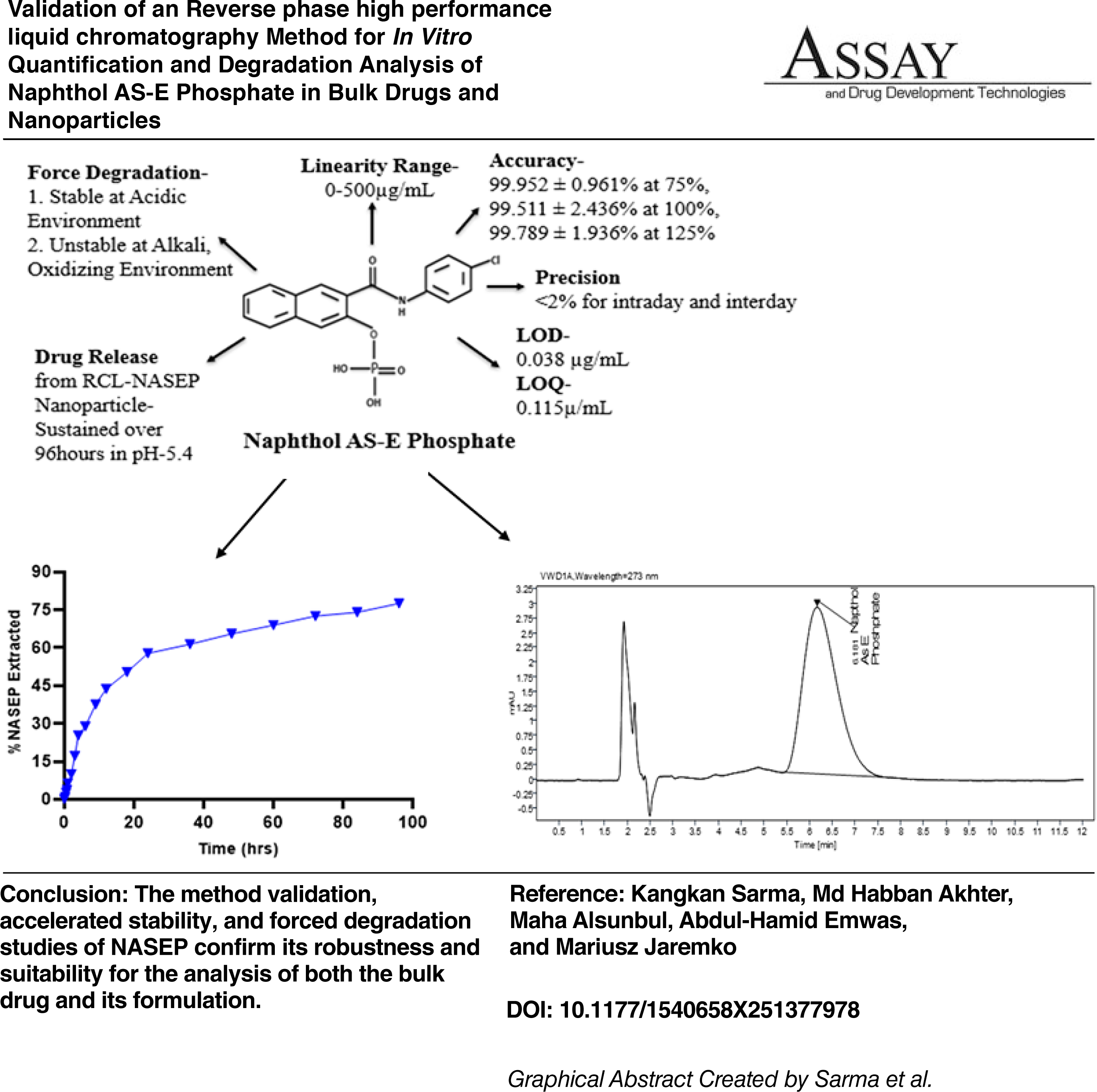
This study presents the first validated High-performance liquid chromatography (HPLC) technique for quantifying naphthol AS-E phosphate (NASEP) in bulk drugs and nanoparticle formulation. A C18 HPLC cartridge (250 × 4.6 mm, 5 µm particle size) served as the stationary phase for quantification. The mobile phase consisted of Milli-Q water with 0.1% trifluoroacetic acid (TFA) in pump A and acetonitrile with 0.1% TFA in pump B, with a flow rate ranging from 0.8 to 1.2 mL/min. A 32 factorial design was employed to evaluate the robustness of the proposed method, using mobile phase composition (X1), flow rate (X2), and column temperature (X3) as independent variables and peak area (R1), retention time (R2), and percent recovery (R3) as response variables. The calibration range curve (10–500 µg/mL) was best fitted by quadratic regression. The linearity was reported in the above-mentioned range. The accuracy was 99.952% ± 0.961% at the 75% level, 99.58% ± 1.483% at the 100% level, and 99.789% ± 1.936% at the 125% level. The coefficient of variation was below 2% for both intraday and interday measurements, and the limits of detection and quantification were 0.038 and 0.115 µg/mL, respectively. The NASEP solution was stable (99.04% ± 0.0251%) for 48 h at 8°C. The forced degradation study also revealed that the NASEP solution remained stable in an acidic environment for 48 h at 40°C but degraded at 80°C (p < 0.046) in a time-dependent manner. In contrast, it was unstable in an alkaline medium, independent of temperature, and degraded in the presence of strong oxidizing agents (p < 0.039). Furthermore, NASEP encapsulated in a Gly-Arg-Gly-Asp-Ser pentapeptide and low-molecular-weight heparin functionalized metal–organic framework exhibited sustained drug release at acidic pH 5.4. The proposed NASEP quantification method was validated and is suitable for routine analysis in pharmaceutical formulations.
Keywords
Get full access to this article
View all access options for this article.
