Abstract
Drug resistant epilepsy is a disorder involving widespread brain network alterations. Recently, many groups have reported neuroimaging and electrophysiology network analysis techniques to aid medical management, support presurgical planning, and understand postsurgical seizure persistence. While these approaches may supplement standard tests to improve care, they are not yet used clinically or influencing medical or surgical decisions. When will this change? Which approaches have shown the most promise? What are the barriers to translating them into clinical use? How do we facilitate this transition? In this review, we will discuss progress, barriers, and next steps regarding the integration of brain network analysis into the medical and presurgical pipeline.
Introduction
Recognizing epilepsy as a network disorder has the potential to aid clinical decision making and improve patient quality of life. However, the practical steps required to efficaciously bring epilepsy network neuroscience into the clinic remain unclear. It is encouraging that great progress has been made in identifying and characterizing epileptic networks across various patient subgroups and analysis methodologies. 1 -7 These findings have implications for medical and surgical management of epilepsy but are typically retrospective and not validated across diverse datasets. Thus, it is time to address the systemic barriers that prevent the advancement of these promising findings and ultimate adoption into clinical care.
This review will begin by briefly outlining promising network neuroscience findings in epilepsy that have the potential to be translated into clinical practice. Specifically, we highlight three critical needs at three stages of clinical care in epilepsy that we believe could be augmented by network analyses: (1) From initial presentation, reduction in the time taken trying different anti-seizure medications (ASMs) to diagnose drug-resistant epilepsy; (2) While evaluating drug-resistant epilepsy patients for advanced diagnosis and surgical treatments, utilization of validated quantitative tools to accurately characterize epileptic networks to minimize misdiagnosis, inform surgical decision-making, and avoid post-surgery deficits, and; (3) During the post-surgical treatment phase, noninvasive monitoring to prognosticate outcomes and inform clinical decision making such as how to alter ASMs post-surgery and when to consider alternative treatments if seizures remain uncontrolled. Fundamental to addressing these critical clinical needs is our ability to accurately identify a patient’s epileptic network and track changes in these networks over time.
We will then discuss the barriers that hinder the adoption of network neuroscience into the clinic. Specifically, we will address barriers of patient representation, external validation, network model inconsistency, equipment and expertise, and general resistance towards change. For each barrier, we will outline a proposal of next steps to facilitate this translation. Overall, it is our hope that this review of progress, barriers, and next steps will serve as an additional motivator to move epilepsy network neuroscience beyond single institutions or datasets findings and coalesce our efforts into practical advancements for the continual benefit to those living with epilepsy.
Progress
Epilepsy has been considered a network disorder for many decades. 8,9 A brain network consists of nodes defined by different brain areas and edges defined by connections between those brain areas. 10 Edges can be structural (e.g., white-matter connections estimated from T1-and diffusion-weighted magnetic resonance imaging (MRI)) or functional (e.g., interactions estimated from electroencephalography (EEG), magnetoencephalography (MEG), or functional MRI). Growing evidence suggests that seizures in epilepsy may emerge due to subtle alterations in the brain network, and seizures harness the altered network architecture of the brain to originate and propagate. 11 Characterization of altered network structure and function could augment clinical decision making at the three important stages outlined below.
Stage 1: ASM Trials
Mapping epileptic networks can identify patients who might be refractory to pharmacotherapy (Figure 1A). Abnormal neuroimaging such as lesions on MRI and specific electroclinical features such as focal to bilateral seizure spread are known risk factors of drug resistance, and these risk factors are also associated with widespread brain network alterations. 12,13 Even in non-lesional newly diagnosed focal epilepsy patients, those with persistent seizures have bilateral structural network impairment compared to patients who were seizure-free with ASMs at 24-months since diagnosis in 1 investigation. 14 In another study examining idiopathic/genetic generalized epilepsy (IGE), drug-responsive patients had reduced diffusion network alterations than drug-resistant IGE patients with bi-hemispheric structural network alterations. 15 These studies indicate that epileptic networks might be more altered in drug-resistant epilepsy patients and measuring these alterations can be a marker for the prognosis of drug resistance in the early stages of epilepsy treatment.
