Abstract
Ultrasound Contrast Agents (UCAs) consisting of gas-filled-coated Microbubbles (MBs) with diameters between 1 and 10 µm have been used for a number of decades in diagnostic imaging. In recent years, submicron contrast agents have proven to be a viable alternative to MBs for ultrasound (US)-based applications for their capability to extravasate and accumulate in the tumor tissue via the enhanced permeability and retention effect. After a short overview of the more recent approaches to ultrasound-mediated imaging and therapeutics at the nanoscale, phase-change contrast agents (PCCAs), which can be phase-transitioned into highly echogenic MBs by means of US, are here presented. The phenomenon of acoustic droplet vaporization (ADV) to produce bubbles is widely investigated for both imaging and therapeutic applications to develop promising theranostic platforms.
Keywords
Introduction
The goal of nanoscale theranostics is to reach targeted tissues and cells for delivering any cargo of interest (drugs, proteins, and DNA) and visualizing in real time the actual achievement of the task. Nanoscale dimensions are required for internalization of the carrier by cells, or for trespassing of anatomical, and/or functional membranes such as the skin, or the blood–brain barrier. Nanocarriers of different materials and characteristics have been so far proposed. Their design can be driven taking into account either the cells to be targeted or the visualization approach, for example, ultrasound (US), 1 magnetic resonance imaging (MRI), 2 and photoacoustic imaging (PAI). 3 -5
Being sensitive to variations in density and compressibility with respect to the surrounding medium, traditional US imaging requires the presence of acoustical scatterers. A very large deal of literature has been devoted to gas-filled microbubbles (MBs). As a matter of fact, MBs have been extensively used in the past as ultrasound contrast agents (UCAs). While their shell has been manufactured using different materials (polymers, lipids, or proteins), their core was mainly composed by inert and nontoxic gases, such as perfluorocarbons (PFCs), which have a very low solubility in blood. Many MBs were approved by the Food and Drug Administration (FDA) in the United States and the European Medicines Agency (EMA) in Europe and are commercially available since 20 years. Consequently, the increasing interest of the clinicians for an extensive application in medicine pushed the US equipment producers to develop ad hoc imaging techniques, such as contrast-enhanced US, to optimize the US backscattered reflection.
Such experience suggested the idea of manufacturing nanosized carriers able to permeate tissues and then to perform some phase change to become visible and monitorable by US medical sonography. Retracing the history of UCAs may be useful to arouse new ideas for future developments.
Ultrasound Contrast Agents
According to the core composition, UCAs can be divided into three main categories: (1) gas core, (2) liquid core, and (3) solid contrast agents. All of them present a density and compressibility substantially different from that of blood and tissues. MBs belong to the first class: they have a gas core surrounded by a shell and their size is usually on the order of a few microns. Upon US application, they not only enhance US backscattering but they can oscillate in the sound field, producing a nonlinear acoustic signal enabling the selective detection of the echoes from the MBs from those of surrounding tissue. 6 Recently, nanosized bubbles have been investigated. 22 Different types of liquid core contrast agents for US imaging have been prepared, tuning composition and size. Liposomes and PFC droplets are the most used nanoscale systems. Liposomes consist of a lipid bilayer with a hydrophilic liquid core. Air pockets within the lipid bilayer can generate acoustic reflectivity. 7 Nanodroplets (NDs) are US-triggered phase-change contrast agents (PCCAs) and their unique properties are discussed in the following sections of this review. Finally, solid nanoparticles (NPs) such as amorphous solid particles, including silica or iron oxide particles, can contain gas cavities in their pore structure. They showed the capability to generate detectable backscatter for US imaging. 8 Due to their size, nanoscale UCAs offer some advantages with respect to MBs, such as their ability to extravasate, providing the opportunity to image targets beyond the vascular system. Concerning liquid/solid UCAs, the relative incompressibility of their core produces a low acoustic reflectivity, making their detection somehow difficult. An overview on UCAs, their main properties, and structure is shown in Table 1 and Figure 1.
Overview on Contrast Agents Used in US Imaging and Their Main Properties.

Abbreviations: US, ultrasound; PFC, perfluorocarbon; SF6, sulfur hexafluoride.

Schematic core–shell structure of contrast agents used in molecular US imaging (not to scale). US indicates ultrasound.
