Abstract
The prevention of human immunodeficiency virus (HIV) infection has recently emphasized the use of pre- and post-exposure prophylaxis (PrEP and PEP), both of which were highly effective in prevention of HIV infection. Since the last published guidance regarding the cotreatment of people with anti-seizure medications (ASM) and antiretroviral treatments (ARTs) in 2012, both fields have numerous new medication options. Historically, cotreatment of HIV and seizures could be challenging with increased risk of virologic failure and barriers in access to health care due to global availability, social determinants of health, and stigma of both HIV and seizures. In this narrative review, we describe the data-driven and expected bidirectional pharmacokinetic (PK) interactions between guideline-based PrEP and PEP treatment and ASM, as well as overlapping side effects. There are many ASMs with no known interaction with PrEP or PEP regimens. The interactions focus on enzyme inducing ASMs, valproate, and lamotrigine. Most prominently, enzyme inducing ASMs lower serum levels of tenofovir-containing PrEP regimens and elements of PEP (dolutegravir, raltegravir, and ritonavir), which increased risk of virologic treatment failure in people with HIV but have unclear clinical significance on the effectiveness of PrEP and PEP. In addition, ritonavir treatment in PEP may significantly lower lamotrigine serum levels even during the 4 weeks of treatment, which may increase risk for breakthrough seizures during PEP and skin reactions after discontinuation of ritonavir. In addition to PK interactions, overlapping side effects are common including osteopenia, hepatic toxicity, and other gastrointestinal effects. This narrative review aims to be a resource for all clinicians prescribing ASMs so that they can create a welcoming environment to enable successful treatment of seizures and reduce the risk of HIV infection in people at risk. In addition, we highlight knowledge gaps and areas of unmet need that can be addressed with future studies.
Keywords
Introduction
Neurologists and epileptologists are experts in the diagnosis and treatment of seizures, but they may not be aware of the fundamental changes that have occurred in the diagnosis, prevention, and treatment of HIV since the last guideline for cotreatment of HIV and epilepsy in 2012. 1,2 Because of these changes, it is important for clinicians treating epilepsy to be aware of current HIV treatments including pre-exposure prophylaxis (PrEP) and post-exposure prophylaxis (PEP), as well as the interactions between these antiretroviral therapies (ART) and anti-seizure medications (ASMs). 3 -6
HIV is a bloodborne pathogen most frequently transmitted through sexual contact, intravenous drug usage, and from mother to child during delivery. 7 Globally, there were an estimated 39.0 million people with HIV in 2022, of which 86% knew their HIV status, 76% were on ART, and 66% were located in sub-Saharan Africa. 8 In the United States, there were 1.2 million people with HIV in 2019, of which 87% knew that they have HIV and 75% were receiving ART. 9 If untreated, HIV influences many systems of the body, most notably the immune system, causing a reduction in CD4+ cells that can increase the risk of life-threatening opportunistic infections. In patients without HIV or other reasons for immunodeficiency, these infections do not tend to pose a threat to human health. The CDC defines stages of HIV infection from acute retroviral syndrome through Stage 3, also known as acquired immunodeficiency syndrome (AIDS), which is defined by a CD4+ cell count less than 200 cells per microliter, CD4+ cells being less than 14% of all lymphocytes, or the presence of an AIDS-defining condition. 10 Currently, there is no cure for HIV, therefore national and international public health efforts aim to reduce transmission. Traditional methods to prevent HIV transmission include abstinence, barrier protection (eg, condoms), male circumcision, proper syringe disposal and hygiene for intravenous drug users, and mother to child transmission prevention programs. 11 However, these methods have limited efficacy when compared to effective treatment of those living with HIV and PrEP or PEP utilization. 7
