Abstract
An antigen binding fragment (BFab) derived from a tumor-associated mucin 1–sialoglycotope antigen (CA6) targeting antibody (huDS6) was engineered. We synthesized a companion diagnostic positron emission tomography (PET) tracer by radiolabeling BFab with [64Cu] to measure CA6 expression on cancer tissues prior to anti-human CA6 (huDS6-DM4 antibody-drug conjugate) therapy for ovarian and breast cancer patients. After chemotherapy, the ovarian patient received PET scan with 18F-2-fluoro-2-deoxyglucose ([18F]FDG: 10 mCi), followed by [64Cu]-DOTA-BFab ([64Cu]BFab; 5.5 mCi) 1 week later for PET scanning of CA6 expression and subsequent surgery. The breast cancer patient was treated with chemotherapy before primary tumor resection and subsequent [18F]FDG-PET scan. 4 weeks later the patient received of [64Cu]BFab (11.7 mCi) for CA6 PET scan. Whole body [18F]FDG-PET of the breast cancer patient indicated FDG-avid tumor metastases to the liver, bilateral hila and thoracic spine, but no uptake was observed for the ovarian patient. Each patient was also imaged by PET/CT with [64Cu]BFab at 1 and 24 hours after tracer administration. The [64Cu]BFab tracer was well tolerated by both patients without adverse effects, and no significant tracer uptake was observed in both patients. Immunohistochemistry (IHC) data indicated CA6 expressions were weak to intermediate and matched with the [64Cu]BFab-PET signals.
Introduction
Antibody-drug conjugates (ADCs) use monoclonal antibodies (mAb) to selectively target and deliver highly cytotoxic chemicals to tumor cells that express specific antigens. 1 -3 Generation of successful ADCs hinge on 4 key components: the expression level of the target antigen, the specificity of the antibody, the efficacy of the cytotoxic agent, and the linker connecting the two moieties. 4 Selecting the ideal target antigen, monoclonal antibody (mAb), and linker-payload chemistry is often determined empirically, 5,6 with recommended dose/schedule established in phase I and II trials. 4 Today, 4 ADCs have received Food and Drug Association (FDA) approval for the treatment of hematologic or solid tumor cancers, including brentuximab vedotin (Adcentis, Seattle Genetics, approved in 2011) targeting CD30, 7 and ado-trastuzumab emtansine (T-DM1, Roche, approved in 2013) targeting human epidermal growth factor receptor 2 (HER2) metastatic breast cancer. 8 Further, more than 60 ADCs presently in clinical trials 9 -12 and several others at pre-clinical stage, and this class of cancer therapeutics is fastly growing.
The chosen antibody and its associated pharmacokinetics primarily influence the ADC efficacy and in vivo toxicity. The antibody must specifically deliver an effective payload to the target compared to normal tissue, have limited cross-reactivity, and possess low immunogenicity. The delivery of the payload within a cell is directly related to antigen density and is thus a driving force in the therapeutic success of ADCs. Cell antigens must (1) be readily available to circulating antibodies, (2) sufficiently expressed on tumor cells compared to little or no expression on normal cells, and (3) able to undergo internalization for processing and payload release. Given the importance of understanding the in vivo actions of ADCs to accurately predict their therapeutic success, methods to assess these important attributes have emerged as a vital part of the drug development process.
Consequently, measuring tumor antigen levels is the most important requirement prior to administering ADC therapy. Traditionally, antigen levels are assessed by an invasive tissue biopsy followed by immunohistochemistry. In this technique, tumor biopsy samples harvested are subject to a sampling error that may not represent the entire antigen expression across the lesion. Further, it is difficult to find samples from multiple metastatic lesions within the body.
Another approach is to employ companion diagnostics such as positron emission tomography (PET) to visualize, characterize, and quantify antibodies uptake in normal and tumor tissues. ImmunoPET has been developed by conjugating a metal chelator (e.g., DOTA) with the CA6 mAb, followed by labeling with PET radioisotope (e.g., [64Cu]). This target specific immunoPET tracer is used for the global assessment of antigen expression, biodistribution, pharmacokinetic, and clearance to help predict ADC treatment responses. Ideally, quantitative biodistribution data obtained from the companion diagnostic would predict ADC uptake for selection of patients who will most benefit from ADC therapy.