MBs: From Diagnosis to Therapy
MBs are blood pool contrast agents because they are transported after intravenous injection in the bloodstream being unable to extravasate due to their size (1-10 µm). 38
According to the Rayleigh description, US backscattering depends on a combination of two factors: the high compressibility of the gas core and the different density with respect to the background. Whenever a sound wave hits such gaseous particles in a liquid medium, the difference between their acoustic impedance becomes remarkable, thus resulting in a stronger echo and greater acoustic energy detectable by standard sonoghaphy. Following insonation, MBs behave as very efficient US scatterers: they start to compress and expand as the gas responds to the pressure oscillations of the acoustic wave. 1,39 When properly modulated, US may elicit nonlinear acoustic signals for selective echoes detection. 6 Since bubble deletion occurs in the bloodstream and may prevent distal detection, the MB stability is crucial, and since 1990, several types of UCAs have been developed to improve bubble lifetime. Albunex (Molecular Biosystems, San Diego, California) was the first UCA available on the market in 1994. It consisted of air bubbles encapsulated by a human albumin shell with the aim of increasing stability. 40 Since then, many other agents have been introduced in clinical practice. Currently, the majority of the UCAs has a lipid coating and contains gases with low diffusivity in blood to further increase bubble half-life. The gases used in commercially available UCAs are sulfur hexafluoride in Sonovue/Lumason (Bracco Imaging, Milan, Italy), 41 octafluoropropane (OFP) (C3F8) in Definity/Luminity® (Lantheus Medical Imaging, North Billerica, Massachusetts), 42 and decafluorobutane (DFB; C4F10) in Sonazoid (Daichii Sankyo, GE Healthcare, Tokyo, Japan). 43 Following Albunex, a new albumin-based formulation was developed encapsulating OFP, named Optison (GE Healthcare AS, Oslo, Norway). 44 However, albumin results in a rather thick and rigid coating, 12 which limits bubble oscillations. On the contrary, lipid-coated bubbles, being the shell more flexible, allow relatively large oscillations, 9 whereas a polymer coating can increase the stability. 10 Nevertheless, polymer-coated contrast agents have more rigid shells, which need to be cracked in order to vaporize the oil core or let the gas escape, providing imaging contrast. 10,11 Both lipids and polymers offer more options to tune the chemical and mechanical properties of the shell, also giving more opportunities for functionalization and targeting of the bubbles.
Since 1990s, UCAs have attracted much research as carriers for drug delivery in order to transport drugs to specific diseased site and safely achieve the desired therapeutic effect, avoiding side effects. The benefit of using US in combination with MBs might be the delivering of the payload in a controlled way, by activating the release process only when the ultrasonic beam is switched on. Moreover, US can provide local real-time imaging. 45 These features allow a more specific delivery of the therapeutic agents, thus reducing the undesired side effects and improving the efficacy.
Generally, two modalities for drug delivery by UCAs are possible. In the co-administration approach, the UCAs are injected in the bloodstream alongside the therapeutic agent. Conversely, local US application can induce a transient increase in endothelial cell membrane permeability, enhancing therapeutic agent uptake by the target cells. 46,47 This method is also known as sonoporation. So far, several MB loading strategies have been investigated and proposed, such as loading drugs into MB core, or incorporating drugs within the shell through covalent or noncovalent interactions (Figure 2). 48 Therefore, MBs can act as a reservoir, and the chemical properties of the drug can affect the release kinetics. 49

Simplified sketch of main drug-loading techniques in microbubbles/nanobubbles (not to scale). Drugs can be embedded in the shell (1), incorporated in the core, in the case of gases or molecules soluble in PFCs (2), covalently attached to the surface (3). PFCs indicates perfluorocarbons.
Different MB formulations have been designed to promote the transport of hydrophilic as well as lipophilic therapeutic agents. The use of US as external trigger to improve drug delivery efficiency has been extensively studied, as reported by Kooiman et al. 50
Hybrid core–shell MBs based on biodegradable cross-linked polymers and phospholipids were described by Capece et al. 51 A self-assembly process starting from the preparation of liquid vesicles was tuned to obtain this system. It involved three steps: (1) the deposition of a phospholipid monolayer around a PFC droplet; (2) the addition of a hydrophilic polymer grafted with a vinyl moiety; and (3) the free radical photopolymerization to cross-link the grafted vinyl side chains. These liquid vesicles, in the presence of US, could successfully undergo the liquid to gas transition, thereby transforming the polymer vesicles into MBs.
A variety of drug-loaded droplets able to undergo a US-driven phase change to gas MBs has been extensively investigated in literature. In vitro results showed the delivery of the lipophilic drug chlorambucil (CHL) encapsulated by albumin/soybean oil-coated microdroplets to hamster ovary cells. 52 The authors found that incubation with the CHL-loaded NDs emulsion exposed to US almost doubled growth inhibition.
For the sake of completeness, we also report the use of MBs as contrast agent in other imaging applications. Interestingly, coupling graphene with MB surface offered the possibility to enhance photoacoustic signals. Paradossi et al designed an hybrid system, made of pristine graphene tethered to poly(vinyl alcohol)-based MBs and demonstrated a very good enhancement in the near-infrared (NIR) spectral region of the photoacoustic signal. 4 The MBs-containing Gadolinium (Gd) complexes can be exploited to obtain a dual imaging strategy, for example, phase contrast imaging and MRI. 53 Superparamagnetic iron oxide NP-modified MBs for US-MRI bimodality imaging have been proposed as well. 54
The Need for Smaller US-Monitorable Carriers
MBs are not able to passively extravasate due to their micron-scaled sizes, but they remain in the circulation until they are taken up by the spleen and the liver 55 or they dissolve in rather short times. 56 To overcome this limitation, nanosized formulations have been designed for US-triggered drug release outside the bloodstream. 49 In addition, MBs are too large to pass through the leaky vasculature of the tumor. Typically, tumor vessels are permeable from particles far lower than 1 µm, suggesting that the cutoff size of the pores is between 400 and 600 nm in diameter. 57 Based on these premises, targeting the tumor cell requires nanocarriers able to escape from the capillaries and enter the defective tumor microcirculation via the so-called enhanced permeability and retention (EPR) effect. 58 This feature can “per se” achieve drug accumulation in tumors without specific targeting ligands.