Central to the treatment of HIV has been the development, access, and use of ART that can induce viral suppression, defined as less than 200 copies of HIV per milliliter of blood. 12 At this level, there is zero risk of sexual transmission of HIV. 13 In the United States, the prevalence of HIV suppression has improved from 19% in 2011 to 66% in 2019. 14 Global estimates of viral suppression are lower and show improving trends over time with availability of ART. 15 Lack of access to and suboptimal adherence to ART is the major driver of low suppression rates both globally and in the United States. In addition to access and adherence, patients taking certain enzyme inducing ASMs were more likely to have virologic treatment failure. 16 -19 This prompted the development of a guideline in 2012 for the cotreatment of HIV in patients taking ASMs. 1,2
Pre-Exposure Prophylaxis and Post-Exposure Prophylaxis
In addition to improving the care of patients living with HIV, current public health efforts emphasize the usage of PrEP and PEP to prevent HIV transmission. 10 Pre-exposure prophylaxis includes use of ART to prevent transmission of HIV to exposed individuals. 7 It has been studied in multiple settings and is safe and effective for individuals with potential ongoing exposure to HIV. The World Health Organization (WHO) has recommended the use of PrEP since 2015 and the United States Preventive Services Task Force/Department of Health and Human Services (US DHHS) has recommended it since 2019. 4,20 Pre-exposure prophylaxis regimens include both oral options and long-acting injectables (LAI) for those at risk of sexual transmission (Table 1A). The oral regimens, but not the LAI regimens have been established for individuals who inject drugs. To reduce the stigma of HIV exposure and treatment and to ensure effective treatment is provided, PrEP should be offered without requiring explicit discussion an individual’s risk for HIV risk. Pre-exposure prophylaxis is indicated in any patient who requests it and is appropriately educated about the risks and benefits associated with use. A systematic review of randomized trials and observational studies demonstrated that the risk of HIV infection reduced from 4.2% to 2.4% with typical use and 1% when adherence was at least 70%. 21
First Line Regimens for Pre-Exposure Prophylaxis (PrEP) From the United States Department of Health and Human Services (US DHHS) and World Health Organization (WHO).a
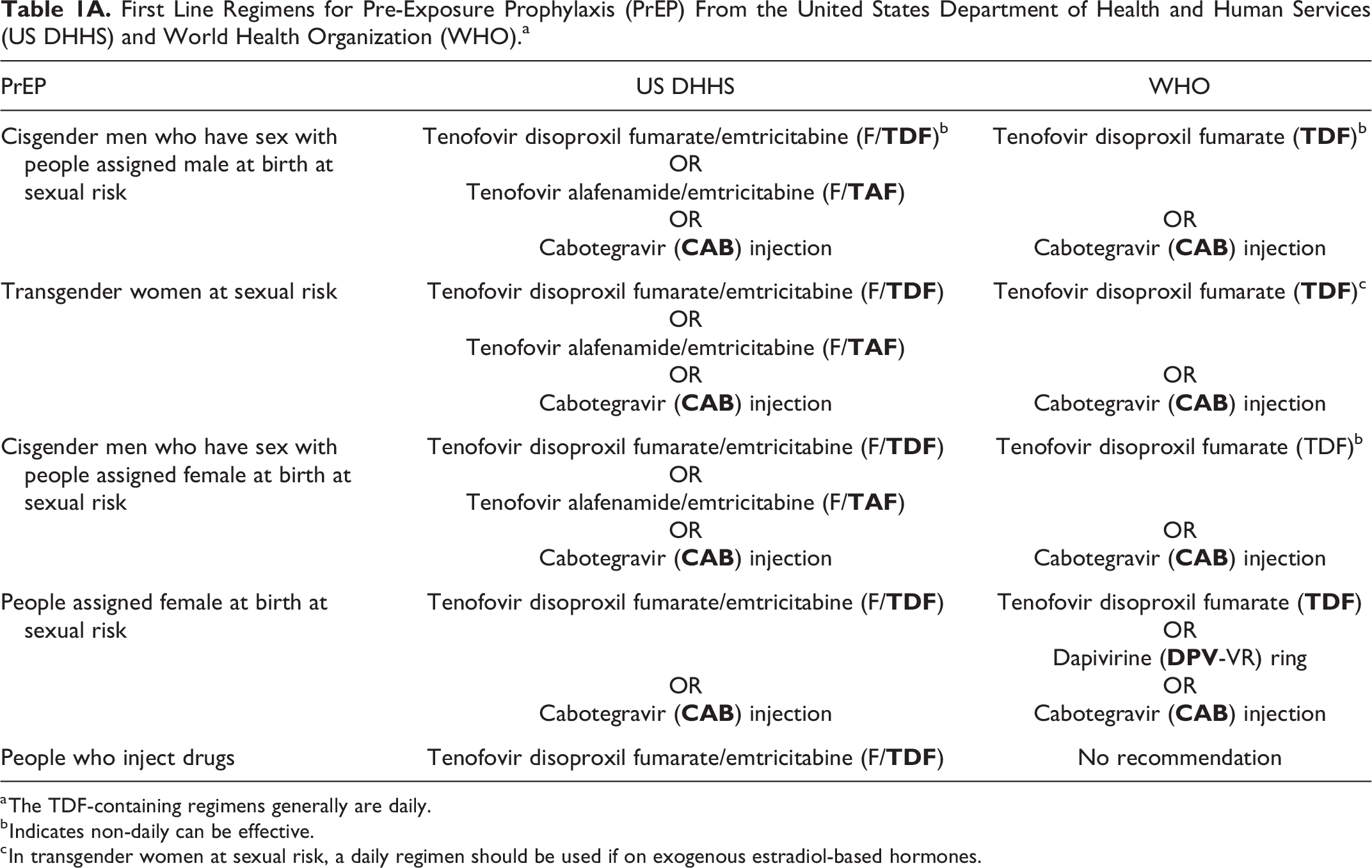
a The TDF-containing regimens generally are daily.
b Indicates non-daily can be effective.
c In transgender women at sexual risk, a daily regimen should be used if on exogenous estradiol-based hormones.
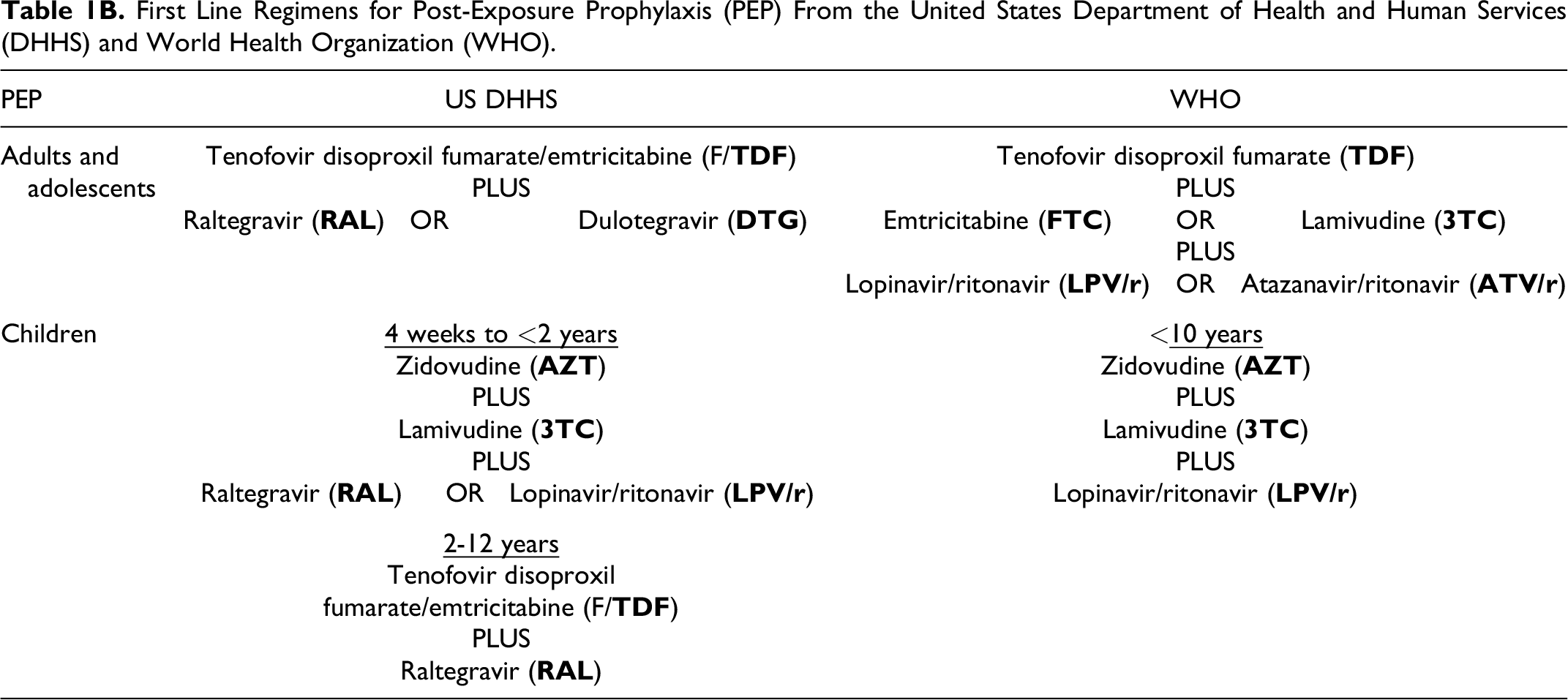
Post-exposure prophylaxis consists of giving an exposed person a fully active HIV regimen within the first 72 hours following exposure to prevent systemic infection. The practice of giving PEP to exposed health care workers began in the 1990s and has expanded to include nonoccupational exposures since the early 2000s, becoming fully incorporated into national guidelines in the United States in 2005 and WHO recommendations since 2014. 3 Post-exposure prophylaxis is used in isolated event driven situations and in infants born to HIV-infected mothers to disrupt vertical transmission. Outside of a health care occupational exposure, PEP can be used after unprotected sex, a condom breaking during sex, sexual assault, and after sharing needles to inject drugs. Sexual assault is not uncommon with 25% of women and 4% of men having experienced completed or attempted sexual assault in their lifetime in the United States. 22 Unfortunately, women with epilepsy or on ASMs reported higher rates of sexual assault than women without epilepsy (53% vs 32%). 23
The use of PEP is supported by animal studies and a single case–control study in exposed health care workers, as well as observational and case reports (Table 1B). Post-exposure prophylaxis may have decreased effectiveness 2 hours after exposure and must be started within 72 hours of possible exposure to HIV. 24 Many studies of PEP report no breakthrough infections on a diversity of regimens.
