Abstract
The study of high frequency oscillations (HFO) in the electroencephalogram (EEG) as biomarkers of epileptic activity has merely focused on their spatial location and relationship to the epileptogenic zone. It has been suggested in several ways that the amount of HFO at a certain point in time may reflect the disease activity or severity. This could be clinically useful in several ways, especially as noninvasive recording of HFO appears feasible. We grouped the potential hypotheses into 4 categories: (1) HFO as biomarkers to predict the development of epilepsy; (2) HFO as biomarkers to predict the occurrence of seizures; (3) HFO as biomarkers linked to the severity of epilepsy, and (4) HFO as biomarkers to evaluate outcome of treatment. We will review the literature that addresses these 4 hypotheses and see to what extent HFO can be used to measure seizure propensity and help determine prognosis of this unpredictable disease.
High Frequency Oscillations and Their Value as Biomarkers of Epilepsy
High frequency oscillations (HFO) have been investigated as new and exciting electroencephalogram (EEG) biomarkers in epilepsy for more than 10 years. The first descriptions and systematic analysis were performed with microelectrodes in animal models or in human mesiotemporal lobe epilepsy. 1 It was natural that early investigation of HFO focused on their value to localize the seizure-onset zone (SOZ) and epileptic tissue. 2,3 There have been many studies analyzing the relationship between the SOZ and HFO occurrence and the correlation between removal of HFO generating tissue and postsurgical seizure freedom. 4 -6 More recent studies cast some doubt on the precision with which HFO can delineate epileptic tissue. 7,8 Up to now HFO are not part of routine clinical investigations in most epilepsy centers.
Alongside these studies, numerous investigations have significantly improved our knowledge about distinct HFO frequencies, the co-occurrence of physiological and epileptic HFO, interactions with epileptic spikes, and underlying mechanisms. These aspects have been highlighted in past review articles 9,10 and will not be the focus of the current overview. In summary, some of these findings explain limitations of the use of HFO in clinical settings, such as the frequency overlap between physiological and epileptic events and our inability to differentiate them.
Another aspect of advances in HFO research is the improved knowledge about technical aspects of HFO recording and analysis. 11 -13 While early investigators doubted that HFO might be visible with standard invasive macroelectrodes, studies have now demonstrated that HFO can be recorded with a wide array of methods including acute intraoperative Eastern Cooperative Oncology Group and scalp EEG. 6,14,15 Modernized automatic detection tools also allow analyzing occurrence and fluctuation of HFO over longer periods of time. 1,16 These improvements, especially the use of scalp EEG, facilitated the investigations which will be reviewed in this article.
While localizing the epileptogenic areas is a key clinical requirement for investigations of focal refractory epilepsy and epilepsy surgery, only a minority of epilepsy patients might profit from this. In many more instances, epileptologists lack diagnostic tools to establish an individualized prognosis, or measure seizure propensity and treatment success. It is well-known that epileptic spikes, as the classical EEG biomarker, can occur in healthy subjects, but their rate does not correlate with seizure frequency and successful treatment does not necessarily result in reduction of spikes. 17 This review will summarize the current knowledge about the ability of HFO to act as an EEG biomarker not to localize but to measure the intensity of epileptic activity at a certain point in time.
We have classified 4 ways in which HFO might be useful in this sense: (1) to predict epileptogenicity in people at risk for epilepsy; (2) to predict seizures; (3) to assess severity of an epilepsy syndrome/pathology; (4) to measure treatment success (see Figure 1). The clinical value of HFO as a specific biomarker in each of the 4 above listed clinical applications would be immense as it could result in tailoring the choice and titration of anti-seizure medication in individual patients. Moreover it would allow identification of individuals at risk who need preventive anti-epileptogenic treatments, prediction of seizure activity, and would aid discrimination of pathology and early prediction of disease severity.
