Abstract
Ultrasonic molecular imaging employs contrast agents, such as microbubbles, nanoparticles, or liposomes, coated with ligands specific for receptors expressed on cells at sites of angiogenesis, inflammation, or thrombus. Concentration of these highly echogenic contrast agents at a target site enhances the ultrasound signal received from that site, promoting ultrasonic detection and analysis of disease states. In this article, we show that acoustic radiation force can be used to displace targeted contrast agents to a vessel wall, greatly increasing the number of agents binding to available surface receptors. We provide a theoretical evaluation of the magnitude of acoustic radiation force and show that it is possible to displace micron-sized agents physiologically relevant distances. Following this, we show in a series of experiments that acoustic radiation force can enhance the binding of targeted agents: The number of biotinylated microbubbles adherent to a synthetic vessel coated with avidin increases as much as 20-fold when acoustic radiation force is applied; the adhesion of contrast agents targeted to αvβ3 expressed on human umbilical vein endothelial cells increases 27-fold within a mimetic vessel when radiation force is applied; and finally, the image signal-to-noise ratio in a phantom vessel increases up to 25 dB using a combination of radiation force and a targeted contrast agent, over use of a targeted contrast agent alone.
Keywords
Introduction
Ultrasound contrast agents, which include microbubbles, nanoparticles, and liposomes, are used to enhance acoustic reflectivity of blood or tissue noninvasively. Site-specific adhesion molecules, such as antibodies, peptides, and polysaccharides, can be incorporated onto the contrast agent, creating “targeted” contrast agents [1]. Targeted ultrasound contrast agents were first demonstrated by Lanza et al. [2] using fibrin-targeted nanoparticles for detection of fibrin thrombi. After injection into the bloodstream, a targeted agent adheres to receptors at the targeted site, allowing disease-specific detection with a clinical imaging system [3]. It is also known that targeted microbubble agents can be displaced from the bloodstream and concentrated along the vascular endothelium by ultrasound driving pulses [4]. The force produced on microbubble contrast agents is due to the absorption of the momentum of the ultrasound wave, and is called primary radiation force. In this article, we demonstrate that ultrasound radiation force used in conjunction with targeted microbubble agents results in significantly increased agent binding and retention under physiologically relevant vessel sizes and flow conditions. This effect is illustrated in Figure 1.
Illustration of the effect of radiation force on targeted imaging with ultrasound. (A) Without radiation force, the majority of the contrast agents fail to contact the target site, and therefore do not bind. (B) Radiation force pushes flowing targeted contrast agents into contact with cells along a vessel wall, where they bind to target receptors.
Targeted Microbubbles for Ultrasound Molecular Imaging
Encapsulated microbubbles are highly echogenic due to large differences in compressibility and density between the encapsulated air and the surrounding tissue or plasma. The compressibility of a microbubble is four orders of magnitude greater than that of the surrounding plasma. A clinical ultrasound system is capable of detecting the echo from a single microbubble, a volume on the order of 0.004 picoliter [5]. Targeted ultrasound contrast agent microbubbles have great potential for molecular imaging because of this high sensitivity. Early proof of principle for molecular imaging with microbubble contrast agents was provided by Klibanov et al., who demonstrated the ability of targeted microbubbles to adhere to a specific ligand. Microbubbles incorporating a biotinylated lipid (0.15–7.5 mol%) adhered to avidin-coated Petri dishes and the biotinylated bubbles remained adherent to the dish under tangential flow rates up to 0.6 m/sec [6]. Unger et al. [7,8] have developed microbubbles labeled with an arginine–glycine–aspartic acid (RGD) peptide analog that bind to GPII-bIIIa receptors on activated platelets. In initial studies, these microbubbles were demonstrated to enhance echogenicity of induced thrombi in canines and human thrombi in vitro [8,9]. Schumann et al. [10] developed a fixed-plate assay for microbubble targeting in which activated human platelets in 96-well plates were incubated with microbubbles, rinsed, and scanned by a visible light plate reader. The optical density of the plates was not significantly changed by nontargeted microbubbles, but increased with an increasing concentration of targeted microbubbles (up to 30 μL per well). The Lindner group has demonstrated that microbubbles targeted to αv integrins (incorporating either monoclonal antibodies against murine αv, or the RGD-containing disintegrin echistatin) are retained in the FGF-2 stimulated mouse cremaster muscle, and can be detected with ultrasound in subcutaneous matrigel plugs [11]. In another study, microbubbles targeted to αvβ3 (incorporating echistatin) were detected in a rat glioma model by both ultrasound and confocal microscopy [12].
Recently, Dayton et al. [13] demonstrated adhesion of αvβ3-targeted contrast agents to cell lines grown in monolayers on acoustically transparent cover slips. Optical and acoustic data illustrate an order of magnitude greater adhesion of targeted agents as compared with control agents, and specificity was demonstrated using blocking. This article demonstrated that the back-scattered echo amplitude increases monotonically with adherent microbubble density, for densities below 15,000 bubbles/mm2, and that the rate of decorrelation between multiple echoes from adherent targeted agents is a function of acoustic pressure and bubble density.
Ultrasound Radiation Force
When a traveling ultrasonic wave is absorbed by a particle, the momentum associated with the wave produces a net primary radiation force. While incompressible objects do experience radiation forces, compressible objects driven at their resonant frequency experience far larger forces and can be observably displaced by low-amplitude ultrasound waves. The effects of radiation force were first reported in 1906 by C. A. and V. F. K. Bjerknes, when they observed the attraction and repulsion of air bubbles in a sound field [14]. Since this initial discovery, many research groups have published theoretical and experimental analyses of radiation forces (also termed “Bjerknes Forces”) on spheres. Only recently, however, have researchers examined the radiation force produced on contrast agent microbubbles with MHz-frequency ultrasound [15,16]. Fortuitously, the resonance frequency of contrast microbubbles is in the frequency range of clinical ultrasound systems, 1–10 MHz. A microbubble driven near its resonance frequency experiences a large net radiation force in the direction of ultrasound wave propagation; pulses of many cycles can deflect resonant microbubbles over distances on the order of millimeters. Thus, it is possible to bring contrast agents circulating in the blood pool into contact with targeting sites on a blood vessel wall. Additionally, when agents are concentrated near a vessel wall, they travel at a reduced velocity compared to those in the center of the flow stream; this was demonstrated using intravital microscopy in Dayton et al. [4]. In addition to primary radiation force, which acts in the direction of acoustic wave propagation, a secondary radiation force for which each individual bubble is a source and receptor causes the agents to attract or repel each other. The result of this secondary force is that a much larger concentration of microbubbles collects along a vessel wall than might otherwise occur.
