Abstract

What are visceral artery aneurysms (VAAs)?
Visceral arteries are blood vessels that branch off the aorta (the main blood vessel in the body) and supply oxygen-rich blood to various organs in the abdomen including the liver, spleen, and intestines. The renal arteries supply blood flow to the kidneys and visceral arteries supply blood to other abdominal organs and intestines. An aneurysm is a bulge or expansion of a blood vessel; it is called an aneurysm when the diameter is more than 50% larger than the vessel’s normal diameter. As shown in Figure 1, aneurysms can be either ‘true’, wherein there is a weakening/thinning of the whole vessel wall leading to ballooning of the artery, or ‘false’, when a breach in the wall causes blood to leak and collect in a confined space near the vessel, also called a ‘pseudoaneurysm’. True aneurysms are further classified as fusiform (the entire artery is dilated) or saccular (one side of the artery is dilated). The pouch-like bulge is referred to as the ‘aneurysm sac.’ Figure 2 shows the location of various VAAs.
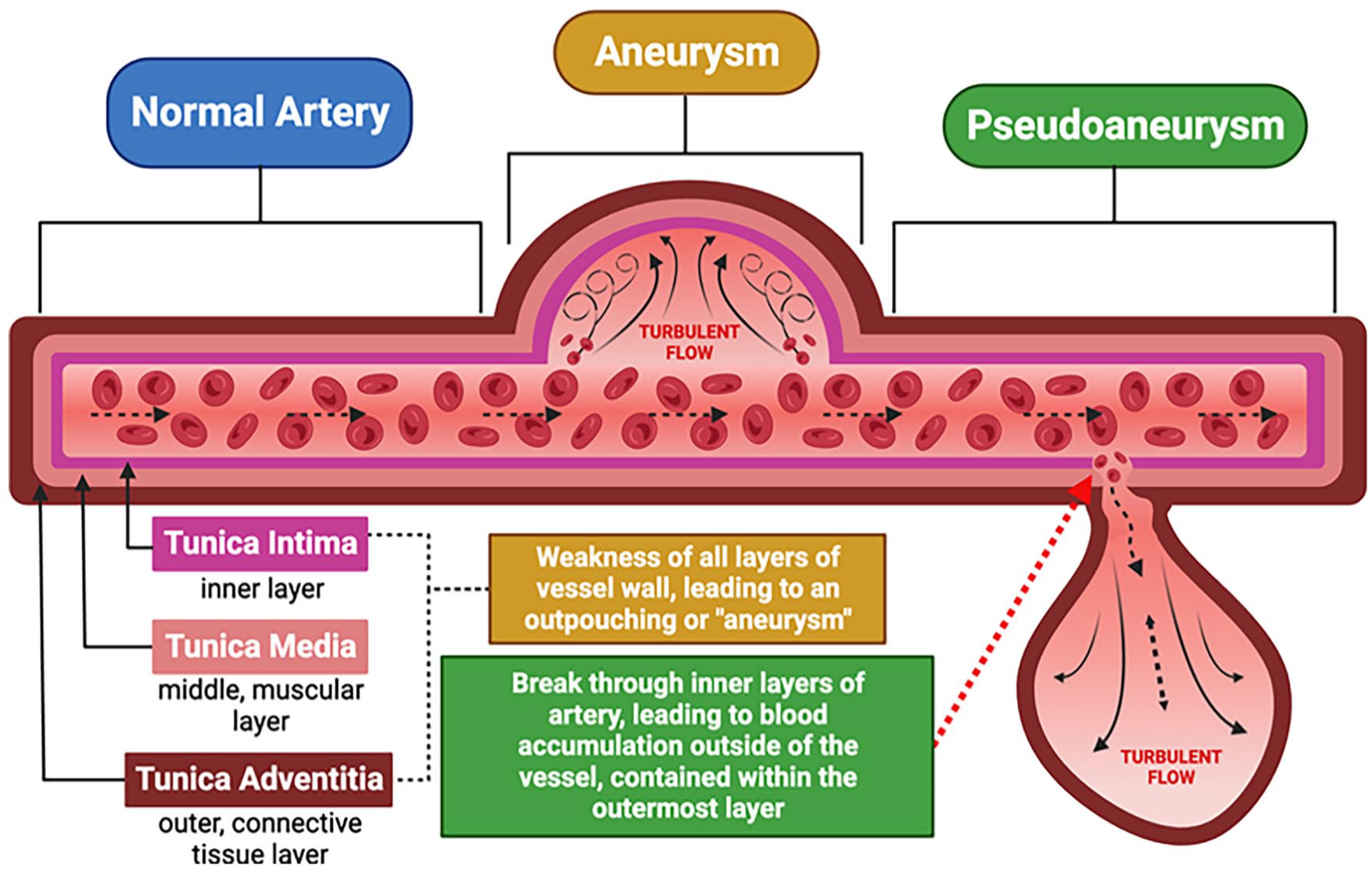
Normal artery appearance, true aneurysm, and pseudoaneurysm.