The carbonic anhydrase 6 (CA6) epitope is found on a variety of solid tumors (e.g. breast, ovarian, cervical, lung and pancreatic tumors) with limited expression in normal adult tissues, 13 and thus creates an ideal antigen for patient stratification and response-assessment. In this study we present a first-in-human companion diagnostic PET tracer to the ADC SAR566658 (i.e., huDS6-DM4 which targets CA6 antigen against the tumor-associated MUC1-sialoglycotope conjugated to the cytotoxic maytansinoid derivative, DM4). 14 Therapeutically, upon binding and internalization of the ADC, DM4 is released and binds to microtubules to disrupt assembly/disassembly dynamics, resulting in mitotic arrest of CA6-expressing tumor cells. The immunoPET tracer was developed by radiolabeling ([64Cu], t1/2 = 12.7 h) an engineered antibody fragment (BFab, ∼72 kDa) which is based on the full-length huDS6 with a significantly shorter half-life in circulation (12 hours vs. ∼7 days for the full-length antibody). 15,16 The [64Cu]-DOTA-BFab ([64Cu]BFab) tracer immunoreactivity, specificity, serum stability, in vivo tracer optimization and targeting ability to CA6 were evaluated using a tumor bearing mouse model and are reported elsewhere. 17 Pre-clinical study results indicated that this tracer demonstrated improved tumor uptake at earlier time points (at 24 h p.i., the uptake ratio in CA6-positive tumors was 1.6-fold higher than that of CA6 negative tumor). BFab also had faster clearance rates from non-tumor tissues compared to the full length mAb.
Our aim was to use the [64Cu]BFab tracer as a companion PET diagnostic for patients receiving ADC therapy (SAR566658) against CA6. However, in this two-patient human study, cancer patients who have not received ADC therapy were selected to evaluate the safety of the tracer, and the utility of BFab as a targeting moiety. Specifically, we investigated (1) the safety and tolerability of [64Cu]BFab; (2) the expression of the CA6 biomarker using PET/CT, and (3) the comparison of [64Cu]BFab to both [18F]FDG PET/CT uptake and correlation with CA6 antigen expression levels in ovarian and breast cancer patients by immunohistochemistry (IHC).
Materials and Methods
BFab
The selection and production of BFab derived from huDS6, a humanized monoclonal antibody against the tumor-associated sialoglycotope human CA6 was described previously. 18 BFab was engineered specifically to bind the cell surface antigen CA6, which is overexpressed in tumors and homogeneous in 96% of breast and ovarian cancers. 19,20 Pharmaceutical grade anti-CA6 BFab was manufactured by Sanofi Aventis. The BFab was engineered from light variable domain (VL), and heavy variable domain (VH) sequences from huDS6 (Figure 1). The BFab construct was produced in bacterial cells with a 6X His tag sequence (GGCGGHHHHHH) at the 3′ end. These cells were lysed and the soluble expression protein was purified using anti-His tag column; and the targeting ability to human CA6 was optimized in both cell culture and pre-clinical models 17 prior to clinical studies.
