Abstract
Heat shock protein expression can be induced by heat shock making it possible to artificially modulate their levels noninvasively in vivo in a spatially and temporally controlled manner. Here, we report the use of the major heat shock protein 70 (HSP70) as an inducible target by using the small molecule deoxyspergualin (DSG) conjugated to the near-infrared fluorophore (Cy5.5). We demonstrate that heat induction in the form of localized hyperthermia of normal tissue in living mice results in sufficient HSP70 overexpression for detection with DSG-Cy5.5 conjugate. This effect is dependent on total energy delivered and reaches maximum fluorescence signal in 6 to 8 hours post heat induction and declines over a period of up to 24 hours. These results suggest that DSG-Cy5.5 agent accumulates in tissue with elevated HSP70 by heat.
Introduction
Heat shock protein 70 (HSP70), is a molecular chaperone, has strong cytoprotective effects and can help in maintaining the proper conformation of other proteins after a large variety of stresses. 1 -6 Stress can be any sudden change in environment such as heat shock, oxidative stress, heavy metals, or anticancer drugs. Stress causes increased amount of damaged proteins by inhibiting their elimination via the proteasome as well as by damaging the chaperones themselves. Heat shock proteins (HSPs) are generally classified according to their molecular weights with the majority belonging to the groups HSP27, HSP40, HSP60, HSP70, HSP90, and large HSPs (HSP110 and glucose-regulated protein 170). 7 Except for the group of small HSPs, HSP proteins are adenosine triphosphate (ATP)-dependent proteins with ATPase activity. 5 Heat shock protein 70 family members possess a C-terminus domain that chaperones unfolded proteins and peptides, and an N-terminus ATPase domain that controls the opening and closing of the peptide-binding domain. 3 It can form stable complexes with cytoplasmic tumor antigens that can then escape intact from dying cells. Heat shock protein 70 proteins have crucial functions in protein folding, the maintenance of protein homeostasis, and enhancement of cell survival following a multitude of stresses. 8 Heat shock proteins are implicated in tumor cell proliferation, differentiation, invasion, metastasis, death, and recognition by the immune system.
Several group reported the roles of HSP70s in cancer. 9 -11 Heat shock protein 70s play important roles in cancer development and are often expressed at abnormally high levels in cancer cells. Heat shock protein 70s promote carcinogenesis by acting as survival factors owing to their tumor-associated expression and antiapoptotic effects. 12 Jäättelä et al reported that HSP70 protected cancer cells from TNF-induced cytotoxicity, and they suggested that HSP70 may increase the oncogenic potential of some cancer cells via an immunological escape mechanism. 13 The roles of HSP70s in cancer development may be a potential theranostic target for cancer therapy. 14 -18
High HSP70 levels are associated with adverse outcomes in breast, endometrial, oral, colorectal, prostate cancers, and certain leukemias. 5,19 -21 Moreover, transgenic overexpression of Hsp70 is sufficient to induce T-cell lymphoma in some models. 20 This observation is important because induction of HSP70 can be misregulated in cancer, potentially mediated by altered activity of the heat shock transcription factor 1. 22 -24 Despite the role of HSPs in cancer biology, the precise mechanism remains largely unknown yet.
In an effort to elucidate the role of heat shock proteins, we set out to generate a new probe selective for the classically defined heat-inducible protein (HSP70). Among the potential chemical ligands specific for HSP70, we have selected a derivative of spergualin, 15-deoxyspergualin (DSG) a potent immunosuppressive agent with antitumor properties, 25 -31 which was also found to biochemically bind to HSP70 and HSP90. 25,32 Although the precise mechanism of action of DSG remains unknown, the antitumor effect of DSG is thought to be mediated by inhibition of cell cycle progression at G1 phase or by affecting angiogenesis. 33 -35 Nonetheless, these pleiotropic effects may also be explained by mechanisms that involve modulation of heat shock proteins. Deoxyspergualin was originally developed in Japan as an immunosuppressive agent but this compound is not commercially available. Several groups synthesized this compound but they reported low yields (7%-12%). 36 -46
In this report, we describe an improved DSG synthesis according to the published methods with some major modification of reaction conditions with high yields (overall 18%-22%). Recently, several groups developed and evaluated the HSP-targeted imaging agents. 47 -50
Using this HSP70-selective ligand, we set out to test our hypothesis for enhancing the intracellular targeting of an inducible protein on demand. We conjugated a fluorophore to the selective ligand (DSG) for in vitro cellular uptake and in vivo imaging studies. We found that this compound accumulates within heat shocked cells and muscle tissue treated with locoregional hyperthermia in mice. We believe these findings have significant implications for developing new HSP-targeted optical imaging agents.
