Abstract
Purpose
To present stent-graft treatment of floating thrombus in the abdominal aorta. A review of the literature about aortic floating thrombus (AFT) was also performed.
Case report
A 56-year-old female with no risk factors for vascular disease but with history of a mild COVID-19 infection in the previous month, for which she had started anticoagulant therapy at a prophylactic dosage, developed an acute ischemia of the lower limbs and was diagnosed with floating thrombosis of the abdominal aorta. The thrombus was excluded from the aortic blood flow by deployment of a stent-graft in the abdominal aorta. At 12 months, the patient was well, and the thrombus in the abdominal aorta appears to be completely excluded by the stent-graft. A review of the available literature from 1980 to 2022 showed 74 cases of AFT located in the aortic arch, in the descending thoracic and in the abdominal aorta. In most cases the AFT involved the aortic arch (38/74, 51.3%) and/or the descending thoracic aorta (30/74, 40.5%), while the abdominal aorta was involved in 6 cases. In 2 of these 6 cases, the patients had a COVID-19 infection. The AFT was mostly approached either medically with anticoagulation/systemic thrombolysis (32/74, 43.2%) or with surgical removal (31/74, 41.9%), while endovascular coverage of the thrombus with an endograft was performed in 6 cases of AFT located in the aortic arch and in the descending thoracic aorta (3 cases each).
Conclusion
There is no consensus about the optimal treatment of AFT. In selected cases, abdominal stent-grafts may be used for stabilization and exclusion of symptomatic abdominal aorta floating thrombosis to prevent progression and recurrent embolization.
Keywords
Introduction
Since the beginning of the pandemic of coronavirus disease (COVID-19), increasing evidence suggests that severe acute respiratory syndrome coronavirus-2 (SARS CoV-2), might produce hypercoagulable state with subsequent arterial and venous thrombosis and thromboembolism, through various pathophysiological mechanisms, such as endothelial dysfunction, activation of coagulation cascade, immobilization, hypoxia and systemic inflammation. 1
Venous thromboembolism has been frequently described in severe cases, 2 conversely there are fewer reports of arterial thrombosis and embolism. 3
While venous thrombosis is usually managed conservatively with appropriate anticoagulation therapy, arterial thrombosis may present with catastrophic clinical pictures requiring urgent surgical treatment.
Here we report a unique case of endovascular treatment of abdominal aorta floating thrombus in a SARS-CoV-2 positive patient presenting with acute leg ischemia. Furthermore, a review of the literature about aortic floating thrombus (AFT) was performed.
Case Report
Informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
A 56-year-old female was admitted to the hospital due to the severe pain in the right leg. Symptoms started few hours earlier and consisted in acute pain of the right lower limb associated with paleness, coldness and numbness. In the previous days the patient complained also for weakness of her left leg.
Her medical history was unremarkable, except for a mild COVID-19 infection in the previous month, for which she had started anticoagulant therapy at a prophylactic dosage (enoxaparin 40 mg/day).
She was nonsmoker and she did not report any previous limb claudication nor arterial or venous thromboembolism.
According to our Institutional protocol based on the COVID-19 outbreak, SARS-CoV-2 throat swab was performed and tested positive; however she was asymptomatic at the admission. The patient was then hospitalized in a COVID Unit.
Blood analysis showed a high level of C-reactive protein (8.9 mg/dL; cutoff < .5), hyperleukocytosis (13’850/μL), high fibrinogen (765 mg/dL; normal range 200-400), low hemoglobin rate (10.4 g/dL; normal range 12-16), normal platelet count (211’000/μL) and normal renal function, while there were signs of hepatic cytolisis. The D-dimer level was high (1.18 μg/mL; cutoff < .5) and the activated partial thromboplastin time (aPTT) was normal. There was no laboratory sign of disseminated intravascular coagulation.
At clinical examination, both legs was pale and cold. Femoral pulses was present, while popliteal and peripheral pulses were not palpable on both side. Color duplex scan revealed total occlusion of the right superficial femoral artery from the middle third of the thigh and extended up to the popliteal artery; tibial vessels showed no Doppler signal.
In addition, a total occlusion of the contralateral superficial femoral-popliteal axis was found with reperfusion flow at the origin of the tibial vessels.
