Abstract
The precise coordination of neuronal activity is critical for optimal brain function. When such coordination fails, this can lead to dire consequences. In this review, I will present evidence that in epilepsy, failed coordination leads not only to seizures but also to alterations of the rhythmical patterns observed in the electroencephalogram and cognitive deficits. Restoring the dynamic coordination of epileptic networks could therefore both improve seizures and cognitive outcomes.
Keywords
Introduction
In a car engine, timing is essential. Throughout the power cycle, each component plays its role at a specific time. Piston compression and extension, intake and exhaust valves, spark plug ignition, all have to be perfectly coordinated in time to ensure proper engine function. When such coordination is altered, as for when a timing belt is failing, problems emerge, ranging from abnormal ticking noises, vibrations, and ultimately engine breakdown.
As for an engine, brain function, that is, cognition also depends on the intricate coordination of its parts (pyramidal cells and interneurons) with regard to specific power cycles defined by neuronal oscillations. Studies of neurological and psychiatric disorders, including epilepsy, also suggest that when such temporal coordination fails, consequences can dreadful. Both in animal models and patients, evidence shows that neuronal coordination and brain rhythms are altered in epilepsy and that seizures involve abnormal but stereotyped dynamics. In this review, I will approach epilepsy from this dynamic perspective and propose that seizures and cognitive deficits result from bad timing within neural networks.
The Ticking Noise: Rhythmopathies and Cognitive Impairments
Neural networks have the natural ability to display oscillatory activity without being forced to do so by external stimuli. Conserved throughout evolution, neuronal oscillations are rhythmical variations of the electroencephalography (EEG) or local field potentials that reflect, for the most part, the synchronized and repetitive activation of synaptic currents among large populations of neurons in a structure. 1 These oscillations can be segregated in classes of similar frequency ranges that progress from low-frequency oscillations (<1 Hz) to ultrafast oscillations (200-600 Hz). In normal conditions, the most known rhythms are δ (0.5-4 Hz), θ (4-8 Hz in humans; 4-12 Hz in rodent hippocampus), α (8-12 Hz in humans), β (15-30 Hz), γ (30-90 Hz), and high-frequency oscillations (HFOs: 90-300 Hz and sharp-wave ripples [SPW-R] in the hippocampus). 2 Evidence suggests that the transmission of information between structures is made possible by oscillations through the principle of “communication by synchrony,” 3 where the firing of input neurons fluctuates rhythmically and in synchrony with the membrane potential of target neurons. Such coincidence, observed only if the 2 structures are coherently oscillating, is more likely to induce firing in the target structure and influence synaptic plasticity. In addition, within oscillatory patterns such as θ cycles or SPW-R, groups of neurons fire in ordered sequences that reproduce, at compressed time scales the past or future experiences of the individual. 4 -7 Finally, γ oscillations are involved in perceptual binding, a process by which the different features encoded by different neuronal ensembles are associated and form a coherent percept. 8 γ alterations are believed to play a major part in the functional deficits observed in schizophrenia, 9 including perceptual binding. Altogether accumulating evidence support the notion that oscillations organize neuronal activity and information processing and allow the emergence of high-level functions involved in planning, perception, and memorization. Alterations of such oscillations are likely involved in neurological and psychiatric disorders. 10
In epilepsy syndromes such as temporal lobe epilepsy (TLE) or epileptic encephalopathy (EE), alterations of naturally occurring oscillations are also observed, leading to the concept of rhythmopathies. Particularly, θ rhythmopathies have gained recent interest 11 and have been proposed as a mechanism responsible for cognitive deficits. In animal models of TLE, θ amplitude and frequency are decreased, even before spontaneous seizures occur. 12,13 Speed modulation of θ amplitude and frequency is also abnormal 14 and the θ coherence (a measure of oscillation synchrony between structures) within the hippocampal formation and between hippocampus and prefrontal cortex is altered. 13,15,16 Finally, the coordination of pyramidal cells and interneurons by θ oscillations is abnormal. 17,18 These changes are correlated with performance deficits in episodic, spatial, or working memory tasks. 12 -14 In addition to θ, slow wave oscillations 19 and γ are also altered in TLE. 20 As for other epilepsy syndromes, TLE is associated with the presence of pathological HFOs (pHFOs with frequencies >200 Hz) which are associated with impaired memory performance. When pHFOs occur during locomotion in animal models, they decrease θ amplitude and alter pyramidal cell spatial coding. 21