Materials and Methods
Reagents and Instrumentations
All reagents and solvents were purchased from Aldrich Chemical Co and used without further purification. Thin-layer chromatography (TLC) was performed on precoated Kieselgel 60 F254 (Merck) glass plates. Proton and Carbon NMR spectra were recorded on a Brucker 300 or 500 MHz spectrometer using tetramethylsilane as an internal reference and hexafluorobenzene as an external reference, respectively, at The University of Texas MD Anderson Cancer Center. The mass spectra were obtained on a LCQ Fleet mass spectrometer using electrospray ionization technique. Microwave synthesis was performed on a Biotage Initiator Eight microwave synthesizer. High-performance liquid chromatography (HPLC) was performed on a 1200 series pump (Agilent), with UV detector operated at 254 or 690 nm, using a semipreparative and analytical C-18 reverse phase column, Luna SCX 100A (5 μm, 250 × 10 mm) or Vydac protein and peptide C-18 column (5 µm, 150 × 4.6 mm).
Chemistry
7-Bromoheptanamide: 1
7-bromoheptanenitrile (3.0 g, 15.78 mmol) was dissolved in 50 mL of concentrated hydrochloric acid and stirred for 8 hours at 40 °C. The mixture was then poured onto ice-cold water (200 mL). The white precipitate was obtained and filtered off. Washed with water and evaporated to dryness. The crude product is crystallized from ethyl acetate-methylcyclohexane solvent mixture to give 2.67 g of the expected product
7-Azidoheptanamide: 2
To a solution of compound
Acetic Acid, [(7-Azido-oxoheptyl)amino]hydroxymethyl ester: 3
Compound
Acetic acid, [(7-azido-oxoheptyl)amino][1-(2-naphthalenyl) ethoxy], methyl ester: 4
Compound
2-[(7-Azido-1-oxoheptyl)amino]-2-[1-(S)-(naphthalen-2-yl)ethoxy]acetic acid: 5
Compound
N
4
-Tert-Butoxycarbonyl-1,4-Butanediamine: 6
1, 4 diaminobutane (4.0 g, 45.38 mmol) was dissolved in tetrahydrofuran (50 mL) under argon, triethylamine (12.76 mL, 90.76 mmol) was added, followed by addition of 4-dimethylamino pyridine (1.7 g, 13.61 mmol). The mixture was cooled to 0 °C then di-tert-butyl dicarbonate (11.89 g, 54.45 mmol) was added via a syringe over a period of 30 minutes. The reaction mixture was warmed to room temperature and stirred for 6 hours when TLC showed that no starting material remained. The solvent was evaporated under vacuum; the residue was dissolved in dichloromethane (150 mL) and washed with water (3 × 150 mL). The organic phase was dried (MgSO4), evaporated to dryness and purified on a silica gel column using 2% methanol in dichloromethane as eluent. The pure compound
N
4
-tert-butoxycarbonyl-N
1
-(2-cyanoethyl)-1,4-butanediamine: 7
A solution of compound 6 (3.0 g, 15.93 mmol) in dry methanol (10 mL) was cooled to −10 °C for 10 minutes under argon. Acrylonitrile (1.01 g, 19.12 mmol) was slowly added via a syringe over a period of 5 minutes. The reaction mixture was stirred at −10 °C for 1 hour, warmed to room temperature and continued for overnight when TLC showed that no starting material remained. Solvent was evaporated under vacuum. The residue was purified on a silica gel column using 2% methanol in dichloromethane as eluent. The pure compound 7, 2.8 g was obtained in 74% yield. 1 H NMR 7 (CDCl3) δ: 1.40 (bs, 9H), 1.46-1.50 (m, 4H), 1.60 (s, 1H), 2.49 (t, J = 6.6, 2H), 2.62 (t, J = 6.6, 2H), 2.88 (t, J = 6.6, 2H), 3.03 (t, J = 7.1, 2H), 7.01 (bs, 1H). MS: M + 1, calculated 242.320, found 242.328.