The electrocardiogram (EKG) was normal. Transthoracic echocardiography showed preserved contractile function of the left ventricle (ejection fraction 59%), without pathological findings on the cardiac valves. No thrombotic formations or vegetations were detected.
Contrast-enhanced computed tomography angiography (CTA) was performed. At the chest focal ground-glass parenchymal thickening associated with fibrotic-disventilative striae was present bilaterally at the lung bases, without any signs of pulmonary embolism. At the level of the abdomen, a floating eccentric thrombosis was found in the aorta, starting about 3 cm below the renal arteries and extending for about 5 cm more caudally, where it occupied almost 50% of the lumen (Figure 1(A)-(B)). The presence of a left inferior renal polar artery was noted (Figure 1(C)). Preoperative angio-CT scan showing the presence of the floating thrombus in the abdominal aorta (1a, on the left side), starting about 3 cm below the lowest renal artery and extending for about 5 cm caudally (1b, in the middle). The yellow arrow on figure 1(c) (on the right) shows the polar renal artery.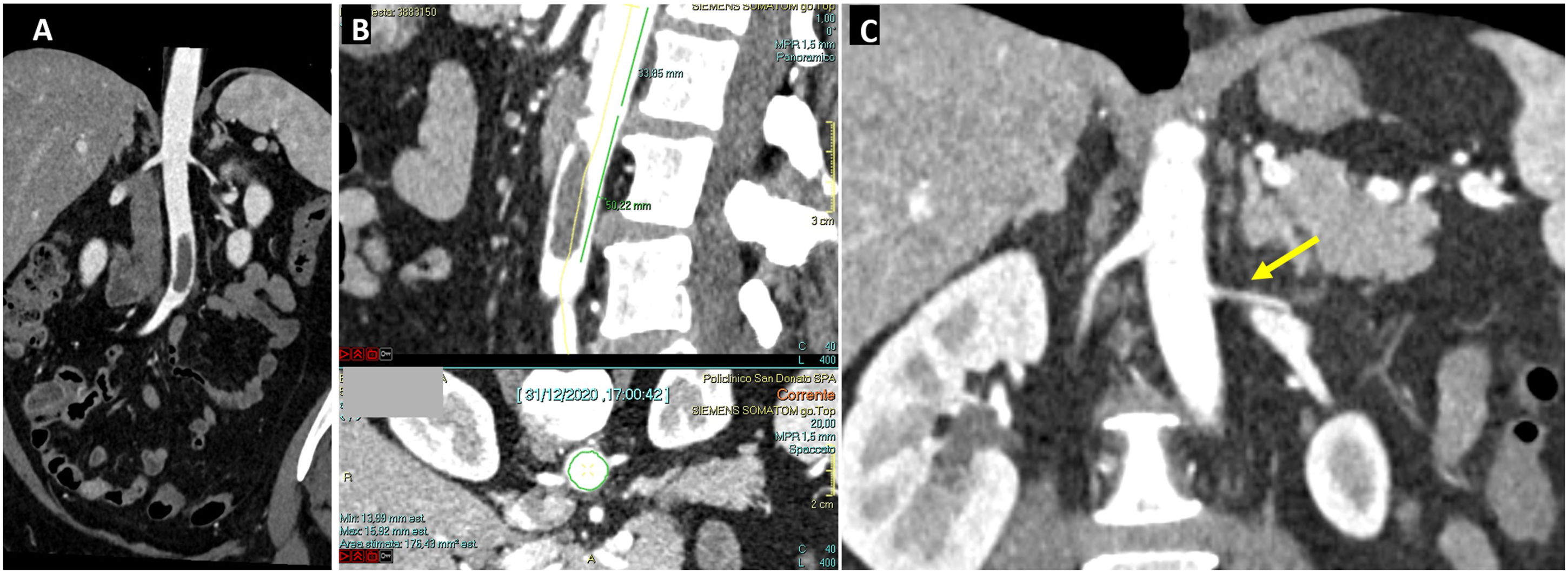
No more embolic lesions affecting the other organs were detected.
Considering the recent history of recurrent peripheral embolism despite anticoagulation therapy, the unstable nature and site of the thrombus, the patient was scheduled for endovascular treatment with Gore Excluder stent-graft (W.L. Gore and Associates, Flagstaff, AZ) to cover the thrombus, besides femoral-popliteal bilateral Fogarty thrombectomy. Planning and sizing were performed on CTA multiplanar, 3-dimensional and centerline reconstructions using 3Mensio image processing software (Pie Medical Imaging, Bilthoven, The Netherlands).
