Abstract
Recurrent spontaneous seizures in epilepsy cause a myriad of structural, circuit-related, and molecular modifications in the brain. The multifaceted molecular changes suggest that wide-reaching epigenetic mechanisms are altered in epilepsy. Indeed, it has been known for more than 15 years that a class of epigenetic regulators called microRNAs—short, noncoding RNAs that control the translation and stability of sometimes hundreds of mRNA targets—are dysregulated after seizures and in epilepsy in human patients and rodent models. Epilepsy-associated microRNAs regulate many different molecular contributors to epilepsy, including ion channels, neuroinflammatory modulators, and proteins critical for neuronal and synaptic structure. In recent years, it has become clear that microRNAs are important at every phase of epilepsy—from the onset of the first seizure to the latent phase and chronic epilepsy. In line with these findings, manipulation of a subset of microRNAs has been shown to alter seizure susceptibility, reduce epileptogenesis, and/or decrease the frequency of spontaneous recurrent seizures in animal models of epilepsy. These studies illustrate the promise of microRNAs as future therapeutic targets in epilepsy and show that specific microRNAs play different regulatory roles depending on the phase and type of epilepsy. In this concise review, we summarize recent findings of microRNAs in epilepsy, emphasizing novel approaches that advance the field. We discuss the insight on underlying mechanisms and disease etiology that can be drawn from these studies and highlight the importance of timing when developing microRNA-based therapeutic strategies.
Introduction
Almost 30% of all patients with epilepsy suffer from recurrent seizures despite multiple medication trials and even (in some cases) resective surgery. A problem contributing to the lack of efficient treatments for many patients is the complexity of pathologic changes in the brain caused by seizures. Most current epilepsy drugs target single molecules, such as specific ion channels, which may not be sufficient to comprehensively treat seizures. Moreover, they are used to treat chronic epilepsy but are not disease-modifying. In recent years, epigenetic mechanisms have come into focus as potential treatment targets in epilepsy. 1 Single epigenetic regulators can alter the expression of many molecules; thus, they are expected to have a broad effect on neuronal pathways and associated brain function. One class of epigenetic regulators shown to be dysregulated in epilepsy is microRNAs. microRNAs are 22–24-nucleotide-long noncoding RNAs that regulate the stability and/or translation of mRNA targets. 2 In the last ∼15 years, many microRNAs have been suggested as biomarkers or potential therapeutic targets for epilepsy. This review highlights the newest developments in the field of microRNAs in epilepsy, with special emphasis on insights into disease etiology and the importance of timing.
microRNA Dysregulation in Epilepsy
Epilepsy can be separated into roughly 3 phases: the epileptogenic insult (eg, an injury, status epilepticus, or a gene mutation), the latent phase, during which no seizures are detected, and the chronic phase, when spontaneous recurrent seizures occur. These phases are characterized by different molecular, cellular, and structural changes that are incompletely understood and believed to occur gradually. The acute changes shortly after an epileptogenic insult involve altered gene expression, neuroinflammation, and cell death. Later, profound structural changes can occur, such as mossy fiber sprouting in the hippocampus. 3 All these processes can be regulated by microRNAs, and microRNAs can be altered at virtually every phase of epilepsy: immediately after seizure, during the latent phase, and in chronic epilepsy.4,5 The hundreds of original research studies analyzing microRNA function in epilepsy support their utility as potential biomarkers and therapeutic targets in epilepsy and provide novel insights into underlying mechanisms and disease etiology.
