Abstract
Background
Visceral artery aneurysms (VAAs) are uncommon vascular lesions, representing less than 2% of all arterial aneurysms, yet they carry significant clinical relevance owing to their frequently silent course and potential for catastrophic rupture. Within this group, celiac trunk aneurysms (CTAAs) and hepatic artery aneurysms (HAAs) constitute a minority of cases but pose distinct diagnostic and therapeutic challenges. Endovascular therapy has gained prominence in recent years; however, open surgical repair remains an essential treatment modality, particularly in complex anatomical settings or when endovascular options are unsuitable.
Methods
We present a case series of 3 patients managed at our institution: 2 with CTAAs and 1 with an HAA. All patients underwent open surgical repair using tailored vascular reconstruction strategies, including splenic artery interposition, aorto-hepato-splenic bypass with a great saphenous vein graft, and superficial femoral artery interposition. Patient selection and operative planning were guided by preoperative imaging, aneurysm morphology, and comorbid conditions.
Results
There was no perioperative mortality. Major postoperative complications occurred in 2 cases, including gastric ischemia requiring total gastrectomy and postoperative hematoma requiring re-exploration. At 1-year follow-up, all patients demonstrated 100% graft patency and preserved end-organ perfusion.
Conclusions
This experience highlights the importance of individualized surgical decision-making in the management of VAAs. While the rarity of these lesions limits large-scale comparative studies, our series reinforces that open surgical repair remains a safe and durable option when tailored to patient-specific anatomy. In the era of endovascular techniques, elective open surgical repair remains associated with low mortality and high technical success when performed by an experienced surgical team, offering satisfactory early and mid-term outcomes.
Introduction
Visceral artery aneurysms (VAAs) are rare vascular entities, accounting for less than 2% of all arterial aneurysms, with the splenic artery being the most commonly affected, followed by the hepatic and superior mesenteric arteries. 1 Celiac trunk aneurysms (CTAAs) and hepatic artery aneurysms (HAAs) are particularly uncommon but clinically significant due to their risk of rupture, which carries high mortality.2,3 The etiology of VAAs is multifactorial, including atherosclerosis, trauma, infection, and connective tissue disorders. HAAs have also been associated with prior endovascular interventions, pancreatitis, and septic emboli.4,5 Many patients are asymptomatic, with aneurysms often detected incidentally on imaging; when present, symptoms may include abdominal pain or gastrointestinal bleeding. 6