Nanoscale carriers may be designed as nanobubbles (NBs) or NDs, depending on whether the core is gaseous or liquid at room temperature. However, NDs show negligible nonlinear acoustic contrast due to the relatively incompressible liquid core. 59 -61 Upon US, they can be phase transitioned into MBs, thus generating acoustic contrast locally. 23,24,62 Unless a recently proposed procedure of “condensation” from micron-sized UCAs, 63 NDs are normally manufactured via sonication, 28 high-shear homogenization, 64 or high-speed mechanical agitation. 52,65 Briefly, an aqueous solution or dispersion of coating material is emulsified by one of the techniques mentioned above in the presence of liquid PFCs. The NDs typically result in a polydisperse size distribution. Lately, microfluidics-based devices have been investigated, which allow a finer control of the droplet diameter and production rate, by controlling gas pressure, liquid flow rate, and device geometry. 66 Sheeran et al described a low-temperature extrusion method to emulsify liquid droplets filled by DFB. Since DFB at room temperature is a gas, droplet generation requires that condensation occurs at reduced temperatures and/or increased pressures. In this study, droplets were first produced by condensing DFB gas at very low temperatures and then by encapsulating the resulting liquid DFB in lipid shells by membrane extrusion. A postsonication step can be carried out to produce NDs with a more uniform size distribution. 24
Sheeran et al investigated how to manufacture PCCAs containing highly volatile PFCs. 26 In their work, they showed that DFB (boiling point −2°C) can be incorporated into metastable liquid submicron droplets with proper encapsulation methods. The resulting droplets are activatable with substantially less energy than other more popular PFCs. Because DFB is a gas at room temperature, producing liquid DFB droplets by using conventional techniques, such as microfluidics-based devices, sonication, or homogenization, may be a challenge. The group of Sheeran proposed an alternative method of generating nanometer-sized lipid-coated droplets based on condensation of preformed MBs-containing DFB, through a combination of decreased environmental temperature and increased environmental pressure. 24 In the liquid state, the reduction in size to nanometer scale increases the Laplace pressure that, in turn, stabilizes the droplets against re-expansion at physiological temperature, unless an external trigger, such as US or heat, is applied. This method is known as “microbubble condensation” (Figure 3). In a subsequent investigation of MB condensation, Sheeran et al revealed the inherent tradeoffs involved with forming PCCAs from low-boiling point PFCs. 67 Martin et al proposed to accumulate perfluorocarbon NDs in a close-packed configuration and let them grow in size in situ, so that the phase-change conversion can occur at lower US pressures compared with isolated NDs. 68 Cavalli et al developed perfluoropentane (PFP)-cored polymer–lipid-coated NBs. They are a hybrid system containing a phospholipid monolayer at the NB interface, coated by a polymer shell to improve stability. 25 This type of formulation is referred to as “nanobubbles” for sake of simplicity, but it must be said that, prior to the US application, it would be more accurate to use the term “nanodroplets” when the core is constituted of PFP, being the PFC liquid at room temperature (boiling point 29°C). 69 The rationale of the hybrid lipid/polymer system is that phospholipid monolayers can adsorb charged polymers, such as polysaccharides exploiting various type of interactions. 70 Moreover, the addition of cosurfactant molecules to the phospholipid monolayer can play a synergic effect on the interfacial packing and surface tension. Shells of various thicknesses can be developed, and multilayer systems can be obtained by the layer-by-layer deposition method.

The exposure of preformed PFC MBs to decreased ambient temperature and increased ambient pressure results in condensation of the gaseous core. The decreased size results in an increased Laplace pressure, which serves to preserve the particle in the liquid state. Once exposed to increased temperature and energy delivered via US, vaporization of the droplet core results in a larger, highly echogenic gas MBs. PFC indicates perfluorocarbon; MBs, microbubbles. Reprinted from Biomaterials, Vol. 33, Sheeran PS, Luois SH, Mullin LB, Matsunaga TO, Dayton PA. Design of ultrasonically-activatable nanoparticles using low boiling point perfluorocarbons, 3262-3269, Copyright (2012), with permission from Elsevier.
NDs need to be stabilized to prevent coalescence. Most PCCAs are stabilized by either a single type of phospholipid, 26 a mixture of them, 27 or are entirely formulated of block copolymer. 28 In 2000, Kripfgans et al reported preliminary results for manufacturing albumin-coated droplets and studied their evaporation as a function of the applied acoustic pressure and frequency, together with a simulation predicting their lifetime based on gas diffusion. 65 Three years later, the idea of producing albumin-coated and other PFC-cored microdroplets undergoing US-mediated cavitation was investigated by Giesecke and Hynynen by in vitro determination of the inertial cavitation (IC) thresholds at various frequencies. 70 A recent study by Lee et al suggested that coating the NDs with a protein–polymer shell can improve their stability. 29 Capece et al studied the interface of hybrid shelled droplets encapsulating 2H,3H-decaperfluoropentane (DFP). 30 Droplet fabrication was based on the deposition of a dextran methacrylate layer onto the surface of surfactants. The droplets have been stabilized against coalescence by ultraviolet curing, introducing cross-links in the polymer layer, and transforming the shell into an elastomeric membrane with viscoelastic behavior. Following US exposure, the DFP core of the droplets vaporized transforming the particles into MBs. The presence of a robust cross-linked polymer shell conferred an unusual stability also during the core phase transition and allowed the recovery of the initial droplet state within a few minutes after switching off US.