Surgery was therefore undertaken on an emergent basis. Under epidural anesthesia and with bilateral surgical femoral approach, a Gore Excluder iliac extension (9.5 cm long and 16 mm in diameter) was advanced and then released from the right access proximally up to the lower edge of the left renal artery and distally immediately above the aortic bifurcation.
Bilateral embolectomy with Fogarty catheter of the iliac and femoral-popliteal arteries was performed, with removal of fresh thrombus on both sides. At the end of the procedure, flushing from the femoral artery showed excellent antegrade flow and backflow.
At the end of the procedure both legs were warm and well perfused at duplex scan, with peripheral pulses that could be appreciated by hand.
The patient was then placed on anticoagulant therapy with enoxaparin (6000 International Units twice a day).
On the following day, signs of recurrent acute right limb ischemia with a cold and pale foot were present. Color-Doppler ultrasound examination revealed recurrence of total occlusion of the right superficial femoral artery from the middle third of the thigh and extended up to the popliteal artery, without any flow at the level of the tibial vessels.
Angio-CT scan was performed, showing regular patency of the visceral and renal arteries without signs of embolic lesions, as well as regular patency of the stent-graft at the level of the abdominal aorta (Figure 2). Postoperative angio-CT scan on the first postoperative day (2A) and 3D reconstruction (2B) showing the patency of the abdominal aorta with coverage of the thrombus.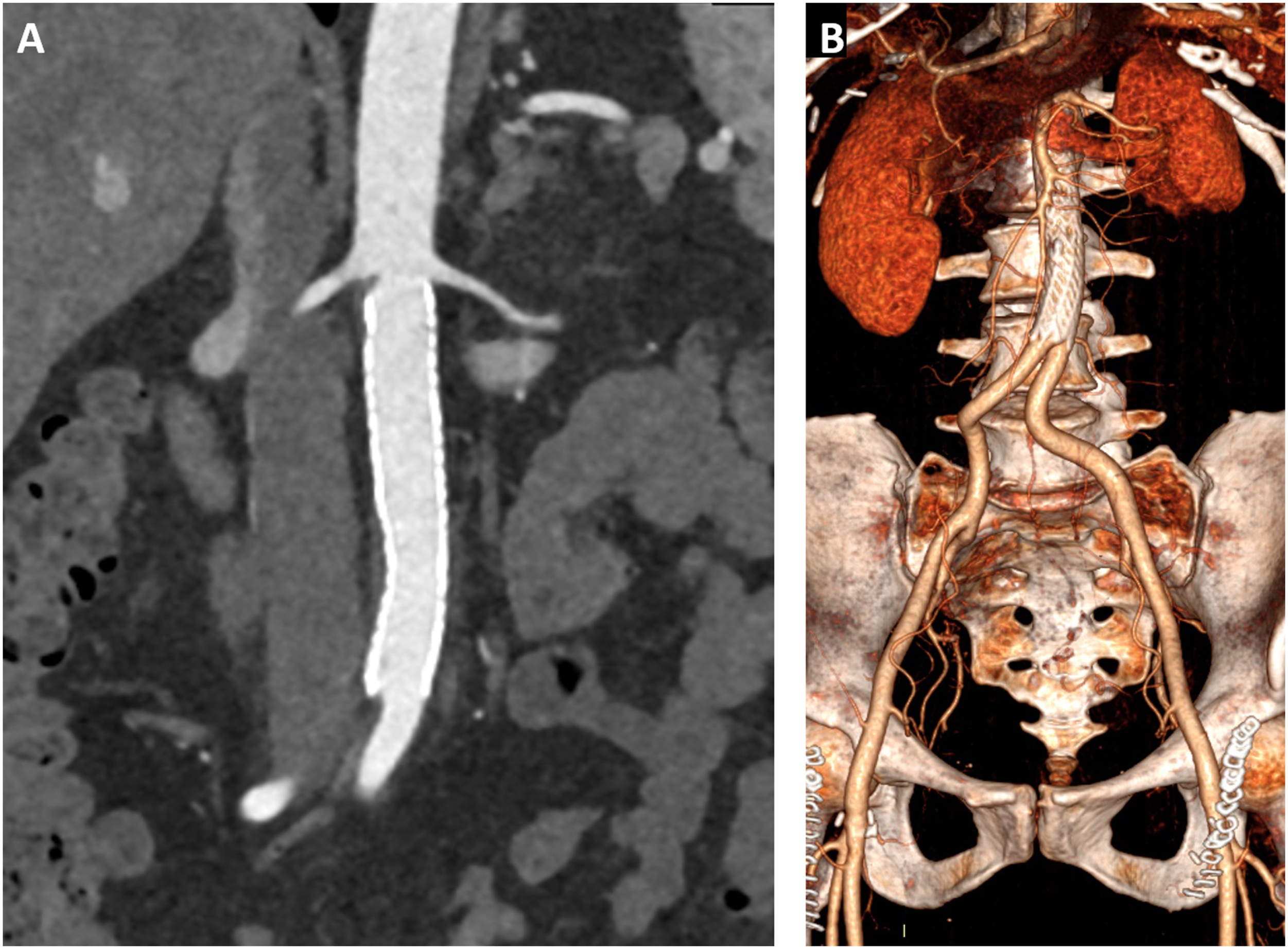
A new thrombectomy via right open popliteal approach was then performed. All thrombi were removed from superficial femoral, popliteal and tibial arteries.
