Abstract
Objective
The presence of a significant left subclavian artery stenosis may occasionally lead to blood flow reversal through a LIMA-to-coronary artery bypass graft during left arm exertion; with “stealing” of myocardial blood supply. The aim of this study was to review our experience with carotid-subclavian bypass in patients with post-CABG coronary-subclavian steal syndrome.
Methods
This is a retrospective review of all patients who underwent carotid-subclavian bypass grafting for post-CABG coronary-subclavian steal syndrome at Mainz University Hospital between 2006 and 2015. Cases were identified in our institutional database, and data were retrieved from surgical records, imaging studies, and follow-up records.
Results
Nine patients (all males, mean age of 69.1 years) underwent surgical treatment for post-CABG coronary-subclavian steal syndrome. Medium interval between original CABG and carotid-subclavian bypass grafting was 86.1 months. There were no perioperative deaths, strokes or myocardial infarctions. At a mean follow-up period of 79.9 months, all patients remained asymptomatic and all carotid-subclavian bypass grafts remained patent. One patient required stenting of a common carotid artery stenosis proximal to the graft anastomosis site, and coronary artery stenting was required in four patients in regions other than those supplied by the patent LIMA graft.
Conclusion
Carotid-subclavian bypass surgery is a safe treatment option even in patients with multivessel disease and severe comorbidities and should be taken into consideration in patients who are deemed fit for surgery and those who would benefit from the excellent long-term patency rates.
Introduction
Coronary-subclavian steal syndrome is becoming increasingly recognized as a potentially serious phenomenon. 1 This condition occurs when the presence of a significant left subclavian artery stenosis proximal to the ostium of the left internal mammary artery (LIMA) leads to blood flow reversal through a LIMA-to-coronary artery bypass graft during left arm exertion; with “stealing” of myocardial blood supply and possible fatal consequences.
There are no set guidelines for the management of coronary-subclavian steal syndrome, and literature concerning surgical treatment is comprised mainly of case reports and a handful of limited series of patients. We present here our experience with a group of patients with post-coronary artery bypass grafting (CABG) coronary-subclavian steal syndrome who were managed with carotid-subclavian bypass grafting.
Patients and Methods
This is a retrospective review of nine consecutive patients who underwent carotid-subclavian bypass grafting for post-CABG coronary-subclavian steal syndrome at Mainz University Hospital between January 2006 and December 2015. Cases were identified retrospectively in our institutional database using relevant key words (namely: subclavian stenosis, subclavian steal, carotid-subclavian bypass and coronary-subclavian steal syndrome), and data were retrieved from surgical records, imaging studies, and follow-up records. Ethical approval was obtained from the institutional ethical committee, and informed consents werewaived due to the retrospective and observational nature of this study.
All patients who were diagnosed with subclavian artery stenosis at our hospital over the past 15 years were reviewed. Subclavian artery stenosis was suspected in the presence of a notable difference in arterial blood pressure between the two arms, and was verified with CT-angiography, MRI-angiography or conventional contrast angiography. A significant obstruction was defined as either a symptomatic stenosis, a stenosis in excess of 70% or a stenosis which was associated with blood flow reversal in the LIMA graft. Decison to carry out surgical management was based on interdisciplinary discussions, and left carotid-subclavian bypass grafting was the sole mode of surgical correction.
Surgical Technique
Surgery was performed under general anaesthesia, with standard cardiovascular monitoring (ECG, pulse-oxymetry, invasive arterial blood pressure and cerebral oxygen saturation). A left-sided transverse supraclavicular incision was performed to expose the left common carotid artery, left subclavian artery and the origin of the left internal mammary artery. Heparin (70 IU/Kg) was given intravenously, and a 6-8 mm polyethylenterephthalat (Dacron®) tube graft was anastomosed end-to-side to the subclavian artery distal to the origin of the left internal mammary artery while the subclavian artery was temporarily clamped beyond the left internal mammary artery origin. The vascular graft was passed behind the sternocleidomastoid muscle and the internal jugular vein, and its opposite end was anastomosed to the left common carotid artery employing a similar technique. Heparin was reversed with an appropriate dose of protamine. Standard cardiovascular monitoring was continued postoperatively in the intensive care unit for 24 hours, and all patients were started on at least one antiplatelet or antithombotic drug in addition to statins. Duplex ultrasound, CT-angiography or MRI- angiography was performed early postoperatively to confirm carotid-subclavian graft patency prior to discharge from hospital. Early follow-up was scheduled at 3 months, and at 6-months’ or 12-months’ intervals thereafter at the discretion of the supervising physician.