Management depends on aneurysm size, location, and clinical presentation, with current guidelines recommending treatment for symptomatic lesions and those larger than 2 cm. 7 Although endovascular therapy is often preferred due to its minimally invasive nature, its use may be limited by unfavorable anatomy or clinical factors such as infection or rupture. 8 In such cases, open surgical reconstruction remains an important treatment option, particularly for complex CTAAs and HAAs involving multiple arterial branches.9,10
Clinical Characteristics, Surgical Management, and Outcomes
Case 1: Celiac Trunk Aneurysm
A 65-year-old male was admitted to our department with progressive abdominal pain, vomiting, and significant weight loss over a 6-month period. Physical examination revealed a thin, malnourished patient with diffuse abdominal tenderness but no palpable mass or peritoneal signs. Laboratory workup showed mild anemia (hemoglobin 11.2 g/dL) and hypoalbuminemia consistent with chronic malnutrition, while liver and renal function tests remained within normal limits. The patient had a history of Billroth I gastrectomy and cholecystectomy, as well as colorectal cancer treated with transanal excision, and remained in remission under regular oncological surveillance.
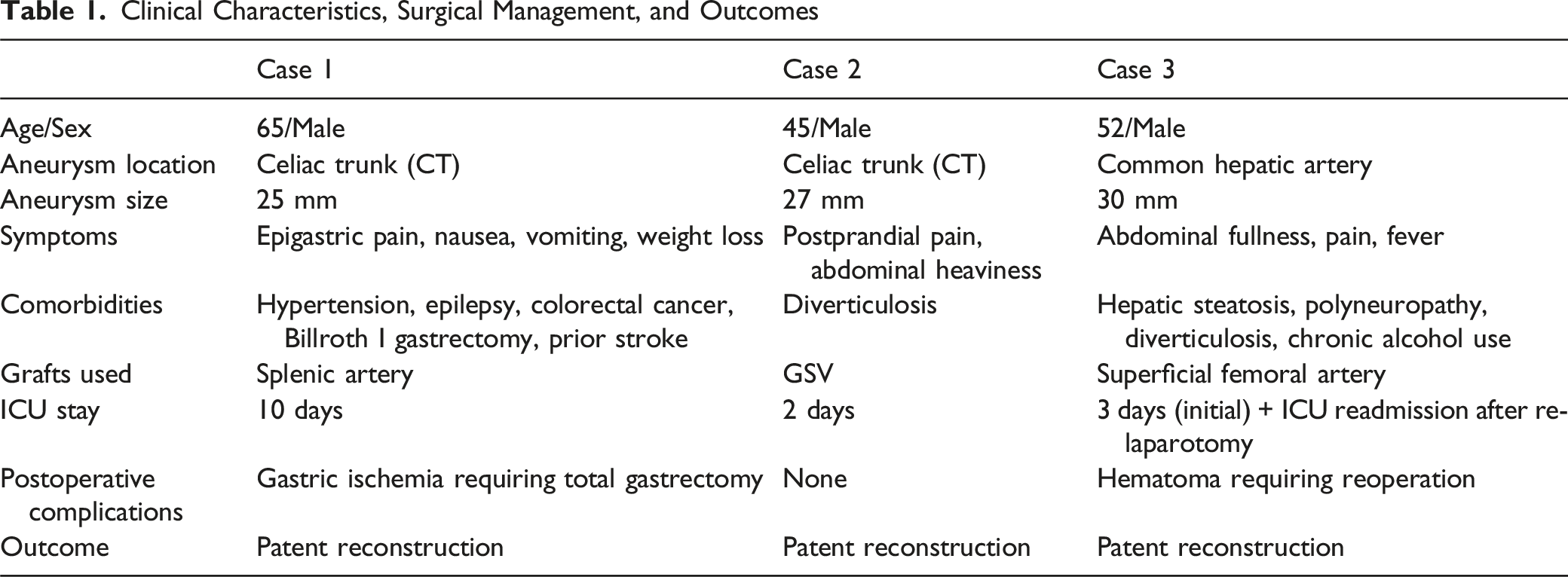
CT of the abdomen demonstrated a 2.5-cm saccular aneurysm arising from the celiac trunk, with partial mural thrombus but no signs of rupture (Figure 1). The common hepatic and splenic arteries originated from the aneurysmal sac, whereas the left gastric artery (LGA) also arose from its proximal margin. No other visceral aneurysms were identified. Elective open surgical repair was performed. Contrast-enhanced computed tomography angiography demonstrating a 2.5-cm saccular aneurysm of the celiac trunk (arrow) with partial mural thrombus. The aneurysm involves the origins of the common hepatic and splenic arteries, while the left gastric artery arises from its proximal margin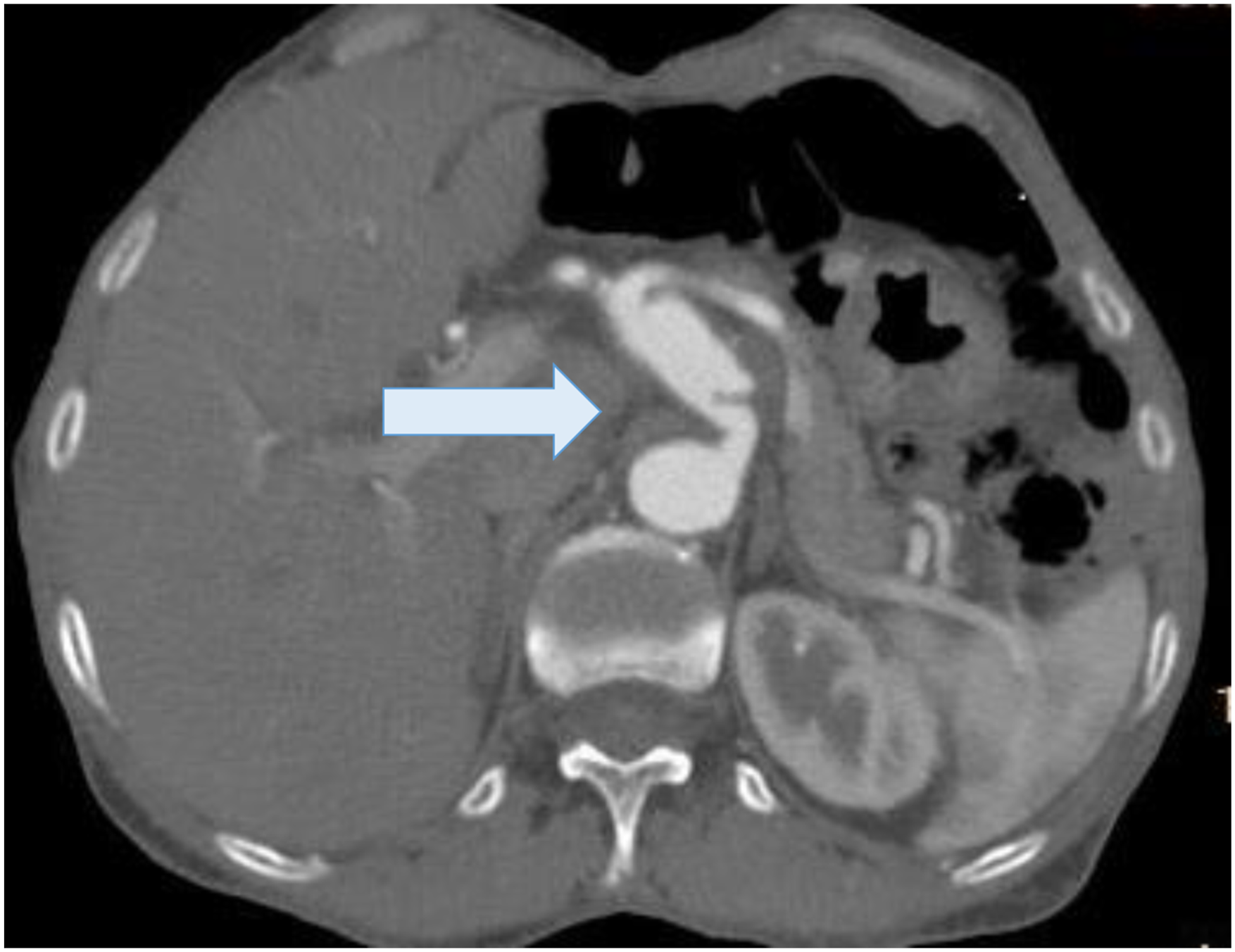