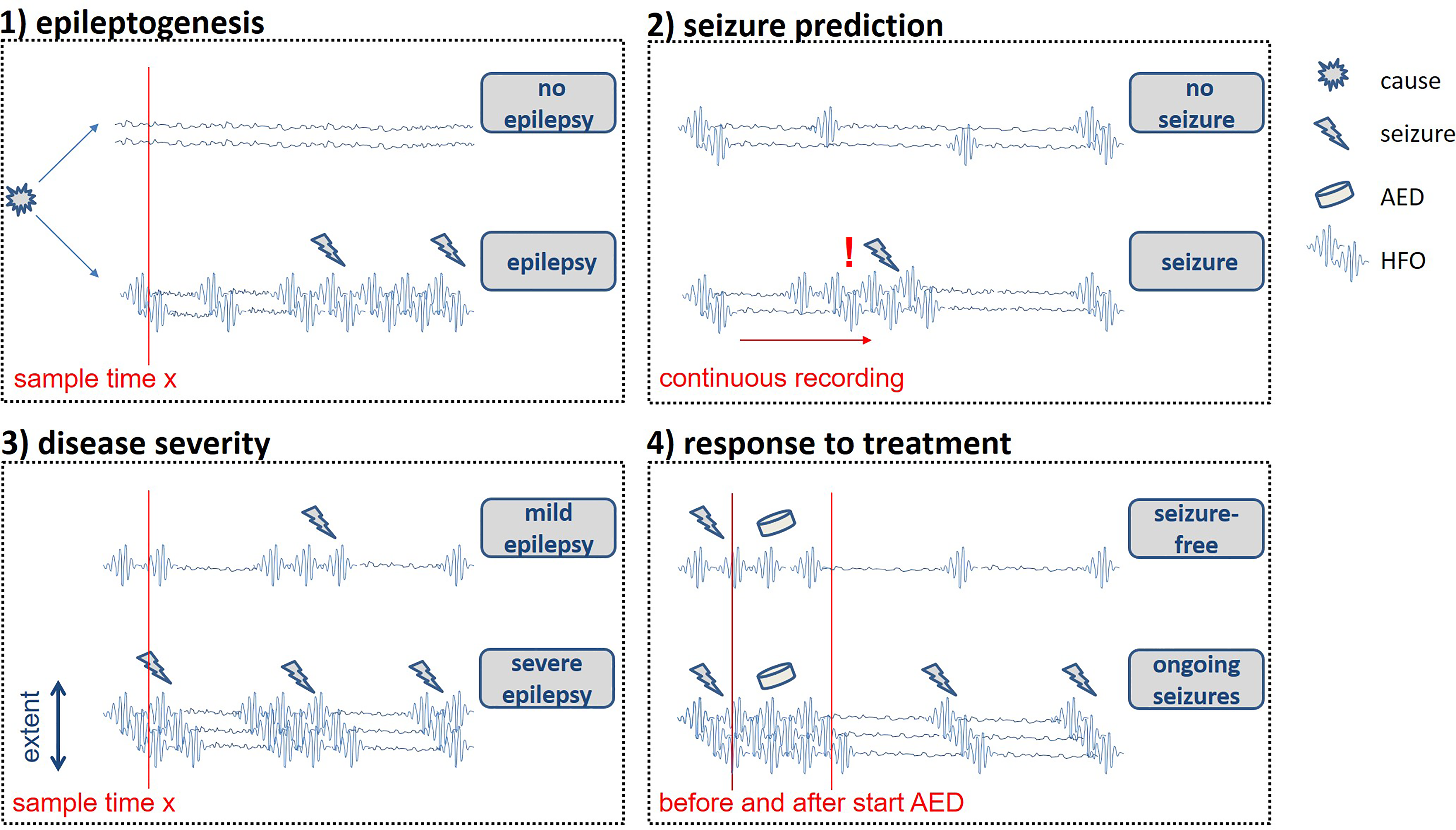
Schematic figure of the scenarios in which HFO can be used as biomarkers for measuring epilepsy activity according to our 4 hypotheses.
We provide 4 tables summarizing the existing literature on the 4 selected topics. As mentioned above, methods and technical knowledge for some of these studies have only become available recently and therefore evidence for some ideas is still limited. Some evidence mostly derives from animal studies and needs to be replicated in patients. It is important to note that modes of recording and the analyzed frequencies are different between studies. This is important to mention as there is currently no evidence that HFO in different frequencies and recorded from different locations have the same underlying mechanisms and thus clinical value. Any discussion about clinical use and applications therefore has to be carefully adjusted to these specifics and they are listed for all studies in the tables.
High Frequency Oscillations to Predict Occurrence of Seizures and Development of Epilepsy in High Risk Populations
According to the current trend of modulating epileptogenesis and preventing epilepsy, measures to identify patients at risk have become more important than ever (Table 1). One of the very first HFO studies in kainate rats suggested that fast ripples visible in the period after status epilepticus only occurred in animals that proceeded to develop chronic epilepsy and not in those that refrained from having further seizures despite suffering from the same first initial hit. 1 More recently a large multicenter effort has been put into predicting epilepsy occurrence after traumatic brain injury (TBI). 18,19 The TBI animal model data suggest that intracranially recorded HFO again occur mainly in animals that develop seizures after the initial insult. 20 Moreover, an analysis that combined sleep spindle and HFO occurrence improved the prediction of spontaneous epilepsy. 21,22 Animals that showed HFO early in the trial and later had a reduced occurrence of sleep spindles were most severely affected by epilepsy. Animal models are incredibly useful in understanding the processes of epileptognesis, especially models in which not all animals develop epilepsy, which seems to mimic the real-life situation of populations at risk quite well. The above data suggest some value of HFO as predictors for epilepsy development.
High Frequency Oscillations (HFO) to Predict Occurrence of Seizures and Development of Epilepsy in High Risk Populations.

Abbreviations: HFO, high frequency oscillations; TBI, traumatic brain injury; TSC, tuberous sclerosis complex.