PFCs have proven to be good candidates for liquid emulsions because they have low solubility in aqueous media and are nontoxic in small doses. 72,73 Due to their immiscibility in water, high-molecular weight, and low-surface tension, PFC-cored droplets can remain stable in circulation much longer than their gas-cored counterparts of similar size. 74 Depending on the PFC of choice, the resulting half-life in vivo ranges from hours to days, 75 which is an appealing feature for applications involving passive targeting and/or drug delivery. Low-boiling point PFCs, such as PFP, are particularly attractive because they allow the NDs to be designed in a metastable, “superheated” state from which they can rapidly turn to gas bubbles in response to US. Table 2 lists selected PFCs that have been used for ultrasonically activatable contrast agents along with their physical properties, whether available, collected from several resources of literature. 76,77 Therefore, these NDs are often called PCCAs 78 and the phenomenon is known as acoustic droplet vaporization (ADV).
Physico-chemical Properties of Selected PFCs Used for Liquid Nanoemulsion.
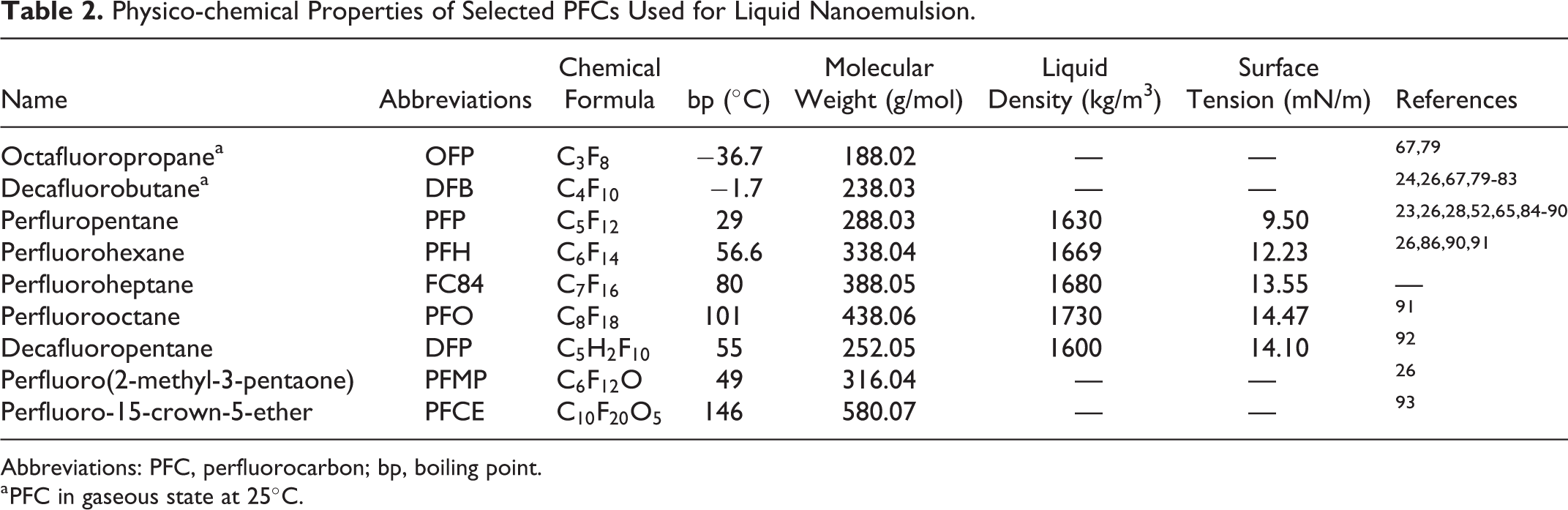
Abbreviations: PFC, perfluorocarbon; bp, boiling point.
a PFC in gaseous state at 25°C.
Finally, many solid nanocarriers have been introduced so far. The use of porous silica NPs to enhance the ND stability has been proposed as well. 94
Applications in Nanomedicine
Molecular Imaging of Cancer
One of the most important goal in tumor diagnosis is the early detection of cancer, for both primary tumors and metastasis. Two techniques can be used to achieve this goal. First, being tumor vasculature defective and characterized by wide fenestrations, leaking of nanocarriers makes the tumor detectable by imaging. 95 Second, the imaging agent can be functionalized with specific antibodies able to identify and bind the cells overexpressing the cancerous phenotype. Theoretically, targeted NDs can penetrate the endothelial barrier and bind those tumor cells. 96
Animal model experiments have demonstrated that ADV-based angiography can be used as real-time monitoring of the effect of the ultrasonic therapy in pancreatic cancer, breast cancer models, and kidney function. 93,97 Matsuura and colleagues used quantum dots incorporated in PFC droplets to show that they can act as additional cavitation nuclei within the core and appear to significantly decrease ADV threshold. 98 They suggested the idea that NP-loaded imaging agent may help in spatially and temporally controlling NP deposition: this approach paved the way to extend ADV-based imaging to other applications.