A midline laparotomy was performed, and exposure was optimized using a Thompson retractor. The supraceliac aorta was exposed by dividing the diaphragmatic crura, providing direct access to the celiac trunk for proximal vascular control. A celiac trunk aneurysm was identified, involving the origins of the LGA, the splenic artery (SA), and the proximal third of the common hepatic artery (CHA) (Figure 2). Surgical management consisted of aneurysm resection, ligation of the left gastric and splenic arteries, and resection of the involved segment of the common hepatic artery with interposition grafting using the splenic artery as the conduit. The graft was anastomosed in an end-to-side fashion to the thoracoabdominal aorta and the CHA (Figure 3). The total operative time was 259 minutes, and the estimated intraoperative blood loss was 400 mL. The patient received 1 unit of packed red blood cells. Intraoperative view of a celiac trunk (CT) aneurysm involving the common hepatic artery (CHA) and splenic artery (SA) Interposition of splenic artery, end-to-end anastomoses with CT and hepatic artery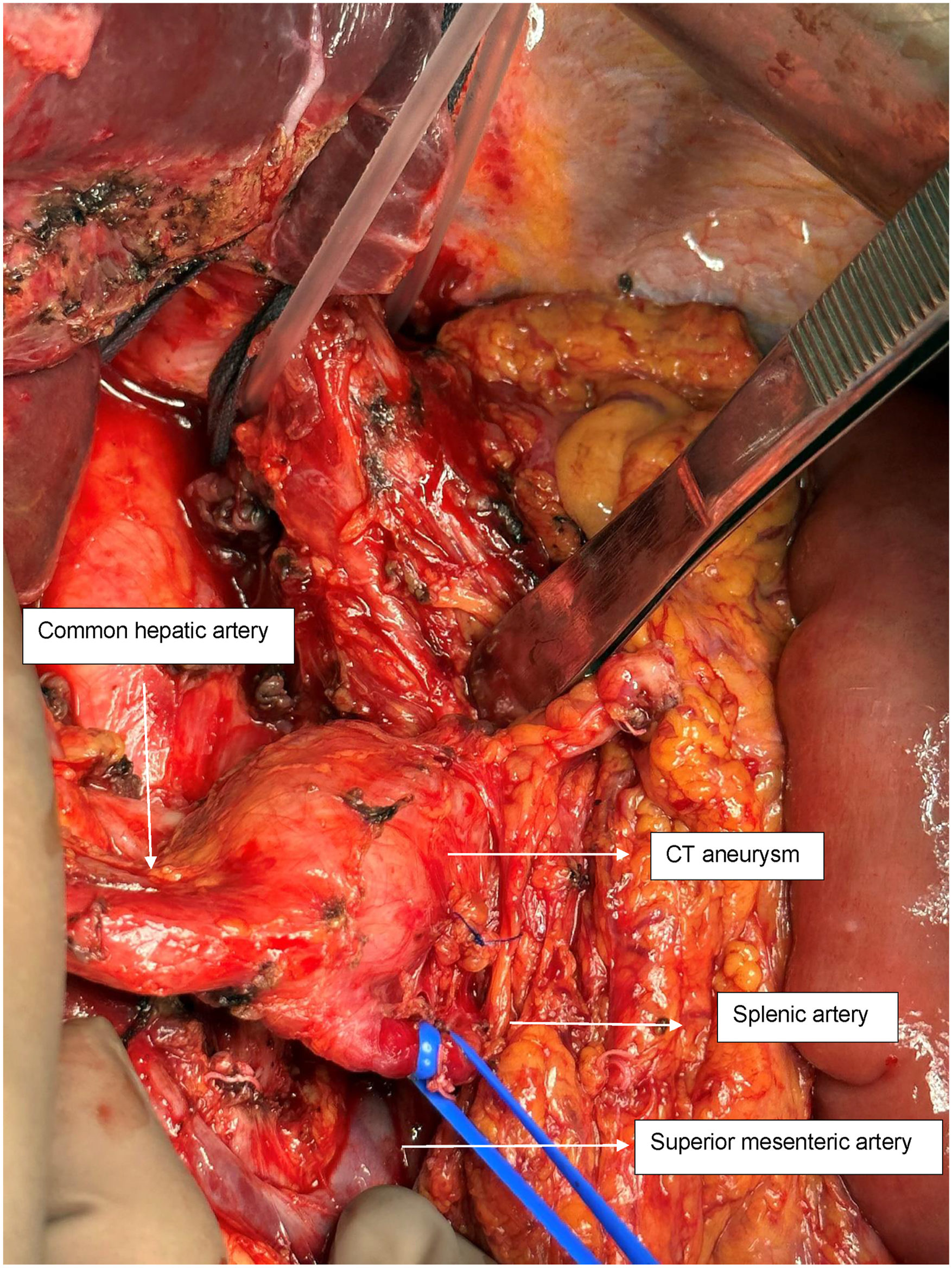
On the first postoperative day diagnostic gastroscopy was performed. It revealed ischemia of gastric mucosa which led to total gastrectomy with Roux-en-Y esophagojejunostomy. The postoperative recovery following the second procedure was uneventful. The postoperative course was prolonged; however, the patient was discharged on the 20th postoperative day with a patent reconstruction. Nutritional rehabilitation was initiated with enteral support, followed by gradual transition to oral intake.
At 1-year follow-up, the patient reported satisfactory recovery with stabilization of body weight and no further postprandial abdominal pain. CT angiography demonstrated patent vascular reconstruction with preserved perfusion of the hepatic artery (Figure 4). There was no evidence of aneurysm recurrence, graft stenosis, or ischemic complications affecting the liver or spleen. Contrast-enhanced computed tomography angiography demonstrating patent vascular reconstruction with preserved hepatic arterial perfusion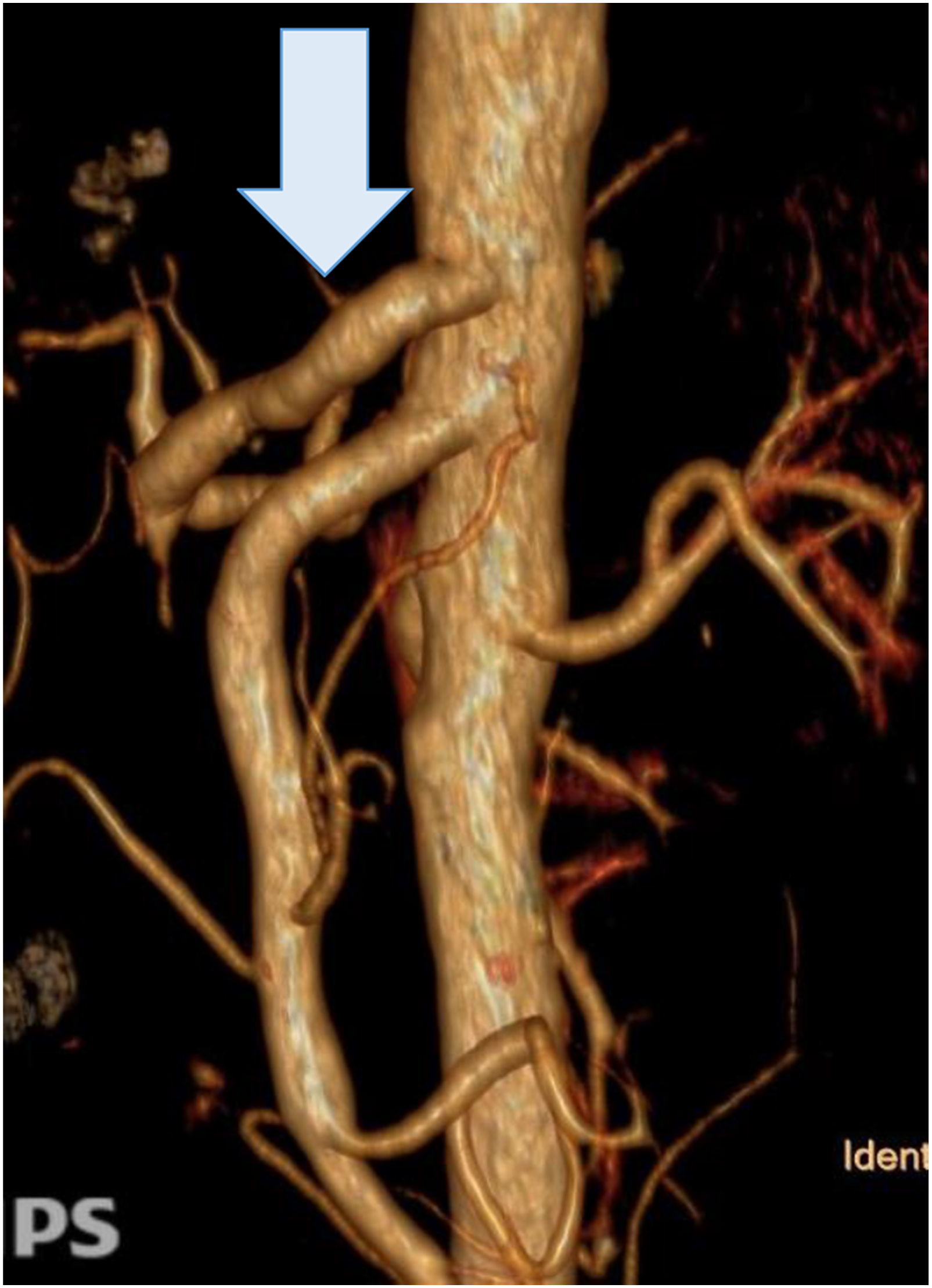
Case 2: Celiac Trunk Aneurysm
A 46-year-old male presented with several months of postprandial abdominal pain associated with early satiety and mild, progressive weight loss. He denied hematemesis, melena, or prior abdominal surgery. On examination, the patient was hemodynamically stable with no palpable abdominal mass. Laboratory findings revealed mild anemia (hemoglobin 11.8 g/dL) but were otherwise unremarkable.