Anticoagulant therapy with low-molecular weight heparin (LMWH: 6000 units twice a day) was continued for the duration of the hospital stay. In addition, a double antiplatelet therapy (Acetylsalicylic acid 100 mg/day and Ticagrelor 90 mg twice a day) was initiated.
The coagulation system screening (platelet count, bleeding time, prothrombin ratio, activated partial thromboplastin time, thrombin time, liver function test) revealed a slight increased level of Xa factor (0,54, normal range <0.2 U/mL), while other tested procoagulants (coagulation factor I-XIII, lupus anticoagulant, anticardiolipin antibodies, anti-β2-glycoprotein) and anticoagulants (Protein C, Protein S, anti-thrombin III) were in normal ranges.
The pathohistological examination of the extracted thrombus confirmed the presence of clots with inflammatory cells, excluding the presence of malignant cells.
The remaining postoperative course was uneventful, and the patient was discharged from the hospital on 27th postoperative day, due to the persistence of the positivity of the throat swab even without any respiratory symptoms. According to the hematologist, therapy at discharge consisted of dual antiplatelet for 6 months and subcutaneous LMWH therapy, which was replaced 2 days after the discharge with a new oral anticoagulant (Rivaroxaban 2.5 mg twice a day). Six months later, Ticagrelor was stopped and the patient was kept on single antiplatelet therapy with Acetylsalicylic acid 100 mg/day in addition to Rivaroxaban 2.5 mg twice a day.