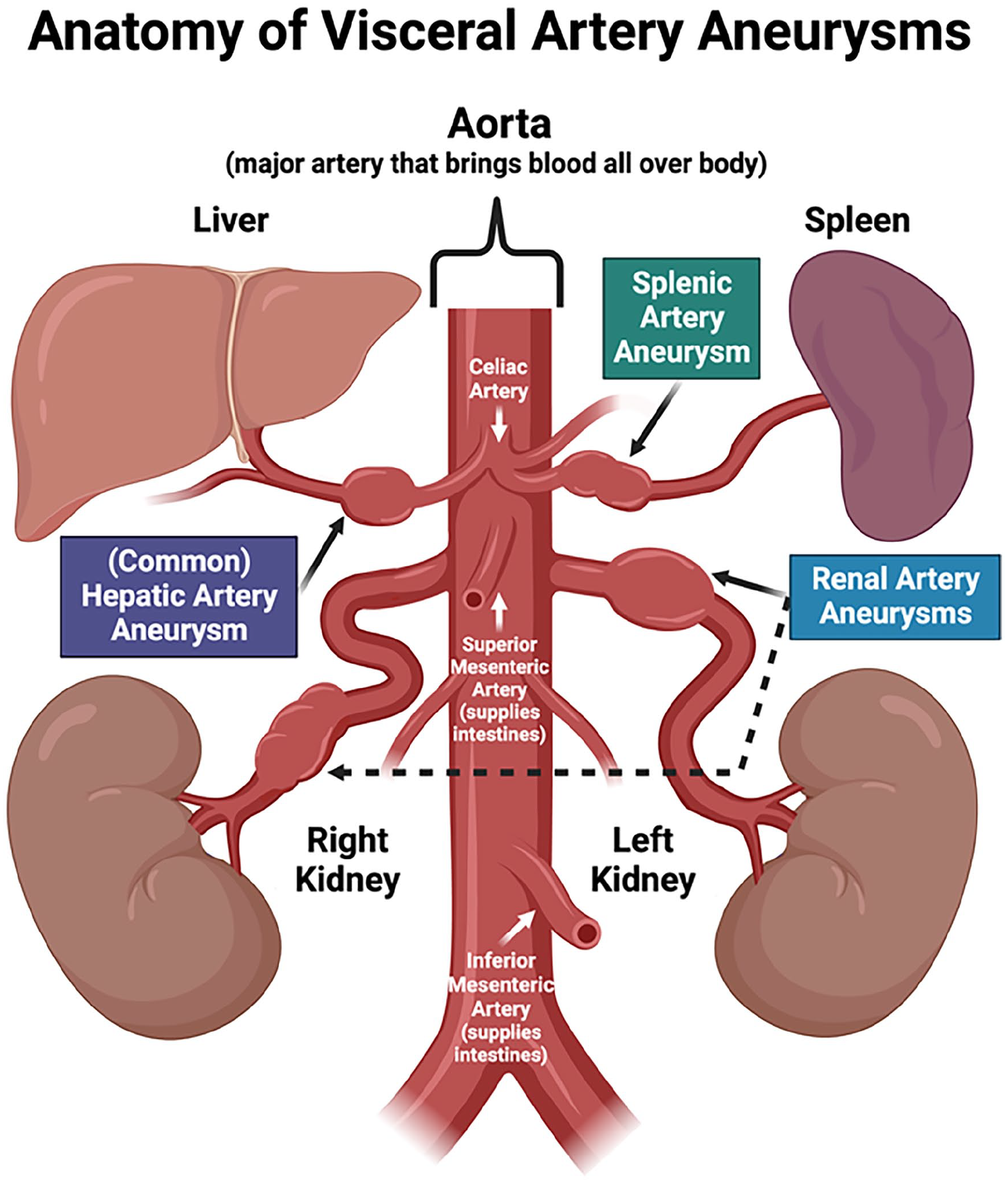
Visceral artery aneurysms (VAAs) based on their location within the abdomen. 2 Visceral arteries include the hepatic artery (supplies the liver), splenic artery (supplies the spleen), and various mesenteric arteries (supply different parts of the intestines). The renal arteries supply the kidneys.