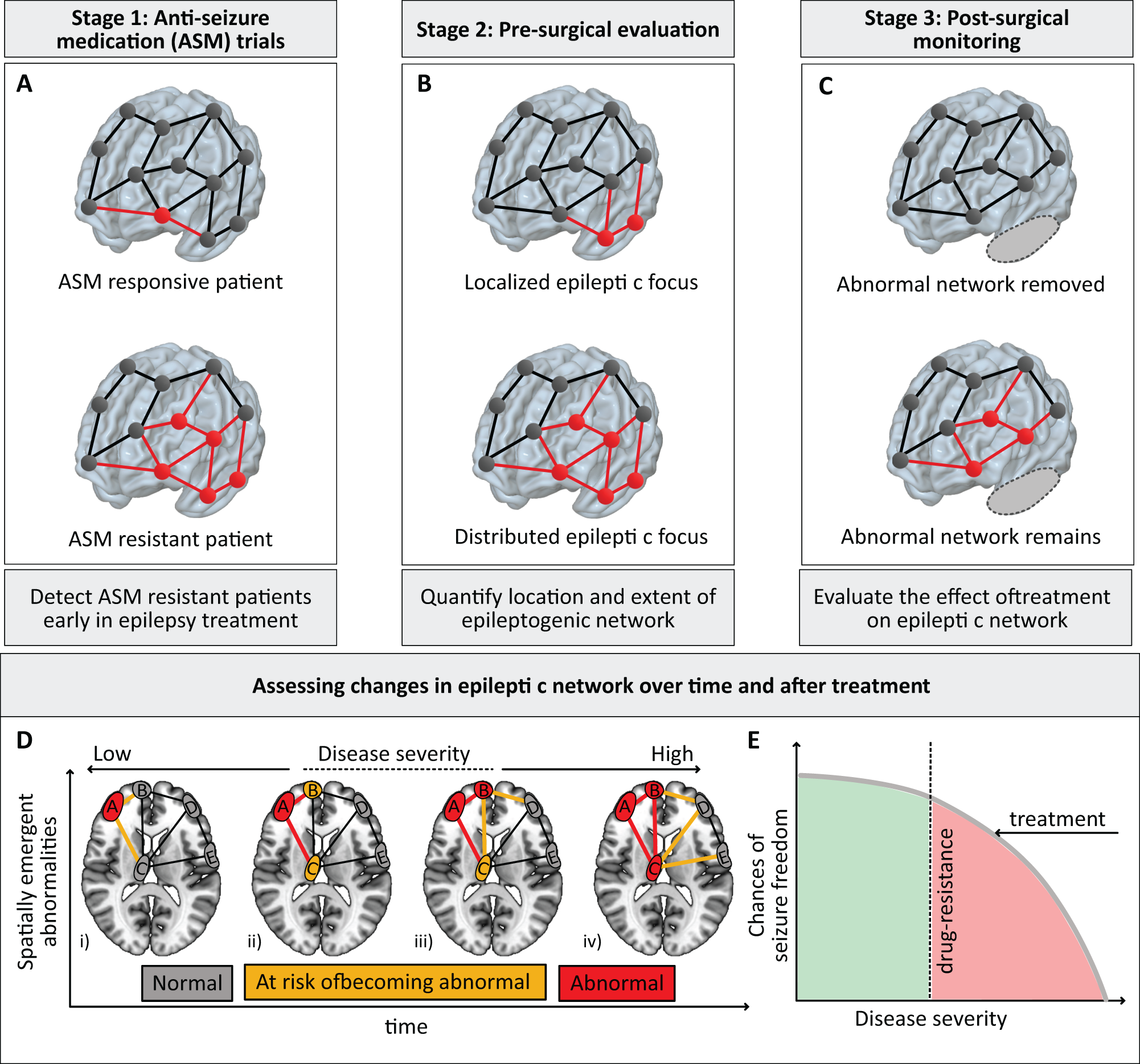
A conceptual representation of the three critical clinical needs that can be addressed by epilepsy network analysis. (A) Compared to the anti-seizure medication responsive patients, evidence suggests that epileptic networks are more pronounced in anti-seizure medication resistant patients. Stratifying patients by mapping and quantifying their epileptic networks can potentially detect patients resistant to anti-seizure medication early in epilepsy treatment. (B) In drug-resistant epilepsy patients, epileptic networks can either localize to a circumscribed spatially contiguous region or be distributed across multiple regions. Accurate quantification of the location and extent of the epileptic network is 1 of the key objectives at the pre-surgical evaluation stage. (C) Epileptic networks can either be removed entirely by treatment, or some of them may remain postoperatively. Assessing changes in the epileptic network after treatment can enable assessing change in disease severity, thus guiding postoperative clinical decisions. Panel (D) conceptually represents the spatial and temporal evolution of epileptic networks with disease progression by harnessing the brain network architecture. An abnormal brain area (in red) predisposes the connections from that brain area at risk of becoming abnormal (in yellow). Recurrent seizures reinforce these excitatory pathways to become abnormal, predisposing the areas connected to the abnormal connections at risk of becoming abnormal. As abnormality load increases with disease progression, the disease severity also increases. (E) A hypothesized profile of chances of seizure freedom as a function of disease severity. In this conceptual model, disease severity has a threshold; below this threshold (green zone), patients are responsive to medication, and the chances of seizure freedom are high. As the disease severity crosses this threshold, the patient becomes drug resistant (red zone), and the chances of seizure freedom drop rapidly. Patients are more likely to benefit by treatment - surgery or neurostimulation - at an earlier time point before the severity of epilepsy has progressed.
Stage 2: Presurgical Evaluation
In patients with drug-resistant epilepsy, prognostic models using clinical characteristics and clinically acquired imaging and electrophysiology studies help predict surgical outcome. 16 -19 Incorporating network analyses in such models could be helpful (Figure 1B). 20 -22 For example, structural networks have shown promise in predicting seizure outcomes after surgery. 23 -27 Specifically, more abnormalities remaining in structural networks spared by surgery correlated with post-operative seizure recurrence. 