Innovative Efforts to Identify Key microRNAs in Epilepsy
miR-146a was one of the first microRNAs found to be altered in epilepsy 6 ; since then, many studies have used various sources of human tissue to identify additional microRNAs that are changed in the disease. 7 These studies were often hampered by small sample sizes and a lack of adequate controls because epileptic brain tissue was retrieved from epilepsy surgery, whereas brain tissue from healthy individuals was collected postmortem. The detection of microRNAs in peripheral blood and cerebrospinal fluid8,9 opened avenues for less invasive biomarker identification in epilepsy. Although individual studies were initially small and found different subsets of microRNAs as potential biomarkers for epilepsy, 10 several microRNAs were identified repeatedly: miR-142-5p, miR-145-3p, miR-153, and miR-199a-3p. 11 To address the issue of reproducibility, researchers have begun to perform multisite, large-scale preclinical studies to identify microRNA signatures that are consistently altered across multiple well-controlled animal models. One of the first studies of this kind identified 9 microRNAs differentially regulated across the hippocampi of 3 different rodent models of chronic epilepsy. 12 This study focused on microRNAs associated with the RNA-induced silencing complex (RISC), instead of assessing total microRNA expression levels, which is advantageous because it only identifies microRNAs that are functionally active. Thus, they captured microRNAs that changed their silencing activity in response to seizures, while overall expression levels may have remained stable. 13 RISC association also serves as validation that an identified short RNA sequence is indeed a microRNA. This avoids mistakenly identifying RNA degradation products as microRNAs, which has been an issue in the past. 14 In another recent study, researchers used a similar approach to identify microRNAs as biomarkers for epileptogenesis after traumatic brain injury (TBI) in rats. 15 Taking advantage of the fact that only about 50% of rats develop chronic seizures after TBI, this international multisite study found several plasma microRNAs that were predictive of epilepsy. If confirmed in humans, this could be a game-changer in posttraumatic epilepsy care because of its potential use to identify TBI patients at risk of developing epilepsy who would benefit from preemptive treatment. While a clear advance compared with previous smaller-scale studies, these preclinical experiments may miss epilepsy-relevant microRNAs that are only expressed in humans but not in mice. 16 Large-scale multisite studies in humans are thus necessary, ideally including prospective designs, to further advance these preclinical findings into clinical practice.
microRNA Therapeutics in Epilepsy—Timing and Disease Etiology Matters
The recent progress in RNA technology and therapeutics in human diseases, including epilepsy, 17 has brought microRNA therapeutics as a potential treatment strategy into focus. Many preclinical studies have tested manipulation of select microRNAs in the brain to reduce seizure susceptibility and frequency or suppress epileptogenesis (Figure 1).
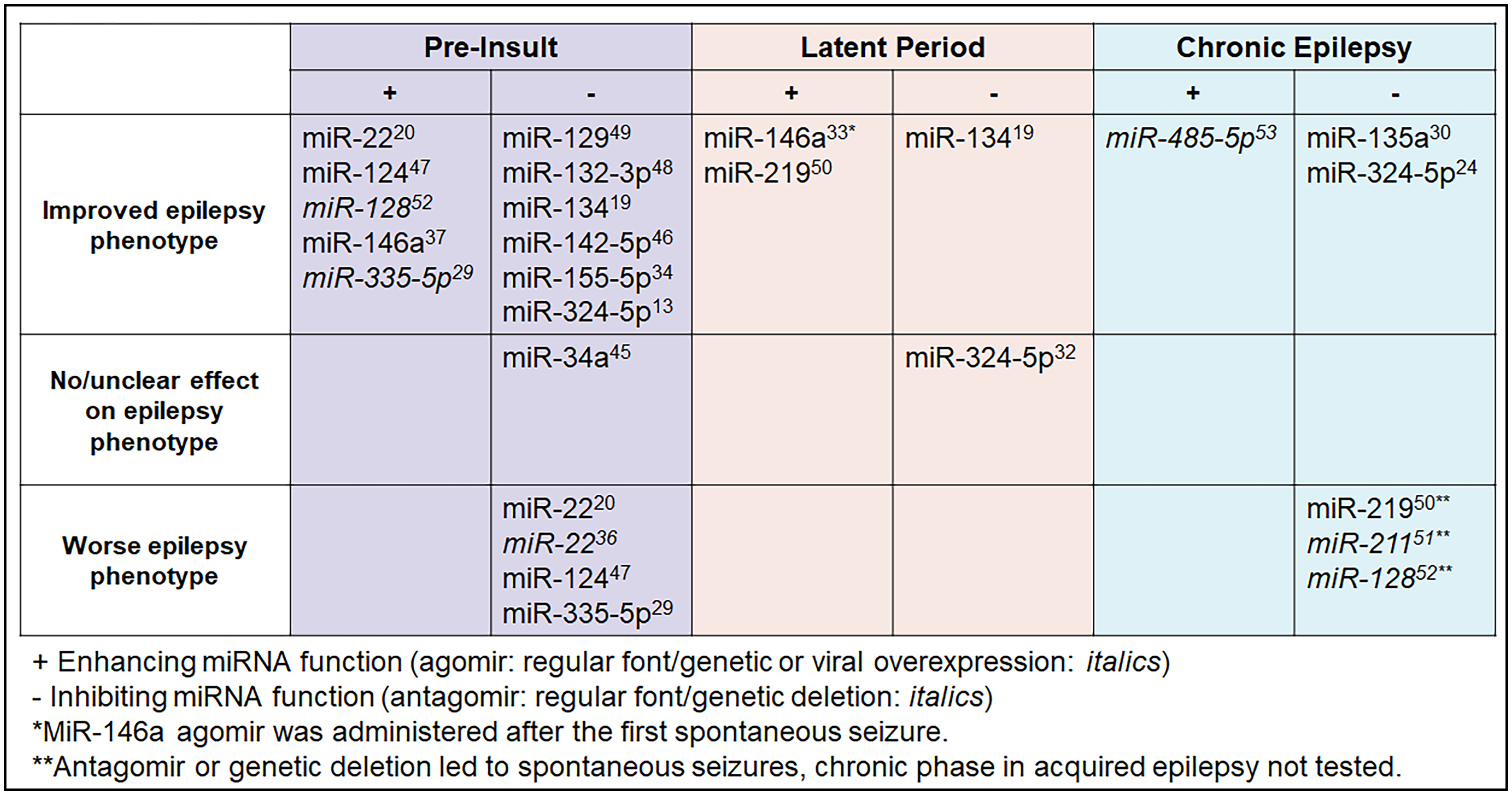