First Line Regimens for Post-Exposure Prophylaxis (PEP) From the United States Department of Health and Human Services (DHHS) and World Health Organization (WHO).
Clinicians should recognize the need and prevalence of PrEP and PEP in all patients, including those who are cisgender and heterosexual (not only men who have sex with men), as well as recognize the disparities in new infections in racial, sexual, and gender minorities. In sub-Saharan Africa and other similar regions, heterosexual transmission remains the main source of HIV infection, where 63% of new infections occur in women of all ages. 8 In the Unites States, 75% of new HIV infections were in non-white patients, including 11% in black women engaging in heterosexual contact, 5% in black men engaging in heterosexual contact, and 3.7% in Hispanic or Latina women engaging in heterosexual contact. In the United States, 7% of new diagnoses of HIV were attributed to injection drug use. 12 Therefore, awareness and appropriate use of PrEP and PEP is needed in all patients irrespective of sexual contacts.
The overlap between patients with epilepsy and undergoing treatment for HIV, or taking PrEP or PEP is not well established. In a neurology-HIV clinic, 23% of referrals were for advice on cotreatment of HIV and epilepsy. 25 While seizures in patients with HIV affects 2% to 14% of children, the anxiety and challenges in management vastly supersede this. 17 We attempted to estimate the degree of overlap from data regarding patients seen at the University of Pittsburgh Medical Center (UPMC) based on International Classification of Disease codes and prescription records. In 2022, UPMC saw 23 300 patients with epilepsy or taking ASMs and 7000 patients with HIV or taking ARTs. There were 115 (0.5%) patients with both epilepsy and HIV as well as 415 (1.7%) patients with epilepsy and a documented exposure to HIV, the latter of which may be an indicator of who may benefit from either PrEP or PEP. When identified based on medication records alone, 950 (4%) patients took both ASMs and ARTs, including 150 (16% of 950) patients taking ASMs with either known or theoretical interaction with ARTs (see below). This highlights that a substantial number of patients are being cotreated with ASMs and ARTs, despite limited guidance regarding this combination since the guideline in 2012. 1,2
We expect that epilepsy specialists and clinicians treating people with epilepsy are likely to care for patients on PrEP or PEP and this rate of cotreatment may increase in the future. Additionally, there may be a stigma against disclosing PrEP or PEP usage to the clinician treating seizures, but there are important interactions between PrEP, PEP, and ASMs, particularly enzyme inducing ASMs (eg, phenytoin [PHY], carbamazepine [CBZ]). 26 Based on findings of increased virologic failure in the treatment of chronic HIV with enzyme-inducing ASMs, 16 -19 failure to account for these interactions theoretically could lead to ineffective prevention of HIV transmission due to reduced effectiveness of PrEP and PEP. However, there have not been studies demonstrating the clinical impact of these interactions on measured effectiveness of PrEP or PEP. Conversely, ARTs can influence ASM treatments. Additionally, adverse effects were more common with cotreatment with some ARTs and ASM. 27 Therefore, improving knowledge of these interactions may improve the health of patients with epilepsy.
ART Regimens Used for PrEP and PEP
Medication regimens used for PEP and PrEP are determined based on patient and local factors and vary in different regions of the world (Table 1). Patient factors to consider include comorbidities (particularly renal disease and hepatitis B), medication interactions (eg, ASMs), tolerability, and the ability to adhere to oral regimens. 3 -6,28 Regional factors include the availability of medications (eg, supply chain limitations in procurement, cost, local licenses, and WHO prequalified status), which varies significantly throughout the world and explains some of the different recommendations between the US DHHS and the WHO. The WHO defines carbamazepine (CBZ), phenytoin (PHY), lamotrigine (LTG), phenobarbital (PHB), valproate (VPA), levetiracetam (LEV), diazepam, lorazepam, midazolam as essential ASMs, but this may not reflect availability. 29 Epidemiological drives of the HIV epidemic differ by region, which also influences the recommendations. To improve readability due to the high number of abbreviations, we list both the full name and abbreviations each time.
The best data on effectiveness of PEP regimens are from animal studies and health care occupational exposure where the risk of HIV transmission from a single exposure was reduced by 81% using zidovudine for 28 days, with hypothesized further risk reduction when a 3-drug PEP regimen for 28 days is used. 30,31 Since the publication of PEP guidelines (Table 1B), newer ARTs, including tenofovir alafenamide (TAF) and bictegravir, became available and studies have shown their efficacy in PEP. 32,33 These newer regimens may be used when available.
Direct and Indirect Pharmacologic Interactions Between ASMs and PrEP or PEP
The Liverpool drug–drug interaction checker and UpToDate are an enduring resource to monitor for emerging evidence as the treatment landscape of HIV and ASMs evolves. 34
Enzyme-Inducing Agents
The primary direct pharmacologic interactions between ASMs and the most used PrEP and PEP regimens consist of an interaction between tenofovir alafenamide (TAF), tenofovir disoproxil fumarate (TDF), and cabotegravir (CAB) with the ASMs known to have the most drug–drug interactions: enzyme inducing ASMs and valproate (Table 2). Tenofovir alafenamide and TDF are both prodrugs that require conversion to their active forms, tenofovir-diphosphate. 35 While the absence of interaction with other ASMs largely has not been studied (with the exception of lamotrigine), there also were limited theoretical interactions.