Rhythmopathies are also observed in other types of epilepsies. Alterations of EEG patterns such as the presence of slow wave activity 22 correlate with neuropsychological scores. In children with Dravet syndrome (DS), there is an overall slowing of the EEG, particularly a decrease of α in favor of θ. 23 In adults with DS, the absence of occipital α is associated with poor cognitive outcome. 24 Induced oscillations are also affected. For instance, in photosensitive epilepsy and DS, there is a decrease in sensory-evoked γ responses in the visual and auditory cortices, respectively. 25,26 In animal models of DS carrying Scn1a mutations, hippocampal SPW-R frequency and their associated HFOs are reduced and the modulation of γ oscillations by θ rhythm is altered. 27,28
The fact that brain rhythms are abnormal in epilepsy and that such alteration is correlated with cognitive performance deficits does not necessarily mean causation. Indeed, both alterations of oscillations and cognitive deficits occur in the context of seizure activity, and seizures could be the main driver of cognitive deficits. To dissociate between the effects of seizures from those of etiology, we developed a series of experiments where etiological features of epileptic syndromes are found in animal models free from seizures. 29 -31 These models consisted in inducing Nav1.1 downregulation, a characteristic of DS networks, in only a portion of the adult brain, therefore allowing us to examine its effects in an otherwise intact brain, that is, devoid of seizures and developmental insults. As for DS models, 32 focal Nav1.1 downregulation in the medial septum, a structure considered as the θ pacemaker, specifically affected the firing of fast-spiking, putative GABAergic neurons. 29 It induced, in the hippocampus, a decrease in hippocampal θ amplitude and frequency, which was correlated with a diminished spatial memory performance. 29,30 When induced in the hippocampus, Nav1.1 downregulation decreased place cell accuracy, altered the θ modulation of pyramidal cells and interneurons and reduced the θ modulation of γ rhythms. 31 Here again, these alterations were associated with spatial performance deficits but were observed in the absence of seizures. Altogether these results suggest that cognitive deficits could directly result from the etiology itself and that alterations of oscillations, caused by GABAergic dysfunction are involved.
To further test the rhythmopathy hypothesis, future studies could artificially induce, in nonepileptic models, the rhythm alterations observed in epilepsy and determine whether the functional and behavioral consequences of such alterations are similar. Additionally, the hypothesis may be supported by manipulations causing the restoration of both rhythmical activity and cognitive function in epileptic animals. That said, oscillations on the EEG merely reflect the dynamic organization of underlying networks. Therefore, the restoration of rhythms considered here should be performed by acting on the mechanisms of network dynamics rather than the EEG pattern itself. To this end, a better fundamental understanding of the processes involved in normal neuronal dynamics is critical.
Engine Breakdown: Stereotypical Progression of Seizures
Since the late 90s investigators noted that EEG signals during seizures followed stereotyped progression patterns, as if they were subjected to a certain deterministic process. 33 -36 To account for this phenomenon, they borrowed from the formalism of the physics of nonlinear dynamics and described neural networks as nonlinear dynamical systems. A dynamical system is a system whose state evolves in time according to fixed rules. Such a system is called nonlinear when the change of its inputs is not proportional to its outputs and its behavior appears unpredictable yet is not random. At the core of this formalism is the concept of an attractor: a state in which the system naturally stabilizes itself even when starting from different conditions. Importantly for epilepsy, attractors can be dynamic: while the system is in constant activity, it forms some kind of recurrent loop out of which it is difficult to exit. In epilepsy, this loop is characterized by the periodicity of spike and wave discharges during seizures. Finally, neural networks can possess multiple attractors and switch from one stable state to another, for example, from a nonictal attractor to the seizure one (Figure 1). How and why systems go from one attractor to the seizure one is a fundamental question in the domain of seizure prediction.