Upon observation of the localization and reduced flow velocity of microbubbles traveling near the wall of a mouse arteriole in vivo, Dayton et al. postulated that radiation force could be used as a technique to assist the adhesion of targeted contrast agents. Assuming that the great majority of microbubbles and targets will reside in capillaries, it may be sufficient to displace the agent only a few microns. Our goal, however, is to displace the agents a distance on the order of hundreds of microns in order to promote receptor–ligand interactions in small arteries and veins as well.
Outline of Approach
In this article, we will first describe the equations used to model ultrasound radiation force, and describe the parameters used in our numerical evaluations. We will then describe several experiments that demonstrate the use of radiation force to enhance adhesion of targeted microbubbles to ligands on surfaces and cell layers. In the Results section, we show how the theory and experiment both indicate that radiation force can deflect targeted ultrasound contrast agents towards vascular endothelium, and that this technique has important consequences for ultrasound molecular imaging.
Materials and Methods
Theoretical Description of Ultrasound Radiation Force Very briefly, the displacement of an ultrasound contrast agent driven by an ultrasound pulse is modeled as follows. First, given information about a fluid and a contrast agent contained in that fluid, it is possible to model the radial oscillation of the agent in response to a given pressure wave. Next, the time-dependent radiation force on the bubble can be determined from the driving pressure wave and the time-dependent volume of the agent (calculated from its radial oscillation). Finally, the translational motion of the bubble in response to the radiation force can be calculated by solving the particle trajectory equation of the bubble. Each of these steps is described mathematically below.
Model for microbubble oscillation. The first step in the prediction of the effect of an acoustic pulse on a contrast agent is the modeling of the radial oscillation of the agent. Our laboratory has developed a model for the oscillation of a shelled microbubble subject to an applied pressure field based on the Rayleigh–Plesset equation [17]. It describes bubble radial oscillation at low driving pressures and has been validated by high-speed optical observation of oscillating bubbles. Given properties of the bubble—including its initial radius, gas composition, and shell stiffness—and properties of surrounding fluid, Equation 1 predicts the radius–time curve of a bubble driven by a pressure wave. The notation for equations described in this article is provided in Table 1.
According to this theory, the linear resonant frequency of a shelled microbubble is given by Equation 2:
Where
Notation

and
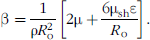
Ultrasound radiation force. Once the microbubble radius has been predicted, it is used to calculate the microbubble's time-dependent volume and thus the primary radiation force experienced by the bubble. The primary radiation force on a compressible sphere is described by Equation 3, which relates the radiation force to the instantaneous volume of the bubble and the gradient of driving pressure [18].

Simplified forms of this equation can be derived for the case of a traveling sinusoidal wave, and the linearized solution found following the methods in Dayton et al. [16] and Leighton [18] is given by Equation 4.

This form is valid for cases in which the microbubble oscillation is small compared with the initial radius.
The damping coefficient in Equation 4 can be rewritten in terms of a dimensionless damping coefficient δtot = 2βtot/ω0, which combines the effects of radiation, thermal, and viscous losses and is assumed to be 0.25 for the range of conditions examined here based on Leighton [18], page 188. For a microbubble driven at resonance, Equation 4 simplifies to Equation 5:

Translation of the microbubble. We have also developed a model for the translational motion of a microbubble in a fluid during insonation [15]. The particle trajectory equation is given by Equation 6.

where FR represents radiation force; FQS, quasi-static drag force; FAM, added mass due to bubble translation; and FRB, added mass due to bubble oscillation. This equation may be derived from first principles for an unsteady Stokes flow around a rigid sphere [19]. The mass of the bubble can be expressed as ρbVb, and the acceleration is the time derivative of the bubble velocity. Thus, the product of the mass of the bubble and its acceleration on the left-hand side of Equation 6 may be expressed as.
In Dayton et al. [15], we demonstrate that Equation 6 can be written as Equation 7, in order to solve for the motion of a bubble translating due to acoustic radiation force.
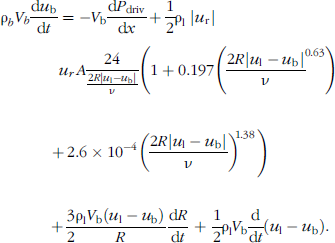
In Equation 7, ur = u1 – ub is the relative velocity between the bubble and the liquid. Other research groups have developed coupled models for oscillation and translation for unencapsulated bubbles [26].
Numerical Evaluation of Equations 1–7
We evaluate Equations 1–7 using experimentally relevant parameters in order to predict the translational velocity and the displacement of a contrast microbubble driven by an ultrasound pulse. The parameters used for the gas core and microbubble shell are those used in previous studies [17,20], and are listed in Table 2. The term μshε in Equation 1 is calculated using the empirical relationship μshε = 1.9Ro – 1.1 [21]. To evaluate the change in radiation force with pressure, we evaluate driving pressures of 14 and 100 kPa; 14 kPa is used in our experiments. In order to evaluate radiation force as a function of the center frequency of the driving pulse, we compare a 3-MHz sinusoidal pulse, a 5-MHz sinusoidal pulse, and a sinusoidal pulse at the resonant frequency of each bubble over the range from 2 to 8 MHz. In each of these cases, we use a pulse length of 3.3 μsec, which corresponds to 10 cycles of a 3-MHz sinusoidal pulse. In the experiments, a much longer pulse (5 million cycles at 3 MHz) is used to facilitate measurement of displacement and translational velocity of the microbubble. Our past experience has shown that the displacement is approximately a linear function of pulse length for low-pressure, long-driving pulses, therefore, we are able to evaluate the response to a short pulse in order to minimize computational time, but we can compare theory and experiment by simply scaling the results by the number of acoustic cycles.