N
1
-Benzyloxycarbonyl-N
4
-tert-butoxycarbonyl-N
1
-(2-cyanoethyl)-1,4-butanediamine: 8
Compound
N
4
-Benzyloxycarbonyl-N8-tert-butoxycarbonylspermidine: 9
A solution of compound
N1, N4-Dibenzyloxycarbonyl-N8-tert-butoxycarbonylspermidine: 10
Compound
N1, N4-bis (benzyloxycarbonyl) spermidine: 11
Compound
Tetraazaheneicosanoic acid, azido[1-(2-naphthalenyl)ethoxy]dioxo[(phenylmethoxy)carbonyl] phenylmethyl ester: 12
Compound
Hexa-azapentacos-2-enedioic acid, 13-[1-(2-naphthalenyl) ethoxy]dioxo[(phenylmethoxy)-carbonyl]-3-[[(phenylmethoxy)carbonyl]amino]bis(phenylmethyl) ester: 13a and Hexa-azapentacos-2-enedioic acid, 13-[1-(2-naphthalenyl)ethoxy]dioxo[(phenylmethoxy)-carbonyl]-3-[[(phenylmethoxy)carbonyl]amino]bis(phenylmethyl) ester: 13b
Compound
7-[(Aminoiminomethyl)amino]-N-[2-[[4-[(3-aminopropyl)amino]butyl]amino]-1-hydroxy-2-oxoethyl]heptanamide-tris hydrochloride: (DSG), 14
Compound
DSG-Cy5.5 conjugates: 15
Compound
In Vitro Cellular Uptake Studies
Immunohistochemical staining procedure
Human lung carcinoma cells (A549) were plated at a density of approximately 10 000 per chamber in a 96-well clear bottom black plate using Dulbecco’s Modified Eagle Media (DMEM) supplemented with 10% fetal bovine serum (FBS), 50 U/mL of penicillin and 50 µg/mL streptomycin, and grown overnight at 37 °C. Fixing was achieved with 4% formaldehyde in phosphate-buffered saline (PBS) for 30 minutes and the subsequent staining procedure was performed as described in the antibody supplier’s instruction manual (R&D Systems, Inc). As an additional positive control, A549 cells were treated with 1 mM CuSO4 for 24 hours as described by Neuhaus-Steinmetz and Rensing 51 followed by fixing and anti-HSP70 antibody staining. Primary antibody detection was performed with a fluorescein-labeled secondary antibody as described by the supplier. Nucleic acid counterstaining with DAPI (4′,6-diamidino-2-phenylindole) was achieved immediately after acquisition of fluorescence microscopy images (Figure 2) while on the microscope stage and then photographed with the appropriate filter.
Cultured cells heat shock procedure
A549 human lung carcinoma cells were grown in a 24-well plate at a density of 400 000 cell/well using DMEM supplemented with 10% FBS and 50 U/mL of penicillin and 50 µg/mL streptomycin in a total of 2 mL. They were allowed to attach for 24 hours prior to heat shock. Cells were heated at 41 °C to 42 °C for 1 hour or 43 °C for 10 minutes (Figure 3) in a humidified incubator with 5% CO2 followed by immediate recovery at 37 °C. During the heat shock period, the temperature was monitored with the use of a micro-thermocouple inserted into a 96-well plate chamber containing the same volume of culture media. Following recovery for approximately 6 hours, DSG-Cy5.5 conjugate was directly added to the culture media at a 3 different concentration of 1, 5, and 10 µM; and incubated for 1 hour at 37 °C. Cells were then washed 3 times with prewarmed PBS (37 °C), followed by scrap harvesting, counted, and diluted to 100 000 cells/mL. Approximately 10 000 cells per well were seeded in 96-well black plates and measured in a fluorometric plate reader (FLUOstar OPTIMA) with a 620/700 nm filter at an optical magnification of ×20. As an additional control, cells treated with CuSO4 were also tested for uptake of DSG-Cy5.5 (Figure 4). Phase-contrast images were obtained with each view.