28 Likewise, functional network analyses have demonstrated that patients with more functional abnormalities persisting after surgery are less likely to become seizure free. 29 -36 Recent studies show that alterations in structural and functional brain networks spread from a traditionally defined epileptic focus to other connected brain regions, and these alterations increase with epilepsy severity and duration. 37,38 Overall, these studies suggest that various methodologies can localize network abnormalities present in surgical non-responders and could help us redefine the brain tissue that is targeted by surgical treatments. Mapping alterations in brain networks can inform how localized or distributed the epileptic focus is, thus augmenting clinical decision making during presurgical planning. With the growing number of treatment options including targeted surgical interventions such as laser interstitial thermal therapy (LITT) and intracranial neuromodulation devices there is a critical need for quantitative tools to guide clinical decision making.
Stage 3: Postsurgical Monitoring
Examining network changes after resection or neuromodulation therapies can explain why patients may respond differently to therapy (Figure 1C). This serial analysis of networks can elucidate whether epileptic networks normalized post-treatment and how this normalization related to surgical outcomes. In a study of pediatric patients with intractable epilepsy, functional normalization of non-resected seizure onset zones after surgery was associated with seizure freedom in 97% of 39 cases. 39 In contrast, postoperative functional network abnormalities did not normalize in a small cohort of adult patients with temporal lobe epilepsy (TLE) who were seizure-free after surgery. 40 In comparison to controls, functional connectivity at the contralateral hippocampus was found to be more deviant from controls, 41 whereas connectivity at some arousal networks normalized towards controls after seizure-free surgery. 42 For structural networks in left TLE, larger reductions between pre and postoperative diffusion networks measuring quantitative anisotropy of white-matter were associated with postoperative seizure-freedom. 43 To our knowledge, there are only a few studies analyzing brain networks after treatment, and more systematic studies are needed to reconcile the contradictory findings highlighted above. Quantifying changes in the epileptic network before and after treatment can elucidate network reorganization, thus identifying patients at risk for seizure recurrence, and may thereby inform clinical decisions such as whether to wean ASMs.