Parameters for numerical evaluation

Contrast Agent Preparation
Two types of contrast agents were used in the experiments described in this article. In the cell adhesion experiments, we used RGD-targeted microbubbles MRX 408, which were provided by ImaRx Therapeutics (Tuscon, AZ) and have been described previously [7]. MRX 408 used in these experiments had a size distribution with a mean radius of 1.0 μm and a standard deviation of 0.8 μm. The remainder of the experiments employed biotin-targeted and untargeted microbubbles, which were made by the tip sonication method with decafluor-obutane gas as previously described [22] less than 1 hr prior to commencing each experiment. All lipids were from Avanti Polar Lipids (Alabaster, AL). 1,2-Distearoyl-sn-glycero-3-phosphocholine (DSPC), 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-2000] (DSPE-PEG2000), and 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[biotinyl(polyethylene glycol)2000] (DSPE-PEG2000-Biotin) were dissolved and mixed in chloroform, dessicated in nitrogen gas and vacuum, and then resuspended in 0.1 M Tris buffer by sonication for 45 min in a bath heated to 60°C. The resulting aqueous cocktail contained 0.75 mg/mL DSPC with 91 mol% DSPC and 9 mol% DSPE-PEG2000 for control experiments, and 82 mol% DSPC, 9 mol% DSPE-PEG2000, and 9 mol% DSPE-PEG2000-Biotin for targeting experiments. The size distribution of these agents is illustrated in Figure 2; they had a distribution of sizes with a mean radius of 0.8 μm and a standard deviation of 0.7 μm. All microbubble counts were measured with a particle sizer (Coulter Multisizer, Fullerton, CA) less than 20 min prior to experimentation to ensure proper dilution.
Bubble resonant frequency as a function of initial radius (left axis, solid line) and size distribution of experimental bubbles (right axis, squares).
Experimental Setup
We employ an experimental system which consists of an optically transparent synthetic vessel submersed in a water bath. Temperature of the water bath was at 25°C for all experiments except for the αvβ3-targeting, where the water bath was maintained at 37°C. A Mikron IV500L (Mikron Instruments, San Diego, CA) microscope interfaced with a video camera allows us to observe and record micron-sized agents inside the vessel. An ultrasonic transducer is also positioned in the water bath such that its acoustic focus overlaps with the optical focus of the microscope. We use transducers with center frequencies of 2.25 MHz (Panametrics V305, Waltham, MA) and 5 MHz (Panametrics V309, Waltham, MA). The 2.25-MHz transducer has a –6 dB bandwidth of 1.5–3.3 MHz, a focal length of 2 in., and an aperture size of 0.75 in. The 5-MHz transducer had a –6 dB bandwidth of 3.7–7.6 MHz, a focal length of 2 in., and an aperture size of 0.5 in. In each case, we use an arbitrary waveform generator (AWG 2021, Tektronix, Irvine, CA) to produce a waveform, which is amplified approximately 55 dB with a radio-frequency power amplifier (3100LA, ENI, Rochester, NY) for transmission through the transducer.
Acoustic pressure calibrations are performed using a needle hydrophone (PZT-Z44-0200, Specialty Engineering Associates, Sunnyvale, CA) and a preamplifier (A17dB, Specialty Engineering Associates, Sunnyvale, CA) connected to a digital oscilloscope (9350, LeCroy, Chestnut Ridge, NY). The hydrophone tip is placed in the optical field of view. Transducer positioning is also performed using the hydrophone—to ensure that the optical and acoustical foci overlap, we maximize the signal transmitted by the transducer and received by the hydrophone while the hydrophone tip is fixed in the optical field of view. Acoustically absorbent rubber was used to line the back wall of the water tank to eliminate standing waves and minimize reflections.
Procedures
Velocity measurement. For measurements of the velocity of contrast agents during the radiation force pulse, individual contrast agent microbubbles were observed in a 200-μm cellulose tube, and a high-speed camera system (Kodak Motioncorder Analyzer 1000) recording at 240 frames/sec was used to record each agent translating in response to ultrasound pulses. The translational velocity was determined off-line using NIH Image (National Institutes of Health, Bethesda, MD).
Avidin–biotin targeting. To demonstrate the effect of radiation force on biotinylated agent binding to an avidin-coated surface, a 200-μm cellulose tube was used. It was cleaned with 100% ethanol, and then pretreated for 30 min with 5 mg/mL avidin. A solution of biotinylated contrast agents diluted to approximately 3000 microbubbles/μL was pumped through the tube. Mean flow velocities of 6, 28, and 142 mm/sec were used; these velocities correspond to wall shear stresses of 0.2, 1.2, and 5.7 Pa. These values were chosen to cover and exceed the shear stresses in the physiologically relevant range for 10–60 μm microvessels, which average ~2.4 Pa in the arterioles and ~1.7 Pa in venules [23]. Before each observation, the tube was translated to allow optical observation of a wall area free of adherent microbubbles. A radiation force pulse approximately 1.67 sec long, with a peak negative pressure of approximately 14 kPa, was then applied, and the number of contrast agents adherent within the 0.035 mm2 field of view was recorded. A microbubble was determined to be adherent if it remained stationary on the tube wall during flow for at least 1 min. At least 10 observations were made under each of four sets of experimental conditions: Using biotin-targeted bubbles and avidin-coated tubing with radiation force, nontargeted bubbles with avidin-coated tubing with radiation force, biotin-targeted bubbles with noncoated tubing with radiation force, and biotin-targeted bubbles and avidin-coated tubing without radiation force.
αvβ3-targeting. For cell targeting experiments, a 1-mm diameter synthetic polymer tube was used. The tube was disinfected in a 10% hydrogen peroxide solution for 30 min and then rinsed three times with PBS. Human umbilical vein endothelial cells (HUVEC) (Cascade Biologics, Portland, OR) were seeded into the tubing at a concentration of approximately 5 × 104 in 30 μL, and allowed to incubate in Media 200 (Cascade Biologics, Portland, OR) for 48 hr. Growth of an adherent monolayer of HUVEC on the luminal side of the tube was confirmed by microscopy. The experiments were performed and analyzed in the same manner as the avidin–biotin targeting experiments, above; in this case the optical fields of view measured 0.07 mm2, and the radiation force pulses had a center frequency of 3 MHz, duration of 1.67 sec, and peak negative pressure of 40 kPa. Ten pulses were administered over 100 sec with pulse repetition rate of 0.1 Hz. Flow velocity in the 1 mm tube was maintained at 5 mm/sec, corresponding to a shear stress of 0.04 Pa. Adhesion of microbubbles after radiation force pulsing was compared with adhesion observed over the same time period without ultrasound.