In Vivo Hyperthermia Imaging
Approximately 6-week-old bagg albino mice (BALB/c) nu/nu female mice were purchased from Charles River Laboratories and maintained on a regular diet. A heating apparatus was constructed to permit localized deposition of heated water to one (right hind) limb of each mouse (Figure 5). For anesthesia, mice were induced with 4% isoflurane and maintained on 2% throughout the entire heating procedure. Each mouse was positioned into the heating apparatus and monitored throughout the entire session. Heating times were varied from 5 to 20 minutes at 43 °C as measured by a thermocouple submerged in the water bath. Upon completion of heating, mice were removed from the apparatus and allowed to recover at ambient temperature for 6 hours. After the recovery period, 5 µM of DSG-Cy5.5 in saline was intravenously administered by tail vein injection and scanned in a Xenogen/Caliper IVIS-200 imaging system using the appropriate Cy5.5 filters with 0.15 seconds exposure time and medium binning. Mice were continuously anesthetized with 2% isoflurane and kept warmed during all image acquisitions.
Immunohistochemistry Procedure
Mice were killed using CO2 gas and harvested mouse muscle samples were fixed in 10% buffered formalin solution and paraffin-embedded for immunohistochemistry using the same anti-HSP70 monoclonal antibody and counterstaining with hematoxylin. Immunohistochemistry procedure was performed by the pathology core service facility of the Methodist Hospital. All animal experiments were performed in strict compliance with a protocol approved by the institutional animal care and use committee of the Methodist Hospital Research Institute.
Results and Discussion
Deoxyspergualin was synthesized according to the published procedures with some modification of reaction conditions.
36
-46
Briefly, the synthetic scheme for DSG-Cy5.5 is illustrated in Figure 1A-C. Orthogonally, protected spermidine derivative is one of the parts of DSG. Several methods leading to the synthesis of differentially protected spermidine have been described but it is not commercially available.
