Abstract
This American Epilepsy Society (AES) official statement provides information and preliminary guidance to Society members related to the U.S. Food & Drug Administration (FDA) December 22, 2021 Emergency Use Authorization for Paxlovid™ for the oral treatment of mild to moderate COVID-19 in adults and children (≥12 years and weighing ≥40 kg). Paxlovid is likely to be widely prescribed, and important considerations for patients on antiseizure medications (ASMs) include key contraindications and potential toxicity or dose adjustments while taking Paxlovid. This statement highlights concerns and provides information about their pharmacologic basis. Of particular concern, concomitant use of Paxlovid with the ASMs carbamazepine, phenobarbital, phenytoin, and primidone is contraindicated, because they are strong inducers of the CYP3A4 isozyme that metabolizes Paxlovid and thereby could cause loss of virologic response and development of resistance. Alternate oral or intravenous COVID-19 treatments should be considered. A second concern is that Paxlovid may increase the plasma concentrations of many ASMs, because it inhibits the CYP3A4 isozyme. ASMs that are metabolized, at least in part, by CYP3A4 include cannabidiol, carbamazepine, clobazam, clonazepam, diazepam, ethosuximide, everolimus, felbamate, lacosamide, midazolam, oxcarbazepine, perampanel, stiripentol, tiagabine, and zonisamide. Patients receiving these medications may warrant closer monitoring while being treated with Paxlovid.
Keywords
The American Epilepsy Society (AES) has compiled U.S. Food & Drug Administration (FDA) information and preliminary guidance for AES members on Paxlovid™, one of the new COVID-19 oral antivirals that is likely to be widely prescribed.
FDA Emergency Use Authorization for Paxlovid
On December 22, 2021, FDA announced Emergency Use Authorization (EUA) for Paxlovid1,2 for the oral treatment of mild to moderate COVID-19. Paxlovid is a combination of nirmatrelvir (a SARS-CoV-2 protease inhibitor) and ritonavir (an HIV-1 protease inhibitor and
Considerations for Patients on Antiseizure Medications
There are two major considerations when Paxlovid is given to patients taking some antiseizure medications (ASMs), consistent with previously documented interactions of ASMs and antiretroviral drugs, including ritonavir.
3
The first major consideration is the effect of some ASMs on Paxlovid. The ASMs
Paxlovid needs to be started soon after diagnosis of COVID-19. Clinically, if patients were to stop taking these strong enzyme-inducing ASMs, the time-dependent process of de-induction of CYP3A4 isozymes would take more time than is likely feasible, so other treatments for COVID-19, including molnupiravir or IV therapies like remdesivir, 5 may be appropriate. The EUA for molnupiravir, 6 another COVID-19 oral therapy for patients ≥18 years old, states that the drug is not a substrate of CYP enzymes or major drug transporters, and in vitro studies do not suggest inhibitory actions.
Antiseizure Medications Enzymatic Considerations.
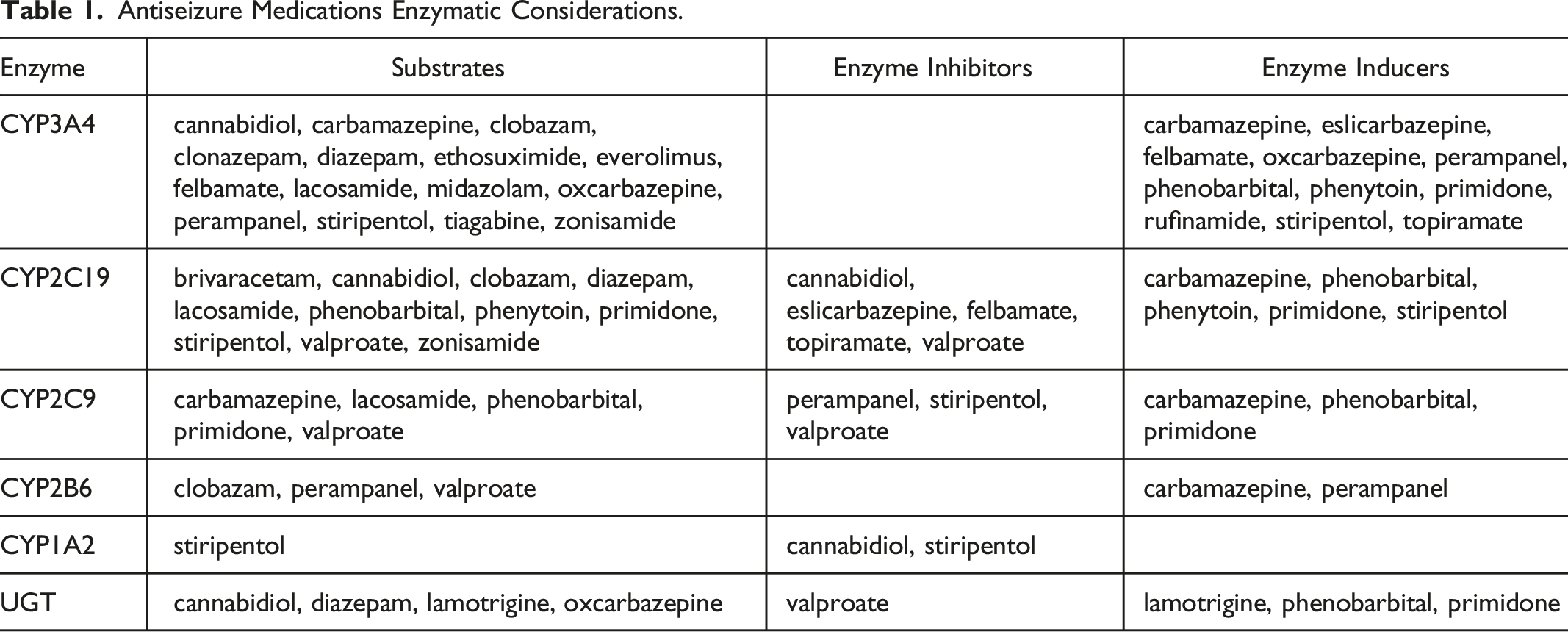
The second major consideration is the effect of Paxlovid on the plasma levels of several drugs