Ultrasonic imaging of adherent contrast agents. Acoustic interrogation of adherent contrast agents was performed using a pulser–receiver system (SP801, BR640, Ritec, Warwick, RI) in conjunction with a diplexer (MDX, Ritec, Warwick, RI). For acoustic experiments, the tube was oriented at an angle with respect to the transducer to eliminate reflections from the tube wall. The 5-MHz transducer was used both for administration of radiation force pulses and for imaging, by switching between the waveform generator/power amplifier system and the Ritec pulser–receiver system. Echoes were recorded using 1.5-cycle pulses at 5 MHz. Received echoes were amplified by 40 dB, bandpass filtered between 1 and 12 MHz, and digitized at 100 MHz using a digital oscilloscope (9350, LeCroy, Chestnut Ridge, NY). Data were recorded on a computer through a LabView interface, and analyzed off-line using MATLAB. A low-pulse repetition frequency of 10 Hz was used to minimize the radiation force and destruction produced by the imaging pulse.
Sets of 100 acoustic echoes were recorded from one location in a 200-μm avidin-coated cellulose tube: First, containing only water; second, containing biotinylated contrast agents in solution; and third, after ultrasound pulses were applied to the agents and the tube was flushed thoroughly with water to remove freely flowing agents. Frequency spectra presented in the Results section were averaged over sets of 100 echoes. Results were corrected for transducer frequency response. Under control conditions, the same procedure was followed with no ultrasound applied. In each case, the flow velocity in the tube was 23 mm/sec, corresponding to a shear stress of 0.92 Pa.
Results
Predictions of Theory for the Radiation-Force Assisted Targeting of Ultrasound Contrast Agents
Several important implications for radiation-force assisted targeting of ultrasound contrast agents can be drawn from the theoretical framework outlined in Equations 1–7 and our numerical evaluations of microbubble behavior in response to radiation force.
Equation 2 shows that the resonant frequency changes as a nonlinear function of the initial radius of the microbubble. Typical ultrasound contrast agent preparations are polydisperse, with radii ranging from less than one micron up to several microns; so there can be a wide range of resonant frequencies. The resonant frequency of lipid-shelled contrast agents with initial radii ranging from 1.8 to 0.6 μm is shown in Figure 2, along with the size distributions of the experimental agents used in this article. It is apparent that the resonant frequency of a single microbubble agent could be anywhere between 2 and 8 MHz, approximately twice the bandwidth of a typical clinical ultrasound transducer.
Equation 4 provides insight as to the effect of ultrasound parameters on the magnitude of radiation force. Radiation force can be increased with increasing driving pressure, increasing pulse duration, or decreasing pulse interval. In Equation 5 we note that the force is proportional to the microbubble radius normalized by the resonance frequency (Ro/ωo), proportional to the resonance radius squared, or inversely proportional to the square of resonance frequency.
Figure 3 gives typical results from the numerical evaluation of radiation force using Equations 1–7. The instantaneous radiation force for a microbubble with an initial radius of 1.0 μm, driven by a 3-MHz 10-cycle pulse at 14 kPa, is shown in Figure 3A. The force oscillates as the driving pressure changes sign, and at this low pressure it is nearly symmetric about zero. The resulting translational velocity of the microbubble is on the order of centimeters per second, but it is not continuously unidirectional; it changes direction with the compressional and rarefactional portions of the acoustic pulse (Figure 3B). A net translation away from the transducer occurs because the oscillation of the bubble is slightly nonlinear (Figure 3C), and this translation occurs slower than the instantaneous velocity, around 2–3 mm/sec. Figure 3D–F illustrate numerical evaluations for the same parameters, except at a higher acoustic pressure of 100 kPa. At this higher acoustic pressure, the instantaneous radiation force (Figure 3D) and resulting translational velocity (Figure 3E) are less symmetric than the 14 kPa case due to greater non-linearities in the bubble oscillation. The resulting translation (Figure 3F) is over 15 times greater than the translation at 14 kPa.
Instantaneous (A) radiation force, (B) velocity, and (C) displacement for a bubble of 1.0 μm radius driven by a 3-MHz, 10-cycle pulse at 14 kPa, and (D) radiation force, (E) velocity, and (F) displacement for a bubble of 1.0 μm radius driven by a 3-MHz, 10-cycle pulse at 100 kPa.
We evaluated the time-averaged magnitude of radiation force for microbubbles of varying initial radius, each driven by a sinusoidal pulse at their respective resonant frequency with a pressure amplitude of 100 kPa (Figure 4). From Equation 5, we expect this radiation force to be inversely proportional to the square of the resonant frequency and this relationship is confirmed in Figure 4. The curve is not smooth due to the fact that the pulses at different frequencies do not all contain integer numbers of cycles (since the pulse length was fixed at 3.3 μsec). The force experienced by microbubbles driven at resonance at 3 MHz is approximately 2.8 times that for resonant microbubbles driven at 5 MHz.