52
-55
We have optimized the reaction conditions of this compound with high yield. Two protecting groups were used for the selective protection of 2 primary amino groups located at N
1
and N
8
and one secondary amino at N
4
in spermidine. Usually, the most popular amino protecting groups, that is, tert-butoxycarbonyl (Boc) and benzyloxycarbonyl (Z), meet the criteria to be used in the orthogonal protection of spermidine. The synthetic scheme for protected spermidine is illustrated in Figure 1A, the first step of the synthesis to obtained orthogonally protected N
4
-tert-butoxycarbonyl-1,4-butanediamine

A, Synthetic scheme of protected spermidine.
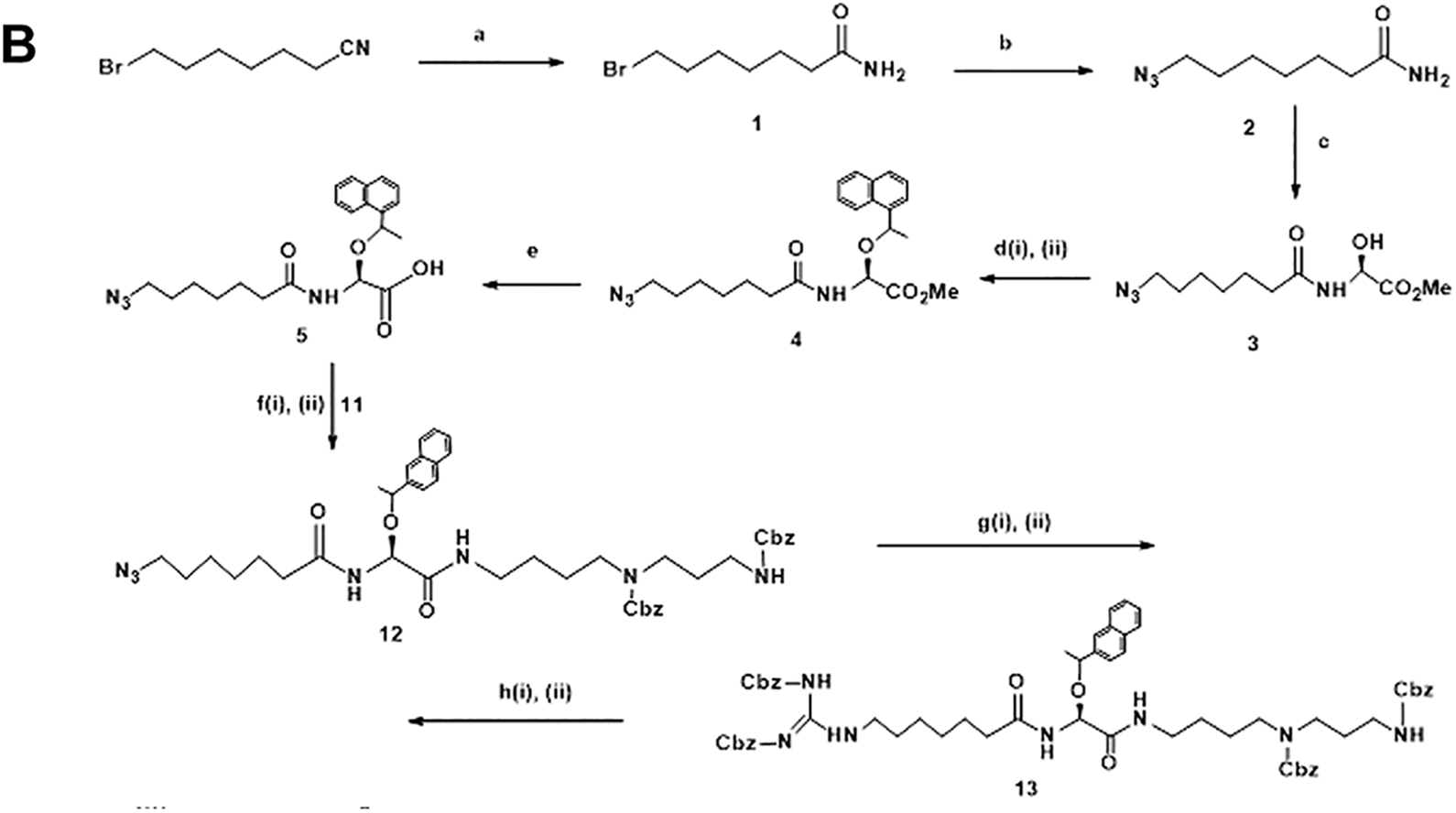
Synthetic scheme of deoxyspergualin (DSG)

C, Synthetic scheme of DSGCy5.5.
The synthetic scheme for DSG is illustrated in Figure 1B, compound
Conversion of
The conjugate

Immunohistochemistry of HSP70 induction by CuSO4 in A549 cells. (A) Bright field phase contrast; (B) Anti-HSP70 staining; (C) DAPI counterstaining; (D) Overlay of anti-HSP and DAPI staining images. DAPI indicates 4′,6-diamidino-2-phenylindole; HSP70, heat shock protein 70.
The deoxyspergualin conjugate (DSG-Cy5.5) was tested for cellular uptake in human lung carcinoma cells (A549) subjected to transient heat shock under aseptic conditions in a tissue culture incubator. As shown in Figure 3, cellular uptake of DSG-Cy5.5 is significantly increased following heat treatment at the concentration tested. A 5 µM and 10 µM concentrations were highly significant (P < .001) when compared with control and heat shock treatment. Cy5.5 alone was used as control at the same molar concentrations showed no significant cellular uptake. Figure 4 also shown that the cellular uptake of DSG-Cy5.5 is significantly increased following heat treatment at the 5 µM concentration in lung carcinoma (A549) cell line.