CT of the abdomen demonstrated a 2.7-cm saccular aneurysm of the celiac trunk, involving the origins of the splenic, common hepatic, and left gastric arteries (Figure 5). No mural thrombus or rupture signs were present. Open surgical repair was performed. Contrast-enhanced axial CT angiography demonstrating a celiac trunk aneurysm (arrow). The adjacent vascular structures include the abdominal aorta (posterior circular structure), the origin of the superior mesenteric artery (anterior to the aorta), and the splenic and common hepatic arteries arising from the aneurysmal segment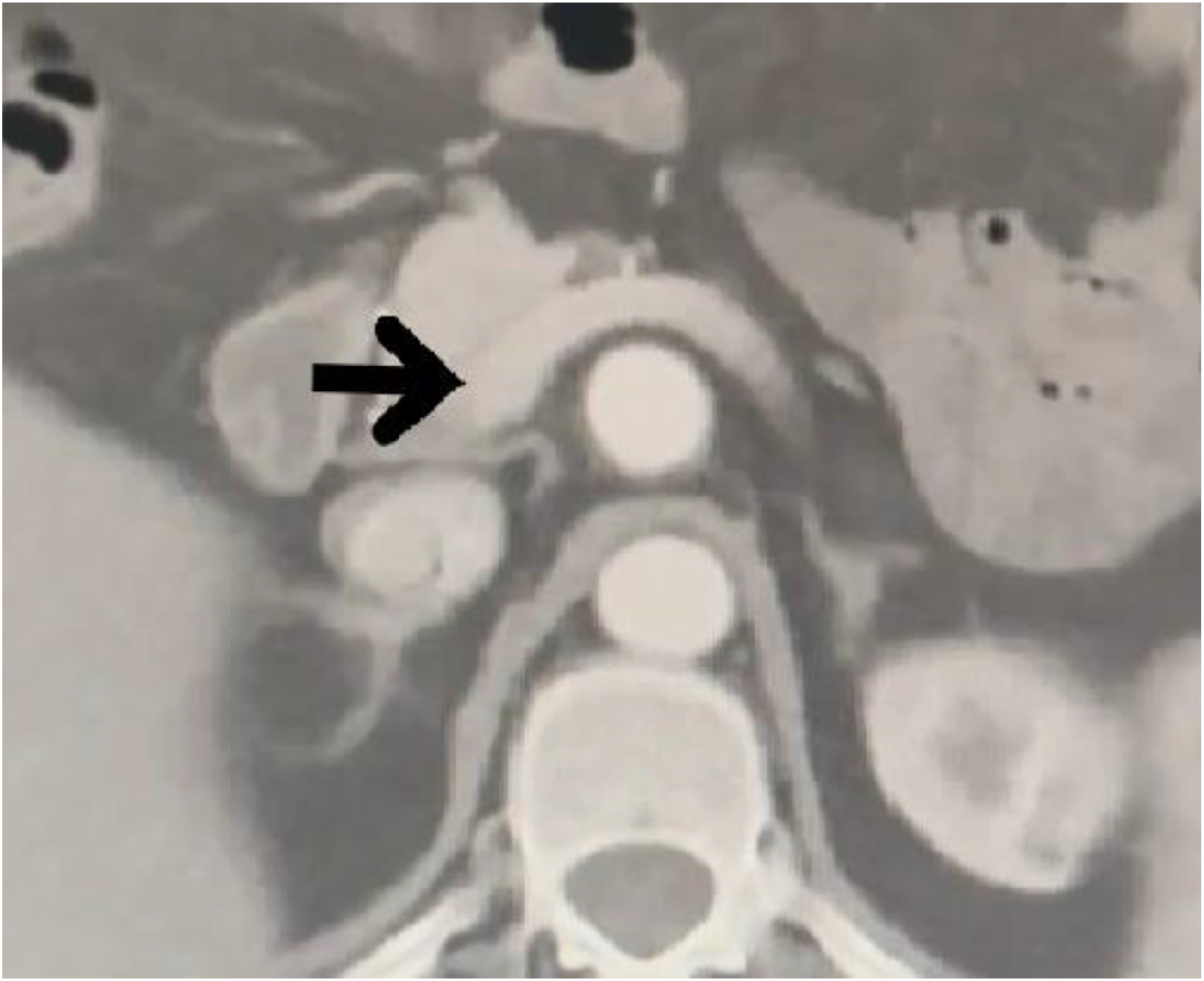
A bilateral subcostal laparotomy with xiphoid extension was performed, providing adequate exposure of the supraceliac aorta and celiac trunk. Exposure was optimized with the use of a Thompson retractor. Intraoperative findings confirmed a celiac trunk aneurysm incorporating the origins of the splenic, hepatic, and left gastric arteries. Following systemic heparinization and after achieving proximal and distal vascular control, aneurysm resection was undertaken. Reconstruction was performed with a descending bypass from the thoracoabdominal aorta to the visceral branches. A reversed right GSV graft was used as conduit. The proximal anastomosis was fashioned in an end-to-side manner to the thoracoabdominal aorta (Figure 6), with distal anastomoses created end-to-side to the common hepatic artery and end-to-end to the splenic artery (Figure 7) - aorto-hepato-lienal bypass. The LGA was ligated due to its involvement at the aneurysmal origin. Intraoperative flow assessment confirmed satisfactory distal pulsatility and preserved perfusion of both hepatic and splenic territories. The splenic artery was preserved. Given concomitant colonic pathology, sigmoid colectomy was performed. Bowel continuity was restored