Time-averaged radiation force for bubbles of different sizes driven by a 3.3-μsec sinusoidal pulse at 100 kPa at their respective resonant frequencies.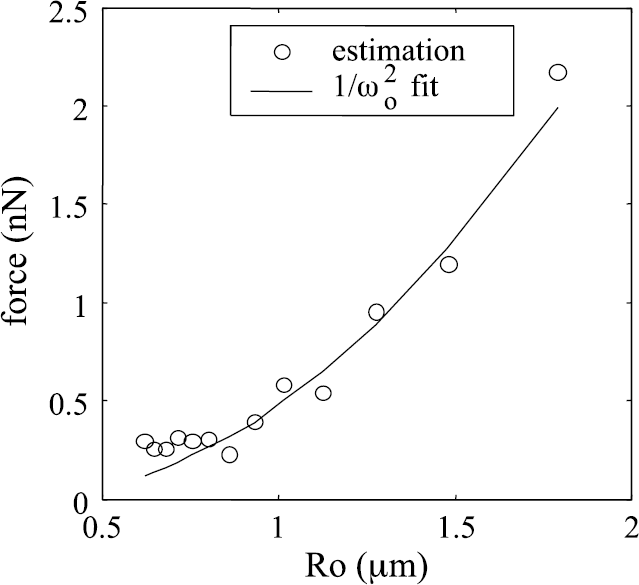
Figure 5 shows the calculated microbubble displacement per cycle for 3.3-μs pulses of 3 and 5 MHz as a function of bubble size along with the maximum predicted displacement as a function of bubble size (using a separate resonant–frequency pulse for each bubble size), for pulse amplitudes of 14 kPa (Figure 5A) and 100 kPa (Figure 5B). The predicted displacement for resonant microbubbles decreases with increasing resonant frequency: the displacement of a microbubble at resonance at 3 MHz is approximately 1.6 times that for microbubbles resonant at 5 MHz at the same pressure. Increasing the driving pressure increases the displacement in a nonlinear fashion. We observe from the results in Figure 5 that the predicted displacement increases from approximately 1 nm/cycle at 14 kPa to approximately 50 nm/cycle at 100 kPa, a 50-fold increase in displacement for a 7-fold increase in pressure. The frequency at which maximum displacement occurs does not exactly coincide with the linear resonance frequency.
(A) Displacement per cycle of bubbles over a range of initial radii, driven by pulses of 14 kPa at 3 and 5 MHz, and also by pulses at their respective resonant frequency. For the multimillion cycle pulses used in the experiments, the peak displacement is predicted to be on the order of mm for insonation at resonance, and thus the experimental pulse is capable of displacing the vehicle across a large vessel. (B) Displacement per cycle of bubbles over a range of initial radii, driven by pulses of 100 kPa at 3 and 5 MHz, and also by pulses at their respective resonant frequency. In this case, a pulse with a length of 100,000 cycles can produce a displacement on the order of millimeters.
In the Experimental section of this article, we use 14 kPa, 3 MHz, and 5 MHz pulses with a duration of 1.67 sec, corresponding to 5 million cycles for the 3-MHz pulse and 8.3 million cycles for the 5-MHz pulse. Based on the previous numerical results for the 3- and 5-MHz driving pulses at 14 kPa, we can predict the translational velocity which a microbubble will achieve when it flows across the ultrasound beam. This velocity is on the order of mm/s for both 3- and 5-MHz driving pulses, with a predicted maximum velocity of 3.3 mm/s at 3 MHz, compared to 2.1 mm/s at 5 MHz (Figure 6). Thus, a resonant bubble can be displaced on the order of millimeters during a 1-sec ultrasound exposure, easily crossing a capillary, venule, or arteriole.
Predicted translational velocity versus radius for microbubbles driven by 14 kPa, 1.67-sec pulses at 3 and 5 MHz.
Translational Velocity of Contrast Agents Experiencing Radiation Force: Comparison of Theory and Experiment
Using a high-speed camera system acquiring images at 240 frames/sec, we measured the translational velocity of contrast agents displaced from the flow stream to the tube wall by radiation force with a transmission pressure of 14 kPa, pulse length of 1.67 sec, and center frequencies of 3 and 5 MHz and compared the velocities to those estimated from the numerical evaluations (Figure 7). Experimentally estimated velocities were on the order of 2.4 ± 1.6 at 3 MHz (n = 9) and 1.9 ± 1.0 mm/sec at 5 MHz (n = 4). These values are in very good agreement with the predicted values for maximum translation velocity: 3.3 and 2.1 mm/sec for 3 and 5 MHz, respectively. The experimental velocities do sometimes exceed the predicted maximum velocities because the prediction only takes into account primary radiation force; secondary radiation forces and other forces on the bubble may cause increased velocities of translation.
Comparison between experimentally measured (black bars) and predicted (white bars) translational velocity of contrast agents during 14 kPa, 1.67-sec pulses at 3 and 5 MHz.
Demonstration of Enhanced Adhesion of Biotinylated Agents to an Avidin-Coated Tube
The adhesion of biotin-targeted contrast agents within an avidin-coated microvessel was observed to be substantially increased by the application of acoustic radiation force (Figure 8). Only sporadic adhesion of agents to the tube wall was observed in the two non-targeted cases (tube not coated with avidin, or bubble not biotinylated), even though the agents were pushed toward the wall with a radiation force pulse. In the targeted case using no ultrasound, an average of three bound bubbles per field of view was observed. With the addition of radiation force, a significant increase in agent binding was observed (p > .05)—a 20-fold increase using a flow velocity of 6 mm/sec (Figure 8A), and a 10-fold increase at 28 mm/sec (Figure 8B).
Adhesion of biotin-targeted microbubbles to avidin-coated tube with and without targeted contrast (TC) and 1.67-sec radiation force (RF) pulses at 3 MHz and 14 kPa. Two negative-targeting controls were performed: in one, the contrast microbubbles were not biotinylated; and in the other, the tube was not avidin-treated. (A) Flow velocity of 6 mm/sec. (B) Flow velocity of 28 mm/sec.
Adhesion assisted by radiation force over a greater range of flow rates was explored, demonstrating that adhesion decreases at higher flow rates (Figure 9A). For a transmission center frequency of 3 MHz, an average of 30, 17, and 1 adherent microbubble per field were observed for flow velocities of 6, 28, and 142 mm/sec, respectively (n = 10 in each case). The number of adherent bubbles was substantially lower using a transmission center frequency of 5 MHz (Figure 9B), with an average of 9, 7, and 2 adherent bubbles under flow velocities of 6, 28, and 142 mm/sec, respectively (n = 20 in each case).