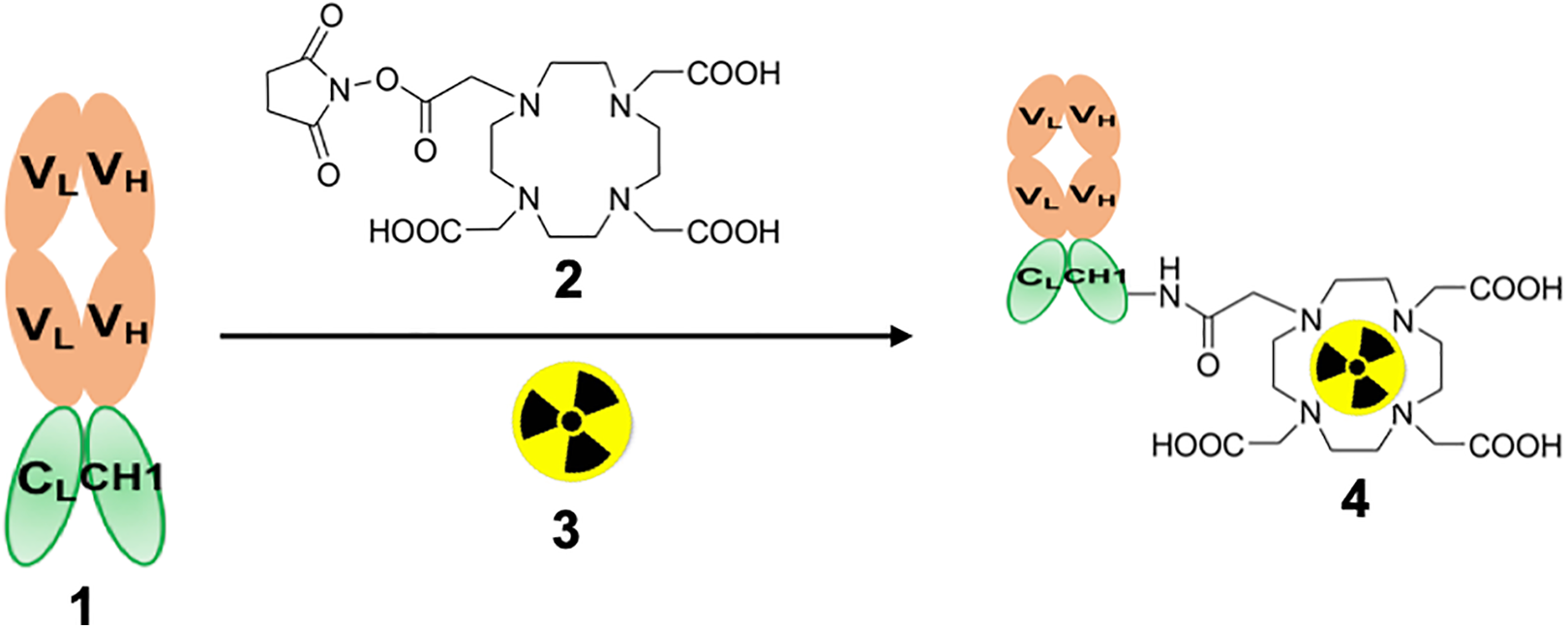
Schematic diagram of the synthesis of
Synthesis of Pharmaceutical Grade [64Cu]BFab Radiotracer
The synthesis and purification of DOTA-BFab and its labeling with [64Cu] via the chelate DOTA, and the resultant product quality tests have been described previously. 21 Synthesis of DOTA-BFab precursor: DOTA-BFab was synthesized using the DOTA-NHS chelator (Macrocyclics, Dallas, TX). Precursor synthesis was performed according to established procedure using DOTA-NHS and BFab of 10:1 molar ratio. 17,21 Radiolabeling: Labeling was performed with [64Cu]Cl2 (approximately 1.7 ± 0.08 GBq in 0.1 M (0.1 mol/L) HCl, (University of Wisconsin - Madison, Madison, WI) and the DOTA-BFab (1 mg in 15-20 mL) were incubated at 37°C in ammonium acetate (200–300 μL, 0.1 M (0.1 mol/L; pH = 5.5) for 1 hour. Sequestration of unlabeled radio metal was achieved by adding ethylenediaminetetraacetic acid (EDTA; 0.5 M; 0.5 mol/L, pH = 8) at room temperature for 15 mins at a final concentration of 0.01 M (0.01 mol/L). This radiochemical mixture was purified via size exclusion chromatography (SEC) high-performance liquid chromatography (HPLC) (SEC-S2000; Phenomenex, Torrance, CA) in sterile condition as per the procedure previously published. 21 The final product of the purified tracer was formulated in phosphate buffer solution (0.1 M; 0.1 mol/L), pH = 7 ± 0.5).
Quality Assurance of the DOTA-BFab, [64Cu], [64Cu]BFab-PET Tracer
The conjugation of mean DOTA chelates (c) per BFab (a) was tested by Matrix-Assisted Laser Desorption Ionization (AB Sciex 5800 TOF/TOF; AB Sciex, Framingham, MA), which was connected to a CovalX high-mass detector. Identity of the [64Cu] was confirmed by the Multi-Channel Analyzer (MCA) analysis, [64Cu] emits positrons with a main energy of 897 keV and an abundance of 22.7%. In addition, non-prompt 909-keV photons are emitted at an abundance of 99.9%. Radiochemical purity was determined by using both SEC-HPLC and instant thin-layer chromatography with Tec-Control Chromatography strips (Biodex Medical Systems, Shirley, NY) developed in saline. The immunoreactivity and the human serum stability assays were performed according to the established procedures. 22 Two batches of [64Cu]BFab radiopharmaceuticals (synthesized on January, 2017 and October, 2017) were assessed for their quality specified in IND with a series of tests listed below (Table 1, Column #2): Immunoconjugate purity, number of DOTA chelates per BFab molecule (c/a), radiopharmaceuticals physical appearance, pH, purity, specific activity, human serum stability, presence of bacterial endotoxin, sterility, and immunoreactivity. These tests results (Table 1, column #3) were compared with the release specifications (Table 1, column #4) required by the FDA for tracer quality.
Summary of [64Cu]BFab Quality Assurance From 2 Batches of Tracer Synthesized for the Ovarian and Breast Cancer Patients.