Human lung carcinoma (A549) cells were grown at 37 °C (dark gray) or heat shocked at 43 °C for 10 minutes in a tissue culture incubator (light gray) and allowed to recover for 6 hours. DSG-Cy5.5 was added to the culture media and allowed to incubate for 1 hour at 37 °C, followed by cell harvest and fluorescence measurements. Relative fluorescence values were plotted for the vehicle (media) and DSG-Cy5.5 at 3 final concentrations: 1, 5, and 10 µM. Error bars indicate standard error of the mean for 6 runs. Among the 3 concentrations of conjugates 5 µM and 10 µM were highly significant (P < .001) when compared with control and heat shock treatment. DSG indicates deoxyspergualin.
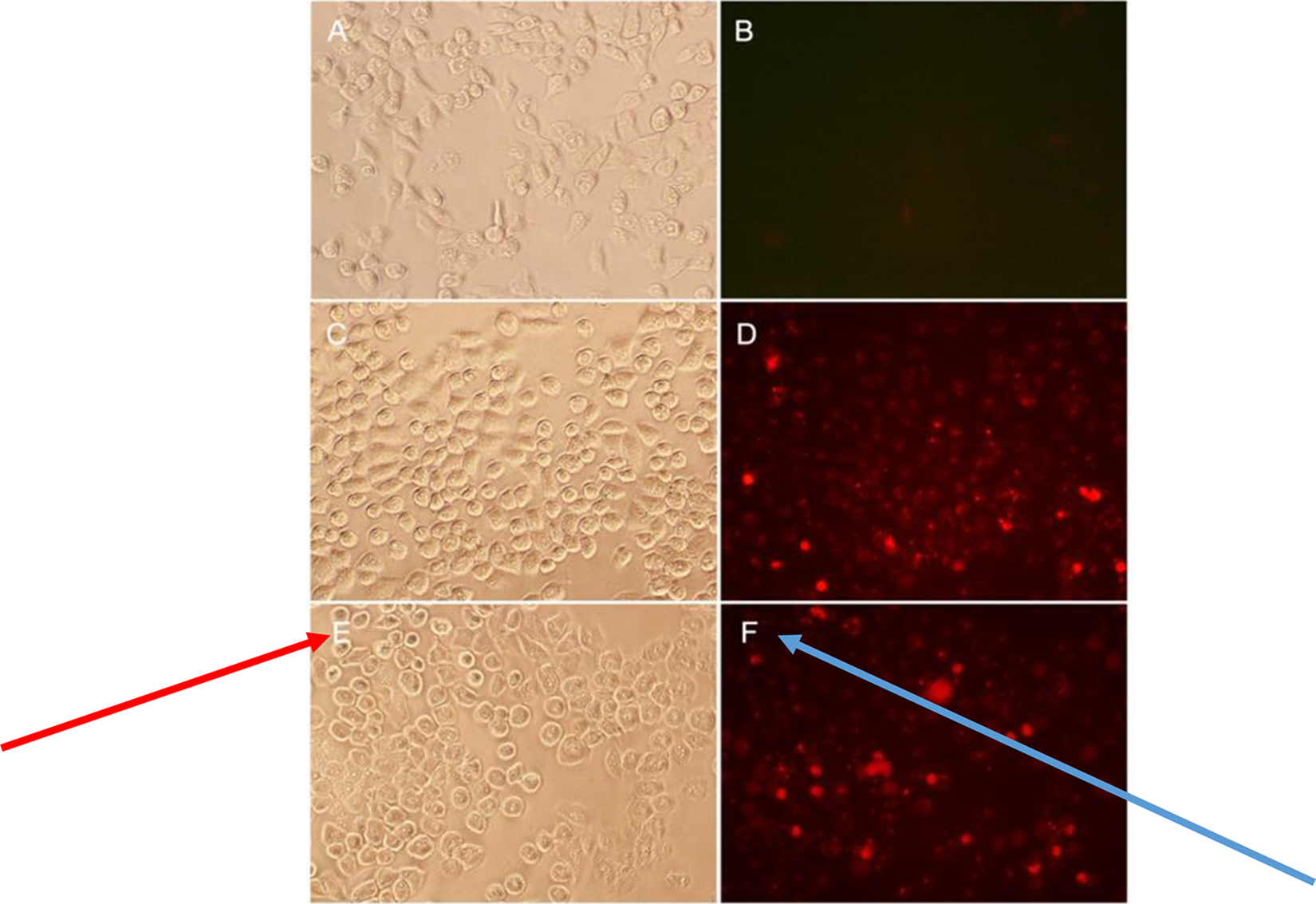
Detection of HSP70 by DSG-Cy5.5 in A549 cells. (A and B) Control at 37 °C, bright field and Cy5 filter, respectively; (C and D) Heat shock at 43 °C for 10 minutes, bright field, and Cy5.5 filter, respectively; (E and F) CuSO4 induction, bright field, and Cy5.5 filter, respectively. DSG indicates deoxyspergualin; HSP70, heat shock protein 70.
