Abstract

Alzheimer’s Pathology Causes Impaired Inhibitory Connections and Reactivation of Spatial Codes During Spatial Navigation
Prince SM, Paulson AL, Jeong N, Zhang L, Amigues S, Singer AC. Cell Rep. 2021;35(3):109008. doi:10.1016/j.celrep.2021.109008 Synapse loss and altered synaptic strength are thought to underlie cognitive impairment in Alzheimer’s disease (AD) by disrupting neural activity essential for memory. While synaptic dysfunction in AD has been well characterized in anesthetized animals and in vitro, it remains unknown how synaptic transmission is altered during behavior. By measuring synaptic efficacy as mice navigate in a virtual reality task, we find deficits in interneuron connection strength onto pyramidal cells in hippocampal CA1 in the 5 relevant human familial mutations (5XFAD) of the mouse model of AD. These inhibitory synaptic deficits are most pronounced during sharp-wave ripples (SWRs), network oscillations important for memory that require inhibition. Indeed, 5XFAD mice exhibit fewer and shorter SWRs with impaired place cell reactivation. By showing inhibitory synaptic dysfunction in 5XFAD mice during spatial navigation behavior and suggesting a synaptic mechanism underlying deficits in network activity essential for memory, this work bridges the gap between synaptic and neural activity deficits in AD.
Commentary
What are memories made of? While this question is open, researchers have some ideas. One hypothesis is that we make memories by establishing associations between the mental representations of events that happened somewhere, sometime. The hippocampus is responsible for making the links. Presumably, activity of hippocampal neurons generates spatial and temporal feature maps during experience, and it is the replay of those sequences that weaves the memory web. In terrestrial rodents, which inspired the theory of the cognitive map, representations arise from theta oscillations (4–12 Hz) in the form of place cells accompanying exploration. Sequence reactivation is brought about by sharp-wave ripple (SWR) oscillations (100–200 Hz), which occur offline when the animal is immobile or sleeping (Figure 1A). The hippocampal code is made up of a patchwork of these representations. Summary of findings. (A) According to the current cognitive map theory, sequences of hippocampal place cells recorded during theta oscillations and representing the order of experience are replayed during sharp-wave ripples (SWR). This replay results from an appropriate interplay between pyramidal cells and interneurons. (B) In Alzheimer’s disease, the connections between interneurons and pyramidal cells become weaker and replay is impaired. Sharp-wave ripples recorded in 5 relevant human familial mutations animals are fewer and shorter. Notably, the effect of epileptiform activities and hyperexcitability should be also considered.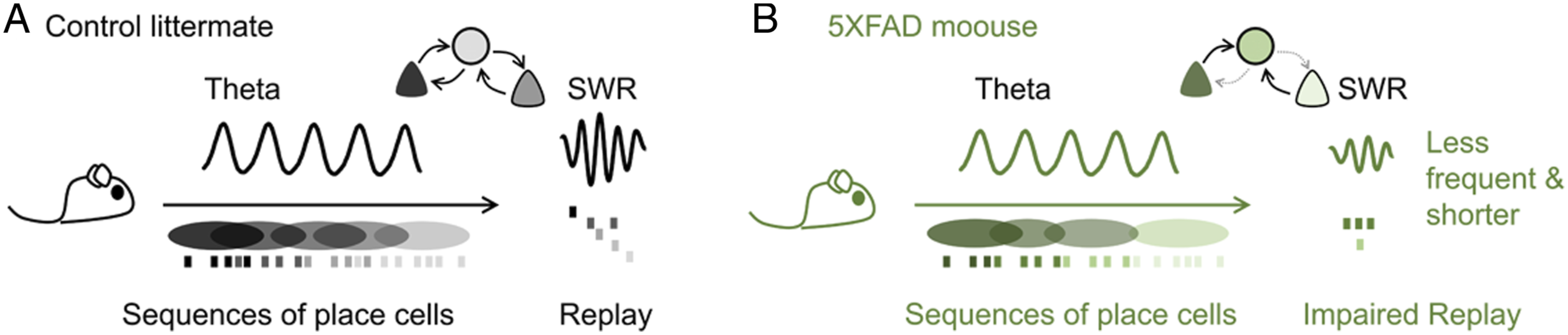
A corollary to this perspective is that in diseases affecting episodic memory, the hippocampal code should be impaired. Alzheimer’s disease (AD) is one of the most common causes of progressive dementia in older people, and synaptic loss is a major correlate of cognitive severity.1 Studying brain diseases has traditionally informed understanding of brain function. If the synaptic threads weaken, the web should fade away and memories sink into the oblivion. A recent paper by Prince et al2 aims to dig into this idea.