However, in the translation to clinical use in patients, investigators face several challenges. First, only a small proportion of at risk populations develop epilepsy and therefore the investigated patient cohorts must be large to reach statistical power. Second, the latency between the underlying cause and the first signs of epilepsy can be long. The most commonly investigated patient group is patients with traumatic or acquired brain injury such as stroke. Also neonates with perinatal brain injury have been discussed. Another potential group is patients with a first provoked seizure such as a febrile seizure. Depending on the patient cohort, the assessed risk for developing chronic epilepsy might vary between 2% and 15%. 18,23 Beside the fact that large patient cohorts have to be collected, HFO studies also struggle with the technical requirements necessary to reliably assess HFO. In contrast to animal studies, HFO need to be assessed on scalp EEG and studies combining invasive and scalp EEG suggest that only 0.1% of all HFO are visible on scalp EEG recordings. 24 High frequency oscillations, in contrast to epileptic spikes, do not propagate; thus, if structural damage/lesions are deep and away from the cortical surface, HFO will not be seen. 25 Also EEGs need to be recorded with high sampling frequencies and low artifact level. This might be especially challenging in populations of interest as they are usually taken care of in emergency department or intensive care unit settings. Improved amplifier and analysis settings might overcome these challenges. 26
Cohorts in whom a genetic predisposition results in a likelihood of developing epilepsy in more than 50% of affected individuals, like tuberous sclerosis complex (TSC) or Angelman syndrome, are more promising for biomarker testing. It is already established that the appearance of epileptic spikes heralds the development of epilepsy in TSC, 27 and the same group could also show that HFO are more frequent in patients with TSC than controls. However, it remains to be investigated if HFO have a predictive value for these high-risk patient groups. This is especially exciting as seizure preventing treatments are discussed for this patient group. 28
Another patient group are those with a first unprovoked epileptic seizure, 21% to 45% of whom develop chronic epilepsy within 2 years. 29 Klotz and coworkers looked at the first EEG of a prospective cohort of children presenting to a first seizure clinic. High frequency oscillations in this first EEG were predictive of later diagnosis of epilepsy, in contrast to epileptic spikes which did not distinguish between patients who did or did not develop epilepsy. 30 These findings are promising but more extensive prospective trials are necessary to assess the value of HFO analysis to predict the development of chronic epilepsy.
High Frequency Oscillations and Seizure Prediction
Because of the unpredictable nature of epileptic seizures, the demand for seizure prediction and detection is high and has led to a tremendous effort to achieve this (Table 2). 31 The analysis of EEG at different time points or continuously is necessary to estimate seizure propensity of an individual at a certain point in time. Electroencephalograms of different people show highly individual dynamics and thus a seizure predictor may only work in a way that is adapted on an individual basis. 32 High frequency oscillations may add information to the knowledge obtained from low-frequency sampled EEG. Several studies investigated whether HFO change prior to the occurrence of seizures and found a subtle and variable increase in HFO rates and a change in relative power of spike-related HFO to post-spike slow waves within the last minute preictally. 33 -35 So far no clear evidence has shown that HFO can be used for individual seizure prediction.
High Frequency Oscillations to Predict Seizure Occurrence.
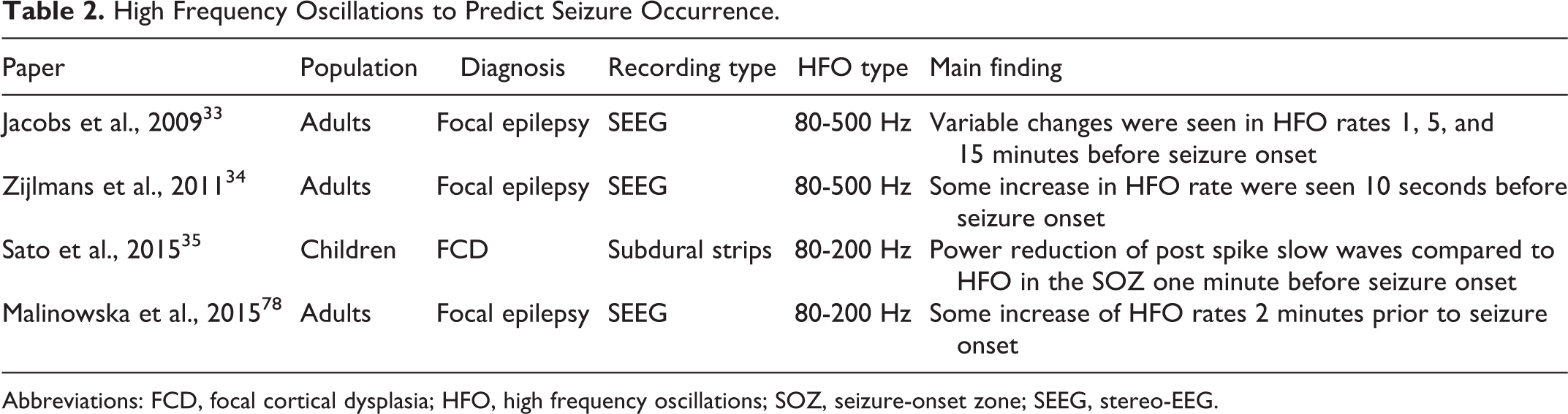
Abbreviations: FCD, focal cortical dysplasia; HFO, high frequency oscillations; SOZ, seizure-onset zone; SEEG, stereo-EEG.