After 12 months, the patients was asymptomatic for limb claudication. Follow-up Angio-CT scan demonstrated patency of the abdominal aortic stent-graft, with complete exclusion of the floating thrombus (Figure 3), and regular patency of both femoral-popliteal arteries, with normal opacification of tibial vessels. No other peripheral embolic events were reported. Angio-CT scan at 1-year follow-up (3A) and 3D reconstruction (3B) showing the correct apposition of the endograft on the aortic wall with complete exclusion of the thrombus.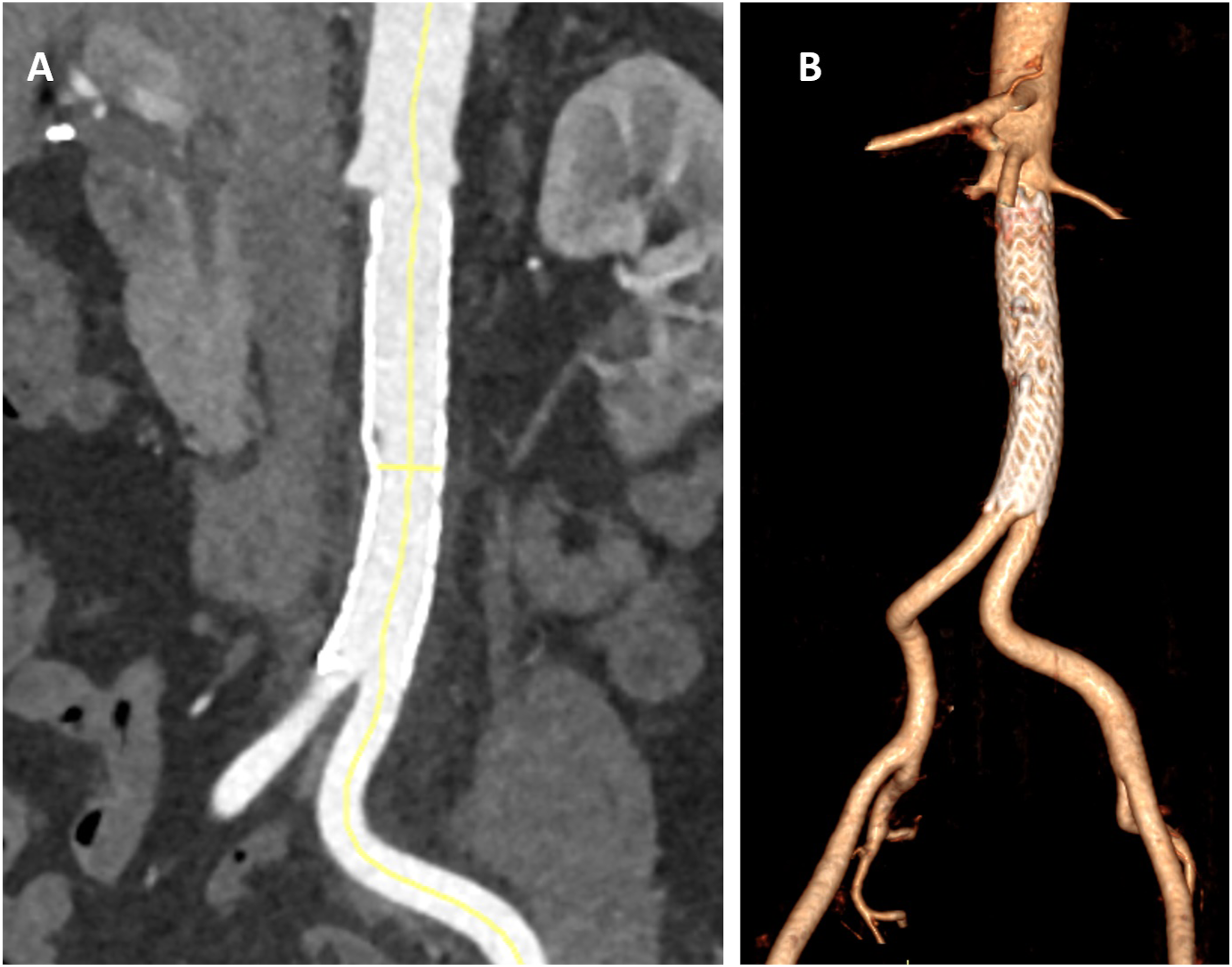
Literature Review
The search was conducted on PubMed using the words “aortic floating thrombus” (AFT) on August 2nd, 2022. All search results were restricted to English, Italian, and French languages.
Studies included case reports and case series about floating thrombus in native aortic arch, descending thoracic and abdominal aorta. Papers reporting floating thrombus after graft placement or aortic dissection or located in the ascending aorta, cardiac valves, cardiac chambers, as well as review, proceedings, letters to editor without data of interest, commentaries and reviews were excluded. Also, papers whose full text was not available or written in other languages than English, Italian, and French were excluded.
Data were collected about patients’ demographics (age, sex), etiology and location of the floating thrombus, clinical presentation, treatment, outcomes.