Therapeutic Delivery
The therapeutic strategy based on the interaction between drug-loaded NDs and US was first proposed in anticancer chemotherapy, for which temporal and spatial control of drug distribution is a goal. With respect to MBs, submicron PCCAs represent a novel approach to achieve localized drug delivery due to their unique properties. The EPR effect in solid tumors assumes that extravasation occurs from the capillaries through their nanometric porous structure (Figure 4).

Schematic representation of chemotherapeutic delivery through the defective tumor microvasculature using drug-loaded ND (not to scale). Hydrophobic drugs can be stored in the NDs cores. The tight junctions between endothelial cells in normal tissues do not allow extravasation of drug-loaded NDs. In contrast, tumors are characterized by fenestrated vasculature with large gaps between the endothelial cells, which allows drug-loaded nanodroplets to pass and accumulate in the tumor interstitium. US causes the NDs to evaporate into MBs, thus locally generating acoustic contrast and triggering the release of the drug. NDs indicates nanodroplets; US, ultrasound; MBs, microbubbles.
Tumor therapy with drug-loaded PFC nanoemulsions combined with US was studied in vitro and in vivo. Gao et al manufactured and characterized multifunctional NPs that combine the properties of polymeric drug carriers, US imaging contrast agents, and enhancers of US-mediated drug delivery. 99 At room temperature, the systems comprise PFC NDs stabilized by biodegradable block copolymer poly(ethylene oxide)-co-poly(l-lactide) or poly(ethylene glycol)-co-polycaprolactone shell. Upon heating to physiological temperatures, the NDs convert into NBs/MBs. Following intravenous injections, a longlasting, strong, and selective US contrast was observed in the tumor volume, indicating NB extravasation through the defective tumor microvasculature, suggesting their coalescence into larger, echogenic MBs in the tumor tissue. Under the action of tumor-directed US, MBs cavitate and collapse releasing the encapsulated doxorubicin (DOX) and dramatically enhanced intracellular drug uptake by breast cancer cells in vivo. Rapoport et al also developed polymeric micelles to encapsulate liquid PFP containing DOX. Increasing the temperature to 37°C caused the PFP nanodrops to vaporize into larger bubbles. It was proposed that US focused on the tumor could generate larger MBs formed by coalescence of the vaporized NDs. The authors showed the feasibility of using polymer-stabilized perfluoropentane NDs to deliver DOX to breast cancer xenograft tumors, proving that tumor growth could be arrested by this method. 19 In vitro US-triggered delivery of paclitaxel (PTX) to monolayers of prostate cancer cells was successfully reported for lipid-coated perfluorohexane (PFH) NDs. 86
In a very recent paper, Cao et al introduced phase-changeable drug delivery NDs with programmable low-intensity focused ultrasound (LIFU) that could trigger drug release significantly, enhancing anticancer drug delivery. Based on the difference of acoustic pressure between soft-shelled and hard-shelled nanosystems, lipid-based and poly(lactide-co-glycolide)-co-poly(ethylene glycol) (PLGA)-based NDs with encapsulated PFP and anticancer drug DOX were manufactured. By accurate acoustic energy deposition and coadministration of multiple NDs, a programmable drug-releasing profile was achieved, which could efficiently increase therapeutic effectiveness and decrease the course of chemotherapeutic treatment, as the authors demonstrated by both in vitro and in vivo studies. 100 The DOX-loaded acoustic phase-change NDs were developed for combined physical antivascular therapy and chemotherapy. The US stimulation can simultaneously induce locally vascular permeability and trigger drug release. 101 Recently, Marano et al published that glycolchitosan NDs loaded with PTX or docetaxel can target taxanes to castration-resistant prostate cancer cell lines. 102 Upon extracorporeal shock waves, NDs entered two different castration-resistant prostate cancer cells (PC3 and DU145), leading to higher cytotoxic and antimigration effect.
Although most of the research was devoted to cancer, other applications were also investigated. Prato et al introduced a new platform of oxygen nanocarriers, with DFP core and dextran shell which showed good oxygen carrying capacity, and no toxic effects on human keratinocytes after cell internalization, suggesting that US-activated nanocarriers might be used to topically treat hypoxia-associated pathologies of the cutaneous tissues. 92,103 Oxygen-loaded nanodroplets (OLNDs) are also internalized by cancer cells (eg, TUBO cells) and are localized only in the cytoplasm compartment (Figure 5). This result indicates that OLNDs are able to deliver their payload directly inside the cancer cells, minimizing the damage to the surrounding healthy tissue.

OLNDs internalization by TUBO cell line. TUBO cells were left untreated (left panels) or treated with FITC-labeled OLND PBS formulation (right panels) for 24 hours in normoxia. After DAPI staining, cells were imaged by confocal microscopy. Results are shown as representative images from three independent experiments. Top panels: Cell nuclei after DAPI staining (blue). Central panels: FITC-labeled OLND (green). Bottom panels: Merged images. Magnification: ×63. OLNDs indicates oxygen-loaded nanodroplets; FITC, fluorescein isothiocyanate; PBS, phosphate-buffered saline; 4′,6-diamidino-2-phenylindole (DAPI).