It is clear that HFO activity increases immediately at the time of seizure onset and thus mirrors seizure activity at that point in time. 34 This change depends on the underlying seizure type. 36 -38
A higher number of ictal fast ripples was found in rats with seizures progressing to status epilepticus than in rats with focal seizures. 39 Using intracranial EEG, Schöenberger and coworkers showed that within the same individuals HFO rates were high in seizures that progressed to secondary generalization compared with those that remained focal. 40 This difference between seizure types was visible prior to the clinical occurrence of secondary generalization.
In summary, a preictal increase of HFO is merely subtle and not consistent, thus is not an obvious marker for seizure prediction. However, HFO components may support seizure prediction algorithms. Ictal HFO may be considered for classifying seizure types and predicting seizure severity.
High Frequency Oscillations Reflecting the Severity of an Epilepsy Syndrome
When it comes to assessing epilepsy severity using HFO, there have been 2 different approaches that partially overlap (Table 3). One can measure disease severity by the number of seizures, assuming that this represents a certain static intrinsic severity, like the average seizure count per year. It is also possible to link HFO to severity of the underlying cause as represented by pathology, magnetic resonance imaging (MRI) changes, or epilepsy syndrome. In both cases a one-time measurement of HFO occurrence is correlated with the presumed epilepsy severity. Using HFO in this context can aid differentiating between patients with and without epileptic seizures, or in comparing individuals with the same etiology but varying degrees of epilepsy severity. The main clinical aim would be to separate individuals with no epilepsy, mild, and severe epilepsy by analyzing an EEG at one point in time and adjust treatment decisions accordingly.
High Frequency Oscillations Reflecting the Epilepsy Severity.
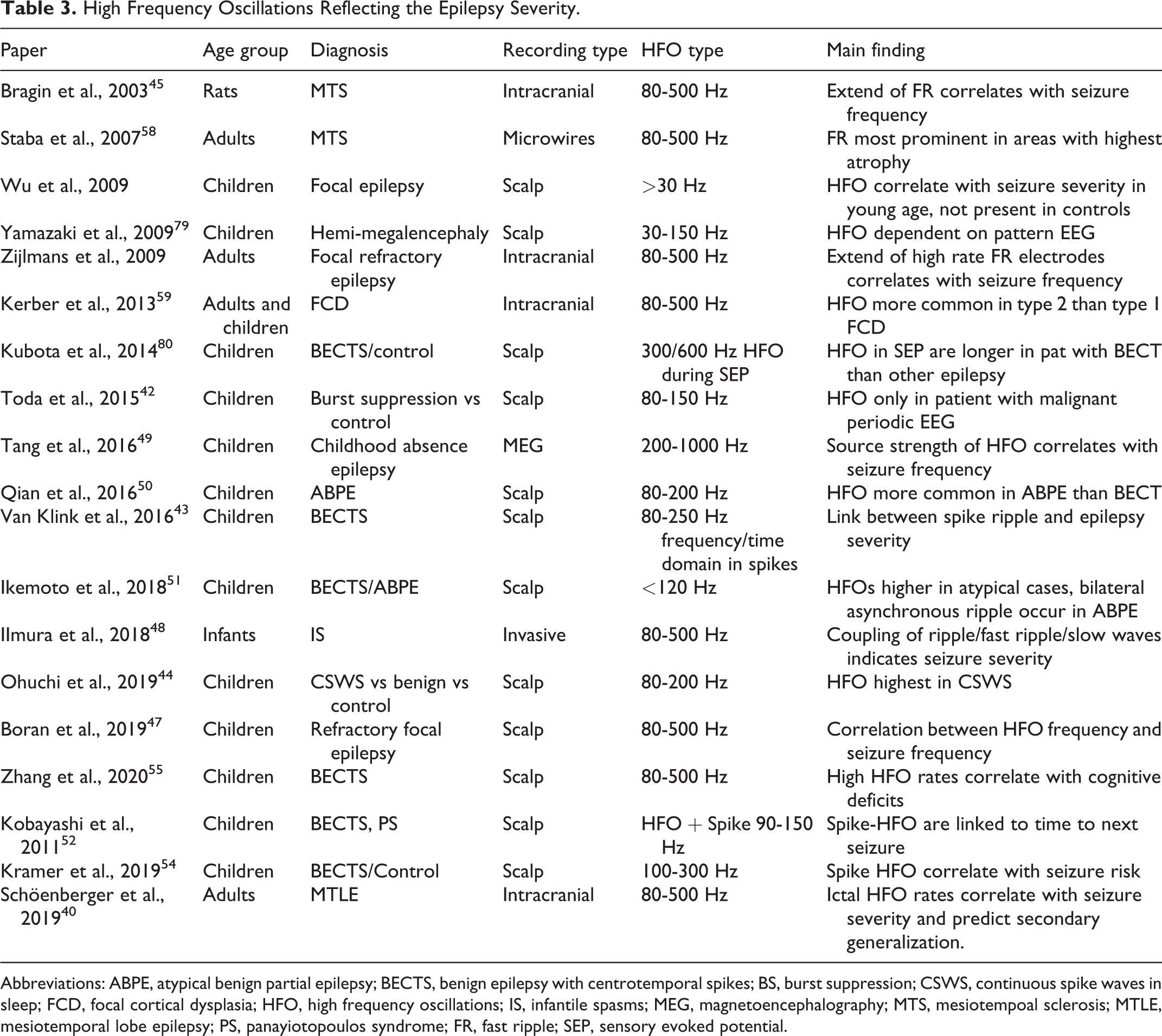
Abbreviations: ABPE, atypical benign partial epilepsy; BECTS, benign epilepsy with centrotemporal spikes; BS, burst suppression; CSWS, continuous spike waves in sleep; FCD, focal cortical dysplasia; HFO, high frequency oscillations; IS, infantile spasms; MEG, magnetoencephalography; MTS, mesiotempoal sclerosis; MTLE, mesiotemporal lobe epilepsy; PS, panayiotopoulos syndrome; FR, fast ripple; SEP, sensory evoked potential.