with primary anastomosis. The total operative time was 350 minutes, and the estimated blood loss was 500 mL. One unit of packed red blood cells was transfused. The patient was admitted to the ICU for postoperative monitoring and remained hemodynamically stable. Extubation was achieved within the first 24 hours, and vasoactive support was not required. He resumed oral feeding on postoperative day 4 and was discharged from the ICU to the surgical ward on day 2. The recovery phase was overall uneventful, although a transient rise in serum amylase and lipase was noted; imaging excluded major complications. Bowel function returned gradually after the colorectal procedure. Aorto-hepato-lienal bypass-proximal anastomosis with thoracoabdominal aorta Aorto-hepato-lienal bypass-proximal anastomosis- End-to-end anastomosis with splenic artery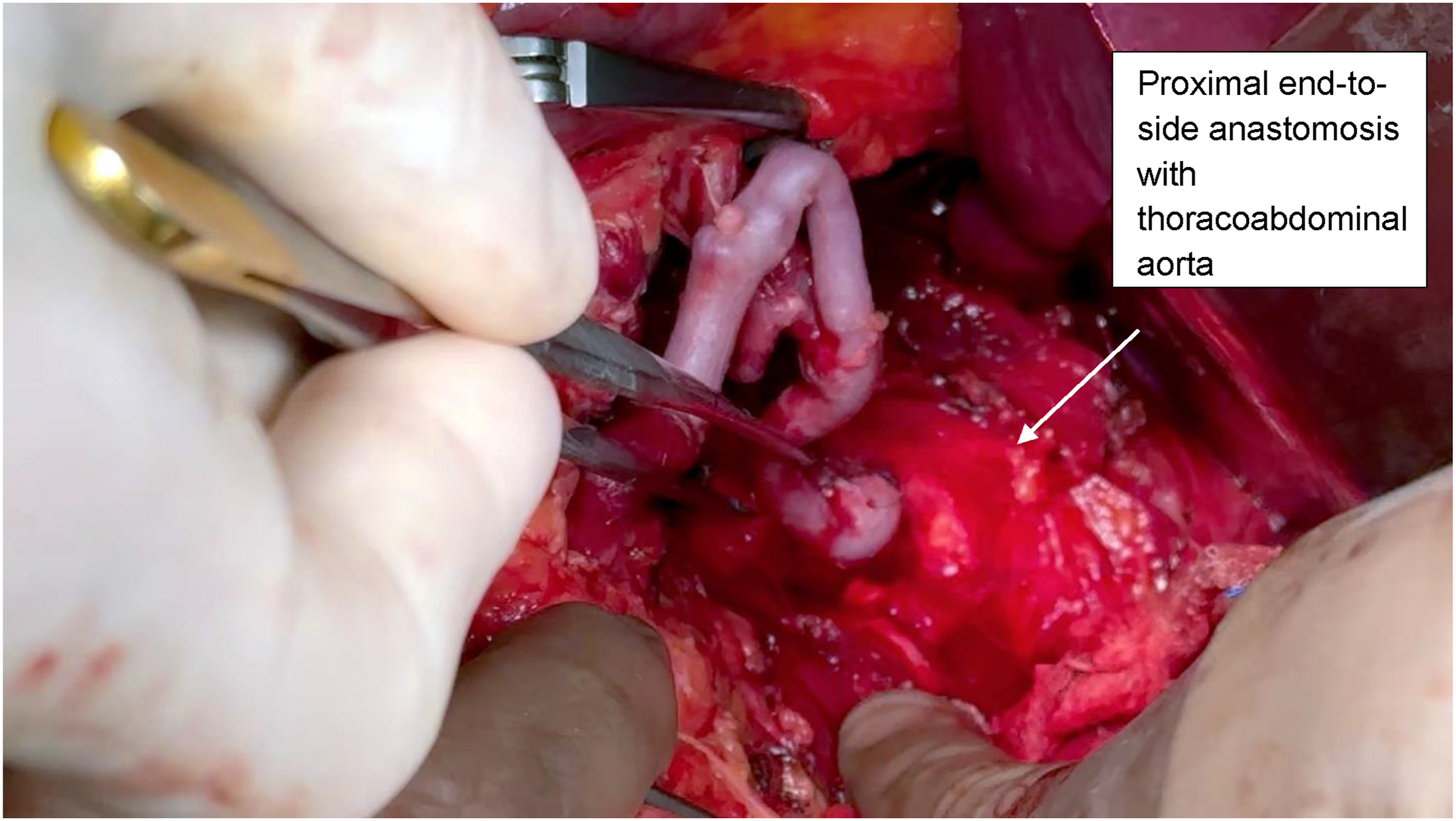
At 1-year follow-up, CT demonstrated durable graft patency without aneurysmal recurrence, stenosis, or thrombosis. The patient underwent CT in an outpatient imaging facility; however, the original disk containing the study was subsequently misplaced and could not be retrieved. No ischemic complications of the liver, spleen, or gastrointestinal tract were observed.
Case 3: Common Hepatic Artery Aneurysm
A 59-year-old male was admitted with recurrent episodes of abdominal pain without associated gastrointestinal bleeding, jaundice, or weight loss. Physical examination revealed mild tenderness in the upper abdomen but no palpable masses. Laboratory studies demonstrated mild leukocytosis and elevated inflammatory markers, while liver function tests remained within normal limits. CT identified a 3-cm saccular aneurysm of the common hepatic artery, located near its bifurcation into the right and left hepatic branches (Figure 8). Surgical repair was