Characterization of [
Patients Study
Patients were selected based on the following inclusion criteria: Patients should be ≥ 18 years old at the time of the tracer administration, a) have proven cancer by CT or biopsy for ovarian or breast cancer and scheduled to undergo surgical excision of the cancerous lesion(s); or b) have biopsy proven for ovarian or breast cancer but is not a surgical candidate. In addition, patients were provided informed consent, to understand and willingness to sign a written informed consent and be able to participate for each imaging procedure. Exclusion criteria: Patients who were pregnant or breast-feeding, could not complete a PET/CT scan or other study procedures or had contraindications for PET/CT or serious uncontrolled concurrent medical illness were excluded from the study.
Safety
The administration of the radioactive substance was given by intravenous injection (i.v.) as per standard procedures established in the clinical PET and Nuclear Medicine clinics adhered with sterile methods. Before and up to 6 weeks after the administration of radiolabeled [64Cu]BFab, routine laboratory analyses were performed, including hemoglobin, hematocrit, mean corpuscular volume, red blood cell count, white blood cell count (including automated differential), platelet count, sodium, potassium, calcium, chloride, creatinine, urea, uric acid, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, γ-glutamyl transferase, albumin, glucose, bilirubin, thyroid-stimulating hormone, and urine sediment. The administration of the radioactive tracer (via i.v.) was performed as per standard sterile procedures established in the clinical PET and Nuclear Medicine clinics. After injection, patients were monitored for injection site reactions, along with following parameters: a) vital signs including pulse rate, blood pressure, temperature, and respiratory rate were recorded before and up to 3 h after injection, and b) observation for any unanticipated adverse effects (AEs) by the Stanford Cancer Clinical Trials Office (CCTO) Safety Coordinator for up to 10 working days. The AEs that were monitored post tracer injection at 1 hour, 24 hours and 7 days include blood pressure, electrocardiogram (EKG), oxygen Levels (%), heart rate (bpm), complete blood count hemoglobin (g/dL), white blood cells (WBC: k/µL), and platelets (k/µL). Results of AEs were recorded in Adverse Events Communication Form. Further, in our previous pre-clinical studies with anti-CA6 mAbs, we have observed that no AEs were reported. 17
PET/CT Imaging of CA6 Expression in Cancer Patients
Rationale
The purpose of clinical radionuclide-based immunoPET imaging is to provide cancer biomarker specific information (e.g., CA6) by imaging patients after they have been injected with a radiotracer. In this study, we have used the novel [64Cu]BFab immunoPET tracer which has high and prolonged tumor binding on cells which express the CA6 receptor. This provided a good tumor-to-background ratio in pre-clinical studies, which is promising result for initiating clinical translation studies. We decided to do PET/CT scanning from head to toe so that all visceral organs or any osseous metastases would be included within the field of view. The scanning at 1-hour post injection time would reveal the blood pool distribution and allow us to determine early tumor uptake, if any. Since the ligand for CA6 is an antibody fragment (BFab), we imaged the patients at 24-hour post injection time to allow binding to CA6-expressing tumors and clearance of the tracers from non-target sites.
PET/CT Scanning Protocol
Two patients were imaged with 2 different PET/CT scanners at Stanford. The ovarian cancer patient was imaged with a GE Discovery 600 scanner (14 minutes per scan), while the breast cancer patient was imaged with a GE Discovery MI scanner (18 minutes per scan). Participants were scanned with a low-dose attenuation correction CT scan (ACCT) followed by vertex to toes PET emission scan at 1hour (Scan #1) post-injection (p.i). After 24 hours p.i. patients received a second low-dose ACCT scan and a second vertex to toes PET emission scan (Scan #2). ACCT scan was performed with 120 kV and 10 mA. Whole body scans were analyzed using MIM Software (MIM, Cleveland, OH) and they were independently reviewed by 2 board certified nuclear medicine physicians.
Rationale for tracer dose
Our aim of this study was to assess the safety and utility of [64Cu]BFab immunoPET tracer for imaging of CA6 expression in ovarian or breast cancer patients. The [64Cu]BFab tracer human dose estimate was 16.5 mCi per scan and 49.5 mCi per year, this was estimated based on the dosimetry study performed with [64Cu]BFab female in Nu/Nu mice. The maximum dose limit was computed based on the international guidelines (21CFR§361.1(b)(3)(i)), which is maximal radiation dose to an adult’s whole body. The upper limits for active blood-forming organs, lens of the eye, and gonads is 3 Rem/single dose (5 Rem/year) and 5 Rem/single dose (15 Rem/year) for the other organs, respectively. However, in this two-patient study, we intended to assess the safety of the tracer, hence we injected the first patient with a low dose of 5.5 mCi. Since the first patient (ovarian cancer) tolerated the injected dose without complication and that no significant [64Cu]BFab signals were detected above background, we increased the dose of [64Cu]BFab for the second patient (breast cancer) (∼11 mCi). The second patient was scanned on our Discovery MI PET/CT scanner since this newer scanner is equipped with digital SiPM technology and thus better image quality and sensitivity compared to the older GE Discovery 600 PET/CT scanner used for the first patient.