The most straightforward question to be asked is whether HFO can distinguish between individuals with and without active epilepsy. Several studies showed clear evidence that scalp HFO rates are higher in individuals with epilepsy and active seizures in different age groups and clinical scenarios. Wu and colleagues first investigated paroxysmal fast activity in young children. 41 This analysis included lower frequencies than usually used in HFO studies but clearly showed that the fast activity was linked to epilepsy and did not occur in children with nonepileptic events. In infants, HFO with a frequency of 150 Hz were linked to burst suppression patterns and did not occur in controls even if these had immature EEGs with trace alternant due to their age. 42 A special characteristic of benign syndromes in children is the occurrence of frequent epileptic spikes in individuals who never experience seizures. Two independent studies showed that these seizure-free children had significantly less or no HFO activity compared to children with similar spike frequency and seizures. 43,44 All these studies suggest that HFO, mostly linked to epileptic activity like spikes or bursts, have a high potential to identify children with epilepsy. High frequency oscillations are distinct from epileptic spikes in epileptic syndromes and might be closer linked to seizure activity than the latter.
Several studies have tried to link HFO occurrence to different degrees of epilepsy severity within groups of patients with similar diagnosis and etiology. Clinical relevance of this topic is obvious, as epilepsy severity varies within the same syndrome diagnosis. In epileptic rats with focal seizures, a correlation was found between the extent of electrodes showing fast ripples and the number of seizures. 45 This finding was confirmed in presurgical Intracranial Stereo-EEG in adults with focal epilepsy. 46 In a group of children with refractory epilepsy and scheduled epilepsy surgery, the rate of scalp HFO was linked to the average seizure frequency prior to surgery. 43 After the surgery, a decline in HFO could be noted. 47 In children with infantile spasms, not the HFO rate alone but the degree of coupling between ripples/fast ripples and slow waves indicated seizure severity and frequency. 48 Tang et al showed a relation between the source strength of ictal HFO in magnetoencephalography (MEG) and seizure frequency in childhood absence epilepsy. 49 Several studies analyzed children with varying severities of benign childhood epilepsies and used slightly different methodological approaches. Three studies showed that spike-linked ripples and ripples are more common in atypical benign partial epilepsy than in typical benign epilepsy with centrotemporal spikes (BECTS) 50,51 (Figure 2). Kobayashi and coworkers analyzed the time between the last recorded seizure and the EEG both in patients with BECTS and Panayiotopoulos syndrome. 52 The time to last seizure was significantly shorter in patients with spikes plus HFO than in those spikes without HFO. A prospective study with more children showed similar results. The likelihood of finding ripples coupled to spikes decreased significantly with every month a patient remained seizure free. 53,54 Specificity and positive predictive value for the occurrence of seizures were significantly higher for spikes with ripples than spikes alone. Qian and coworkers also showed that patients with refractory continuous spike-wave during sleep (CSWS) have higher HFO rates than more benign versions of CSWS. 50 Additionally it could be shown that high HFO rates correlate negatively with cognitive function in patients with BECTS. 55