The search of the Literature retrieved 247 results from 1980 to 2022. Of them, 143 were excluded from the title and 12 more after the reading of the abstract. Of the remaining 92, 59 papers were included in the study (Figure 4). Flow diagram of the results from the literature search and of the studies included in the review, according to the PRISMA statement.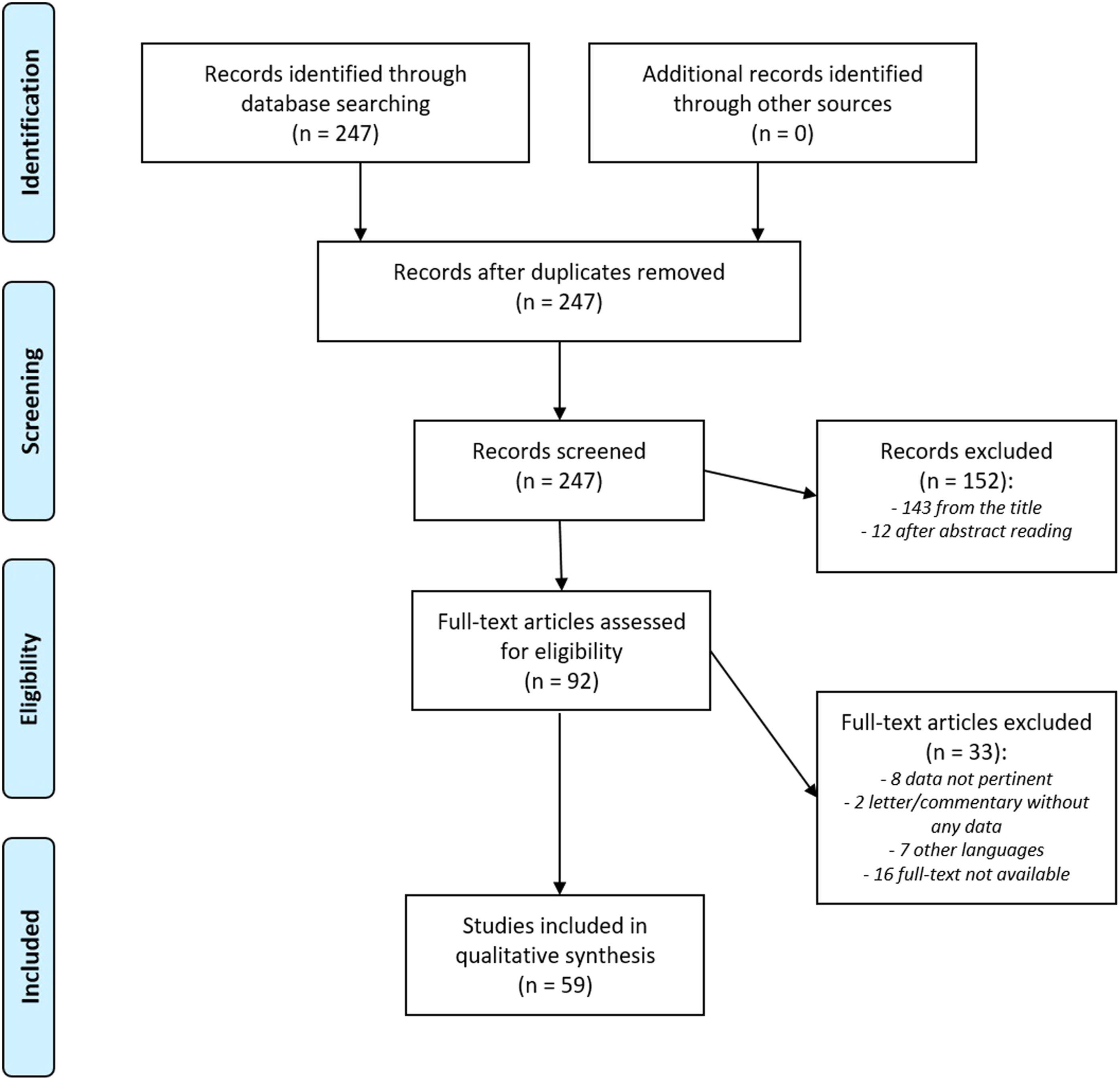
A total of 74 cases of AFT were described (37 males, 50%). Patients had a mean age of 56.2 ± 12.1 years (range 21-92 years).