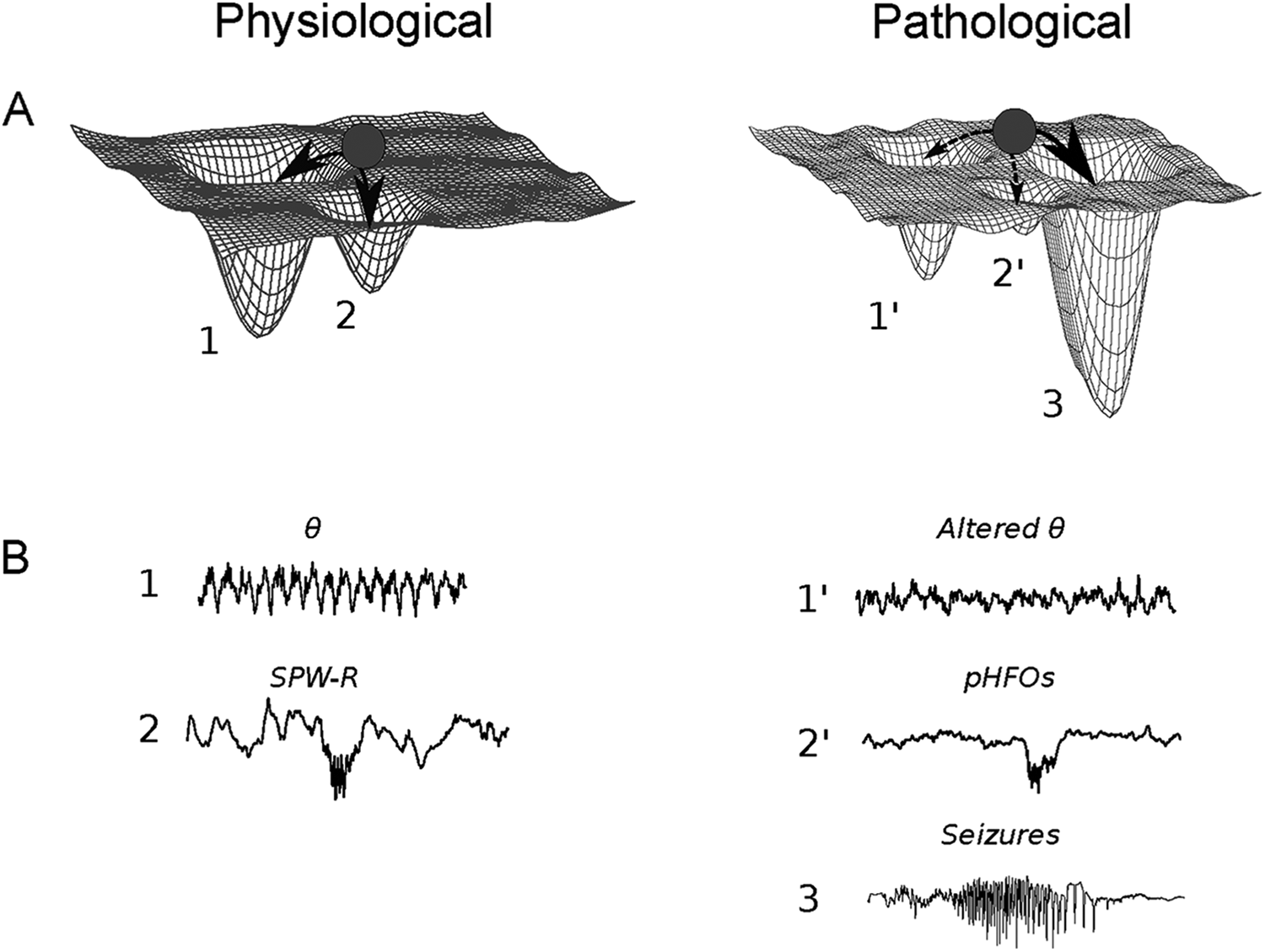
(A) Attractor dynamics. The state of a network can be illustrated by a ball evolving on a surface containing wells to which the ball is naturally attracted. In physiological conditions, attractors consist of brain rhythms like θ oscillations (1) or sharp-wave ripple (SPW-R) (2). In epilepsy (right), these attractors are eroded (1′ and 2′) and a new, powerful attractor appears: the seizure state (B) θ oscillations (1) and SPW-R (2) in physiological conditions (left). In addition to seizures (3), epilepsy (right) is characterized by rhythmopathies, that is, alterations of brain rhythms. Here, θ amplitude and frequency decrease and pathological high-frequency oscillations (pHFOs) occur.