Results
Patients‘ demographics and risk factors.
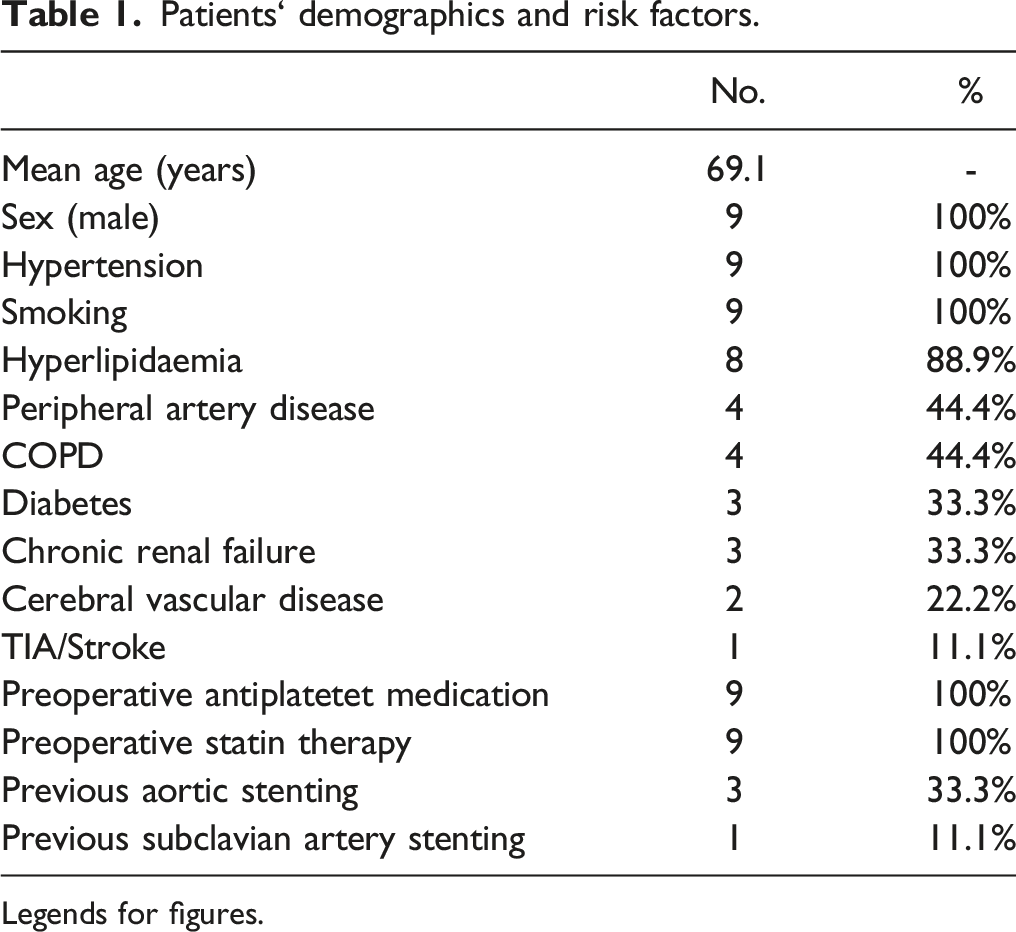
Legends for figures.