What are renal artery aneurysms (RAAs)?
Renal artery aneurysms are sometimes discussed separately from VAAs in the medical literature because of their unique symptoms and associated medical conditions. RAAs are most commonly seen in patients with fibromuscular dysplasia (FMD), 1 a rare vascular condition in which the walls of certain arteries undergo abnormal changes causing vessels to narrow, dilate, or, rarely, develop a tear (dissection). In patients who have an RAA, around 68% are diagnosed with FMD. 2 High blood pressure can occur with RAAs, which is not the case with other VAAs. For simplicity, in the discussion that follows, we have grouped RAAs under VAAs as they share a similar approach to diagnosis and treatment.
How common are VAAs?
VAAs are uncommon and account for around 5% of all aneurysms in the abdomen. 2 Abdominal aortic aneurysms 3 are much more common and can be seen in association with VAAs in 20% of cases. 4 The most common locations for VAAs are the renal arteries, followed by the splenic artery, and then the hepatic artery. Splenic artery aneurysms (SAAs) are more common in women of childbearing age. 4 RAAs are most frequently reported in middle-aged White women. Hepatic artery aneurysms (HAAs) are commonly seen in men between 40 and 60 years of age.2,4
What causes VAAs?
Although the exact cause is often unknown, certain medical conditions and risk factors can increase the chances of developing a VAA. Factors that can lead to the development of VAAs are listed below:4,5
Atherosclerosis
Medial degeneration
Abdominal or surgical trauma
After an artery dissection
After an abdominal or blood infection (sepsis, endocarditis, or pancreatitis)
Inflammation of the blood vessels (vasculitis, systemic lupus erythematosus, polyarteritis nodosa)
Fibromuscular dysplasia (FMD)
Connective tissue disorders and hereditary diseases (Marfan, Ehlers–Danlos, Osler–Weber–Rendu syndromes)
Pregnancy
Idiopathic (unknown cause).
True aneurysms are most commonly caused by atherosclerosis, which is the build-up of cholesterol plaque in the artery; the vessel wall weakens, leading to the formation of an aneurysm. Loss of the elastic fiber and smooth muscle cells within the wall of the vessel (called ‘medial degeneration’) is the second most common cause. Injury to the vessel wall after trauma or surgery, or after a dissection (tear in the artery), can also cause a visceral or renal artery aneurysm. Other less common causes include vasculitis (inflammation of the blood vessel), FMD, 1 and inherited vascular connective tissue disorders (such as Marfan syndrome, vascular Ehlers–Danlos syndrome, or Loeys–Dietz syndrome). Cardiovascular risk factors like smoking and high blood pressure can promote aneurysm formation and expansion.4,5 Increased blood flow through vessels and hormonal changes during pregnancy can also increase the chances of aneurysm development.
What are the symptoms and potential complications of VAAs?
Most patients with VAAs have no symptoms. The aneurysms are often found incidentally on abdominal imaging done for another health concern. Symptoms of VAAs, in general, may include vague abdominal pain, nausea, or vomiting. In special situations like RAAs, the most common symptom is new or uncontrolled high blood pressure. Patients with RAAs may also experience pain in the flanks (sides and back of the abdomen) and, rarely, blood in the urine.
Rarely, a patient can present for the first time with a ruptured (burst) aneurysm, which is potentially fatal. In certain cases, patients may have a ‘double rupture phenomenon,’ where the aneurysm bursts but the rupture is initially contained, giving time to obtain medical assistance. Without intervention, if the aneurysm ruptures a second time, it can be fatal. When presenting as an emergency with a ruptured aneurysm, patients may experience severe, sharp abdominal pain, collapse, and/or loss of consciousness from blood loss.