In summary, the field of network-targeted epilepsy treatment is rapidly evolving and shows great promise for clinical translation. Understanding epileptic networks can help predict the predisposition to drug resistance and prevent delays in surgical assessments, localize epileptogenic tissues for targeted interventions, predict surgical outcome, and elucidate post-intervention network reorganization for follow-up assessments (Figure 1D and 1E). 44
Barriers and Next Steps
Although promising, these network-based findings are far from clear clinical translation. The French clinical trial (EPINOV NCT03643016) is now recruiting 350 patients from 11 epilepsy centers to test the capacity of the Virtual Epileptic Patient algorithms to improve clinical outcomes. 45 However, most network neuroscience findings in epilepsy face multiple systemic barriers (Table 1) that obstruct the road to clinical translation. To address these barriers, we must examine the stages prior to randomized controlled trials and/or clinical adoption. Specifically, how can we homogenize our efforts to enable large-scale consensus on key network findings?
Barriers and next steps to translating epilepsy network neuroscience findings into clinical practice.

Patient Representation
To begin, an important barrier to direct clinical translation of network neuroscience findings is incomplete patient representation. Many network studies focus on specific homogenous subgroups of patients (e.g. mesial TLE) to avoid statistical confounds from analyzing a heterogeneous cohort. While there is obvious value in studying homogenous patient groups, these datasets often do not accurately reflect the heterogeneity of epilepsy patient care. For example, a network analysis to localize seizure onset zones developed exclusively in mesial TLE patients is of less value early in the diagnostic evaluation of focal epilepsy than one validated in a heterogeneous patient population. Furthermore, single-center study populations are often biased towards the demographic and socioeconomic features of a particular geographic region.
To address these issues, studies with broad inclusion of epilepsy patient subtypes should be encouraged and carefully considered at the editorial level, to complement continued investigations in more homogeneous patient subsets. An active effort should be made by investigators to design studies intended to capture patients with diverse demographic backgrounds, and diversity in geographical regions should be considered when planning multisite investigations. Finally, advanced statistical analyses (e.g. mixed effects modeling 46 ) should be employed to account for patient heterogeneity and encourage inclusive patient selection.
Network Model Inconsistency
The specific network models used by research groups vary tremendously. It is often difficult to contrast and compare network findings because the fundamental assumptions, data preprocessing, and statistics vary widely between studies. A formalized review of the literature to identify studies with the most promising network approaches may be a valuable first step. If we could establish a rubric upon which each study would be evaluated, then each research group would be motivated to provide the necessary elements for their individual study. Some suggested elements in the rubric could be the use of pre-defined MRI and EEG acquisition and preprocessing protocols, specific neuroimaging atlas usage, network model types, and categorization of clinical decision-point that the study is attempting to address. These elements would require considerable discussion amongst interested parties, and do not need to be overly rigid, but the field could benefit highly from this type of agreement.
To facilitate model homogenization and adoption of a formalized rubric, increased effort could be given to producing streamlined analysis pipelines that groups could utilize with their data with minimal preparation. This federated approach could serve as an additional tool to that of large, shared databases and would not require data-sharing agreements. The oversight for this process could be undertaken by existing societies which could systematically analyze current progress and provide suggestions. For example, the International League Against Epilepsy (ILAE) Surgical Therapies Commission recently created the Epilepsy Surgery Networks task force specifically tasked with making recommendations on these issues. The purpose of the formalized review system would not be to restrict the exploratory analyses that groups are conducting, but rather to encourage the community to additionally consider utilizing standardized/semi-standardized pipelines on their data for the benefit of the field.
External Validation
A common critique of promising findings in epilepsy network neuroscience is the lack of external validation across multi-institutional datasets. There are existing efforts to collect multi-institutional data, such as the Enhancing Neuro Imaging Genetics through Meta Analysis (ENIGMA) consortium, among others. 47 These data-sharing initiatives address the issues of underpowered studies and patient heterogeneity. However, there are still barriers to external validation that include raw data heterogeneity, lack of comprehensive clinical variables, and publication considerations. For raw data heterogeneity, there are existing measures that can be taken to combine and homogenize large datasets. 48,49 Regardless, prospective agreement on data collection protocols would best address this issue, but institutions often remain resistant to changing the protocols that they have used for years. Furthermore, not all institutions collect the necessary clinical variables to properly use that cohort for validation. In terms of publication considerations, if every clinician were to contribute to these common databases, then who would serve as an unbiased peer reviewer? Further, how can we avoid groupthink when 1 study finds positive results from the database, and other groups do not question the findings because their data was included in the analysis? We believe contributions to these databases are important, but we also believe that parallel preparation at the level of individual institutions can help ameliorate persistent barriers to external validation.