Surgical Data and Early Results
Median interval between original CABG and carotid-subclavian bypass grafting was 86 ± 65.7 (sd) months (range 2-213 months). All carotid-subclavian bypass grafts were established between the left common carotid artery as the inflow source and the left subclavian artery distal to the origin of the left internal mammary artery. Vascular graft diameters were chosen to match grafted vessels, and those were 6 mm (1 patient), 7 mm (5 patients) and 8 mm (3 patients). Mean operating time was 150.8 (±30) minutes, and mean carotid artery clamping time was 13.6 (±6) minutes. One patient underwent concomitant thoracic endovascular aortic repair of penetrating aortic ulcer and one patient underwent repeated coronary revascularization.
There were no incidents of perioperative death, stroke, myocardial ischemia, peripheral perfusion deficit or surgical site infection, nor did we encounter phrenic nerve injuries or lymph leaks. Median in-hospital stay following surgery was 7.0 ± 3.1 (sd) days. Graft patency was confirmed in all cases prior to discharge with CT-angiography (6 patients), MRI-angiography (1 patient) and duplex ultrasound (1 patient) [Figure 1]. Postoperative 3D-CT reconstruction of aorta (volume rendering) showing the aortic arch, left carotid-subclavian bypass and LIMA-coronary bypass graft.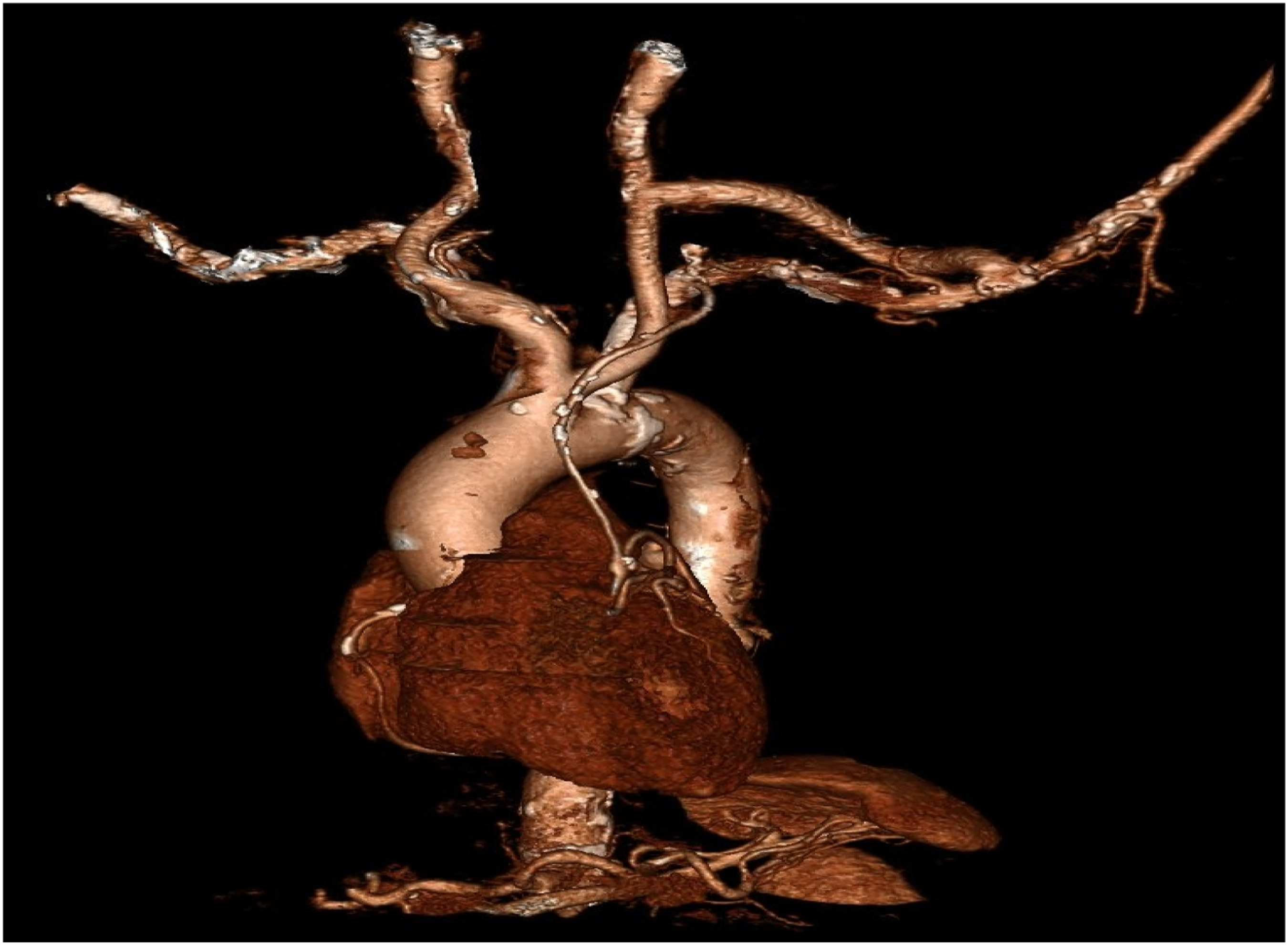
Long-Term Follow-Up
Mean follow-up time was 79.9 months (median 69 ± 57.5 months, range 12-188 months). Follow-up data was complete for 8 patients, while one patient was lost to follow-up 33 months after the surgery, hence his last visit at our institution was considered as the latest follow-up.