Known and Theoretical Drug–Drug Interactions Between Anti-Seizure Medications (ASMs) and Anti-Retroviral Treatments (ARTs) Used in Pre-Exposure Prophylaxis (PrEP) and Post-Exposure Prophylaxis (PEP).
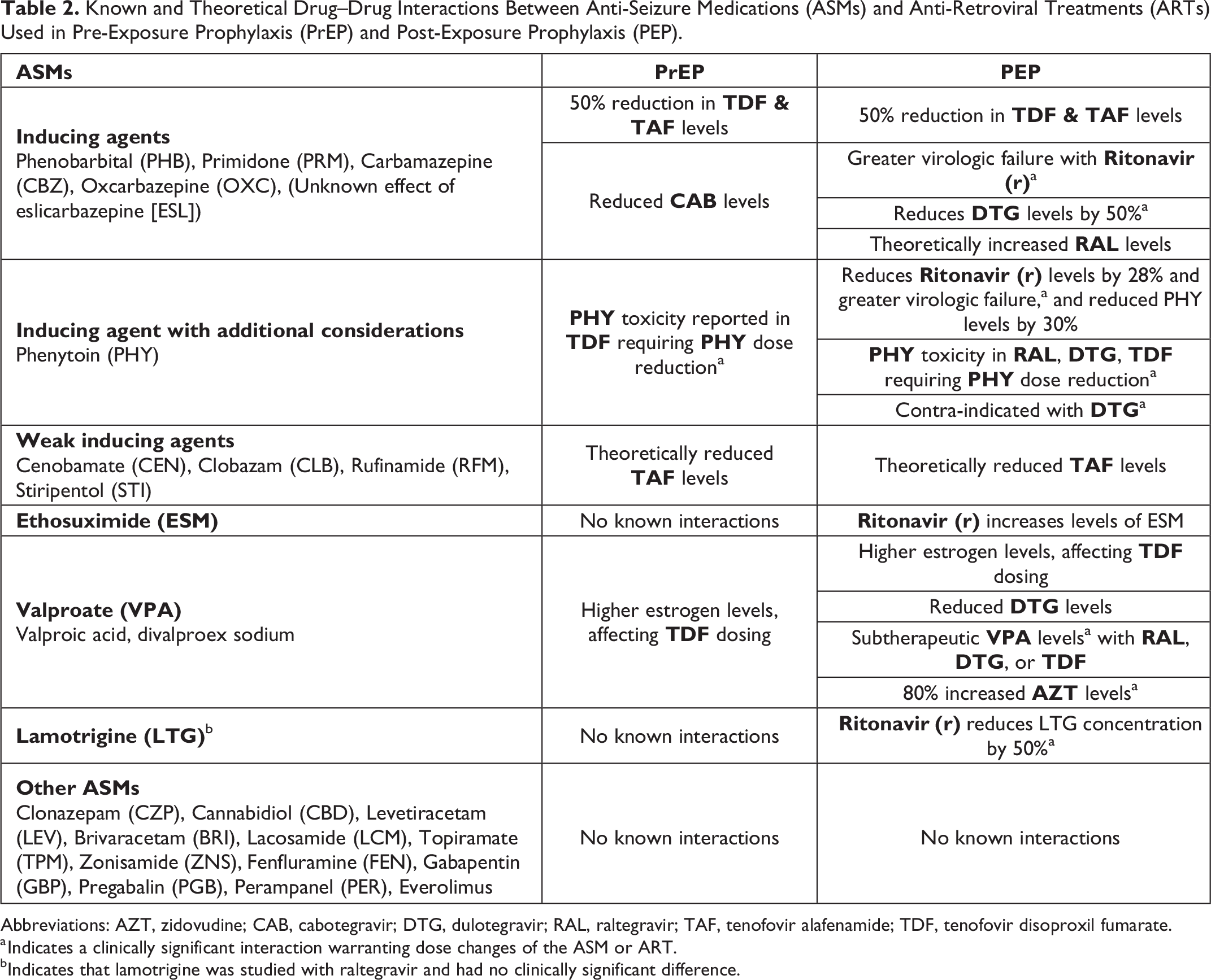
Abbreviations: AZT, zidovudine; CAB, cabotegravir; DTG, dulotegravir; RAL, raltegravir; TAF, tenofovir alafenamide; TDF, tenofovir disoproxil fumarate.
a Indicates a clinically significant interaction warranting dose changes of the ASM or ART.
bIndicates that lamotrigine was studied with raltegravir and had no clinically significant difference.
For patients with epilepsy who are eligible for PrEP, the primary drug–drug interactions focus on the interaction of TDF, TAF, and cabotegravir with the enzyme inducing ASMs, including phenytoin, phenobarbital, primidone, carbamazepine, and oxcarbazepine (Table 3). 36 These ASMs can induce CYP3A4 and other enzymes causing increased metabolism of the ARTs (Table 4). Tenofovir alafenamide or cabotegravir are components of all PrEP regimens; therefore, we recommend that these agents be avoided in patients with epilepsy eligible for or taking PrEP. The serum levels of TDF and TAF were 50% reduced in patients co-treated with carbamazepine or oxcarbazepine. 37 While therapeutic serum concentrations for TDF exist, TAF is metabolized mostly intracellularly by cathepsin A to tenofovir therefore the relevance of serum level reductions is unclear. 38 The effect of eslicarbazepine has not been studied but is theoretically expected to be similar in mechanism to carbamazepine and oxcarbazepine. 39 The magnitude of CYP450 induction is stronger for carbamazepine than oxcarbazepine or eslicarbazepine. Cenobamate, clobazam, rufinamide, and stiripentol also induce CYP3A4 enzymes to a lesser extent than the traditional inducers, which theoretically have a similar effect. 40 In addition, valproate treatment can be associated with higher estrogen levels, which can influence TDF dosing due to protein binding.