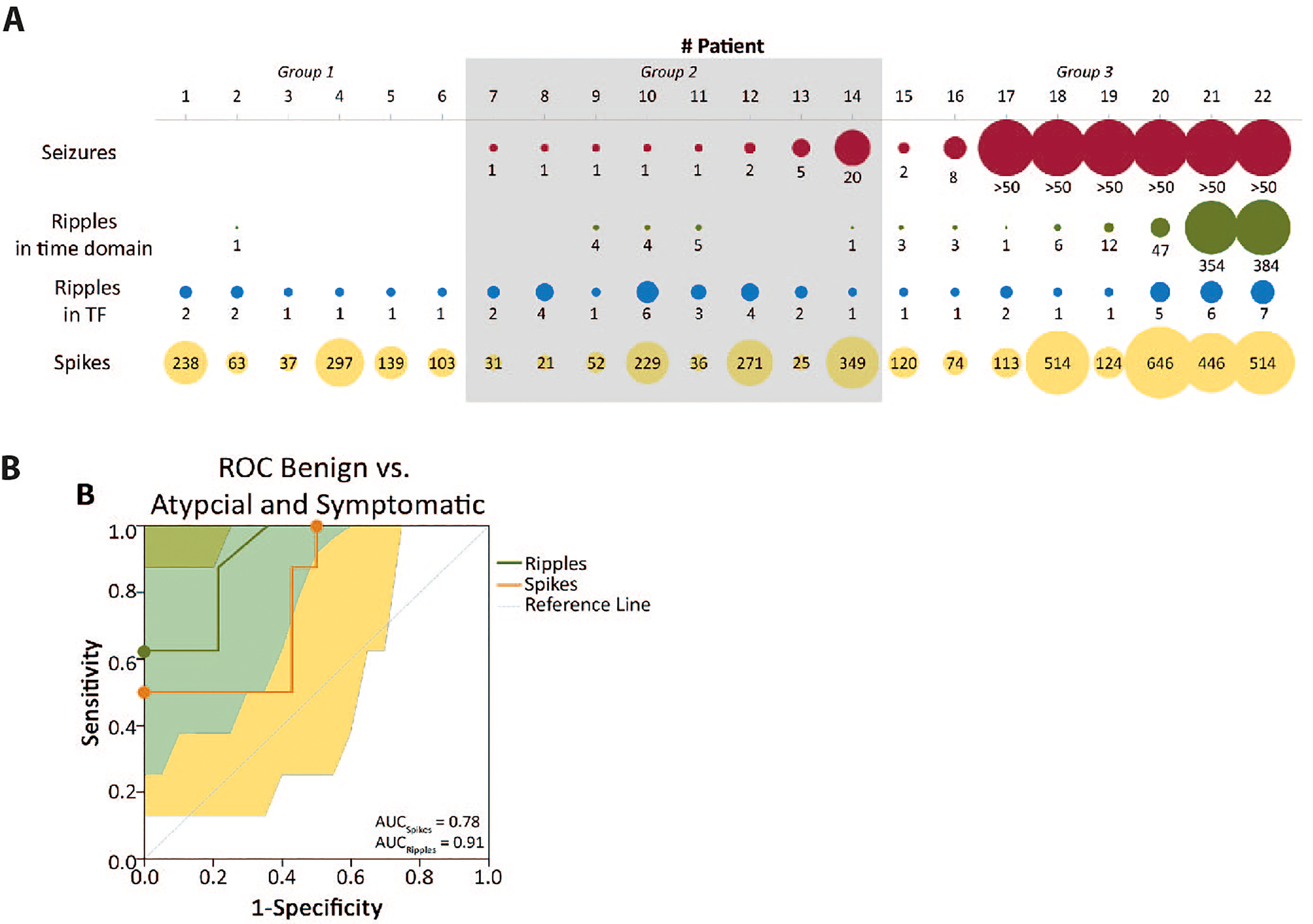
Example for HFO as a predictor of epilepsy severity in patients with BECTS modified after Van Klink and coworkers (2016). A, Overview of the number of seizures, number of ripples in time domain, number of channels with ripples in time-frequency domain (TF), and the number of spikes in individual patients. The size of the circle is proportional to the number of events. For visualization purposes, the circles for Seizures and Ripples in TF were 10-fold larger than for Ripples and Spikes. Groups: (1) Rolandic spikes but no epilepsy, (2) Typical Rolandic epilepsy, (3) Symptomatic epilepsy, (4) Atypical childhood epilepsy with centrotemporal spikes. B, Receiver operating characteristic (ROC) curves for differences between benign courses and atypical BECTS, based on the number of spikes and number of ripples in the EEG. Ripples are a better predictor for both seizures and severe seizures. The highest sensitivity and specificity combinations are indicated with a dot. AUC indicates area under the curve; BECTS, benign epilepsy with centrotemporal spikes.
Several studies have addressed the relationship between HFO occurrence and different structural brain abnormalities. It is generally assumed that HFO are more common in lesional areas that are part of the SOZ than in those that do not show ictal onset. 53 However, there is some evidence that certain brain lesions generate HFO more commonly than other. 56,57 Two studies have addressed the question of whether the degree of structural change as a measure of disease severity correlates with the amount of HFO. In early work using intracranial microelectrodes, Staba and colleagues demonstrated that fast ripples were most common in those parts of the mesiotemporal structures with the most atrophy. 58 Detailed morphometrics of the patient’s MRI and precise localization of the microwires allowed for a correlation within the substructures of the hippocampal formation. In another study, classification of different types of focal cortical dysplasia (FCD) using pathology was correlated with the occurrence of HFO. 59 High frequency oscillations rates were higher in the more dysplastic type 2a& b lesions compared to FCD type 1 a&b. Interestingly, in these patients, overall HFO rates correlated with the overall seizure burden as measured over the last year rather than with the acute seizure burden during the long-term EEG recording.
In summary, the presence of HFO relates to seizure risk and in patients with inter-ictal epileptic spikes more precisely measures this risk than other EEG findings. This directly relates to HFO being a measure of seizure susceptibility at a certain point in time. Moreover, in specific lesions, the HFO rate reflects the epileptogenicity of the lesion and the resulting epilepsy severity.
High Frequency Oscillations and Treatment Response to Anti-Seizure Drugs
Many clinicians might expect that rates of epileptic spikes increase when anti-seizure medication is decreased (Table 4). Data from serial EEGs and longterm EEG monitoring however suggest that the rate of spikes does not show systematic changes with reduction of medication. 17 While a clear EEG response to treatment is expected for some pediatric syndromes like West syndrome, this is not necessarily the case for most patients with epilepsy. It is therefore not a trivial question whether treatment—and especially successful treatment—results in a reduction of HFO occurrence.