This in vitro result prompted us to evaluate the in vivo uptake of this compound by testing for accumulation in normal tissue subjected to localized hyperthermia. Results from a study testing 43 °C for 6 to 10 minutes are shown in Figure 6A-E. Image processing was performed on the Living Image v3.0 software without background subtraction or autofluorescence correction. As evident from the fluorescence images, there is increased signal intensity in the right hind limb indicated by arrows. Prominent signal intensity is noted at approximately 7 minutes at 43 °C and imaged at 7 to 8 hours post-heat treatment. The majority of the nonspecific thoracic and abdominal fluorescence signal is largely due to autofluorescence (and likely from high-chlorophyll content diet) as determined by imaging of uninjected animals (data not shown). After approximately 24 hours, a second intravenous dose was administered and imaged immediately and at 1 hour postinjection resulting in detectable signal in the treated limb (Figure 7). This result corroborates the persistent HSP70 staining observed in the tissue culture experiment (Figure 4D). Following completion of imaging studies, mice were killed and soft tissue from the hind limbs were harvested and fixed in 10% formalin, sectioned, and immunostained with anti-HSP70 antibody. Localized disruption of the skeletal muscle fibers was observed with heat treatment at 43 °C for 10 minutes, suggesting thermal injury (Figure 8). Tissue harvested at the time of killing 6 hours post heating were fixed and stained for HSP70 protein. Robust immunohistochemical staining for HSP70 was observed in the heated tissue (Figure 8B) and not in the untreated control (Figure 8A). This observation provides supporting evidence for the postulated localization mechanism of DSG-Cy5.5 to the site of heat shock. Nonetheless, the experimental results present in this work support the hypothesis that accumulation of a composite small molecule may be controlled “on demand” by the induction of endogenous intracellular targets.

Small animal heating apparatus. (A) Top oblique view of mouse placed on platform with anesthesia nose cone (B) top view.

DSG-Cy5.5 fluorescence imaging of 5 mice treated in the right hind limb at 43 °C for: (A) 6 minutes, (B) 7 minutes, (C) 8 minutes, (D) 9 minutes, and (E) 10 minutes. DSG-Cy5.5 was administered at 5 hours post heating. Each mouse was imaged at 5 (immediately after injection of DSG-Cy5.5), followed at 6, 7, and 8 hours post heat treatment. Arrows indicate increased fluorescence signal in the treated limb. DSG indicates deoxyspergualin.