By exploiting a mouse model of AD carrying 5 relevant human familial mutations (5XFAD), the authors explore neuronal representations from the CA1 hippocampal region. 5XFAD mice develop high levels of amyloid beta in the subiculum and layer V of the entorhinal cortex in less than 2 months of life. By 12 months, amyloid pathology has expanded all over the brain. 3 This is the timeframe when Prince et al started working with these animals. Notably, reports suggest aberrant cortical excitability and impaired hippocampal oscillations in 5XFAD mice, as seen in other experimental AD models and in people suffering from AD.4-6
Using multi-electrode recordings in mice running head-fixed in a virtual reality task, the authors evaluated the neuronal firing and local field potentials (LFP) from the dorsal hippocampus of older 5XFAD mice and their wild-type littermates. They managed to separate pyramidal and interneuron-like firing extracellularly and looked at their potential monosynaptic connections by examining their correlative activation. Their first idea was to check for the strength of the threads.
Identifying functional connectivity between neuronal pairs using firing rate data is not an easy task. You need to see the pre-synaptic cell firing and check for the effect in the presumptive postsynaptic cell. However, cells may fire close in time, not because they are necessarily connected through excitatory synapses. Since neurons are embedded in a circuit, they could simply follow the ongoing collective dynamics. If you look for inhibitory connections instead, you need to infer them indirectly by looking at firing decreases of the putative postsynaptic cell. When a postsynaptic cell fires poorly, how to be sure about inhibition? Moreover, because the neuronal firing rate is influenced by running speed and motivation, there may be some confounding factors in the analysis. Therefore, careful statistical design is required to avoid these biases.
The authors first checked that mice from the 2 groups behaved similarly to acquire a water reward in virtual reality. Moreover, they confirmed mice were using visual cues to drink because they got similarly lost when cues were removed. Using the head-fixed experimental approach is especially useful to reduce the number of animals, by enabling multiple longitudinal recording sessions. It is also simpler and less demanding than chronic implants, where each mouse is instrumentalized. By allowing several penetrations at different locations in the brain, the yield of single cells increases one order of magnitude. The work entailed collecting over 1360 cells from 5XFAD mice and more than 1000 cells from the control group.
In 5XFAD mice, the authors found weaker inhibitory connections from GABAergic cells to pyramidal cells located in CA1, but not the other way around. They found this trend both during periods of running and immobility, when neuronal firing rates are fundamentally different. After controlling for variances of spike numbers, firing rate, and other confounders, the authors found that functional connectivity in 5XFAD mice was about 60% smaller during SWRs and 40% smaller during theta. In all cases, they found inhibition was especially poor in a window of about 5 milliseconds after the pre-synaptic GABAergic cell fired. This is exactly 1 cycle of reactivation at 200 Hz.
During sequence replay, neurons fire in a chain, and it is proposed that it is the interplay between pyramidal cells and interneurons that pace the ripple. 7 Therefore, if the interneuron-to-pyramidal cell thread weakens, the chain should be broken. Prince et al found shorter and fewer numbers of SWRs in 5XFAD mice (Figure 1B). They looked at the consecutive spatial maps of the cells while mice ran in virtual reality and found they were just marginally different. Notably, the clearest alteration emerged when comparing replay of pairs of cells that fired closely together during theta oscillations. These very same pairs were poorly reactivated during ripples in 5XFAD mice. The hippocampal patchwork was frayed.
In recent years, accumulating data support the idea that the cognitive map theory of hippocampal function applies to dysfunction as well. In diseases affecting this region, such as AD and temporal lobe epilepsy, cognitive impairment may be caused by rhythmopathies and microcircuit alterations of hippocampal representations. 8 Understanding how intrinsic, synaptic, and circuit dysfunction contribute to these deficits is an emerging direction. For instance, altered intrinsic excitability of GABAergic cells caused by sodium channel loss-of-function contribute to impaired GABA release and epileptiform activity in the hippocampus of a different AD mouse model. 6 Yet, synaptic dysfunction is considered a major mechanism resulting from the alteration of signaling pathways. 9 The paper by Prince et al aims to link these different levels by showing how deficits of inhibitory function at monosynaptic pairs associate with impaired place cell reactivation. Importantly, the very same mechanisms contribute to hyperexcitability and subclinical seizures, suggesting potential commonalities between AD and epilepsy.
The study remains correlative though. Since mice were just running for water in a virtual world, there was no explicit test of their memory abilities or a causative link between their frayed monosynaptic threads and ripples. Yet, the changes observed in SWR dynamics call for a fundamental mechanism of impairment. Similar single cell reactivation shuffling caused by an impaired excitatory–inhibitory ratio was described in epileptic rats and linked to their poor episodic-like memory. 10 Other AD models have reported alterations of sharp-wave ripple physiology as well. 11 The effect of epileptiform activities and hyperexcitability in place cells should also be considered.
The study cannot reveal changes occurring at CA3 to CA1 monosynaptic circuits nor inputs from entorhinal cortex, all of which influence SWR dynamics. Independent of the complexity of the underlying mechanism, early synaptic deficits of the hippocampal synapses and interneuronal dysfunction have been recently associated with memory impairment in AD models.12,13 Unwinding the AD skein may start from pulling up the monosynaptic threads.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