How are VAAs diagnosed?
VAAs can be diagnosed using a variety of vascular imaging techniques, 6 which can be invasive or noninvasive (Table 1). The best method for detection of most VAAs is computed tomography angiography (CTA), which involves using contrast (dye) to visualize the blood vessels and aneurysm structure. Magnetic resonance angiography (MRA) is a reasonable alternative for individuals who have an iodinated contrast allergy, concern about risk of radiation exposure, or chronic kidney disease. Depending on the aneurysm location, duplex ultrasound is a helpful tool for follow up once an aneurysm has already been identified.
Advantages and disadvantages of vascular imaging techniques for VAAs.

CTA, computed tomography angiography; MRA, magnetic resonance angiography; VAA, visceral artery aneurysm; US, ultrasound.
Catheter-directed angiography is a more invasive technique, but it provides the most detailed view of the aneurysm and can help with treatment planning. 2 A catheter is inserted into a vessel in the wrist or groin and threaded up to the aneurysm. Contrast is inserted through the catheter, allowing for visualization of the architecture of the vessel and its branches on an X-ray (Figure 3).
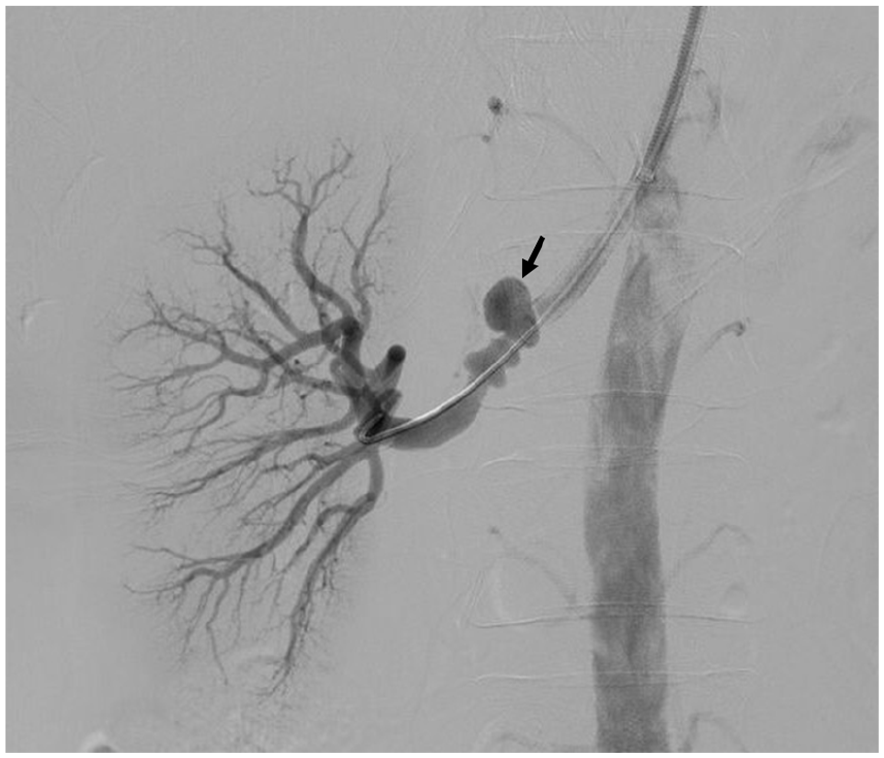
Renal artery angiogram of a mid-right renal artery aneurysm (arrow). Contrast (dye) shows the architecture of the renal artery and its branches, which can be useful for planning an intervention. This 38-year-old woman had uncontrolled hypertension, resulting in the diagnosis of fibromuscular dysplasia (FMD). The aneurysm was treated with a covered stent (not shown).
After diagnosis, imaging also plays an important role in monitoring the aneurysm(s) over time and in detecting other aneurysms. A one-time complete screening with a CTA may be performed when a VAA is found, as patients may have aneurysms in other blood vessels as well. 7
What is the treatment for VAAs?
Management depends on the patient’s symptoms as well as the size, location, and other features of the aneurysm. The patient’s age, risk factors, and other medical conditions also need to be considered. 5 Small aneurysms that remain stable and do not grow rapidly can be managed with medical treatment options (as below) and watchful monitoring. Large, rapidly growing aneurysms and aneurysms that cause symptoms often require invasive treatment.