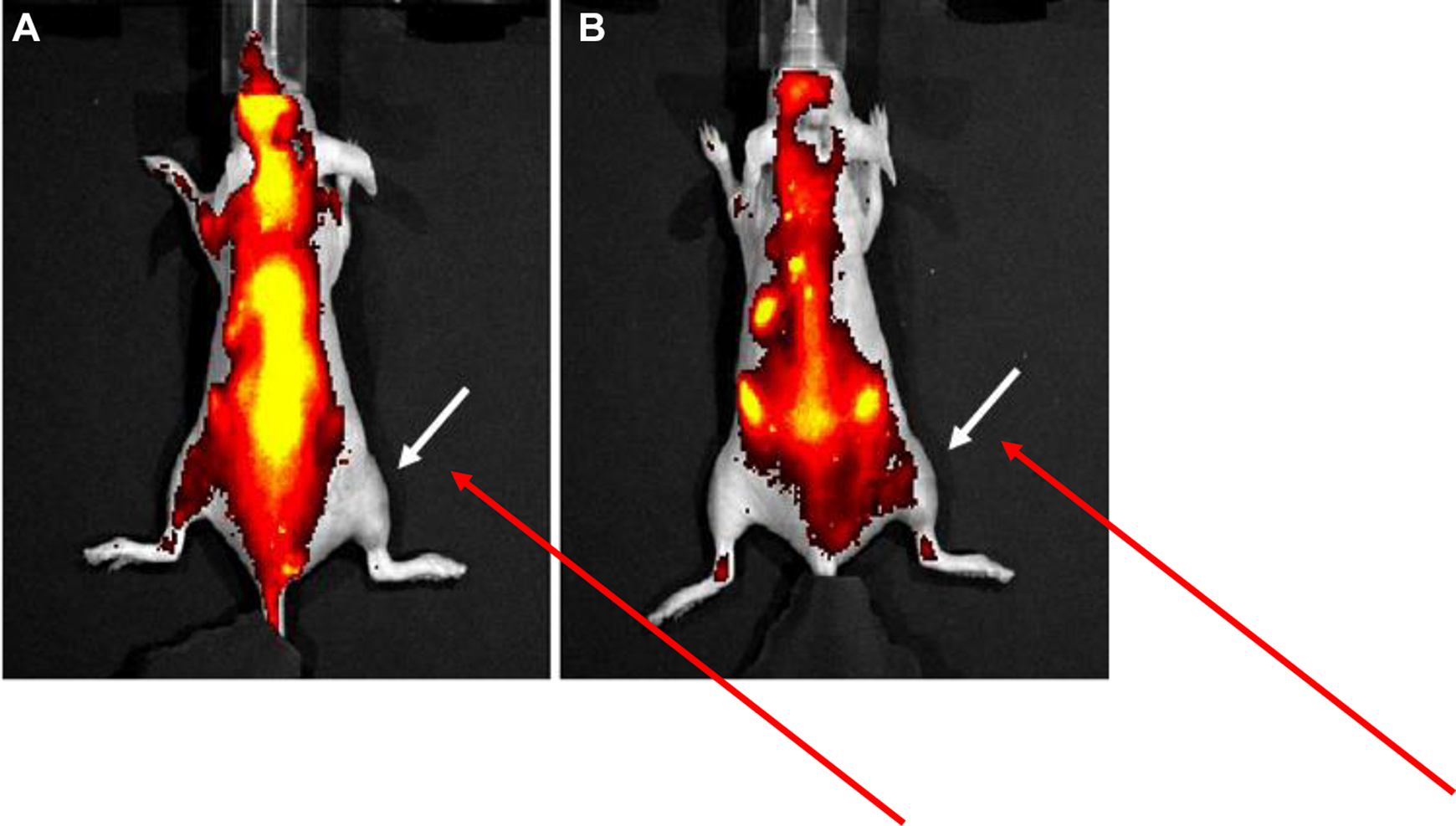
Detection of DSG-Cy5.5 at 24 hours. Mice were injected with a second dose of DSG-Cy5.5 and immediately imaged (A) followed by delayed imaging at one hour (B). Arrow points to the region in the right hind limb that continues to exhibit some fluorescence activity at 24 hours post heating. DSG indicates deoxyspergualin.

Immunostaining for HSP70 in soft tissue harvested 6 hours post heating at 43 °C for 10 minutes. (A) Unheated contralateral limb; (B) Heated limb. HSP70 indicates heat shock protein 70.
In this study, DSG-Cy5.5 was significantly accumulated both in tissue culture and in experimental mice with comparable findings. Further translational evaluation of DSG-Cy5.5 is warranted.
Conclusion
This study demonstrates proof of concept for the novel approach of imaging an inducible target using an endogenous protein involved in stress response. According to our findings, DSG-Cy5.5 may serve as a valuable optical probe for imaging and also to investigate the role of HSP in cancer biology in future.
Footnotes
Acknowledgment
This project was supported by The Methodist Hospital Research Institute, the M.D. Anderson Foundation, and the Vivian L. Smith Foundation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