Four patients died after a medium of 66.8 months (median 50 ± 53.2 months) following surgery, and none were related bypass surgery. All patients remained free of symptoms of coronary-subclavian steal or left arm ischemia, and all carotid-subclavian bypass grafts remained patent during follow-up, as confirmed by CT-angiography, MRI-angiography or duplex ultrasound [Figure 2]. One patient required stenting of a common carotid artery stenosis proximal to the graft anastomosis site, and coronary artery stenting was required in four patients in regions other than those supplied by the patent LIMA graft. Finally, two patients suffered late strokes in regions other than those supplied by the left carotid artery. (A) Pre-operative MRA showing a severe stenosis of left subclavian artery proximal to the LIMA (yellow arrows). (B) Postoperative MRA showing a left carotid-subclavian bypass grafting.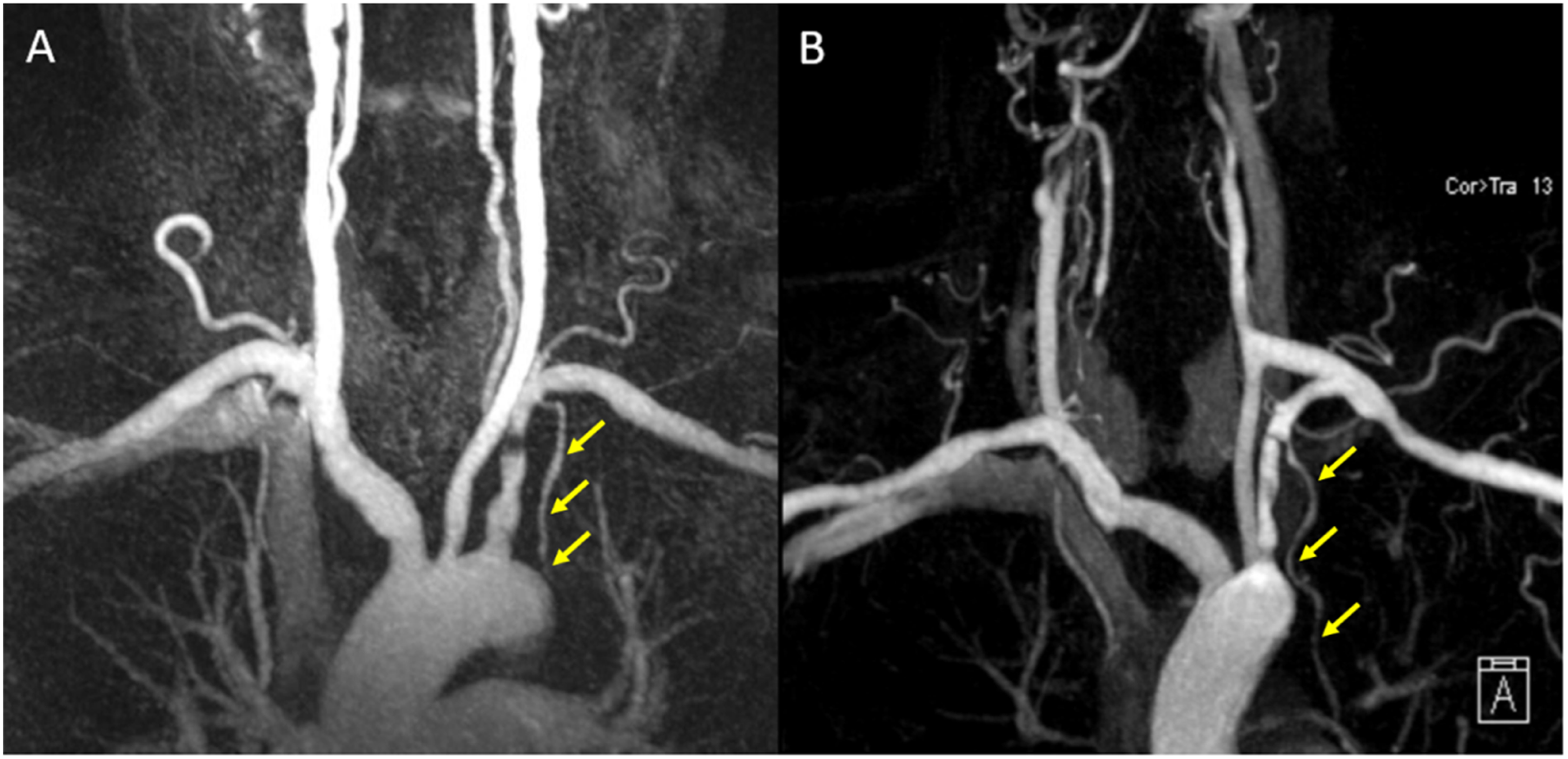
Discussion
Coronary-subclavian steal syndrome is reported to occur in .2% to 6.8% of patients with LIMA coronary bypass grafts several years after undergoing coronary surgery.1–6 Subclavian stenoses may be due to atherosclerosis, vasculitis, thoracic-outlet syndrome or sequelae of radiation therapy. 6 Iatrogenic occlusion of left subclavian artery can also occur during stenting of aortic aneurysms or dissections. 7
While endovascular stenting is possible in many cases, stenotic subclavian lesions (especially complete occlusions) may occasionally require surgical intervention, with comparable perioperative stroke and mortality rates. 2 Importantly, patency rates following stenting of the left subclavian artery are reportedly inferior to those after bypass surgery. AbuRahma et al conducted a comparative study involving 172 patients, and showed that patency rates following stenting were 93% at one year and 70% at 5 years while patency rates following bypass surgery were 100% and 96%, respectively. 