Ultrasound as external trigger to enhance the gene transfection efficiency has been investigated as well because of its safety and noninvasiveness for site-specific targeting, overmatching the other physical methods. 104 Gao et al developed an US-triggered phase-transition cationic ND based on a novel perfluorinated amphiphilic poly(amino acid), which could simultaneously load PFP and nucleic acids. 105 Cavalli et al developed a polymeric ND formulation, consisting of a chitosan-based shell and a PFP core for DNA delivery. 64 To obtain a nanoscale system, tetradecylphosphoric acid (C14) was added to the formulation. This amphiphilic molecule can localize to the PFP–water interface, lowering the surface tension. Chitosan was selected for the ND shell because of its low toxicity, low immunogenicity, and excellent biocompatibility. DNA-loaded NDs showed the ability to complex with and protect DNA. Transfection of COS7 cells in vitro was triggered by US, without affecting cell viability.
Theranostics
Theranostic systems provide imaging support to a therapeutic treatment, offering the potential to image the pathological tissues and simultaneously to monitor the delivery kinetics and biodistribution of a drug. The MBs/NBs have been proposed as multifunctional theranostic agents with the capability to provide US imaging and US-triggered therapy. Oddo et al designed a multimodal theranostic platform based on poly(vinyl alcohol)-shelled MBs coupled with superparamagnetic iron oxide NPs and an NIR fluorescent probe (indocyanine green) in order to support MRI and fluorescence imaging capability. 106 Various PLGA-shelled NB formulations have been investigated as theranostic system for imaging and tumor drug delivery. 107 -111 Rapoport et al have proposed a multifunctional imaging and treatment platform using nanosized PCCAs bearing a drug in the polymer shell in conjuction with drug-loaded micelles. Ultrasound, causing the PCCAs to vaporize, enhances local drug delivery, and the newly formed MBs can be used to monitor “on-the-fly” the effectiveness of the therapeutic treatment. 19 Cavalli et al developed a chitosan-based NB formulation, containing prednisolone phosphate as model drug and the paramagnetic complex 1, 4, 7, 10-tetra-azacyclododecane-N,N′,N′′,N′′′-tetrakis (methylenephosphonic acid) (Gd-DOTP) as T1-MRI agent. 25 In a recent work, porphyrins were combined with pluronic NBs to obtain an US-activated theranostic agent that exploits the sonodynamic activity in vitro. 112 Recently, cell-penetrating peptide targeted 10-hydroxy camptothecin-loaded lipid NPs were combined with LIFU for precision theranostics against hepatocellular carcinoma. A hyaluronic acid-mediated tumor accumulation was observed and after irradiation by LIFU, NPs turned into MBs by ADV, thereby enhancing US imaging and promoting local release of antitumor drug. 113
The Physics of ADV
The physical principles underlying ADV have not been completely clarified yet. Concerning the basic thermodynamics, in any closed system at a given temperature, the vapor pressure is defined as the pressure at which the liquid phase of a substance is in equilibrium with its vapor phase.
When the surrounding pressure is larger than the vapor pressure, the liquid substance remains in its condensed form. Conversely, when the surrounding local pressure drops below the vapor pressure, the liquid molecules will quickly escape from the gas phase (boiling) without any necessary changes in temperature. 114 The acoustic waves can modify the local pressure of the liquid, thus triggering the phase transformation, either from liquid to gas or backward from gas to liquid.
The Role of the Laplace Pressure
Being the ND a confined system, it experiences a Laplace pressure, which is defined as the pressure upon the interior fluid generated by the surface tension (or interfacial energy) between the two immiscible phases that compresses the liquid or gas inside the droplet:

where

Schematic plot showing the vaporization of the liquid droplet as soon as the local acoustic pressure drops below the pressure difference between the vapor pressure and the Laplace pressure (not to scale).
Finally, other authors reported subpressurization of liquid PFC droplets with no gas formation. 19,28,65,82 This phenomenon could be referred to “apparent subpressurization” (analogous to the apparent superheating) in which the Laplace pressure was sufficiently large that the vapor pressure at a temperature above the normal boiling point was still not greater than the local pressure inside the droplet. This may be due to the absence of a nucleation event. 114,115
The Impact of the US Parameters
Much discussion in the literature has been devoted to the mechanisms forcing a droplet to undergo a phase transition when US is applied and, in particular, to define its threshold.
ADV will be favored by increasing US frequency, pulse length, and peak-negative pressure (PNP) of the acoustic wave and by decreasing the Laplace pressure (both due to lower surface tension or larger droplet radius). Several authors experimentally confirmed these findings. 71,79,116 All these factors should be taken into account to optimize ADV process.
The relation between ADV and IC threshold, that is, when the sudden expansion and then rapid collapse of a gas bubble in response to a PNP occurs, was investigated by different groups. 71,91,117 -119 Their experiments focused on albumin- or lipid-coated microdroplets and demonstrated that the ADV threshold was lower than the IC threshold, meaning that the droplet-to-bubble transition occurs in less extreme conditions than those required for IC. The IC threshold was monitored through passive acoustic detection of albumin-coated microdroplets loaded with different PFCS, including those with boiling point higher than that of PFP (ie, PFH and perfluoromethylcyclohexane). 71 The PFC droplets of higher molecular weights and boiling points did not show larger IC threshold and thus superheating is not required to cavitate the droplets with US bursts. 71 This result was later confirmed by Rapoport et al in experiments with perfluoro-15-crown-5-ether NDs: the US pressures required to trigger their vaporization were only slightly higher than those for PFP NDs. 93 Kawabata et al proposed to use the sequential phase shifting of multiple liquids with different boiling points. 119 They considered NDS containing a mixture of PFP and DFP that could be vaporized at diagnostic US frequency: by changing the ratio of DFP to PFP, the US intensity required to induce sonographically significant vaporization changed. The authors hypothesized that vaporization of DFP with higher boiling point was favored by the fact that PFP was already vaporized. 119 In 2009, Rapoport et al showed that indeed US-induced droplet-to-bubble transition was substantially catalyzed by large preexisting bubbles irradiated by low-frequency US. 28,120 These results suggested a new strategy for improving the efficiency of droplet-to-bubble transition not only by mixing PFCs of different boiling point but also by using polydisperse droplet size distributions.