High Frequency Oscillations and Treatment Response.a
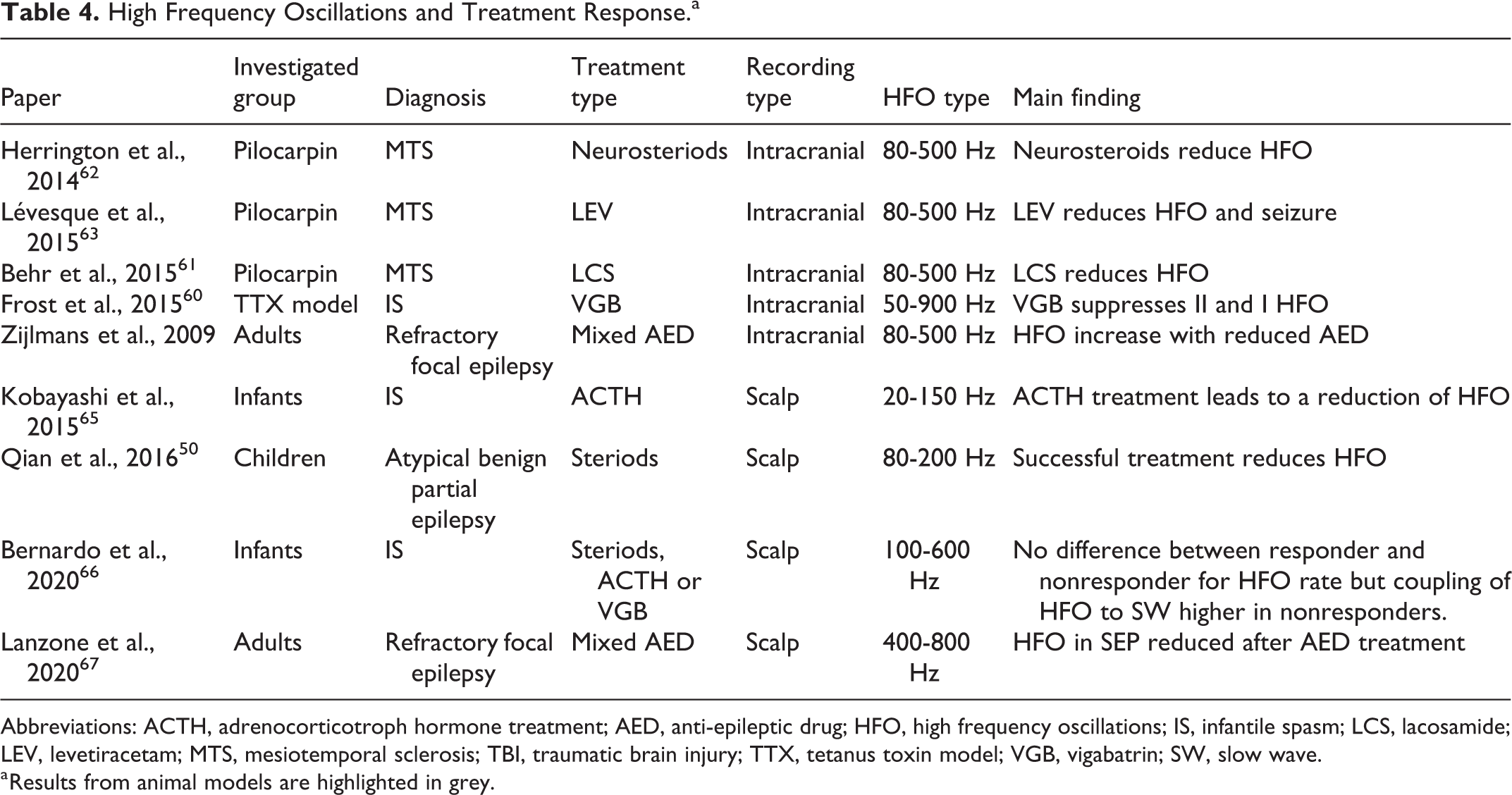
Abbreviations: ACTH, adrenocorticotroph hormone treatment; AED, anti-epileptic drug; HFO, high frequency oscillations; IS, infantile spasm; LCS, lacosamide; LEV, levetiracetam; MTS, mesiotemporal sclerosis; TBI, traumatic brain injury; TTX, tetanus toxin model; VGB, vigabatrin; SW, slow wave.
a Results from animal models are highlighted in grey.
Several studies in rodents suggest that HFO occurrence decreases under treatment with commonly used anti-epileptic drugs (AED). Frost and coworkers used the tetanus toxin model for infantile spams and showed a reduction of HFO following vigabatrin therapy. 60 A mesiotemporal lobe epilepsy model showed a reduction of HFO following treatment with lacosamide and neurosteriods. 61,62 The same group showed that treatment with levetiracetam directly after status epilepticus could reduce the likelihood of spontaneous seizures and that this was reflected in reduced rates of HFO in the treatment group. 63 Thus there is some evidence from animal models that suppressing seizure activity is reflected in reduced HFO occurrence independent of type of epilepsy and treatment. It is however important to note that the above studies often had much higher anatomical resolution than is possible in humans and found different HFO responses in distinct brain regions, such as CA3 and subiculum. These kinds of distinct and focal HFO changes might not be recorded and assessed easily in humans.