The first patient with ovarian cancer had her [18F]FDG-PET/CT (dose: 10 mCi) scan (GE Discovery 600 scanner) performed on January, 2017 demonstrating no definitive evidence of active disease and a mildly FDG avid left paraortic lymph node that was likely reactive. The ovarian patient was intravenously injected with the tracer (5.5 mCi/0.25 mg of DOTA-BFab) and imaged at 1 hour and 24 hours post injection. [64Cu]BFab-PET/CT scan (GE Discovery 600 scanner) was performed on January 2017. However, the patient had biopsy (biopsy on January 2017) proven disease in omentum, myometrium, serosa, fallopian tube, and ovaries.
The second patient with breast cancer had her [18F]FDG-PET/CT scan (GE Discovery MI scanner) performed on September 2017. [64Cu]BFab-PET/CT scan (GE Discovery MI scanner) was performed on October 2017. The breast patient was injected with 11.7 mCi (0.38 mg of DOTA-BFab) and imaged at 1hour and 24 hours post injection. PET/CT image analysis was performed by using a picture archiving and communication system (PACS) software (Carestream Health, Inc, Rochester, NY).
Immunohistochemistry
For the ovarian cancer patient, immunohistochemistry staining for the CA6 receptor was performed on the peri-pancreatic lymph node tissue sample acquired on November 2016 (prior to FDG PET scan) by endoscopic ultrasound guided fine needle aspiration that was initially positive for adenocarcinoma. Patient 1 had 3 neoadjuvant chemo cycles on every 2 weeks between November to December 2016. On January 2017, patient underwent [18F]FDG-PET and [64Cu]BFab immunoPET scans. On January 2017 (4 days after [64Cu]BFab immunoPET scan) omentum from peritoneal tumor was excised for IHC staining of CA6.
For the breast cancer patient, IHC staining for the CA6 receptor was performed on the liver core biopsy (September 2017), after [18F]FDG-PET scanning on September 2017. Both of these patient samples were fixed and processed into paraffin blocks according to standard hospital procedures. Blocks or 5-µm slides were sent to Sanofi for immunohistochemical evaluation. 5-µm thick sections were prepared from the blocks as needed. Immunohistochemical procedure was carried out on a Discovery XT platform (Roche Ventana, Tucson, AZ) by following previous published methods. 23,24 CA6 expression was determined with SAR566658 (Internal Sanofi reagent) at 5.3 µg/mL as the primary antibody, and a mouse biotinylated anti-DM4 antibody (internal Sanofi reagent) at 5.16 µg/mL as the secondary antibody. Detection was achieved with DABMap (reference: 760-124, Roche Ventana) and counterstaining included hematoxylin (Catalog#: 790-2208, Roche Ventana) and bluing reagent (Catalog#: 760-2037, Roche Ventana). For each sample a set of 3 slides was prepared. The first slide was stained for CA6 using SAR566658, the second slide was incubated with an isotope control reagent (HuIgG1-SPDB-DM4, Sanofi reagent) and the last slide did not receive any specific reagent (buffer only). Additional slides were hematoxylin-eosin-saffron stained to support evaluation. IHC slides were evaluated by a pathologist by light microscopy. Percentage of tumoral cells stained and intensity of staining for membrane and cytoplasmic staining were scored. Results were summarized as H-score calculated by following formula: H-score = [(1 * percentage of cells staining at 1) + (2 * percentage of cells staining at 2) + (3 * percentage of cells staining at 3)]
Results
Radiolabeling Efficiency, Specific Activity, and Quality Control (Table 1)
Synthesis and quality testing of the pharmaceutical grade [64Cu]BFab tracer (Figure 1) was established previously. 17 Prior to radiolabeling the immunoconjugate purity by HPLC shown >99.5% with 1 DOTA chelate per B-Fab molecule (c/a). After radiolabeling both batches of radiopharmaceuticals, physical appearance was very clear solution, indicated 6.8 pH and yielded >99.5%. Radiolabeling efficiency of the [64Cu]BFab tracer was >90%, as measured by both TLC and HPLC. HPLC-purified tracer was >95% assayed by TLC and HPLC. Radiopharmaceutical Purity (by HPLC) and Specific Activity (by TLC) measured were >99.0% and 2.0 Ci/µmol, respectively. The results of the human serum stability, bacterial endotoxin, and immunoreactivity (mean ± SD) tests were reported as, >90%, < 2.5 EU/mL, and 69.0 ± 6 percentage, respectively. Sterility tests results clearly shown no growth, even after 14 days, observed no growth for any contamination. Overall tracer quality was summarized in Table 1.