Metabolism of Agents Used in PrEP or Post-Exposure Prophylaxis (PEP).a
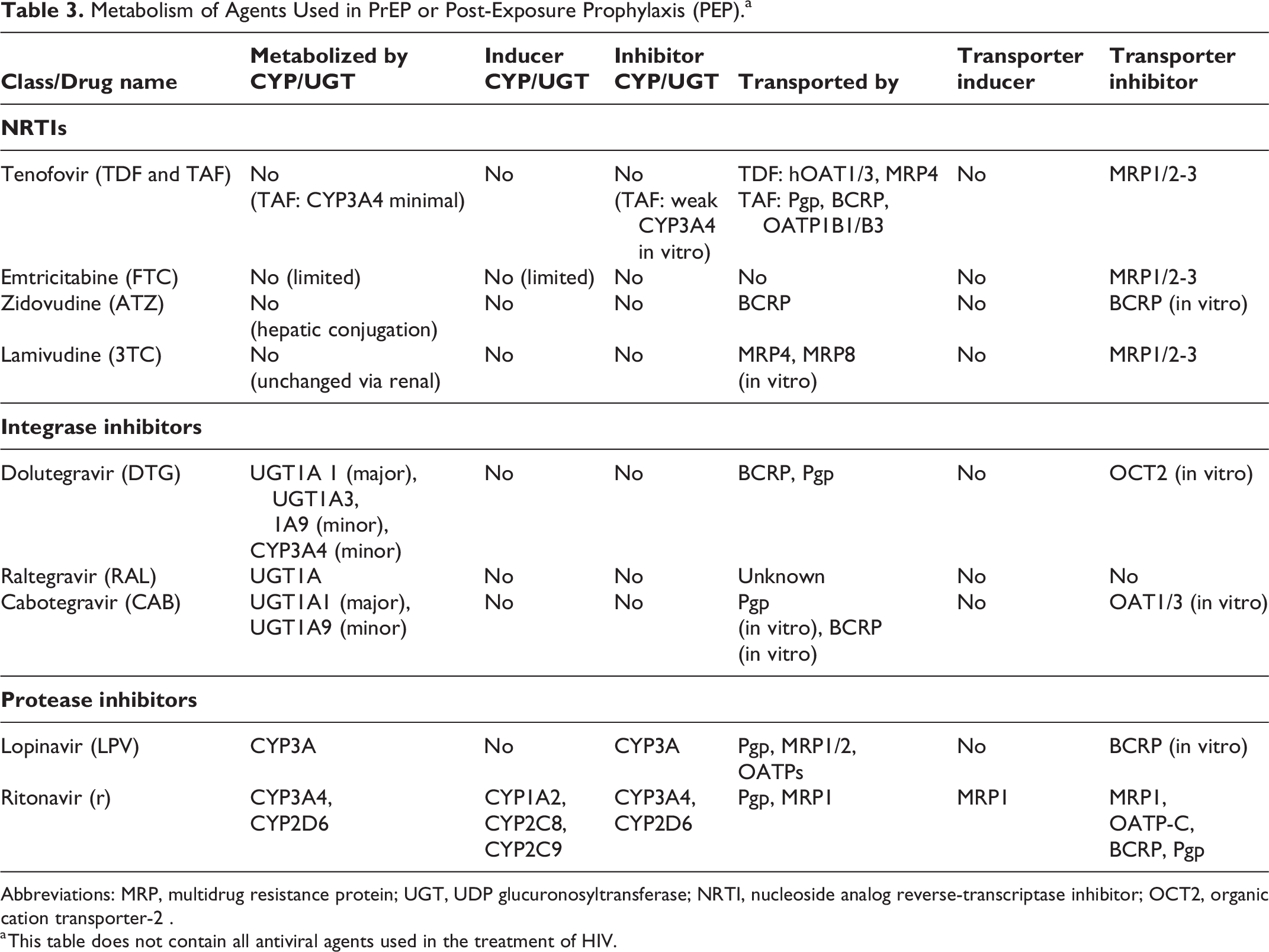
Abbreviations: MRP, multidrug resistance protein; UGT, UDP glucuronosyltransferase; NRTI, nucleoside analog reverse-transcriptase inhibitor; OCT2, organic cation transporter-2 .
a This table does not contain all antiviral agents used in the treatment of HIV.
Potential Impact of Anti-Seizure Medications (ASMs) on PrEP/Post-Exposure Prophylaxis (PEP) Medications—Specific Drug Metabolizing Isozymes and Transporters.
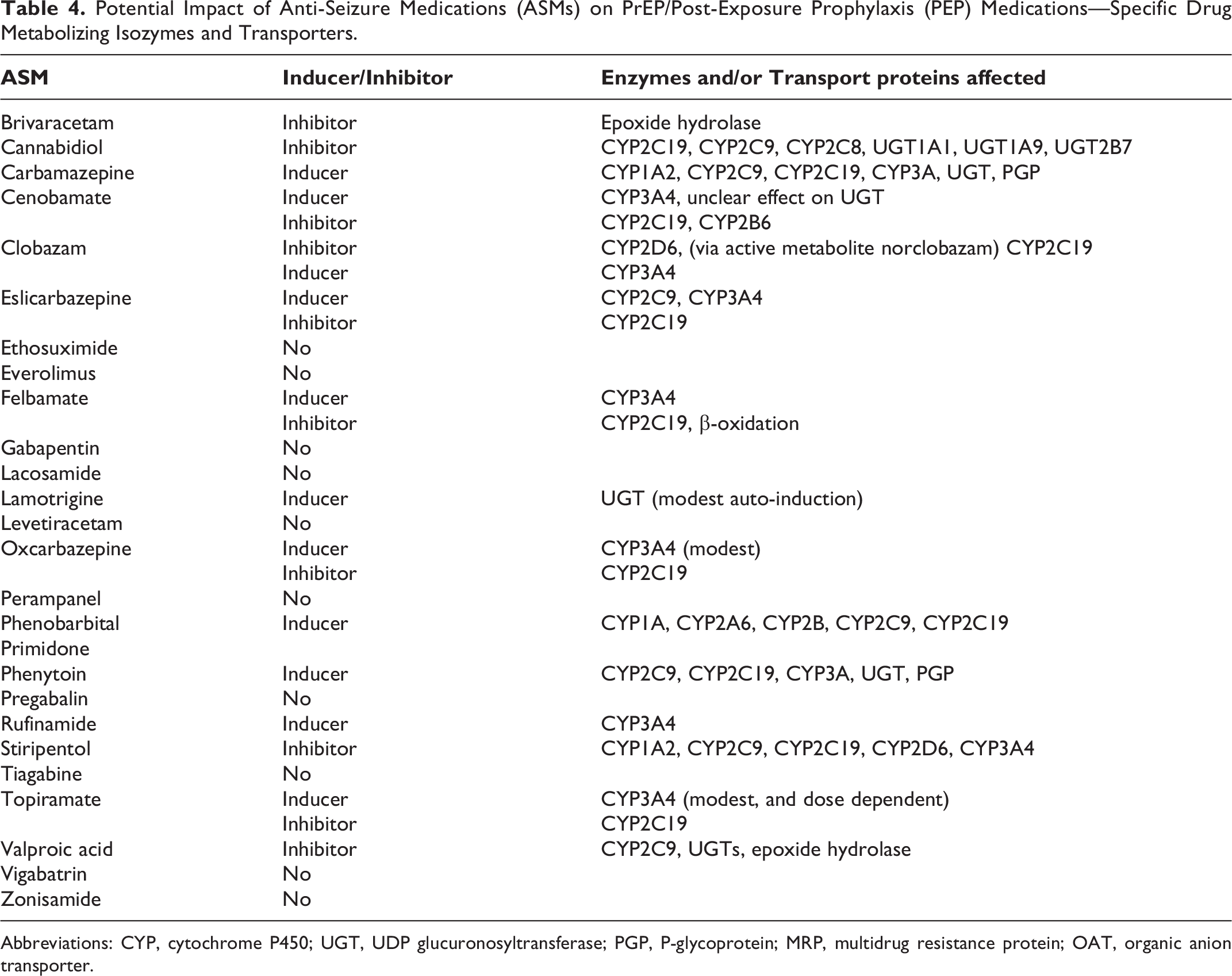
Abbreviations: CYP, cytochrome P450; UGT, UDP glucuronosyltransferase; PGP, P-glycoprotein; MRP, multidrug resistance protein; OAT, organic anion transporter.