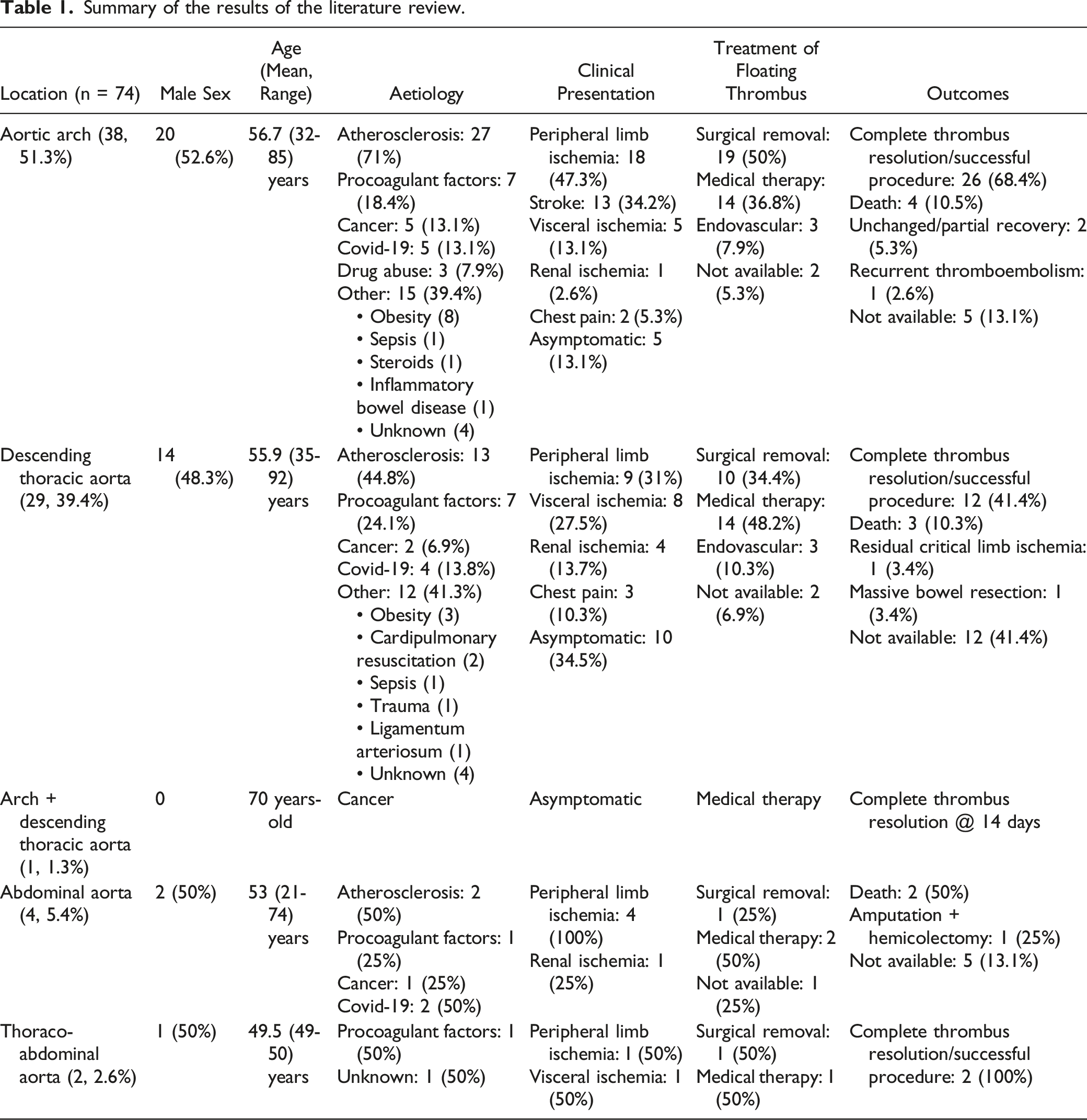
Summary of the results of the literature review.
Details of the 6 cases of floating thrombus involving the abdominal aorta.

CIA = Common Iliac Artery; OAC = Oral AntiCoagulant; n.a. = not available.
The floating thrombus was diagnosed mainly when searching for a possible embolic sources in patients who presented with stroke (17.6%), renal (8.1%), visceral (18.9%) or peripheral limb ischemia (43.2%). In 16 cases (21.6%) it was an incidental finding being the AFT asymptomatic, especially when it was located in the descending thoracic aorta (Table 1).
The AFT was mostly approached either medically with anticoagulation/systemic thrombolysis (32/74, 43.2%) or with surgical removal (31/74, 41.9%).
In 46.8% of the cases treated with medical approach the thrombus was completely resolved within a reported period ranging from 2 days 10 to 17 months. 11 In 1 of the reported cases, a 85-year-old man who arrived with a stroke due to cerebral embolism from an AFT located in the arch was treated with apixaban, but the AFT remain unchanged at 1 month follow-up, 12 and in another similar case of a 50-year-old man who was deemed to be unfit for surgery the AFT still remained a source of recurrent thromboembolism despite the oral anticoagulation therapy. 11 In 2 cases of AFT located in the descending thoracic aorta, the oral anticoagulation therapy was unsuccessful and both patients finally underwent open surgical removal of the thrombus through thoracotomy.13,14
Five cases of aortic arch and descending thoracic aorta involvement were successfully treated with endovascular coverage using an endograft (2 and 3 cases respectively,15–18).