Patients
Two patients who participated in this study were identified for proven focal ovarian lesions or breast lesions by CT and IHC from biopsy. Evaluation of [64Cu]BFab immunoPET was performed to measure the CA6 expression levels in 2 female cancer patients. Both patients were previously diagnosed with their malignancies, had initial resections of the primary tumor and have undergone multiple chemotherapy treatments prior to PET scanning.
The first ovarian patient was a 62 y/o female BRCA negative widely metastatic high-grade serous Mullerian adenocarcinoma that was diagnosed in November 2016 by [18F]FDG PET/CT and IHC of the biopsy. Three cycles of neoadjuvant carboplatin and paclitaxel, optimal interval debulking with robotic assisted hysterectomy, bilateral salpingo-oophorectomy, omentectomy, and lymph node dissection was completed by December 2016. This patient was scanned for [18F]FDG PET/CT by January 2017 and followed by [64Cu]BFab immuno-PET was performed a week later in January 2017. This patient received [64Cu]BFab tracer dose intravenously i.e. 203.5 MBq [5.5 mCi] / 0.25 mg of DOTA-BFab.
The second patient was a 47 y/o female with history of left intraductal breast carcinoma that was diagnosed in 2010. She was status-post lumpectomy, sentinel lymph node biopsy, adjuvant chemotherapy and radiation therapy. In 2014, she developed a low axillary recurrence (1 to 3 axillary lymph nodes (N1) that was treated with wide excision and a second round of chemotherapy. Later she was diagnosed with a second recurrence in the low left axilla that was treated with a complete left axillary lymph node dissection on April 2016 and post-operative radiation therapy in May 2016. This patient had [18F]FDG PET/CT scan on September 2017 demonstrating at least 4 FDG avid hepatic metastases, FDG avid bilateral hilar metastases, and an FDG avid osseous metastases at the T5 vertebra. Possible FDG avid lung metastases was also observed in the lower left lobe. For immunoPET scan, [64Cu]BFab tracer (433 MBq [11.7 mCi] / 0.38 mg of DOTA-BFab) was administered with on October, 2017.
Safety
Two board certified NM physicians reviewed the imaging data and they were in agreement with the following results: [64Cu]BFab PET scans were negative for both ovarian and breast cancer patients. With respect to [18F]FDG PET scans, ovarian cancer patient had no disease related uptake, while the breast cancer patient had positive [18F]FDG PET scan with multiple metastases. Further, the [64Cu]BFab tracer was well tolerated by both patients at the given doses with no safety concerns or adverse events. After administration of this novel immunoPET tracer, even after 7 days neither patients had significant changes in blood and urine parameters (Supplemental Table 2 S).
Tracer Distribution and Whole-Body PET/CT Imaging of [18F]FDG and [64Cu]BFab
The [18F]FDG and [64Cu]BFab tracers were utilized in both ovarian and breast cancer patients. The visual quality of the immunoPET images varied slightly between the 2 patients (Figures 2F and 3F). After 1 hour, [64Cu]BFab radioactivity was mostly confined to the blood pool, SUV (19 and 21 for patients #1 and #2, respectively) from aorta, as expected with an antibody fragment-based radiotracer. 5 After 24 hours post injection, [64Cu]BFab uptake (measured as SUV) could be observed in the liver, kidneys, and spleen were 11.4, 65, and 11.4, respectively for ovarian patient. The immunoPET SUV of 24 hours p.i. for breast cancer patient was 14, 77, and 11, for liver, kidneys, and spleen, respectively.

PET/CT images of the ovarian cancer patient for CA6 expression. Maximum intensity projection (MIP) images using [18F]FDG-PET (A) performed 1 week prior to the [64Cu]BFab-PET (D). [18F]FDG-PET scan from mid skull to thigh (A-C), and the whole body [64Cu]BFab-PET (D-F) scan were performed 24 hours after administration of the tracers. The MIP-[18F]FDG-PET, CT, PET/CT (A-C) and MIP-[64Cu]BFab-PET, CT, PET/CT (D-F) images showed no significant tumor uptake. The [18F]FDG uptake in panel A [18F]FDG-PET corresponded to brown fat distribution.