performed. Preoperative blood cultures were positive for Escherichia coli. Definitive repair was therefore delayed for 2 weeks, during which the patient received targeted intravenous antibiotic therapy until sterilization of the bloodstream was confirmed. Contrast-enhanced axial CT angiography demonstrating a saccular aneurysm of the common hepatic artery (arrow) at the level of its bifurcation. The abdominal aorta and adjacent vascular structures are shown for anatomical orientation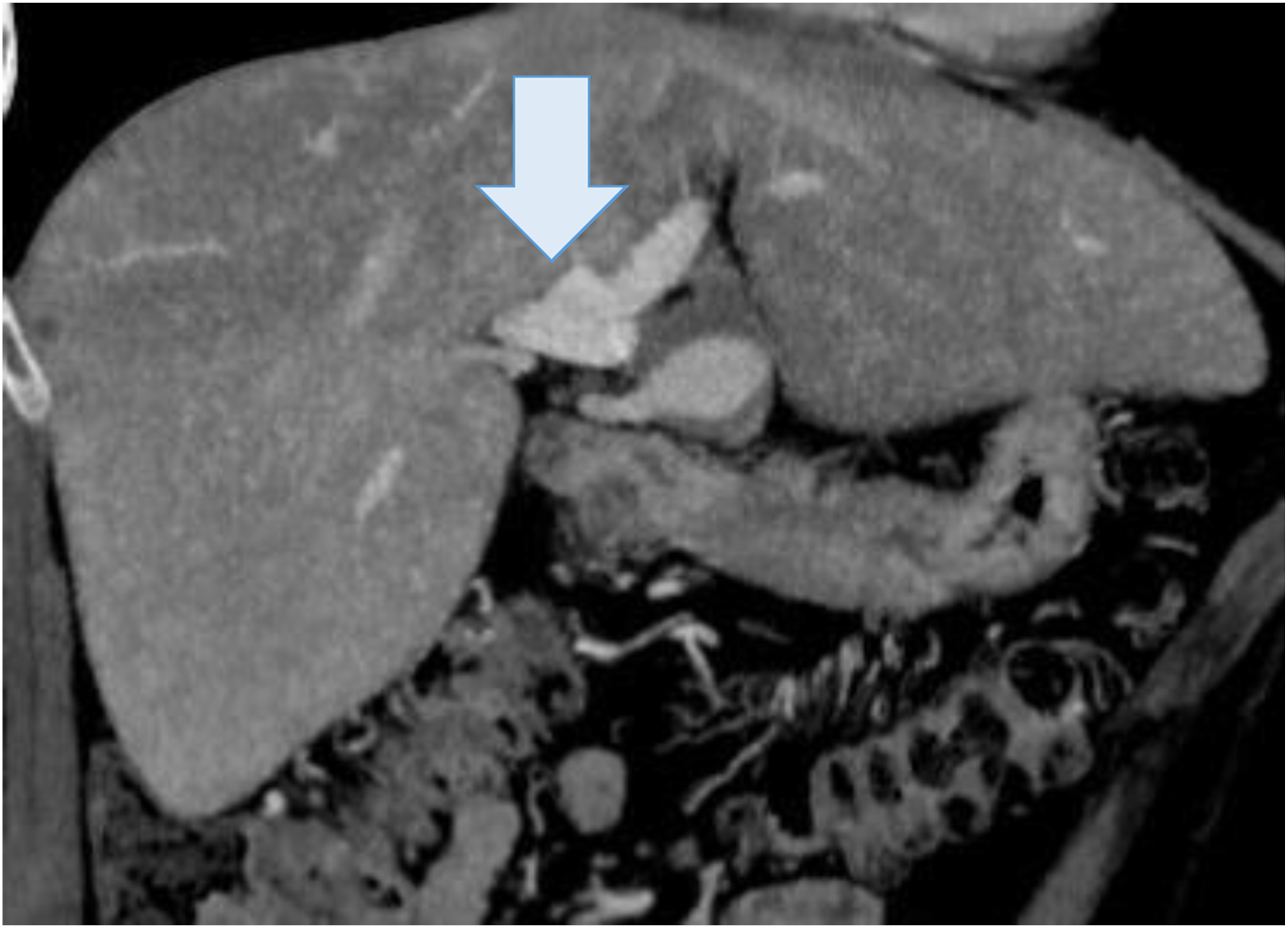
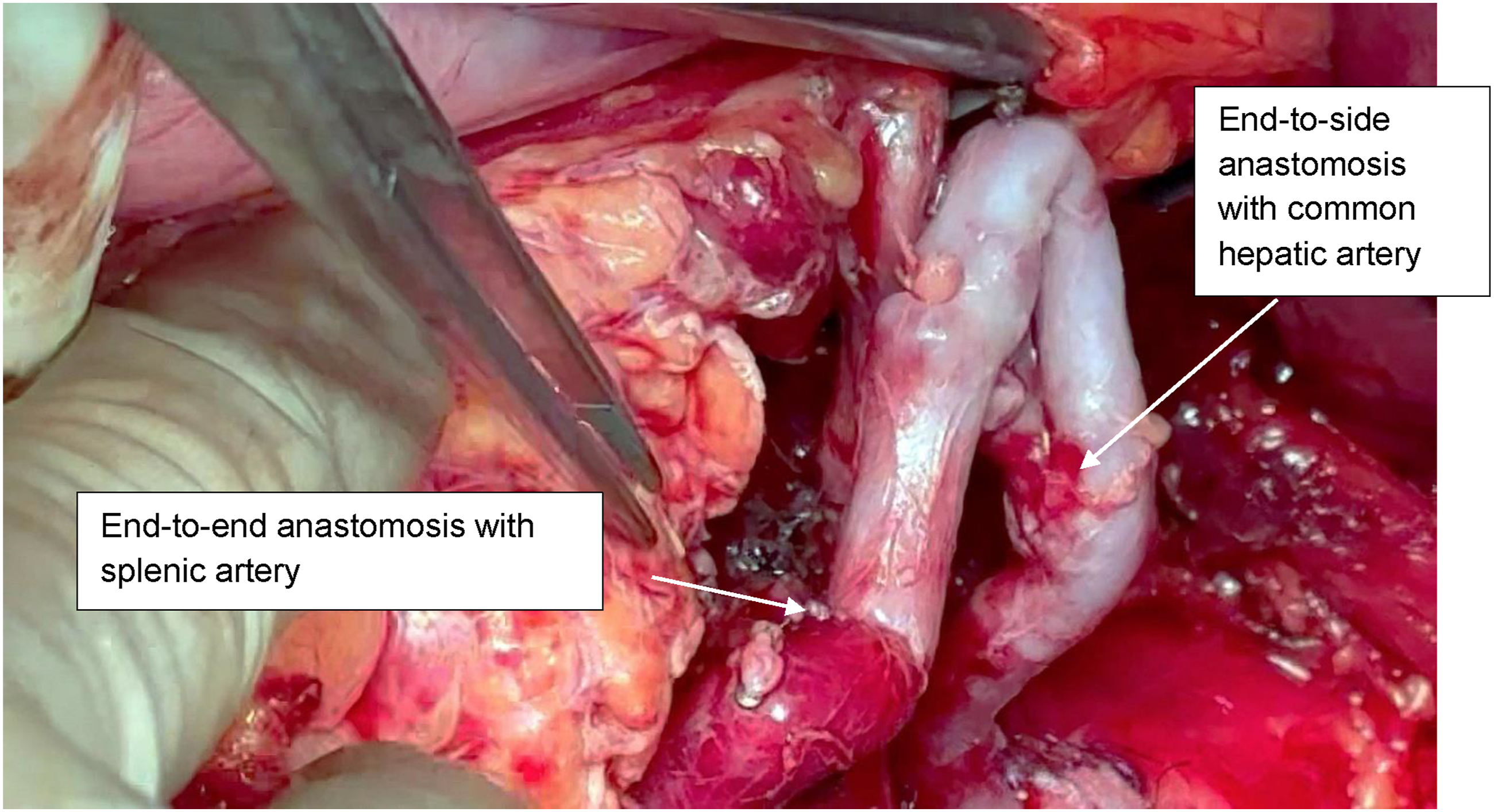
Surgical exploration was performed through a bilateral subcostal incision with xiphoid extension. Exposure was optimized with the use of a Thompson retractor. Dense adhesions surrounding the aneurysm were carefully dissected. Vascular control was achieved proximally at the celiac trunk and distally at the right and left hepatic arteries, the splenic artery, and the left gastric artery. The aneurysm was resected. Reconstruction included interposition of the superficial femoral artery (SFA) from the celiac trunk to the right hepatic artery with end-to-end anastomoses. In addition, the left hepatic artery was reimplanted (Figure 9). Intraoperative Doppler confirmed satisfactory flow in both grafts. The total operative time was 360 minutes, and the estimated blood loss was 600 mL. Two units of packed red blood cells were transfused intraoperatively. The patient was admitted to the intensive care unit (ICU) postoperatively for close hemodynamic monitoring. Initially, the patient remained hemodynamically stable with preserved liver function. However, on postoperative day 1, he developed progressive abdominal distension and paralytic ileus. CT demonstrated a large left-sided abdominal hematoma, prompting urgent re-exploration. The patient was taken back to the operating room for