The outcome of an AFT after treatment depended on the patient’s clinical presentation and on patient’s pre-existing pathologies but was favorable in most cases irrespective of the type of approach (Table 1).
Discussion
The finding of a floating thrombus in the thoraco-abdominal aorta is rare life-threatening disease, mostly observed in the descending aorta or at the level of the aortic arch. As described in some case series in the literature,19,20 the etiology of AFT is unclear.
To the best of our knowledge, this is the first described case of an abdominal aortic floating thrombus treated with endovascular approach in a patient with SARS-CoV-2 infection.
In our case, coagulation screening detected a slight increased level of Xa factor in the patient. However, other procoagulants and anticoagulants were in normal ranges, therefore antiphospholipid syndrome, systemic lupus and malignancies were excluded as potential causes of this hypercoagulable state. Furthermore, the patient had no risk factors for atherosclerosis, and her arterial system did not show any sign of disease.
In the literature a total of 6 cases of AFT involving the abdominal aorta have already been described,4–9 2 of them in patients who had a concomitant COVID-19 infection.6,7 One of these cases regarded a 55-year-old male who presented with renal infarction and lower limb ischemia 6 and was managed with Fogarty embolectomy and anticoagulation. In the second case 7 a 74-year-old female presented with a dramatic clinical picture of aortic barrage but died before any treatment.
There is no consensus regarding the management of AFT, especially if located in the abdominal aorta. Of course, when embolic events occur resulting in splanchnic infarctions or limb ischemia, urgent treatment is mandatory.
Anticoagulant or thrombolytic medical therapy, open surgical therapy or endovascular treatment are all considered valid options of treatment, depending on clinical presentation and on the location of the thrombus. 20
Use of anticoagulant or thrombolytic medical therapy as the primary therapy appears reasonable and may lead to the complete thrombus resolution even within few days, 8 but the risk of disruption and consequent embolic events is increased. With this paradigm, surgical or interventional therapy would then be reserved for the initial embolic complications and the occasional patient who despite adequate anticoagulation therapy shows persistence of the aortic thrombus or recurrent embolization. 21
In our case, the patient had already started anticoagulant therapy about a month before. However, considering its ineffectiveness for the recurrent episodes of peripheral embolization, we opted for early endovascular treatment with the coverage of the thrombus, in addiction to an antiplatelet and anticoagulant medical therapy. Open surgical approach with mechanical thrombus removal was excluded due to its invasiveness, the need for general anesthesia with subsequent monitoring in intensive care and the increase of postoperative morbidity. 22
As for endovascular approach, the choice of the proper stent-graft can take into account some factors, since current endografts are designed for use in the setting of abdominal aortic aneurysms.
Adequate preoperative planning and special care during the deployment are required to prevent distal embolization. Operators should ensure that an adequate proximal neck (>20 mm) is present, just as in EVAR procedures, to avoid inadvertent migration of thrombotic material in the renal artery ostia. 23
For this case, we opted for an iliac extension, since aortic diameters were normal. Furthermore, the bilateral femoral surgical exposure allowed the performance of bilateral embolectomy and prevention of further peripheral embolism that could be consequent to the endograft deployment with thrombus dislodgement.
In our opinion, the placement of stent graft in the abdominal aorta to exclude a floating thrombus from the systemic circulation can be a safe and minimally invasive alternative when anticoagulation therapy has not been effective in preventing embolization. 18
Conclusion
SARS-CoV-2 infection may induce hypercoagulable state that can induce venous and arterial thromboembolism, such as the occurrence of abdominal aortic floating thrombus. There is no consensus about the optimal treatment. Endovascular coverage with a stent graft may represent an effective mini-invasive option, when anticoagulant or thrombolytic medical therapy may not be adequate in preventing complications.
Footnotes
Authors’ Contributions
MG: conception and design, data collection, writing the manuscript, critical revision, final approval; DM: conception and design, data collection, analysis and interpretation, writing the manuscript, critical revision, statistical analysis, final approval; PR: analysis and interpretation, critical revision, final approval; GN: data collection, critical revision, final approval.
Declaration of conflicting interests
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