Other Open Problems
The main reason why the physical mechanisms of ADV still remain elusive relies in the large mismatch between the US wavelength and the droplet size. Nevertheless, such discrepancy may be overcome by a new recently proposed mechanism. 121 This study reveals that ADV is initiated by a combination of two phenomena: highly nonlinear distortion of the acoustic wave before it hits the droplet and focusing of the distorted wave by the droplet itself. At high excitation pressures, nonlinear distortion causes the formation of superharmonics, whose wavelengths are of the order of magnitude of the droplet size. These superharmonics strongly contribute to the focusing effect; therefore, the proposed mechanism also explains the observed pressure thresholding effect. This interpretation was validated with experimental measurements of the positions of the nucleation spots captured with an ultrahigh-speed camera, and an excellent agreement with the theoretical prediction was observed.
Another critical issue depends on the initial vapor nucleation required for ADV to occur. The basic explanation for the formation of a gaseous nucleus in an infinite liquid is provided by the homogeneous nucleation theory. It is usually assumed that the initial cavity formation takes place instantaneously. 122 Due to this very fast transition and the extremely small size of the initial vapor seed (on the order of 10 nm), 89 the nucleation pocket may entirely vaporize the droplet before the event becomes observable with the detection systems currently available. Experimental results actually show that some time is required: the probability of homogeneous nucleation of a growing gas bubble is proportional to the time window (at constant subpressurization) and increases exponentially with the magnitude of subpressurization. In 2016, Mountford and Borden pointed out that the mechanism of homogeneous nucleation may be an alternative explanation for the metastability of superheated NDs. Interestingly, they compared the homogeneous nucleation theory with some ADV results, 123,124 showing that a possible explanation was due to the subharmonic focusing of the incident US waves at the droplet–water interface, 121,125 which would increase the local PNP experienced within the droplet and facilitate the phase transition.
Theoretical Modeling
Theoretical modeling of ADV has been carried out as well. One of the first analytical approaches has been proposed by Qamar et al, 126,127 who modeled bubble evolution from microdroplets contained in a rigid tube. Their results were compared with the experimental data of Wong et al for PFP droplets. 125 The simulations by Qamar et al showed an interesting pattern by which under certain conditions, droplet evolution may be characterized by an overexpansion and oscillatory settling to a final diameter. Moreover, they assumed that the PFC mass evaporation rate was constant over the entire process of vaporization, which is a strong simplification, especially when the ambient pressure changed in response to the applied US field. Additionally, Qamar et al used the ideal gas law for describing the vapor bubble. More recently, Shpak et al proposed a simple model to describe the growth of a vapor bubble inside a PFP droplet, 128 hypothesizing that the vapor temperature inside the bubble is constant and equal to the boiling point for PFP (29°C at atmospheric pressure), and the bubble growth is driven by heat transfer from the surrounding liquid medium which has a temperature higher than that within the bubble. To compute the heat transfer, Shpak et al assumed a temperature gradient only within a thin (relative to the bubble radius) thermal boundary layer, whose thickness increases proportionally to the square root of time. 129,130 To take into account for the impact of US, they also included an effect called “rectified heat transfer.” 131 In another study, Shpak et al 132 modeled the growth of a vapor bubble by numerically solving the Keller-Miksis equation 133 in combination with the standard partial differential equations, describing the temperature field in the surrounding liquid, the heat transfer between the bubble and the liquid, and the gas diffusion through the bubble surface. 134 Regarding thermal processes, their model reproduces the approach used by Hao and Prosperetti for vapor bubbles in ordinary liquids. 135 Both models of Shpak et al assume that a vapor bubble is embedded in an infinite medium of liquid PFP, preventing the study of the bubble oscillations. The droplet-to-bubble transition has been modeled by Pitt et al who proposed a modification of the Rayleigh-Plesset equation. 136 Conversely to the models mentioned above, they considered a vapor bubble formed around a PFC droplet immersed in water, that is, the gas bubble formation initiates as a spherical vapor layer between the droplet surface and water and consists of a mixture of PFC and water vapor.
US Imaging Applications of ADV: Limitations and Perspectives
Many strategies using nanoscale systems have been proposed in order to improve the current formulations. As a matter of fact, some critical issues related to ADV at the nanoscale should be taken into account for future diagnostic and therapeutic applications. According to Laplace law, the smaller the droplet, the larger the internal pressure, thus increasing the saturation pressure required to trigger the droplet-to-bubble transition to higher temperature and acoustic pressure that are potentially harmful in clinical applications. The problem has been handled using different strategies ranging from droplet design to optimization of the US activation parameters.