What are the medical treatment options for VAAs?
Patients who have small, uncomplicated aneurysms without symptoms can be managed by controlling risk factors and routine imaging evaluation to monitor the size. Blood pressure control is essential to avoid the build-up of pressure within the aneurysm, which can lead to expansion and rupture. Statin medications are recommended for patients with atherosclerosis or high cholesterol, and smoking cessation is critical to prevent aneurysm growth and further weakening of the vessel wall. Overall, lifestyle changes aimed to improve cardiovascular health are important, such as a well-balanced diet and regular mild- to moderate-intensity exercise. In certain cases, aspirin may be recommended to avoid clot formation within the aneurysm sac and to prevent the migration of blood clots to other parts of the body. 2 Periodic imaging with CTA, MRA, or duplex ultrasound is used to monitor the size and rate of growth to determine if more invasive treatment is required.
When is invasive treatment required?
Guidelines for the management of VAAs have been published to assist vascular specialists in providing optimal treatment for patients with VAAs. The Society for Vascular Surgery clinical practice guidelines 2 provide recommendations on close clinical observation versus invasive treatment for the management of VAAs. The choice of procedure for repair is determined on the basis of location and features of the VAA.
General indications that warrant repair of VAAs:
All ruptured VAAs
All pseudoaneurysms of any size
Large size of VAAs (exact size is dependent on the affected artery)
Rapidly growing VAAs based on serial imaging over time
VAAs associated with symptoms (regardless of the aneurysm size).
Special situations that may warrant repair of VAAs:
RAAs when associated with significant narrowing of the artery (stenosis), which can be seen in FMD and can lead to difficult-to-control hypertension