There are clinical situations where these drug–drug interactions cannot be avoided. One practical dosing modification is that dosing of TDF for PrEP can be less than daily in specific situations. However, if the patient is taking an enzyme inducing ASM, we recommend daily dosing due to these potential interactions and otherwise good tolerability of daily TDF dosing.
In all patients, there is a paucity of data to demonstrate that these interactions have a clinically meaningful impact upon breakthrough HIV infections with PrEP or PEP. Historical data to suggest that cotreatment of HIV with enzyme inducing ASMs was associated with increased risk of virologic treatment failure, but these data primarily involve patients cotreated for HIV with boosting elements of PEP (ritonavir [/r], dolutegravir [DTG], and raltegravir [RAL]) as well as protease inhibitors, and non-nucleoside reverse transcriptase inhibitors. The exception was one report of phenytoin toxicity with cotreatment with TDF that resolved with phenytoin dose reduction. 41 Therefore, the clinical significance of the interaction between enzyme-inducing ASMs and breakthrough HIV infections with PrEP is unclear and is an area of future study.
The interactions of enzyme inducing agents with boosting agents of PEP are more established. Enzyme-inducing agents may reduce dolutegravir (DTG) levels by 50%. For carbamazepine specifically, it is recommended to increase dolutegravir (DTG) dosing from once daily to 50 mg twice daily in adults and twice daily of the weight-based dosing in pediatrics based on data supporting safety and efficacy of this dosing. 42 This recommendation for twice daily dosing was generalized to other strong inducers of the CYP3A4 and UGT1A1 enzymes, including phenobarbital, primidone, but the safety and efficacy of more general recommendation was not specifically studied. Phenytoin is also a strong inducer of CYP3A4, but cotreatment with dolutegravir (DTG) is contraindicated. Phenytoin also has clinically significant interactions with raltegravir, TDF, and ritonavir (r). 1 There are no data regarding the combination of PEP components with moderate or weak enzyme inducing ASMs (eg, oxcarbazepine, clobazam, cenobamate).
Cotreatment of HIV with ritonavir (r), which is part of the PEP WHO regimen, together with these enzyme-inducing ASMs also was associated with greater virologic failure in long-term HIV. 18,19,26 This interaction also is theoretically present with the US DHHS alternative of RAL.
These complex interactions highlight the challenge in providing the short-term treatment of PEP or longer-term PrEP in patients on long-term enzyme inducing ASMs. While the need for PEP cannot always be predicted, we recommend avoidance of these enzyme inducing ASM regimens, if possible, when patients are anticipated to need PrEP or PEP.
Valproate
In addition to traditional enzyme-inducing agents, valproate has complex drug–drug effects on TDF, a component of both PrEP and PEP. 43 Increased estrogen levels associated with valproate may also warrant daily TDF dosing for PrEP, similar to the recommendation for transgender women taking exogenous estradiol-based hormones. 4,41,44 -46 Additionally, there is a single case report of lower valproate serum level and effectiveness in bipolar disease with ritonavir. 47 When PEP is needed, there can be bidirectional interactions that can reduce both ART levels as well as valproate serum levels, resulting in theoretical concerns regarding both breakthrough HIV infections and seizures. There is mixed evidence between peer-reviewed publications and the Liverpool drug–drug interaction checker regarding the details of interactions between valproate and individual elements of PEP. We did not identify empiric evidence or case reports regarding the clinical significance of these theoretical concerns for seizures, so we cannot confidently recommend empiric dosing changes of valproate, PrEP, or PEP.
For children under 10 years using the WHO regimen for PEP and children between 4 weeks and less than 2 years using the US DHHS regimen for PEP, valproate increased the levels of zidovudine (AZT). 48 Because PEP treatment is typically temporary for 4 weeks, patients requiring both valproate and PEP could receive both treatments, but providers should monitor for increased adverse effects related to zidovudine (AZT) including hematologic toxicity, anemia, and neutropenia. In absence of observed adverse effects, we cannot confidently recommend empiric dosing modifications.
Ritonavir Interactions With ASMs
Another potential clinically relevant interaction exists between lamotrigine (LTG) and ritonavir (r), an element of the WHO recommendation for PEP. Ritonavir (r) can reduce lamotrigine (LTG) concentration by 50% within days, therefore lamotrigine dosing may need to be modified during short-term PEP treatment containing ritonavir (r). 1,34,49 Irrespective of potential lamotrigine adjustments or decreases during PEP, the removal of ritonavir (r) may contribute to a rise in serum lamotrigine concentration. This may be associated with both transient increased adverse effects of lamotrigine, similar to those during the placebo week of estrogen-containing oral contraceptives, as well as increased risk of Steven’s Johnson Syndrome (SJS). 50 Data from withdrawal of lamotrigine during long-term inpatient monitoring suggest the risk of SJS is low if lamotrigine dosing was lower for less than 7 days, but the duration of ritonavir (r) in PEP typically lasts 4 weeks. Therefore, during this period of PEP and afterward, the dosing of lamotrigine may need to be increased and additional counseling provided regarding monitoring for SJS. 51 In addition to lamotrigine, the European Medicines agency recently issues a warning regarding the interaction with everolimus and ritonavir, which also is an element of treatment for Covid-19 in combination with nirmatrelvir in Paxlovid. 52
Other ASMs and Other ARTs Without Known Interactions
In addition to highlighting the presence of known or theoretical interaction between certain ASMs and many of the ARTs used in PrEP/PEP, we emphasize that there are several ASMs without known or limited interactions. When possible, patients with anticipated need for PrEP/PEP should aim to achieve seizure and side effect freedom with these noninteracting ASMs. However, one third of patients with epilepsy have medication-resistant epilepsy, defined by failure by 2 or more appropriately prescribed and tolerated ASMs, therefore this may not be possible. 53 Additionally, while levetiracetam (LEV) was recently added to the WHO essential medications list, these modern ASMs often are not widely available globally due to cost, regulatory approval, and supply chain challenges. Analogously, modern ARTs typically have fewer drug–drug interactions; therefore, they may emerge as preferred elements of PrEP and PEP regimens in people taking ASMs.