8 Similarly, in a group of 252 patients, Song et al reported similar results with 1-year patency rates of 91% following stenting and 99% following bypass surgery, with a growing margin reaching 49% and 89% after 10 years of follow-up, respectively. 9
Our experience shows that carotid-subclavian bypass surgery is a safe treatment option, even in patients with multivessel disease and severe comorbidities. None of our patients suffered from perioperative myocardial infarction or stroke and all patients remained symptom-free 79.9 month after surgery. We believe that carotid-subclavian bypass should be taken into consideration in patients who are deemed fit for surgery and those who would benefit from excellent long-term patency rates. With comparably low perioperative morbidity and mortality rates and superior long-term patency and freedom of re-intervention, its arguable whether a “surgery-first” approach should be recommended for patients with coronary-subclavian steal syndrome.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Ethical approval was obtained from the institutional ethical committee, and informed consents were waived due to the retrospective and observational nature of this study.
Central Message
Carotid-subclavian bypass is a safe and feasible treatment option in patients with coronary-subclavian steal syndrome, and should be taken into consideration in patients who are deemed fit for surgery and those who would benefit from the excellent long-term patency rates.
Perspective Statement
The coronary-subclavian steal syndrome is a rare but potentially life-threatening condition, and should therefore be known to physicians who are managing patients following coronary artery bypass surgery.