Effects of manipulating select microRNAs on seizure phenotypes in rodent models depending on the timing of intervention. Top: Acquired epilepsy can be divided into 3 phases: preinsult (left), latent period (middle), and chronic epilepsy (right). Bottom: microRNA effects on epilepsy phenotype (defined as seizure onset, seizure severity, and/or seizure frequency). MicroRNAs in Italics were genetically deleted or overexpressed, while regular fonts indicate the use of antagomirs or agomirs. Only select microRNAs tested preinsult are shown (based on availability of electroencephalogram (EEG) to confirm seizures, testing in multiple models). For a more complete overview of microRNAs in epilepsy, see previous comprehensive reviews.22,44
MicroRNA activity can be inhibited using antagomirs, which are modified antisense oligonucleotides that block the binding of microRNAs to their target mRNAs. 18 Vice versa, sense oligonucleotides (agomirs or microRNA mimics) can be used to “overexpress” microRNAs, enhancing their function. Most microRNAs studied in the context of epilepsy are thought to be proconvulsant; that is, inhibition of the microRNA reduces seizure susceptibility or severity. This may be a reporting bias, however, because antagomirs have been shown to be stable and effective for at least a month after intracerebroventricular injection, 19 whereas agomirs may only be stable for hours. 20 Moreover, as with every overexpression approach, the correct dosing of agomirs to be effective without inducing toxicity, for example, by oversaturating the RNA-induced silencing machinery, is challenging. 21 Expression studies after seizures or during epilepsy in mouse models and human patients have identified both up- and downregulated microRNAs, suggesting that microRNAs can be pro- or anticonvulsant.12,22 Here, we will focus on a subset of microRNAs that have been tested in multiple epilepsy rodent models and at different phases of disease progression, providing insight into underlying mechanisms and the disease etiology of the epilepsy disorders tested. A more comprehensive overview of microRNAs tested in epilepsy models is shown in Figure 1.
Neuronal Morphology and Neuroinflammation are Common Targets of microRNAs With Therapeutic Potential in Epilepsy
Several microRNAs repeatedly shown to be upregulated in epilepsy animal models and human epilepsy have proconvulsant effects in rodent models, including but not limited to miR-134 and miR-142-5p.19,23 However, even microRNAs not consistently identified as potential biomarkers show potential as modulators of epilepsy.13,24 With the first indication that microRNAs may be therapeutic targets in epilepsy, efforts began to identify the underlying mechanisms. Initially, most studies that tested microRNAs in epilepsy manipulated microRNAs before the induction of status epilepticus using chemoconvulsants (Figure 1). While these studies showed the importance of specific microRNAs for seizure susceptibility, the insight into the potential significance as treatment targets in epilepsy was limited because it is currently not possible to predict when a patient will have a seizure. Below, we highlight a few studies that went beyond testing microRNAs’ effects on seizure susceptibility. Through the identification of critical mRNA targets and testing microRNA manipulation at different time points and in different epilepsy models, these studies reveal common mechanisms of microRNA regulation in epilepsy, most prominently involving neuronal morphology and neuroinflammation, and provide useful insight into disease etiology. Specifically, neuroinflammation has frequently been shown to be a promising microRNA-regulated target for seizure protection.