Pregnant women or women of childbearing age
Patients undergoing liver transplant
Certain conditions that cause inflammation of the blood vessels (vasculitis).
What are the invasive treatment options for VAAs?
Options for treatment range from open surgery to minimally invasive repair called endovascular repair. The goal of VAA treatment involves blocking/cutting off the connection of the aneurysm sac to the vessel while preserving blood flow through the rest of the artery.
Open surgical repair
In the traditional open surgical approach (Figure 4), the surgeon makes an incision in the abdominal wall to visualize the aneurysm. The artery is temporarily clamped to stop blood flow and the aneurysm is clipped/removed. Depending on the situation, the rest of the artery may then be repaired using a vein or synthetic graft to replace the portion of the artery that was removed. In some cases, if the body part being supplied by the artery is compromised, that organ may need to be removed; for example, there may be a need for partial or total removal of the spleen with SAA. Open surgical repair may be used to treat ruptured VAAs and in situations with complex aneurysms. Though this approach is riskier with a longer hospital stay and longer recovery period, it is sometimes the best approach. 4
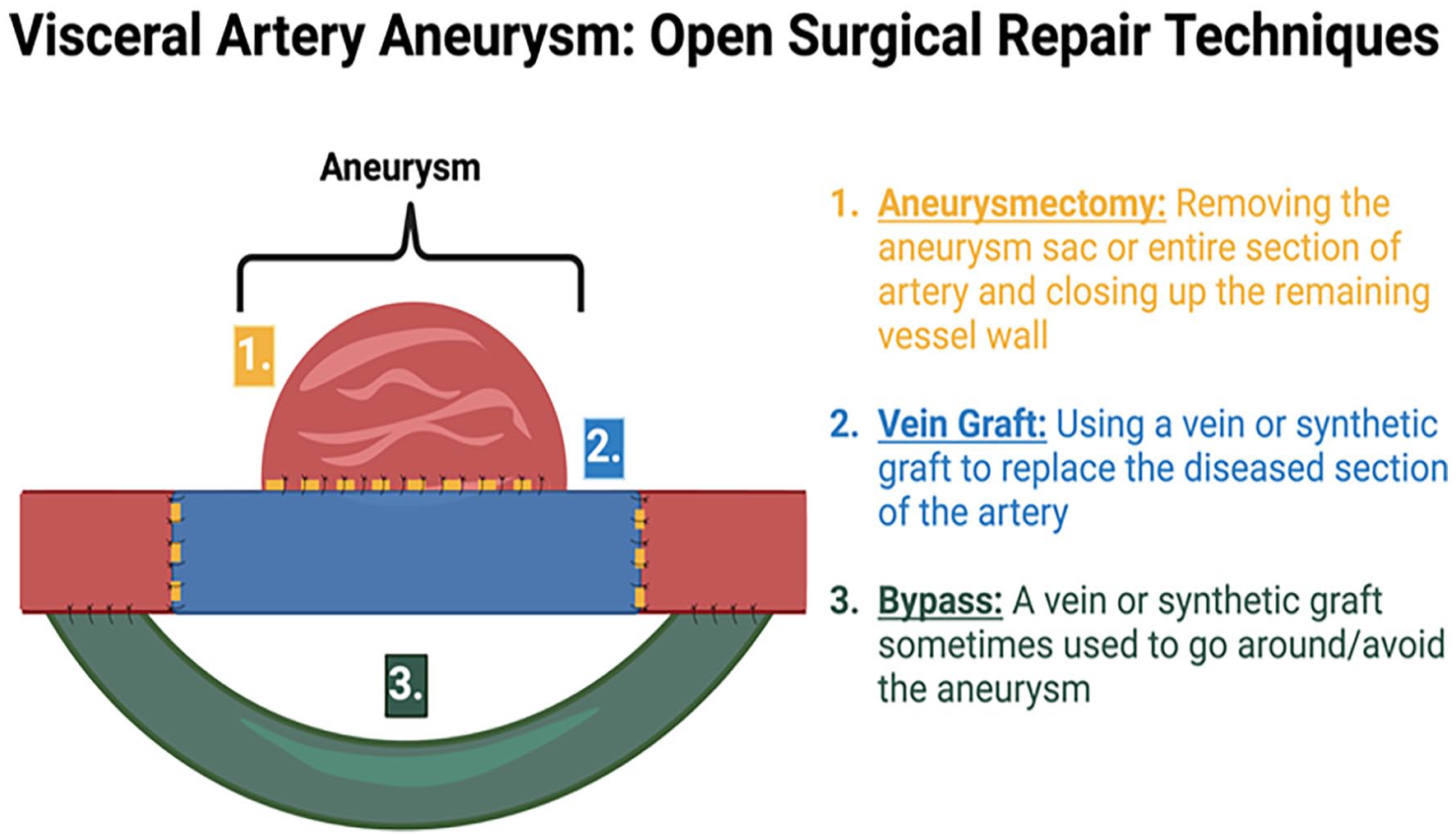
Open surgical repair of an aneurysm involving different techniques to isolate and remove the aneurysm sac from the rest of the artery.
Endovascular repair
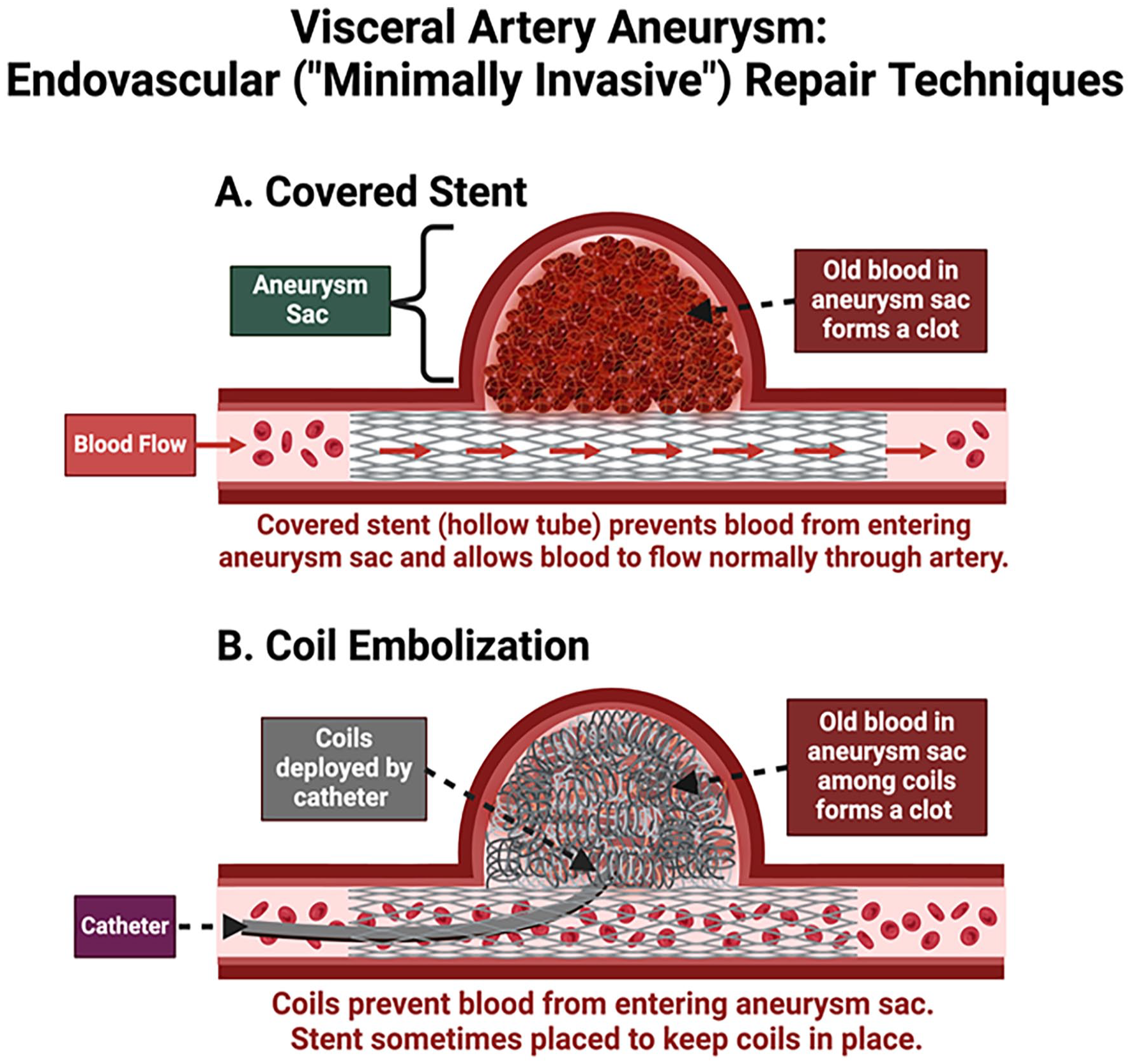
Endovascular repair (Figure 5) is a minimally invasive procedure to insert a catheter through a vessel in the groin which is directed to the aneurysm under guidance of X-ray. Two techniques can be performed using this approach:
Embolization: This technique involves the introduction of material (glue or coils) into the aneurysm to clog the aneurysm sac. The material can fill the aneurysm causing it to form a clot, thus blocking off its connection to the artery.