Adherent bubbles after 1.67-sec, 14-kPa radiation force pulses in tubes with flow velocities of 6, 28, and 142 mm/sec at (A) 3 MHz and (B) 5 MHz.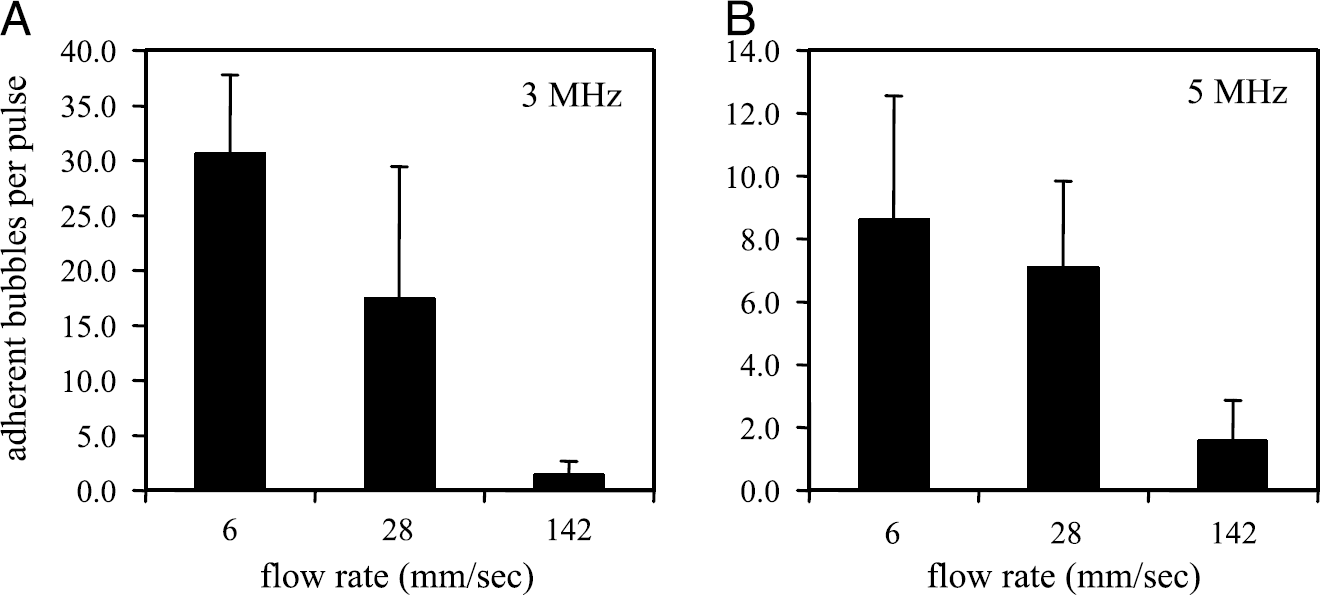
Demonstration of Enhanced Adhesion of RGD-Targeted Contrast Agents to avβ3 Expressing HUVEC Cells
The potential for radiation force assisted adhesion of RGD-targeted contrast agents to αvβ3-expressing HUVEC was evaluated using endothelial cells grown on the inside lumen of a small mimetic vessel. Previous studies have demonstrated the affinity of the MRX 408 RGD targeted agents for αvβ3 expressing HUVEC, and flow cytometry demonstrated 145,300 ± 33,400 αvβ3 binding sites/cell [13]. Agents flowed through the vessel at a mean velocity of 5 mm/sec. Adhesion of microbubbles after radiation force pulsing was 27-fold higher than adhesion without ultrasound (p > .05, <i>n = 3 in each case) (Figure 10) and the targeted agents were observed to remain adherent as the flow velocity was increased to 50 mm/sec. The increased adhesion of the agents is also shown in fluorescent micrographs of the tube wall with (Figure 11A) and without (Figure 11B) radiation force. In Figure 11A, the white spots in the image are from fluorescent adherent agents. The microbubbles appear blurry due to the fact that they were in different focal planes due to the curvature of the vessel. During the experiment, the number of adherent agents was counted by adjusting the focus.
RGD-peptide targeted bubbles adherent to avβ3-expressing HUVEC cells without (left) and with (right) a 1.67-sec, 14-kPa radiation force pulse at 3 MHz. Flow velocity of 5 mm/sec. Fluorescence photomicrographs showing a portion of a microvessel flow phantom. Each image is approximately 100 × 60 μm. (A) Fluorescent RGD-targeted microbubbles adherent to αvβ3-expressing HUVEC after 10 radiation force pulses over 100 sec. (B) Minimal adhesion of fluorescent RDG-targeted microbubbles to HUVEC is seen without application of radiation force over the same period.

Ultrasonic Imaging of Adherent Targeted Agents
For examination of the echo spectrum from contrast agents adherent to the phantom vessel wall, biotinylated contrast agents were targeted to avidin-coated tubing using radiation force as in the experiments above, using a flow velocity of 23 mm/sec. Optical microscopy confirmed the presence or absence of adherent microbubbles in each case. Spectra were obtained from water alone, from freely flowing bubbles in the tube, from bubbles adherent to the tube wall after rinsing with no radiation force applied, and from bubbles adherent to the tube wall after application of radiation force and rinsing. Echoes were obtained using imaging pulses of 440 kPa (Figure 12A) and 690 kPa (Figure 12B).