Antagonizing miR-134 Shortly Before or After Seizure Induction Reduces Seizure Severity and Frequency of Spontaneous Recurrent Seizures
The first functional study suggesting that select microRNAs may be treatment targets in epilepsy showed that a miR-134-specific antagomir injected intracerebroventricularly 24 h prior to seizure induction by intraamygdala kainic acid reduces overall seizure severity and seizure-induced cell loss in the hippocampal CA3 region. 19 Subsequently, miR-134 antagomirs were shown to be effective in reducing seizures in several rat and mouse models of chemoconvulsant-induced acquired epilepsy, 25 and in reducing audiogenic seizure susceptibility in a mouse model of Angelman syndrome (AS). 26 miR-134 was inhibited prior to or shortly after the induction of a seizure in these studies, but it is unknown if inhibition in the chronic phase of epilepsy, when an epileptic network is already established, would be successful. By contrast, the miR-134 antagomir does not reduce thermally induced or spontaneous seizures in a mouse model of Dravet syndrome (DS). 27 miR-134 is upregulated in the cortex and hippocampus of mouse models of acquired epilepsy and in humans with epilepsy, but not of DS or AS mice, suggesting that miR-134 upregulation does not predict the therapeutic efficacy of miR-134 antagomirs.19,26,27 Efficacy of the miR-134 antagomir in AS may be mediated through loss of the Ube3a1 transcript, which contains miR-134 binding sites, potentially leading to imbalances of miR-134 activity; however, there was no clear experimental support for this hypothesis. 26 Thus, the causes for the varying therapeutic success of miR-134 antagomirs in these models are not fully understood yet, but could be due to differences in the mechanisms of ictogenesis or a lack of anticonvulsant effects of miR-134 inhibition once an epileptic network has formed. miR-134 targets that are upregulated by its antagomirs—Limk1, Creb1, and Dcx—play major roles in dendritic spine morphology and neuronal migration.19,28 The selective lack of effect in the DS mouse model, in which a sodium channel is mutated, suggests that genes with structural functions do not play a key role in DS disease etiology. It will be interesting to test if miR-335-5p, an anticonvulsant voltage-gated sodium channel regulator, 29 has therapeutic potential in DS.
miR-135a Inhibition Reduces Seizures in Chronic Epilepsy
Although testing microRNAs for their ability to alter seizure susceptibility when manipulated prior to the insult provides important insight into their function in the brain, a more translational approach is to test if they can affect spontaneous recurrent seizures. One of the first studies assessing the effect of microRNA inhibition at later stages of epilepsy showed that inhibition of miR-135a 2 weeks after pilocarpine-induced status epilepticus, when spontaneous recurrent seizures occur, significantly reduces seizure frequency. 30 As a potential underlying mechanism, Mef2 was identified as a target essential for miR-135a's effect on dendritic morphology. This suggests that, similar to findings reported with miR-134, altered neuronal morphology could play a role in miR-135a-mediated seizure suppression.
miR-324-5p Inhibition is Anticonvulsant but may not be Antiepileptogenic
miR-324-5p inhibition using antagomirs prior to insult increases latency to kainic acid-induced seizures, and antagomir treatment after the onset of spontaneous seizures reduces their frequency in the pilocarpine model.13,24 Effects are partially mediated through the potassium channel Kv4.2.13,24 Genetic deletion or antagomir-mediated knockdown of miR-324-5p also reduces dendritic spine density, 31 suggesting that the anticonvulsant effect could additionally be mediated by altering dendritic morphology. Interestingly, miR-324-5p antagomirs do not block epileptogenesis when injected 24 h after intrahippocampal kainic acid, during the latent period, 32 suggesting that miR-324-5p antagomirs are anticonvulsant but not antiepileptogenic or disease-modifying. Alternatively, the role of miR-324-5p in ictogenesis and epileptogenesis may differ depending on the type of epilepsy or the timing and duration of miR-324-5p suppression. Further studies are needed to delineate the role of miR-324-5p in seizure susceptibility, epilepsy, and epileptogenesis.