Overlapping Side Effects Between ASMs and PrEP or PEP
In addition to direct and indirect pharmacologic interactions between ASMs, it is also important to consider side effects that overlap between these medications and therefore may synergistically worsen with dual treatment (Table 5). 54 This worsening of adverse events was observed with enzyme inducing ASMs and historical ARTs that are not part of the current PrEP or PEP regimens. 27,43 While there are no data to support increased monitoring, these should be elements of counseling and can influence the selection of either ASMs or PrEP regimens. Because of the short duration of PEP treatment, the benefit of PEP treatment to avoid HIV infection outweighs the temporary adverse events during treatment.
Overlapping Side Effects Between Anti-Seizure Medications (ASMs) and Anti-Retroviral Treatments (ARTs) Used in Pre-Exposure Prophylaxis (PrEP) and Post-Exposure Prophylaxis (PEP).

Abbreviations: 3TC, lamivudine; AZT, zidovudine; CBD, cannabidiol; CBZ, carbamazepine; CEN, cenobamate; CLB, clobazam; CYP, cytochrome P450; CZP, clonazepam; DTG, dulotegravir; ESL, eslicarbazepine; ESM, ethosuximide; GBP, gabapentin; LCM, lacosamide; LTG, lamotrigine; MRP, multidrug resistance protein; OAT, organic anion transporter; OXC, oxcarbazepine; PGP, P-glycoprotein; PER, perampanel; PGB, pregabalin; PHB, phenobarbital; PHY, phenytoin; PRM, primidone; RAL, raltegravir; RFM, rufinamide; TAF, tenofovir alafenamide; TDF, tenofovir disoproxil fumarate; TPM, topiramate; UGT, UDP glucuronosyltransferase; VPA, valproate; ZNS, zonisamide.
a Ending ritonavir (r) treatment may lead to elevations in LTG serum level, thereby increasing risk for Stevens Johnson Syndrome (SJS).
The primary overlapping adverse effect between TDF-containing regimens and ASMs was increased risk of osteoporosis and osteopenia. The American Epilepsy Society recommends monitoring for osteopenia and osteoporosis with DEXA scans after at least 10 years of treatment with a broad array of ASMs, as well as every 5 years thereafter (Table 3). 55 Similarly, the PrEP and PEP regimens containing TDF have been associated with osteopenia and osteoporosis. 56 -58 In cisgender men and transgender women at sexual risk, we recommend TAF, which has less impact upon bone health. 59 In all patients on PrEP for sexual risk, cabotegravir long-acting injections also do not have a known impact on bone health. However, neither TAF nor cabotegravir (CAB) have been studied in people who inject drugs.
The second most common overlapping adverse effect was hepatic and gastrointestinal (GI) adverse effects. Tenofovir disoproxil fumarate, TAF, and dolutegravir were associated with mild transaminitis, diarrhea, as well as other gastrointestinal effects. The zidovudine (AZT) and lamivudine (3TC) in PEP for children has been associated with severe hepatomegaly with steatosis and GI distress. This overlaps with the hepatic and gastrointestinal adverse effects seen in valproate, cannabidiol, ethosuximide, topiramate, zonisamide, felbamate, carbamazepine, phenytoin, phenobarbital, lamotrigine, and perampanel. Therefore, patients on these ASMs may opt for cabotegravir long-acting injections for PrEP due to sexual risk. 4,20,21
The next overlapping side effect was weight gain and dyslipidemia with TAF and dolutegravir as well as some ASMs. 60,61 Weight gain and metabolic syndrome can be meaningful side effects to valproate, clonazepam, clobazam, cannabidiol, gabapentin, and pregabalin. Additionally, the enzyme inducing ASMs have been associated with increased cardiovascular risk that may be compounded by increased by weight gain and dyslipidemia. Therefore, non-TAF and non-dolutegravir regimens may be preferentially selected in patients on those ASMs. In contrast, TDF was associated with weight loss and improved lipids, similar to topiramate and zonisamide. 62
While Stevens-Johnson-Syndrome (SJS) and rashes are rare both for ASMs and PrEP regimens, they represent meaningful overlapping concerns but there is no evidence that the risk of SJS increases when treated with multiple medications each independently associated with SJS. The concern for SJS is one of the reasons why lamotrigine requires a titration period when initiated, slow dose changes when doses are increased, and good medication adherence; but SJS was uncommon in patients on stable levels of medications. Increased SJS risk on lamotrigine may occur after discontinuation of ritonavir at the end of WHO-guideline driven PEP, therefore cotreatment with lamotrigine and ritonavir needs to be supervised carefully both during and after PEP. Additionally, SJS can occur during the first 3 months of treatment in patients with HLA-B*1502 taking carbamazepine, oxcarbazepine, or eslicarbazepine. 63 -65 While not associated with SJS, we also highlight that cabotegravir (CAB) in PrEP is an injection medication and can be associated with injection site reactions that could be misinterpreted as the initial rash of SJS. Raltegravir in PEP also has case reports of association with SJS. The PEP alternative to raltegravir is dolutegravir, which also was associated with a rare hypersensitivity reaction including rash and hepatotoxicity in less than 1% of participants in phase 3 trials. 66,67 There also have been postmarketing reports of rashes and SJS with ritonavir, which is an element of some PEP regimens. Rashes also can be a common side effect of zidovudine-based PEP in children and the alternative PrEP regimen of rilpivirine.
The last commonly overlapping adverse effect of both ASMs and medications used in PrEP and PEP was dizziness. Dizziness is a common adverse effect across many ASMs and was most prominent in sodium channel blockers, especially with polytherapy of multiple sodium channel blockers. 68 Dizziness also was noted in TAF, used in PrEP, as well as raltegravir and lamivudine, both of which are elements of PEP.