Representative images of immunoPET from both patients at 24 hours post injection of [64Cu]BFab and 1 h after [18F]FDG are presented in Figures 2 and 3 as the maximum intensity projection (MIP), CT, and PET/CT images of [18F]FDG-PET (A-C) and [64Cu]BFab-PET (D-F). Mid skull to thigh [18F]FDG-PET scan for the ovarian cancer patient post chemotherapy treatment (Figure 2A-C) was performed 1 week prior to the [64Cu]BFab-PET (Figure 1D-F). Mid skull to thigh [18F]FDG-PET (Figure 3A-C) scan for the breast cancer patient post chemotherapy treatment was performed 1 month prior to the [64Cu]BFab-PET (Figure 2D-F). In Figure 2A, the [18F]FDG-PET MIP image of ovarian cancer patient with mid skull to thigh scan shows no tumor uptake but with brown fat distribution. The same ovarian cancer patient had [64Cu]BFab-PET scan performed at 24 hours after tracer administration and no tracer uptake for tumor lesions was observed (Figure 2D). There were no lesions that were seen in the [18F]FDG-PET scan at 1 hour post tracer injection (Figure 2).

PET/CT images of the breast cancer patient for CA6 expression. Maximum intensity projection (MIP) images using [18F]FDG-PET (A) performed 1 month prior to the [64Cu]BFab-PET (D) tracer. [18F]FDG-PET mid skull to thigh (A-C), and whole body [64Cu]BFab-PET (D- F) scans were performed 24 hours after administration of the tracers. The images of MIP-[18F]FDG-PET, CT, PET/CT (A-C) and MIP-[64Cu]BFab-PET, CT, PET/CT (D-F) corresponds to [18F]FDG and [64Cu]BFab-PET scans, respectively. (A) [18F]FDG-PET uptake was indicated by arrows indicating the FDG avid metastases in the liver, bilateral hila, and thoracic spine. The corresponding SUVmax of FDG uptake were identified by the serial numbers (1 = 4.8, 2 = 10.9, 3 = 8.3, 4 = 12.7, 5 = 6.9 and 6 = 6.5) next to arrow. (D) Same breast cancer patient with whole body [64Cu]BFab-PET scan 24 hours after administration showed no tumor uptake above the background.
In Figure 3A, MIP image of an [18F]FDG-PET scan of indicated avid metastases in the liver, bilateral hila, and thoracic spine in the breast cancer patient. The corresponding SUV values are identified by the serial numbers (1 = 4.8, 2 = 10.9, 3 = 8.3, 4 = 12.7, 5 = 6.9 and 6 = 6.5) next to the arrows. A month after the [18F]FDG- PET scan, the same breast cancer patient had [64Cu]BFab PET scan performed at 1 and 24 hours after administration. The [64Cu]BFab-PET scan after 24 hours p.i. no tracer uptake (Figure 3D-F) was observed for any tumor lesions, but tracer uptake was noted to be very high in kidneys.
Tumor Tissue Staining Pre- and Post- Tracer Administration to Evaluate the CA6 Expression Level in the Ovarian Cancer Patient
Figure 4 displays the IHC staining for CA6+ and HES of pre- and post- therapy of the ovarian patient. Ovarian patient tissues from pre- (lymph nodes) and post- therapy (omentum from peritoneal tumor) were analyzed for CA6 expression that indicated expression levels were intermediate and weak, with an IHC H-score of 80 and 35, respectively. IHC H-score results of the liver metastasis in the breast cancer patient post chemotherapy was 35 (Figure 3S). Overall, IHC data indicated weak expression of CA6 in both cancer patients.

CA6-expression of ovarian cancer patient biopsies. Immunohistochemistry was carried out to evaluate CA6 expression in the ovarian adenocarcinoma patient (High-grade serous carcinoma = HGSC). Representative tissue specimens from the same patient of peripancreatic lymph node (A, pre-treatment) and omentum from peritoneal tumor (B, post-treatment of chemotherapy, surgery, and adjuvant-chemotherapy). Tissues were stained for CA6 expression (top panels) using the validated anti-human CA6 antibody SAR566658 (Internal reagent from Sanofi) and hematoxylin eosin saffron (HES) staining (bottom panels) for the evaluation of cell morphology. IHC analysis for pre- and post- treatment tissue specimens indicated H-scores were 80 (medium) and 35 (weak), respectively for CA6 expression. Original magnification was indicated on images as 10×, scale bar = 25 mm.