Spectra from echoes scattered from a microvessel phantom in response to imaging pulses of (A) 440 kPa and (B) 690 kPa. Both plots show spectra from water only (dotted line), from freely flowing bubbles (thin solid line), after rinsing with water after applying radiation force pulses (thick solid line), and after rinsing with water with no radiation force application (dashed line). The narrowband spectrum of adherent bubbles after application of radiation force pulses can be observed at both pressures.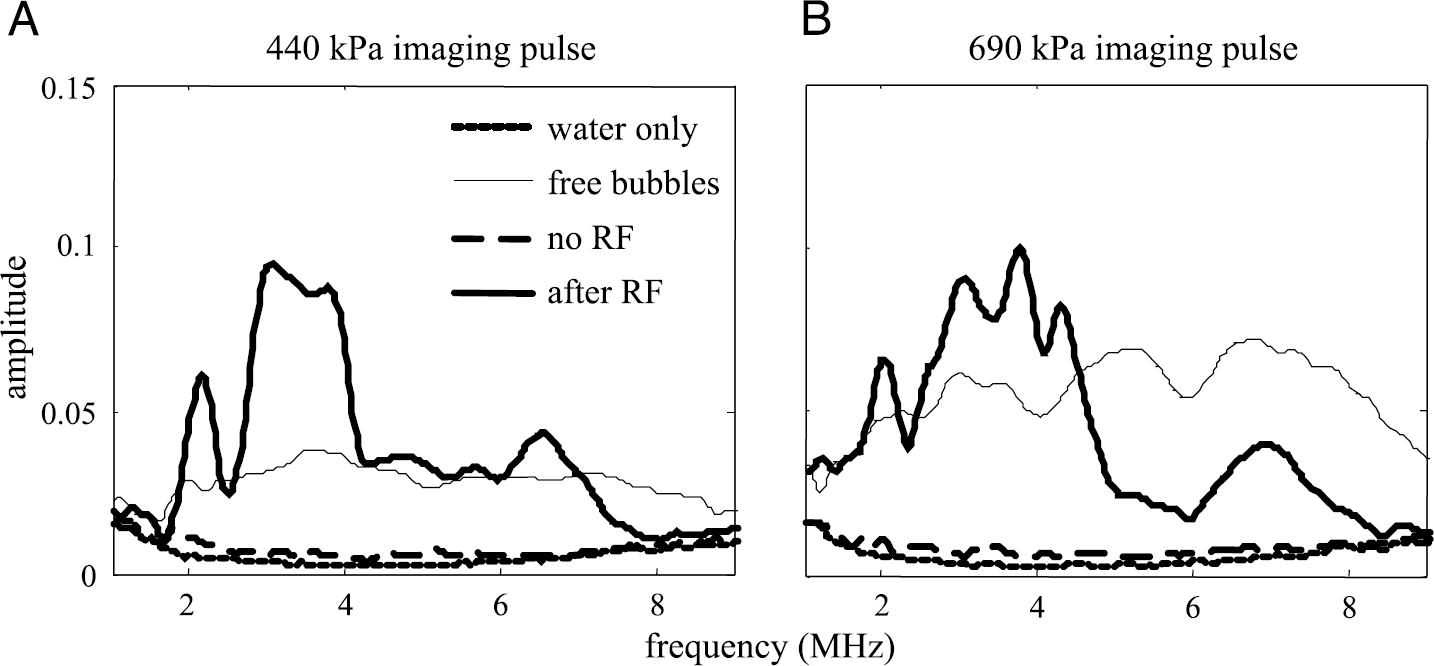
The spectrum from the received echo from the tube with water only (dotted line in Figure 12, parts A and B) provides a baseline noise floor for this study. The received echo spectrum from a solution of contrast agents pumped through the tube (thin, solid line) is typical of the broadband echo produced by freely oscillating microbubbles [21,24]. The no-ultrasound control (thick dashed line) is obtained from echoes from adherent microbubbles, recorded after freely flowing bubbles were allowed to pass through the tube for 4 min with no ultrasound application and then were rinsed. This spectrum is low in amplitude and corresponding optical observations did not show adherent bubbles. In contrast, echoes recorded following radiation force assisted targeting (thick solid line) show a distinct spectrum for the relatively dense microbubble adhesion confirmed optically. The spectrum from these adherent agents includes a narrowband component centered near 3.5–4 MHz, and a harmonic component approximately 6 dB below the amplitude at the fundamental frequency. The peak amplitude of the adherent agents does not increase with increasing transmitted pressure, and shows a decreasing amplitude with each 690 kPa imaging pulse due to the effect of microbubble destruction over the pulse train. Since the freely flowing agents are able to refill the vessel between pulses, we note that the spectral amplitude from nonadherent agents increases relative to the adherent agents as the transmission pressure increases. Exposure of adherent bubbles to ten 5-MHz, 2.2-MPa high-pressure imaging pulses reduced the echoes from the adherent bubbles nearly to the background level, and optical observation confirmed destruction of the adherent bubbles (data not shown).
Discussion
In the results above, we have demonstrated that ultrasonic radiation force can greatly increase the number of microbubbles that bind to a specific molecular target, enhancing the scattered signal from the target site. Radiation force allows targeted microbubble agents to be concentrated in a desired region, without increasing nonspecific adhesion of agents to the target surface. The significance of these results for ultrasound molecular imaging is clear: The application of ultrasound radiation force pulses to a tissue during administration of a targeted contrast agent should increase binding of the agent to its intended receptors, increasing the number of agents bound to the target site of interest and therefore the signal-to-noise ratio of subsequent ultrasound imaging. However, the theory of this interaction remains incomplete, and there may be some barriers to application of this method in vivo; these issues are discussed in the remainder of this section.
Limitations of Theory
Wall effects. The theory of radiation force on a microbubble presented in this article assumes that the bubble is suspended in an infinite medium. In reality, the oscillation of a microbubble is affected by the vessel wall, and its motion as it approaches the wall is complex. The bubble's radial oscillations, which involve wall velocities on the order of 10–1000 m/sec, will become asymmetrical as the bubble approaches a boundary, with unknown consequences. In this work, we observed microbubble behavior in agreement with theory despite this limitation: The net translational velocity of microbubbles in response to a long pulse was 2–3 mm/sec for a pressure of 14 kPa, in agreement with theory, and we observed that the contrast agent approached the wall at an angle determined by the fluid velocity and displacement velocity, with no observable interference due to wall proximity.
Mechanism of enhanced binding. A related limitation of the theoretical framework presented here is that it provides no insight into the mechanism of increased targeted agent binding through the use of radiation force. The increase in the number of bound microbubbles clearly demonstrates that targeted adhesion is increased, but it is unclear whether this increased adhesion is due only to the increased proximity of the ligands and receptors, or whether the complicated combination of radial oscillation (involving wall velocities on the order of meters/second), translational oscillation (centimeters/second), and net motion of the microbubble towards the wall (millimeters/second) also somehow facilitates the binding of the receptor and ligand.