Considerations for the Treatment of Specific Populations
It is of utmost importance to ensure that whichever medications are chosen for HIV prevention and seizure management are affordable and accessible because of pervasive barriers in access to health care and systematic determinants of health that influence both HIV and epilepsy care. 69 Due to associated stigma and concern for judgment regarding HIV care, it is highly important to avoid assumptions and present a welcoming, safe environment so anyone may feel comfortable disclosing if they are using PrEP/PEP so that they can avoid preventable medication interactions. The guidelines for PrEP regimens specifically describe differing treatments based on use of injection drugs, sexual behaviors, and people who are transgender/gender nonconforming/nonbinary/two-spirit or any variation and may be on feminizing hormones (Table 1A). The discussion with patients should focus on medication choices and not a detailed discussion of the reason why PrEP or PEP is needed. To assist in these conversations, clinicians may seek additional training in providing an open and welcoming environment for sexual or gender minorities and familiarizing themselves with epilepsy care in transgender patients. 70
There are many special populations to keep in mind when prescribing PrEP or PEP for HIV prevention. Most commonly, TDF/TAF dosing and use can change in individuals with preexisting renal or hepatic disease and alcohol or substance dependence. Regular monitoring of liver enzymes and renal function is advised for those on PEP, but serious hepatic complications have not been observed. In medically complex patients, collaborative consultation with HIV medicine, neurology, and other relevant specialties should occur, especially for patients who are immunosuppressed (eg, organ transplants, multiple sclerosis), undergoing chemotherapy, or considering any surgical intervention. PrEP is not explicitly contraindicated in any of these situations, but an individualized medication interaction check should occur (eg, Liverpool drug–drug interaction checker and UpToDate).
As for individuals of childbearing potential, gravid state, breast/chest-feeding, or expressing milk/lactating, there are no contraindications to PrEP use with TDF and TAF-containing regimens. 71,72 There is a lack of data regarding cabotegravir in the peri-partum time, but pharmacokinetic data suggests cabotegravir (CAB) levels are adequate during pregnancy. In people who are breast/chest-feeding, there may be small transfer of medication to the infant through milk, but this risk must be weighed against the risk of acute HIV infection. In people of childbearing potential who need PEP, prenatal vitamins can inhibit RAL and DTG absorption, so doses should be more than 2 hours apart. LactMed is an updating resource to evaluate for medication considerations for lactation. 73
Unmet Needs and Knowledge Gaps
The theme of this guide for the cotreatment of epilepsy and use of PrEP or PEP was the lack of data regarding interactions their clinically meaningful impact on breakthrough HIV infections. We have highlighted both objective and theoretical interactions between commonly used ASMs as well the common regimens for PrEP and PEP, but there are limited studies to address if these interactions impact the efficacy of PrEP or PEP in patients with epilepsy on these regimens. If there is reduced efficacy of PrEP or PEP in patients taking certain ASMs, then further studies should evaluate both the safety and efficacy of potential dosing modifications to allow for safe and effective cotreatment. For example, the safety and efficacy of twice daily dolutegravir (DTG) dosing in patients taking carbamazepine (CBZ) has been established, but similar studies for tenofovir disoproxil fumarate (TDF) or tenofovir alafenamide (TAF) do not yet exist. These further studies could support development of formulations with appropriate dosing, as well as insurance coverage for potential modified dosing recommendations.
Lastly, there are several options for noninteracting ASMs in the treatment of and prophylaxis for HIV. The presumed safety of these ASMs is largely based on a paucity of evidence to the contrary, but empiric data should be collected to support this presumption. Further studies on the overlap between epilepsy and the need for PrEP or PEP are needed, as well as studying the impact of switching ASMs to avoid interactions with PrEP or PEP.
Conclusions
This practical narrative review of the known and theoretical pharmacologic interactions and overlapping side effects between ASMs with PrEP and PEP highlights many clinically meaningful considerations when these treatments need to be used in combination. This can guide in selection of both ASMs and PrEP/PEP regimens to avoid or account for those considerations. In addition, we highlight that there are numerous ASMs without known or theoretical interactions with PrEP and PEP regimens. Therefore, there are viable cotreatment options for both epilepsy and provide appropriate prophylaxis for HIV infection. This is a marked development from the prior treatments, where interactions created challenges in cotreatment. 16 However, these current ASM and ART treatments are not globally available; therefore, providers should be aware of the known interactions. Due to the profound impact of HIV infection, patients taking ASMs and at sexual risk for HIV or who inject drugs should both be offered and counseled about the value of PrEP and PEP.
Footnotes
Acknowledgments
The authors would like to acknowledge the invaluable contributions of the American Epilepsy Society Treatments Committee and the Council Activities in providing support, expertise, and guidance throughout the development and review of this manuscript. Their collective efforts have significantly enriched the content of this manuscript and contributed to its overall quality and relevance in advancing epilepsy care. The authors thank Lauren Orciuoli for her support throughout the process of creating this manuscript.
Author Contributions
Dr Alick-Lindstrom proposed this work and supervised all phases. Dr Kerr organized the team, wrote the first draft, and provided expertise on seizures. Dr Gidal systematically evaluated the pharmacologic interactions with additional insights by Dr Eyal. Dr Wilmshurst provided dual insight on epilepsy and HIV. Drs McAnaney, Eley, and Avedissian provided expertise in PrEP, PEP, and HIV both in the United States and globally. All authors reviewed and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: No pharmaceutical company contributed to or reviewed this manuscript in any stage of development. Dr Kerr received honoraria for his work as Associate Editor for Epilepsia and review articles for Medlink Neurology, is a paid consultant for SK Life Sciences (cenobamate), UCB (levetiracetam and brivaracetam), EpiTel, Jazz Pharmaceuticals (cannabidiol), Cerebral Therapeutics (intra-ventricular valproate), and Biohaven Pharmaceuticals, has collaborative or data use agreements with Eisai (perampanel), Janssen (topiramate), Radius Health, and GSK (levetiracetam, lamotrigine, cabotegravir, lamivudine, zidovudine, dolutegravir, rilpivirine). Prof. Wilmshurst has received honoraria for her work as Associate Editor for Epilepsia and Chief Editor for the Pediatric Neurology subsection of Frontiers in Neurology, and as a member of the national South African advisory boards for Novartis (carbamazepine, oxcarbazepine) and Sanofi (valproate). Dr. Eyal was a consultant for TrueMed, Israel (eslicarbazepine), BioPass, and the Israeli Ministry of Health. The remaining authors have no conflicts of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Kerr’s research time was funded by the US National Institute for Neurological Disorders and Stroke (NIH R25NS089450, NIH U24NS107158), the American Epilepsy Society, the Epilepsy Foundation, the American Academy of Neurology, the American Brain Foundation, and the Epilepsy Study Consortium. Dr Eyal’s research time is funded by the Israel Science Foundation and the Israeli Ministry of Science and Technology.