In order to prevent the increase in ADV thresholds that occurs when the droplet size decreased, the use of volatile PFC has been proposed. The advantage in the vaporization thresholds obtained by increasing PFC volatility is, however, counterbalanced by a decrease in thermal stability (as the vaporization pressure and temperature are governed by the Clausius-Clapeyron relation). The stability of NDs can be improved with surfactant or stabilizing agent in the external media, 19,28,96,96,137 but the in vivo stability is still critical, due to the rapid dilution of the surfactants upon injection. In an early study by Kawabata et al, the authors showed that the ADV threshold can be finely tuned by mixing miscible PFC. 119 Some years later, Sheeran et al manufactured droplets composed of 1:1 mixture of DFB and OFP and showed that the resulted droplets had intermediate vaporization thresholds as well as intermediate stability. 67 Another strategy to lower the phase-transition threshold is to increase the size of the NDs. 71,116,118 Larger droplets are characterized by smaller Laplace pressure 138 and are more easily vaporized into bubbles at lower pressure with respect to nanoscale droplets. 26,97,139,140
Decreasing the US pressure threshold has been explored by changing the US parameters in terms of frequency 65,117 or pulse length. 118 Minimizing the mechanical index is often preferred in order to circumvent undesired side effects, but this can compromise the activation of the PCCAs in the acoustic field at depth due to attenuation. A technique called activation pressure matching has been proposed by Rojas et al to deliver the required pressure in order to produce uniform ND vaporization and to limit the delivered amount of energy. 141 They showed that such approach increases the time needed by a single bolus of NDs to generate useful contrast and to provide consistent image enhancement in vivo. Interestingly, Lin et al investigated the contrast enhancement produced by ultrasonically activatable PCCAs, either free or confined in a microvessel or microchannel phantom after US application. 142 Their experimental results indicated more than 1 order of magnitude less acoustic vaporization in a microchannel than that in a free environment taking into account the attenuation effect of the vessel on the MB scattering. This finding may improve the knowledge and the understanding in the applications of PCCAs in vivo.
Beside US, microwave-activated ND vaporization was investigated to overcome the critical issues of traditional ADV. Novel folate-targeted lipid-shelled NDs cored with a mixture of PFCs (ie, PFH, PFP) were prepared for highly efficient percutaneous US imaging-guided microwave ablation of tumors. 143
Much research has been devoted to the possibility of visualizing the NDs or inducing their vaporization adding other imaging modalities to US. Concurrent optical observation and PAI are the most popular approaches, 81,83,144,145 together with the use of surfactants detectable with MRI. 146,147 Interestingly, phase-change agents have been recently proposed as promising photoacoustic contrast agents. Highly absorbing optical contrast agents, such as gold NPs, can be encapsulated in MBs and NBs for multimodal imaging contrasts. Gold nanospheres have been encapsulated in bovine serum albumin shell of PFC droplet to obtain a dual-contrast agent capable of providing image contrast enhancement for both US and PAI modalities. 148 Other authors reported the preparation of gold NP-templated MBs, filled with various PFC gases, that can generate NDs by a condensation approach. 149
Very recently, NDs production from homemade, preexisting MBs, based on the use of cosolvents 150 or the acoustic destruction of MBs, 151 has been described by many researchers.
A very appealing idea for future development consists in manufacturing PCCAs directly from already commercially available MBs, such as Definity (Lantheus Medical Imaging) 152 and MicroMarker (Bracco, Geneva, Switzerland and VisualSonics, Toronto, Canada). 63 This strategy is very promising since off-label use of commercially available MBs may be simpler than obtaining FDA or EMA authorizations ex novo.
It is worth noting that the interaction of US pulses with the drug-loaded MBs produced by ADV involves a number of mechanisms, such as acoustic cavitation, heating, radiation forces, and sonoporation. 153 Stable bubble cavitation generates strong shear stress close to the bubble surface, thus impacting on cell membranes. The IC produces shock waves and high-speed microjets, which also disrupt cell membranes. The transient increase in cell membrane permeability allows the uptake of drugs, genes, and peptides from a variety of carriers (polymeric micelles, liposomes, and nanoemulsions). 114
Intriguingly, the internalization of targeted or nontargeted drug-loaded droplets into cells and the subsequent vaporization could lead to new tissue-specific therapy, allowing the controlled release of drugs directly to the diseased cells. Studies have shown that lipid-coated MB and liquid PFC droplets can be internalized into neutrophils, macrophages, and tumor cells. 154 -157 Preliminary investigations from Kang and colleagues demonstrated the PCCA uptake into peritoneal macrophages. 157
Conclusions
In summary, future design of theranostic platforms should address many challenges, such as good visualization, high payload, target delivery, and toxicity issues. A number of nanostructures have been developed as effective US imaging tools. Among them, PCCAs might be interesting as multifunctional systems for their specific physicochemical properties and flexible composition. However, further understanding of the NDs behavior as well as the physical mechanisms of ADV either in vitro or in vivo is still needed for the clinical translation.
Footnotes
Abbreviations
Acknowledgments
The authors thank Dr Klazina Kooiman and Prof Dr Ir. Nico de Jong from Erasmus Medical Center in Rotterdam for many fruitful discussions and Prof Umberto Lucia from Politecnico of Torino who provided insight and expertise that greatly assisted the research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Caterina Guiot and Roberta Cavalli were founded by University of Turin (ex 60%).