urgent re-exploration via a midline laparotomy. Hematoma evacuation was performed, with no active bleeding identified. Following re-intervention, the patient’s postoperative course stabilized. He remained in the ICU for 3 additional days, with gradual return of bowel function and no evidence of hepatic ischemia. Interposition of SFA from the celiac trunk to the right hepatic artery with end-to-end anastomoses; reimplantation of the left hepatic artery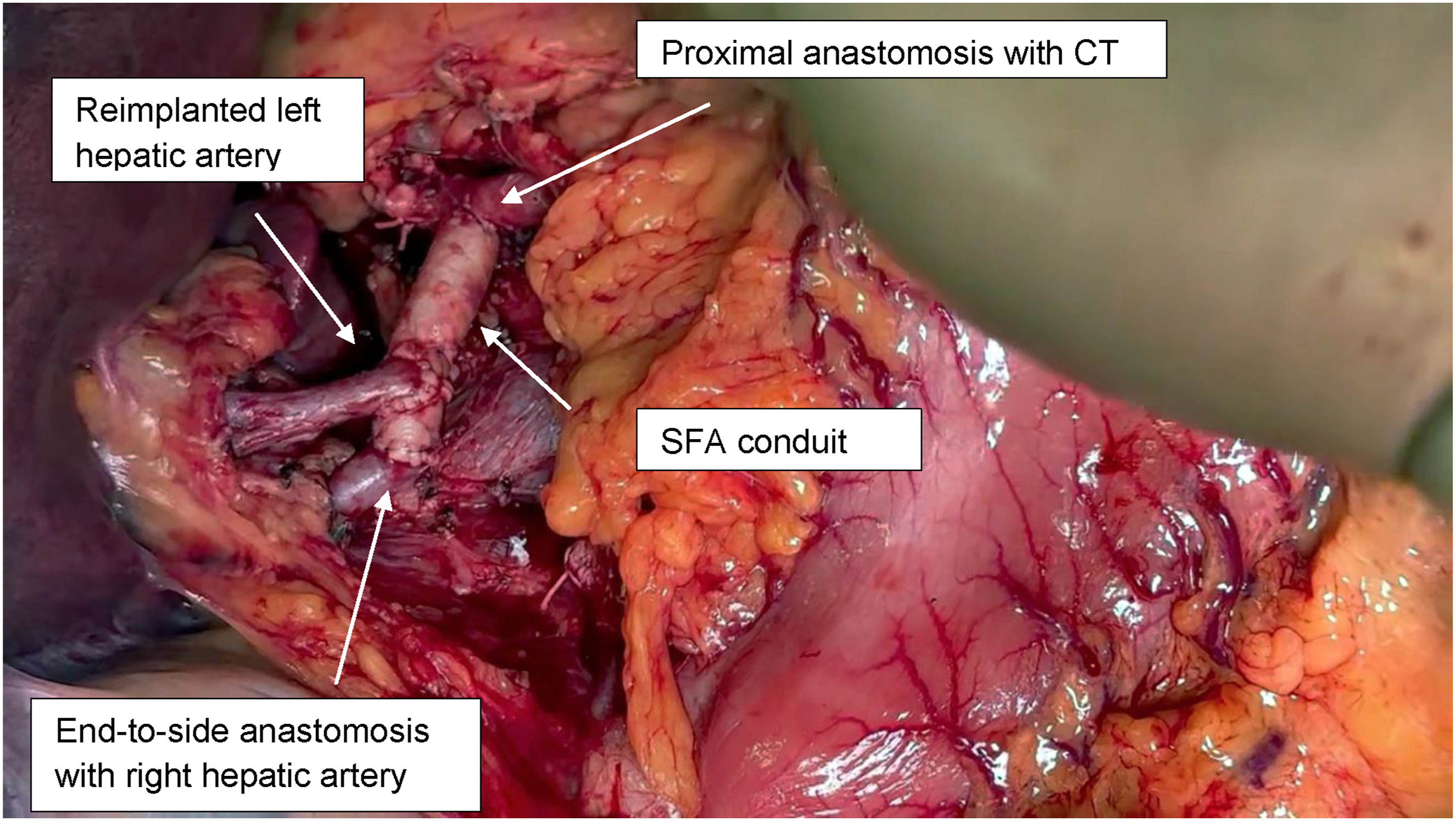
At 1-year follow-up, CT confirmed patency of the reconstruction, with preserved perfusion of the right and left hepatic lobes (Figure 10). No recurrent aneurysm, graft thrombosis, or new complications were observed. Contrast-enhanced computed tomography angiography at 1-year follow-up demonstrating patent vascular reconstruction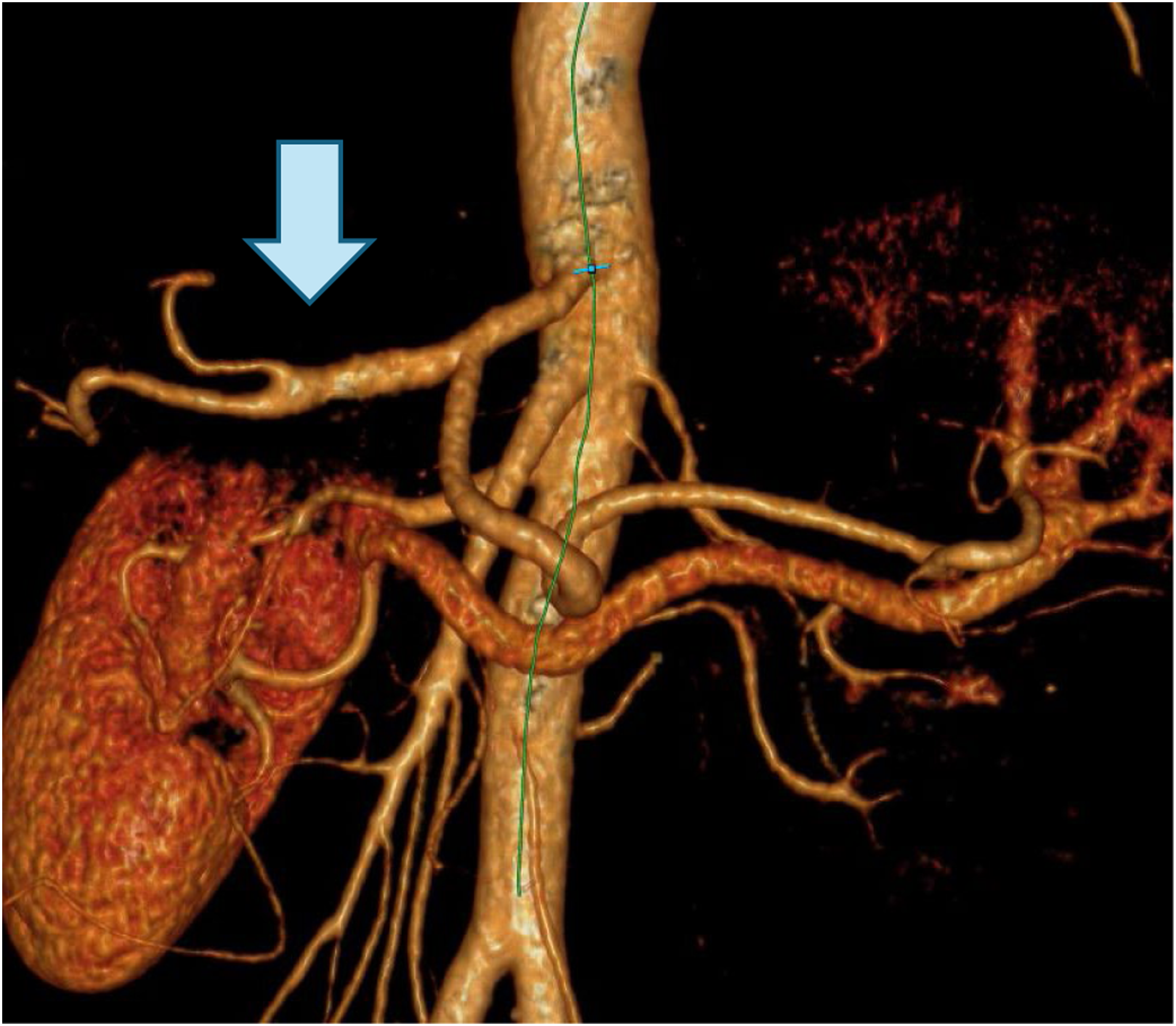
Discussion
Visceral artery aneurysms are rare but clinically significant due to the risk of rupture and associated high mortality.1,3,11 Most are asymptomatic and increasingly detected incidentally, while symptomatic cases may present with abdominal pain or gastrointestinal bleeding. 5
Current guidelines recommend treatment of symptomatic aneurysms and those larger than 2 cm. 7 Endovascular techniques are increasingly used due to lower perioperative morbidity, 8 but their applicability may be limited by unfavorable anatomy, involvement of major branches, or infection. In such cases, open surgical repair remains an important treatment option.9,10
In our series, all 3 cases demonstrated anatomical features unfavorable for endovascular repair. In Cases 1 and 2, involvement of the CT trifurcation precluded preservation of branch perfusion, while in Case 3, the distal location at the hepatic artery bifurcation and associated infection made endovascular treatment unsuitable.