The relationship between microbubble binding and ultrasound frequency. Within the experiments detailed here, it was observed that more agents bound to the vessel wall when assisted by a 3-MHz transmitted pulse than by a 5-MHz transmitted pulse (Figure 9). Given that there are a larger number of bubbles with a resonance frequency near 5 MHz (0.9 μm) than with a resonance frequency near 3 MHz (1.3 μm), this observation cannot be attributed simply to the distribution of sizes and resonance frequencies within the contrast media. Numerical evaluation shows that both the radiation force applied on a microbubble driven at resonance and the displacement produced by this force is larger at 3 MHz than at 5 MHz, so it is possible that the smaller agents resonant at 5 MHz were not forced against the vessel wall as strongly. An alternative explanation is that the difference is due to the difference in lateral beam widths of the 3- and 5-MHz transducers. The lateral beam width of the 3-MHz transducer is approximately 1.7 times that of the 5-MHz transducer, and therefore radiation force is applied over a wider region as the particles move across the beam. In support of this hypothesis, increasing the flow rate also decreases the time over which the force is applied to particles moving across the beam, and we did observe that increasing flow rate decreased the number of bound particles. However, we did not observe agents attach when they were rolling along the vessel wall, as if the time they spent forced to the wall increased the probability of finding an appropriate binding site; in all observed cases, the agents appeared to bind as soon as they hit the wall. This question is related to the mechanism of binding, and we hope that further research will elucidate to what extent the complicated motion of the ligand on the microbubble relative to the receptor on the vessel wall, which is strongly dependent on frequency, enhances binding.
Practical Considerations
The effects of microbubble size distribution. Ultrasound contrast agents are polydisperse, with diameters ranging from below one micron to several microns. As shown in this article, this range of diameters leads to a wide range of resonance frequencies, and microbubbles driven off-resonance experience greatly decreased radiation force: numerical evaluation of Equations 1–7 demonstrates a substantial decrease in radiation force magnitude and displacement for particles whose diameters are far from the resonance size (see Figure 5). For example, a particle with a diameter of 1.2 μm driven by a narrowband pulse at 3 MHz is expected to be displaced 1/50th of the displacement of a resonant particle with a diameter near 3 μm. Therefore, it is important to choose the ultrasound frequency carefully to correspond to the agent size. However, it may not always be ideal to choose the resonance frequency of the mean bubble diameter. In Ref. [25], it was shown that in a shear gradient adhesion of a particle to the surrounding vessel wall increases with decreasing diameter. Thus, it may be more important to deflect small microbubbles because they experience fewer shear forces and so may bind with greater frequency in vivo. Large agents can be prevented from binding to the vascular endothelium by the large shear forces they experience, even if they are efficiently pushed against the vessel wall.
In order to produce a substantial force on all particles, either an engineered pulse with a high time–bandwidth product must be applied, or a mono-disperse contrast agent must be created. Smaller particles are desirable, as discussed above, however, the resonance frequency varies rapidly for small particles with small changes in size, and therefore efficient use of radiation force is likely to require the creation of engineered pulses that can deflect all agents within a given distribution with an equivalent force. Such a pulse might use a chirp with varied amplitude and phase modulation. In our experiments without the use of an engineered pulse, we estimate that approximately 5–10% of the particles that moved through a vessel were captured. With the design of an efficient pulse, it is possible that the majority of the contrast agents moving through a vessel can be bound to a receptor target, further increasing the signal-to-noise ratio of ultrasound molecular imaging techniques.
Limitations on acoustic parameters. Safety concerns dictate limits for the transmitted pressure, pulse duration, and pulse interval due to the potential for mechanical and thermal damage to the surrounding tissue, although the current limits are based on historical precedent, rather than evidence of damage. However, radiation force is effective at very low ultrasound driving pressures, relative to the output of clinical ultrasound scanners, so regulatory issues should not be a barrier to the implementation of this method. The pressures demonstrated to be effective in this manuscript for radiation force assisted targeting are on the order of only 14 kPa, compared to the hundreds to thousands of kPa used in clinical ultrasound. In fact, the most stringent limitations on the driving pulse may be imposed by the fact that even a moderate ultrasonic pressure (~hundreds of kPa) will result in microbubble destruction; simply increasing the pressure to increase results will be ineffective. Even with extremely long pulse lengths, the integrated spatial-peak temporal-average power for the several-million cycle pulses used in our experiments is on the order of 10 mW/cm2, which is well below the FDA limit of 720 mW/cm2.
Orientation of ultrasound source and blood vessels. Using a clinical ultrasound system and a micron-sized contrast agent, radiation force typically displaces microbubbles away from the ultrasound source. Hence, the use of radiation force to enhance adhesion on the side of a vessel closest to a transducer may not be possible. However, given that we are often targeting capillaries and very small arterioles and venules, which are spaced by less than our resolution distance, and these vessels are not likely to express targets uniquely on one side, this seems unlikely to affect our application. The use of radiation force assisted targeting for individual lesions in larger vessels may be limited by the orientation of acoustic windows available for ultrasound transmission into a patient.
Detection of bound microbubbles. The scattered echoes from a layer of bound microbubbles include a narrowband component near the transmitted center frequency, in addition to higher frequency components. This differs from the echoes from freely flowing microbubbles, which typically produce broadband responses. However, differentiating bound agent echoes from those produced by surrounding tissue in vivo may be challenging. Ultimately, detecting bound agents may require high system bandwidth or the use of pulses which consistently destroy the contrast agent, with detection based on the transient change in signal characteristics.
Drug delivery. This method shows great potential for improving the signal-to-noise ratio in ultrasound molecular imaging techniques. And, while we focus on ultrasonic molecular imaging in this article, there is also great interest in developing ultrasound-enhanced drug delivery using targeted agents. These agents may or may not have a gas core; future research will address the question of whether nonmicrobubble agents can also be translated significant distances by radiation force pulses.
Conclusion
The use of radiation force to assist targeted imaging has been shown to have a significant impact on the number of targeted bubbles binding in a synthetic vessel over a short time period. We have illustrated this technique with both biotin-targeted agents adhering to avidin-coated microvessels, and RGD-targeted agents adhering to HUVEC monolayers grown in synthetic vessels. Numerical evaluation of a model for microbubble displacement confirms that displacement of microbubble agents at physically relevant velocities is consistent with theory, and we find that the predicted velocity is similar to that observed in vitro. In addition, we show that the echo amplitude from the vessel wall is greatly increased when bound agents are present, and that the echo spectrum of bound agents is different from that of freely flowing agents.
We believe that radiation force assisted binding of targeted microbubble contrast agents is a method that has the potential to substantially improve ultrasound molecular imaging.
Footnotes
Acknowledgments
We acknowledge the support of NIH 1R21CA980692. Additionally, we thank Hanako Zeidenberg for the assistance with data analysis. We also thank Terry Matsunaga, Patricia Schumann, and Reena Zutshi at ImaRx Therapeutics for supplying ImaRx contrast agents.