For efficacious and efficient external validation, large-scale data sharing agreements at the level of individual institutions would be valuable to actively engage all stakeholders. To facilitate this, the funding mechanisms such as the Brain Research Through Advancing Innovative Neurotechnologies (BRAIN) Initiative 50 could support network neuroscience research across multiple institutions with prospective data sharing agreements to enable rapid external validation. This paradigm would expedite research turnover, highlight robust replicable results, and promote the development of data necessary for confident clinical translation.
Equipment and Expertise
Just a few decades ago, there was a lack of standardization for the equipment and experience recommended to medically manage epilepsy and evaluate patients for surgery at U.S. epilepsy centers. 51 For example, the availability of 3T MRI for epilepsy neuroimaging was inconsistent, and no formal recommendations regarding subspeciality training of neurologists and neurosurgeons treating surgical patients existed. Over time, epilepsy organizations recommended standard equipment and expertise for epilepsy centers. 52 This standardization has improved the consistency of care across institutions, but how do we adapt these recommendations for the modern era of network neuroscience?
Network neuroscience will bring new considerations to the equipment and expertise required to evaluate patients medically and surgically. For example, (i) functional and diffusion-weighted MRI protocols could be collected in addition to anatomical images in epilepsy protocol MRIs, (ii) the formatting of clinical variables necessary to quantify and qualify network analysis results could be better homogenized across epilepsy centers, (iii) a formalized database at each center could be recommended to capture clinical and research data, such as MRI and EEG recordings, and the data could be stored in a generalizable de-identified format such as iEEG-BIDS (intracranial EEG Brain Imaging Data Structure) that could be made available upon reasonable request, 53 iv) computational resources necessary to conduct network analyses could be standardized and recommended, (v) an engineer or computational neuroscientist could be recommended to serve on multidisciplinary epilepsy teams to run standardized network analyses and interpret them for clinicians. Overall, foreseeing these resource barriers and addressing them with standardized recommendations may smooth the incorporation of network neuroscience into the epilepsy clinic.
Resistance to Change
As Kuhn outlined in The Structure of Scientific Revolutions (1962), incremental progress is episodically disrupted by paradigm shifts in thinking. 54 While current tools for epilepsy evaluation will continue to be valuable, network neuroscience has the potential to facilitate a paradigm shift in the clinical care of epilepsy. For example, utilizing network analyses to predict ASM failure, predict the surgical success for a patient with a normal MRI, or understand specifically why a patient failed epilepsy surgery may significantly alter our diagnostic and treatment algorithms over time. Mounting evidence suggests that we are not accurately modeling the medical and surgical management of epilepsy patients. People still fail medications after many years of trials despite new agents, and surgeries fail to prevent disabling seizures at similar rates for the past several decades. Even if network models may sometimes only offer subtle improvements to clinical outcomes, we must not be afraid of a shift in epilepsy care to include network neuroscience. As Kuhn outlines, this shift in thinking could open the door to large future improvements potentially unobtainable within our current paradigm. Presently, clinicians collect a range of variables based on the patient’s history, semiology, electrophysiology, and radiological findings. Thus, the actionable suggestion is to benchmark novel quantitative network metrics alongside routinely used clinical variables to encourage clinicians to adopt the new quantitative tools into their clinical workflow. Importantly, not every question raised in the “Progress” section above requires a randomized controlled trial. Rather, by systematically addressing the barriers outlined in this review, we can take the next steps necessary to translate network neuroscience into the clinic and realize a shift in epilepsy care.
In summary, the field of epilepsy network neuroscience has many promising findings that could likely improve patient care if adopted into the clinic. We have outlined some of the barriers we believe are hindering the path to translation and suggested practical steps to begin addressing these barriers. We are optimistic about the future of clinical care for patients with epilepsy and believe epilepsy network research will play a pivotal role in improving patient quality of life.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NINDS: R01NS116504 (Davis and Sinha), F31NS120401 (Johnson), R01NS110347 (Davis), R56NS099348 (Davis), R01NS110347 (Englot).