Preservation of Visceral Perfusion and the Choice of Conduit
The overarching principle in open reconstruction of VAAs is the preservation of hepatic and splenic perfusion. In Case 2, preservation of the splenic artery was necessary to maintain perfusion of the spleen and distal pancreas, particularly given the absence of a well-developed collateral network such as Kirk’s arcade. In these situations, maintenance of splenic arterial flow is critical to avoid ischemic complications, as collateral circulation may be insufficient.
Autologous vein grafts, particularly reversed GSV, are widely favored due to their biocompatibility, durability, and resistance to infection. 2 In Case 1, splenic artery interposition was used successfully to maintain perfusion, while in Case 2, a GSV conduit facilitated an aorto-hepato-splenic bypass. The literature supports the use of autologous grafts as the first-line material when reconstructing visceral circulation, especially in younger patients or those with infection risk. 12
Previous reports have described the use of the splenic artery for visceral revascularization.13,14 Nevertheless, the use of the splenic artery must be carefully considered. Potential risks include splenic ischemia and pancreatic injury. Preoperative imaging and intraoperative assessment are essential.
Our case contributes to this limited body of evidence by demonstrating the feasibility of using the splenic artery as an interposition graft following celiac trunk aneurysm resection. In this context, the artery provided a well-matched conduit for hepatic arterial reconstruction, preserving perfusion and maintaining long-term patency at follow-up.
To the best of our knowledge, Case 3 is the first reported case in which resection of a visceral artery aneurysm was followed by arterial reconstruction utilizing SFA as conduit.
Gastric Ischemia Risk
In Case 1, the patient developed gastric ischemia requiring total gastrectomy with Roux-en-Y reconstruction. This complication was likely related to ligation of the left gastric and splenic arteries in the setting of prior Billroth I gastrectomy, which may have compromised the collateral gastric circulation. Normally, gastric perfusion is maintained by a robust collateral network between the left and right gastric, gastroepiploic, and short gastric arteries. 15 Patients with previous gastric surgery are at increased risk of ischemia following ligation of the left gastric or celiac artery, as the normal collateral network may be disrupted.15-17 In Case 2, ligation of the left gastric artery was well tolerated, with no ischemic consequences.
Complications and Reintervention
Open repair of VAAs carries inherent risks, particularly in anatomically demanding cases. Case 3 illustrates this well: the patient developed a postoperative hematoma requiring early re-laparotomy. Although no active bleeding was detected, prompt surgical reintervention was essential to evacuate the hematoma, relieve mass effect, and confirm graft integrity. The literature consistently emphasizes that postoperative complications—including bleeding, graft thrombosis, and ischemic sequelae—must be anticipated, and that early recognition and timely re-exploration are critical to patient outcomes.17,18
Role of Multidisciplinary Management
Management of visceral artery aneurysms often requires a multidisciplinary approach. In our series, coordination between vascular surgeons, gastroenterologists, anesthesiologists, and intensive care specialists was essential, particularly in cases involving combined vascular and gastrointestinal procedures. Such collaboration is important in complex reconstructions to optimize perioperative management and outcomes.19,20
Importance of Preoperative Imaging
Preoperative imaging, particularly contrast-enhanced CT angiography, was crucial to diagnosis and surgical planning. In addition to confirming aneurysm size and morphology, CT allowed delineation of arterial anatomy, detection of anatomic variants, and assessment of collateral circulation. This information was invaluable in planning revascularization strategies and anticipating ischemic risks. Advanced imaging has been repeatedly shown to improve surgical decision-making and reduce intraoperative surprises.21-23
Several retrospective studies have reported favorable outcomes following open repair of VAAs, with low perioperative mortality and durable long-term patency.24,25 Our results are consistent with these findings, with all patients demonstrating patent reconstructions and preserved visceral perfusion at follow-up.
This study has several limitations. It represents a small, single-center case series of 3 patients, which limits the generalizability of the findings. In addition, the absence of a comparison group precludes direct evaluation against endovascular approaches. Therefore, our observations should be interpreted with caution and viewed as illustrative of surgical strategies rather than definitive evidence.
Conclusion
Open surgical repair remains an important treatment option for complex visceral artery aneurysms when endovascular approaches are not feasible. Careful preoperative imaging is essential for accurate anatomical assessment and operative planning. Individualized reconstruction strategies, including the use of autologous conduits, allow preservation of visceral perfusion in challenging cases. Particular attention should be given to collateral circulation, especially in patients with prior gastric surgery, to reduce the risk of ischemic complications. Multidisciplinary management and vigilant postoperative surveillance are important for optimal outcomes. While limited by the small number of cases, this series highlights practical surgical considerations and supports individualized management of VAAs.
Footnotes
Acknowledgements
The authors would like to thank the multidisciplinary team involved in the management of these patients, including anesthesiologists, intensive care specialists, and nursing staff, for their contribution to perioperative care and patient outcomes.
Ethical Considerations
The patients provided written informed consent for the use of photographs obtained during their surgical treatment.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