The 3 promising therapeutic candidate microRNAs discussed above, miR-134, miR-135a, and miR-324-5p, all play a role in neuronal or dendritic morphology in addition to regulating seizures. This suggests a common mechanism in microRNA-mediated regulation of seizure susceptibility via altered neuronal morphology that warrants further exploration.
Antiinflammatory microRNA Treatments may be Disease-Modifying in Epilepsy
Chronic neuroinflammation is one of the hallmarks of epilepsy and is thought to contribute to disease progression. microRNAs targeting neuroinflammatory pathways have been consistently identified as potential biomarkers in epilepsy (eg, miR-142-5p, miR-146a, and miR-155-5p) and are thus of special interest. In a mouse model of acquired epilepsy, miR-146a mimics given every 3 days for 16 days after the first spontaneous recurrent seizure was detected, that is, in the early phase of chronic epilepsy, reduced seizure frequency for at least 10.5 weeks through suppressing the inflammatory interleukin-1R1/TLR4 pathway. 33 This long-lasting effect after agomir treatment suggests that miR-146a could be disease-modifying; however, it is possible that agomir treatment simply delayed epilepsy progression and that seizure frequency would have “caught up” if mice were monitored longer. Another important microRNA involved in the regulation of postseizure neuroinflammation is miR-155-5p. In the kainic acid-induced rat model, cell type-unspecific inhibition of miR-155-5p reduces seizure-induced hippocampal damage 34 ; however, deleting the Mir155 gene selectively in microglia of an Alzheimer's mouse model increases seizure susceptibility, suggesting that cell type matters. 35 Studies targeting neuroinflammatory microRNAs also illustrate the importance of timing in microRNA manipulation and the complexity of their role in regulating neuroinflammation: miR-22 antagomirs intracerebroventricularly delivered 24 h before intraamygdala kainic acid-induced status epilepticus led to increased mRNA expression of select neuroinflammatory markers in the contralateral hippocampus and increased frequency of spontaneous recurrent seizures 5 days later, suggesting that elevated neuroinflammation can exacerbate the phenotype of acquired epilepsy. 20 Accordingly, enhancing miR-22 function with an agomir transiently reduced seizure frequency. 20 Vice versa, a Mir22 knockout mouse lacked the inflammatory response usually seen after kainic acid-induced status epilepticus but displayed an exacerbated epilepsy phenotype, suggesting that the immediate inflammatory response is protective and that miR-22 can have pro- and antiinflammatory roles. 36 It is important to consider that some of the observed effects in the Mir155 and Mir22 knockout mice may be due to changes during development, further complicating the interpretation. Additional studies are necessary to test the disease-modifying effects of manipulating neuroinflammatory microRNAs in epilepsy.
Conclusion and Future Directions
MicroRNAs play diverse roles in epilepsy, depending on phase and etiology. Preclinical studies manipulating select microRNAs provide mechanistic insights and support microRNAs as therapeutic targets in epilepsy. The translational potential of microRNAs was strengthened by recent studies showing the feasibility of systemic intravenous, intraperitoneal, and intranasal delivery of microRNAs in epilepsy models.37–39
This concise review highlights the importance of timing and suggests that microRNA treatments that are anticonvulsant are not necessarily beneficial in preventing the development of spontaneous seizures. However, as illustrated in Figure 1, very few microRNAs have been tested as treatment targets in the latent phase, and there is most likely an underreporting bias of negative results. Another underexplored aspect of microRNA dysregulation in epilepsy is the occurrence of pathogenic mutations: only very few mutations in microRNA genes have been associated with epilepsy.40–43 Pathogenic mutations in microRNA genes are expected to increase in number and recognition as more whole-genome sequencing data become available. Together with the success of RNA therapeutics in neurological diseases, this will further raise interest in microRNAs in epilepsy.
Footnotes
Declaration of Conflicting Interests
C.G. is a co-Inventor on US patent 9,932,585 B2. All other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