Covered stent: A covered mesh-like tube called a stent can be introduced into the vessel, lining the artery above and below the aneurysmal portion, thus blocking off the connection of the aneurysm to the vessel. The hollow tube-like nature of the stent allows blood to continue to flow through the vessel but not into the aneurysm. The blood left behind within the aneurysm eventually forms a clot, thus sealing off the aneurysm sac from the vessel.
(
Endovascular approaches have lower complication rates and are usually the first procedure offered if the patient’s aneurysm size, location, and architecture could be best addressed by endovascular repair. The procedure is done under sedation and local anesthesia, with a faster recovery period and shorter hospital stay. Disadvantages of endovascular repair include a higher potential for failure and the risk of obstruction of blood supply to the organs that the artery supplies.
Summary
VAAs are uncommon dilations of arteries that supply blood to the various organs in the abdomen. They are frequently diagnosed on imaging done for another reason or for nonspecific symptoms like vague abdominal or flank pain. Although most patients do not have symptoms, these aneurysms, if large enough, can cause an acute emergency due to rupture. CTA is the recommended imaging modality to diagnose and monitor VAAs. The treatment plan depends on symptoms, the aneurysm’s location, size, and growth rate, as well as other patient characteristics. Small, stable aneurysms without symptoms can be regularly monitored with imaging and treatment of risk factors. However, ruptured aneurysms, pseudoaneurysms, symptomatic aneurysms, and large visceral aneurysms warrant repair.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Dr Elizabeth Ratchford’s work was supported in part by the generosity of David Kotick (1926–2021).