Discussion
The purpose of this study was to assess the safety profile of the tracer ([64Cu]BFab) in human patients, and to evaluate the uptake in CA6 antigen positive tumors to select patients for future immunotherapy using ADC. 18 Overall, the tracer preparation was of high quality with respect to purity, stability in human serum, and binding affinity in CA6+ cells (Table 1). Since most of the mAbs, antibody fragments, and conjugates are stable in blood, and show good PK properties, 25 hence we did not perform the tracer stability in serum of each patients. [64Cu] was used to label BFab (∼71 kDa) because of half-lives of each component. Since the half-life of [64Cu](12.7 hours), and BFab (<24 hours) are longer than commonly used small molecules or peptide-based PET radiotracers, it allowed patients’ immunoPET to be performed 24 hours post injection to visualize tumor specific signals upon clearance in normal organs and tissues that do not express CA6.
We feel compelled to report our results in this pilot safety study, even though we are unable to recruit additional participants, because the immunoconjugate shelf-life date was expired, and the sponsor (Sanofi) was not able to provide a new lot of the conjugate. Our tracer was administered into 2 patients diagnosed with ovarian or breast cancers. In both patients, the tracer was found to be safe and well tolerated. After administration of the [64Cu]BFab tracer, it cleared from most normal organs except for the kidneys during the first 24 hours. Although the quality of the [64Cu]BFab PET images was visually acceptable, the uptake of the individual organs was not appreciable (low [64Cu]BFab signals compared to the background) to accurately delineate individual organ ROIs. At the same time point, [64Cu]BFab did not show significant tumor uptake in both patients, compared to that of normal organs. Metastatic tumor uptake was observed in the breast cancer patient using [18F]FDG (Figure 3A). Subsequently this patient received chemotherapy prior to [64Cu]BFab tracer imaging and thus preclude the direct comparison of [18F]FDG vs. [64Cu]BFab PET scans.
Our [64Cu]BFab-PET imaging results were grossly correlated with IHC scoring, in which there was no significant CA6+ expression observed in the omentum from peritoneal tumor in the ovarian cancer patient (Figure 4), or in the liver metastasis in breast cancer patient (Supplemental Figure 2S). The lower detection limit of CA6 by immunoPET compared to that of IHC may be due to the partial-volume effects of PET for very small tumors that remained post chemotherapy treatment. 26 In addition, the clinical PET scanner we have used in this study has 4-6 cm spatial resolution, which may not be optimal to detect small tumor lesions. To overcome this limitation, the newer scanners with improved detector technology including small pixel size, better depth resolution and increased axial field-of-view in combination with enhanced image-reconstruction algorithms can be used for future studies. In spite of all these limitations, [64Cu]BFab immunoPET has the potential to become an important tool in the detection and treatment monitoring of CA6 positive-cancer. Theoretically, we can identify patients with high tumor uptake of [64Cu]BFab for ADC therapy using huDS6-DM4. Future clinical evaluation with this immunoPET tracer is warranted.
Conclusions
In this report, we presented the safety of [64Cu]BFab radiotracer in 2 human cancer patients. Overall, our novel [64Cu]BFab tracer was found to be safe and well tolerated. Further, quantitative analyses of the expression level of cell-surface CA6 in lesions by IHC staining (H-score) are in agreement with the overall weak immunoPET signals in both patients.
Supplemental Material
Supplemental Material, 02_Suplemental_data_Final_060420 - Two Patient Studies of a Companion Diagnostic Immuno-Positron Emission Tomography (PET) Tracer for Measuring Human CA6 Expression in Cancer for Antibody Drug Conjugate (ADC) Therapy
Supplemental Material, 02_Suplemental_data_Final_060420 for Two Patient Studies of a Companion Diagnostic Immuno-Positron Emission Tomography (PET) Tracer for Measuring Human CA6 Expression in Cancer for Antibody Drug Conjugate (ADC) Therapy by Arutselvan Natarajan, Shyam M. Srinivas, Carmen Azevedo, Lacey Greene, Anne-Laure Bauchet, Erwan Jouannot, Anne-Sophie Lacoste-Bourgeacq, Isabelle Guizon, Patrick Cohen, Anne-Laure Naneix, Ohad Ilovich, Jordan Cisneros, Krithika Rupanarayan, Frederick T. Chin, Andrei Iagaru, Frederick M. Dirbas, Amer Karam and Sanjiv S. Gambhir in Molecular Imaging
Footnotes
Acknowledgments
The co-authors would like to dedicate this work to Dr. Sanjiv Sam Gambhir (1962-2020) for his excellent mentoring and tremendous support through the years. We acknowledge Dr. Lucia Baratto for assistance with PET/CT image data analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We would like to acknowledge the funding from Sanofi Aventis (SSG), and National Cancer Institute (P50CA114747, SSG), for their support in funding of this research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