In addition to EEG patterns, evidence for such attractor dynamics in epilepsy is also observed at the network level through calcium imaging or single-unit recordings. 37,38 In both acute preparations and chronic epilepsy models, seizures preferentially recruit specific groups of neurons that fire in stereotyped order, from ictal spike to another, and even from seizure to seizure. As for attractor dynamics in kindled seizures, 36 such sequences are driven by GABAergic activity, particularly the activity of fast-spiking, putative inhibitory interneurons. 37,38 Altogether, these data suggest that epilepsy can be considered as a disease of dynamical systems where neuronal networks transition between normal attractor states to extremely strong seizure attractors. As for brain rhythms, GABAergic inhibition is critically involved in this process.
The Failing Timing Belt: GABAergic Neurons in Motion
In a large part, the generation, modulation, and coordination of rhythmical patterns involve GABAergic activity. Through the strong influence of fast-spiking, perisomatic targeting interneurons on the timing of action potential firing, the filtering influence of dendritic targeting cells on synaptic processing or the coordinated influence of long-range projecting GABAergic cells, inhibition forms a critical part of the brain machinery. For example, during θ oscillations in the hippocampus, each interneuron subclass, targeting specific portions of the pyramidal cell, is activated at a specific time with regard to the oscillation cycle and inputs. 39 Although the functional role of this intricate timing relationship is still not completely understood, evidence suggests that it is critical for the organization of information in episodic and working memory. 40,41
Importantly, a wide range of epilepsy syndromes, including TLE and EEs are associated with GABAergic dysfunction 42 when GABAergic transmission is corrupted or interneurons are missing. This is particularly the case for interneuronopathies, a group of syndromes with severe epileptic activity and cognitive impairment, where the development, migration, or function of inhibitory interneurons is impaired. 43 Inhibition can also be altered indirectly, when the excitability or fast-spiking properties of interneurons are altered or interneurons lose excitatory inputs. Given the central role of interneurons in the brain machinery, GABAergic alterations in these syndromes would cause rhythmopathies and lead to information processing deficits and cognitive impairments. Of course, abnormal excitation is involved in other epilepsy syndromes, and evidence shows that it also contributes to altered dynamics and rhythm coordination. 44 -47 Therefore, rhythmopathies can arise from multiple failing parts.
Conclusion: Fixing the Timing Belt?
Although the concept of rhythmopathy helps explain the mechanisms involved in cognitive dysfunction, it is important to keep in mind that oscillations, as seizures, are just the signatures of neuronal networks in action. If these networks are abnormal in epilepsy, so are their dynamic signatures. When GABAergic activity fails or abnormal excitatory loops emerge, this leads to abnormal rhythms and/or to the extreme recurrent patterns observed during seizures.
This dynamic systems perspective can offer new therapeutic opportunities that are oriented toward the restoration of neuronal dynamics and rhythms rather than just influencing the excitation–inhibition balance. One possibility is to replace the timing belt of neural circuits, that is, the interneurons. This approach has been quite successful in animal models where embryonic GABAergic progenitor cell grafts prevent seizures and restore cognitive performance. 48,49 Interestingly, grafted progenitors mature and integrate into the hippocampal networks as expected from normally developing interneurons. 50 Other approaches may consist of pharmacologically altering synaptic or intrinsic properties of neurons to restore network dynamics or directly influencing key players in the timing of the brain machinery via optogenetic or chemogenetic methods.
Footnotes
Author's Note
Pierre-Pascal Lenck-Santini is also affiliated with Neurological Sciences, University of Vermont, Burlington, Vermont.
Acknowledgments
The author thankful to Najoua Biba, Sophie Sakkaki, and Gregory Richard who provided the electrophysiological recordings shown in the figure; Rustem Khazipov and Jerome Epzstein for helpful comments on the manuscript.