Paxlovid (specifically ritonavir) can also inhibit P-Glycoprotein (PGP), a drug transport protein, which may impact bioavailability of some ASMs.7,8 Patients receiving these medications may warrant closer monitoring while being treated with Paxlovid. In particular, use of Paxlovid with everolimus (which is a substrate of both 3A4 and PGP) may increase the risk of toxic adverse effects and may require a reduction in the dose of everolimus.
Ritonavir is a weak inducer of CYP1A2, CYP2C19, and CYP2C9, a moderate inducer of CYP2B6, and an inducer of UGT1A4. Notably, ritonavir combinations can significantly reduce lamotrigine plasma concentrations, presumably via induction of glucuronidation; however, data for Paxlovid are not yet available, and the clinical relevance during the short duration of Paxlovid treatment is unclear. Monitoring of lamotrigine plasma concentrations may be warranted in patients receiving Paxlovid therapy. Formulations that contain ritonavir may reduce plasma concentrations of other ASMs that are substrates of these isoenzymes, as specified in Table 1.
Additional important information for AES members about Paxlovid and ASMs is highlighted in Table 1, with the caveat that specific pharmacokinetic/pharmacodynamic (PK/PD) data and clinical outcome data are not yet available on Paxlovid for people with epilepsy.
A more complete summary of other ASM pharmacokinetics and drug interactions may be found in the AES 2020 update of A Summary of Antiseizure Medications Available in the United States. 9
Guidance to American Epilepsy Society Members and American Epilepsy Society Monitoring
Given the known interactions, AES members should closely monitor patients taking any of these several ASMs while taking Paxlovid for signs and symptoms of toxicity and make appropriate dose adjustments.
In addition to the fact sheet on the Paxlovid EUA, 2 available FDA resources include a provider letter 10 and FAQ, 11 information for patients with moderate renal impairment,10,12 and the EUA scientific review information,13,14 as well as a fact sheet for patients, parents, and caregivers, available in English and Spanish. 15
The AES Treatments Committee will continue to monitor the latest information on known interactions for Paxlovid and other newer antiretroviral drugs and will alert AES members as new information or further guidance becomes available. Updates to other AES COVID-19–related documents are underway and will be disseminated to members after review by the committee and Council.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In addition to employment with affiliated institutions, authors report the following relationships: J. Cokely – Speakers bureau for UCB Biosciences, Inc.; Co-author on manuscript for Neurelis Pharmaceuticals, Optimizing Counseling and Treatment Management of Patients With Epilepsy Experiencing Seizure Clusters: The Role of the Pharmacist, a review for pharmacists of all seizure cluster medications currently in use for seizure rescue therapy. B. Gidal – Speaking honoraria from Eisai, Greenwich, and SK Life Science; consulting with UCB, Eisai, Greenwich, and Aquestive. J. Keller – no conflicts reported. D. Vossler – Past clinical trials results write-ups for SK Life Science and UCB Pharmaceuticals; current clinical trials PI, with salary support via payment to employer, for SK LIfe Science, Xenon, Longboard, and Neuroelectrics.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