The first study to assess this question was performed using intracranial EEG in patients with focal refractory epilepsy. 64 For this reason, the analyzed time period was limited to a few days. However, this study clearly showed that the reduction of AED resulted in an increase of HFO while spike rates remained unchanged. This observation did not depend on whether patients also developed more seizures after AED reduction. Later studies profited from the discovery of HFO on the scalp and are limited to pediatric populations. Kobayashi and coworkers analyzed high frequency changes and not distinct HFO in a population with hypsarrhythmia and infantile spasms. 65 It could clearly be shown that HFO activity was reduced after adrenocorticotroph hormone treatment (ACTH) treatment. This reduction was seen in all patients and no analysis was performed to differentiate between responders and nonresponders. Bernardo and coworkers replicated this finding of reduced HFO during treatment of infantile spasms with treatment of steroids, ACTH, or vigabatrin. 66 However, they could not find a difference in HFO reduction between treatment responders and those who failed treatment. This study also investigated coupling of HFO with slow waves and found that this coupling is stronger in nonresponders compared to treatment responders, in whom the coupling decreases with successful treatment. It would be interesting to know whether these changes in coupling or also the HFO occurrence correlate with long-term seizure and cognitive outcome, but no data for these questions is currently available. One other study looking at patients with atypical benign partial epilepsy showed a clear difference in HFO reduction between patients who improved under treatment with steroids and those who did not. 50 The use of AED lowers physiological evoked HFO associated with sensory evoked potentials in the surface EEG, which was suggested to represent a reduction in overall excitability of the brain. 67
In summary, it is interesting to see that all studies suggest some relationship between changes in antiepileptic treatment and HFO occurrence and these changes seem to be unspecific in regard to type of treatment. At this point in time, it is however unclear whether these HFO changes can be used to tailor treatment or predict treatment success. Additional studies are needed to confirm the effect of all different types of drug mechanisms on HFO occurrence.
Challenges in the Use of HFO as a Clinical Tool
Most of the above studies are retrospective analyses and many are based on group analysis to accomplish statistical significance. For an EEG biomarker, like HFO, several hurdles must be cleared to allow reliable use that will change clinical practice.
First, it must be proven that HFO reflect epileptic activity in an individual’s brain better than chance and that clinical conclusions derived from this additional information are beneficial for the patient. Current evidence level is far from providing this certainty and prospective studies and, as a next step, treatment trials tailored by HFO are necessary.
Second, the technical and methodological requirements for HFO analysis must be defined, accessible, reliable, and affordable. While the good news is that most companies that sell amplifiers have moved toward high sampling rates, which allow HFO recording, and software providers adapted their EEG reviewing software, many other technical challenges are still unsolved. Several centers have developed automatic detectors for HFO recorded intracranially 68 -70 ; the same is true for platforms that display HFO rates across brain regions. 71 At this point, the application of these techniques still requires more technical support than most centers could afford. For the topics of the current review, however, scalp HFO are much more important. Some studies published scalp HFO or scalp Spike-HFO detectors, but many of them still need visual verification and show low sensitivity and specificity. Scalp HFO detection is challenging, as the HFO events are frequently of lower amplitude and artifact levels on the contrary might be high. Topics covered by this review aim to use HFO in a routine clinical setting and under these circumstances noise levels, impedances, data quality, and as a result detector performance might largely vary. Even with perfect EEG quality, some question marks about scalp HFO remain. Some studies suggest that high-density EEG coverage improves the recording 24,72 ; others suggest low noise amplifiers or special electrode types are needed for scalp HFO identification. 73,74 Especially for the use of HFO as a tool to detect patients who are developing epilepsy, one must bear in mind that physiological HFO can not only be seen intracranially but also on the scalp. 75
The third and final step will be training epileptologists on using HFO, finding the correct interpretation and drawing best practice conclusion. Data for this clinical training can only come from the necessary clinical trials mentioned above.
What Comes Next!
The new generation of EEG amplifiers will increase the circle of researchers and clinicians who can record EEG data with a high sampling rate that enables HFO analysis. If data can be collected in a routine clinical setting, acquiring the missing EEG data for the questions discussed above will be facilitated. A trustable and uniform method of HFO recording and analysis is needed to pull together HFO information from different clinics. 76 High frequency oscillations per se may not be the optimal biomarker, but using HFO information in combination with information on spikes, artefacts, and several other EEG derivatives, like DC-shifts, cross-frequency coupling, network analysis, and other signal parameters that can be derived from (surface) EEGs, may together lead to definition of optimal biomarkers. Artificial intelligence may find optimal solutions based on many surface EEGs. Combining surface EEG with transcranial magnetic stimulation, MEG, or other modalities like functional MRI may further lead to gain of information needed to enable steps toward early diagnoses, disease prediction, and treatment evaluation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JJ was supported by German Research Foundation Grant JA 1725/4-1, MZ was supported by ERC starting Grant 803880.
