Abstract
In epilepsy research, the analysis of rodent electroencephalogram (EEG) has been performed by many laboratories with a variety of techniques. However, the acquisition and basic analysis of rodent EEG have only recently been standardized. Since a number of software platforms and increased computational power have become widely available, advanced rodent EEG analysis is now more accessible to investigators working with rodent models of epilepsy. In this review, the approach to the analysis of rodent EEG will be examined, including the evaluation of both epileptiform and background activity. Major caveats when employing these analyses, cellular and circuit-level correlates of EEG changes, and important differences between rodent and human EEG are also reviewed. The currently available techniques show great promise in gaining a deeper understanding of the complexities hidden within the EEG in rodent models of epilepsy.
Introduction
The electroencephalogram (EEG) is a direct window into electrical activity generated by the brain and an invaluable diagnostic tool for the clinical diagnosis of epilepsy. In many respects, rodent and human EEGs are very similar, including the relationship between power (amplitude) and frequency (P = 1/f), frequency shifts with state change, and various forms of epileptiform activity resulting from a plethora of etiologies including genetic disorders and focal lesions. In this review, the approach to the analysis of rodent EEG will be examined, with a focus on (1) advanced techniques that have more recently become accessible to those who may not have a background in signal processing, (2) caveats to keep in mind when these techniques are employed, and (3) key differences between rodent and human EEGs.
Before analyzing EEG data, the integrity of the recording must first be verified. The American Epilepsy Society (AES) and International League Against Epilepsy (ILAE) Translational Task Force have published methodological standards for adult and immature rodents 1,2 as well as basic guidelines for data acquisition and software-based analysis 3 to help standardize rodent preclinical EEG experiments. Of note, acquisition and analysis of EEG in neonatal rodents, though technically challenging, has been performed in rats for decades 4 and is now more commonly being performed in mice. 5 These AES/ILAE guidelines discuss essential issues that contribute to obtaining high-quality rodent EEG, including use of screws versus wire electrodes, tethered versus wireless approach, epidural versus subdural versus intracranial targeting, reference and ground placement, inclusion of an EMG electrode (recommended for sleep staging), sampling rate (at least 2 kHz is recommended), and electrode size and material.
Once the data are acquired, which software should be used to analyze the data? While there are a number of commercial software options which are often bundled into a package with hardware, 3 there are also freely available, peer-reviewed software platforms that offer a wide range of modern analytical tools. For those with programming skills, these can be customized further for more specific experimental applications. Within MATLAB (available through most academic institutions), there are graphic user interfaces (GUIs) including EEGLab, 6 Brainstorm, 7 and FieldTrip 8 with additional integrated toolboxes for statistical analyses. 9 These GUIs are user-friendly and run within the MATLAB environment but otherwise require no additional programming skills. Python has a similar GUI called PyEEG. 10 Electroencephalogram obtained by most systems can be exported into the European Data File (.edf) format, which is universally recognized by these GUIs.
Once the EEG has been loaded and visualized, it should first be inspected for quality and subsequently preprocessed using a standard algorithm. The main goal of this step is to remove artifact. This can be done manually by simply removing time periods where the EEG shows significant artifact based on visual inspection. Alternatively, this process can be standardized by processes adapted from artifact removal in human scalp EEG and reviewed elsewhere, 11 including independent component analysis, 12 fully automated unsupervised programs, 13 or machine learning techniques. 14 After artifact has been sufficiently removed, analysis can proceed to evaluate both the epileptiform (including ictal activity and interictal sharp waves or spikes) and nonepileptiform (background) EEG. The modern approach to both epileptiform and nonepileptiform EEG analysis includes a myriad of commonly used techniques (Table 1).
Examples of Common EEG Analysis Techniques.a
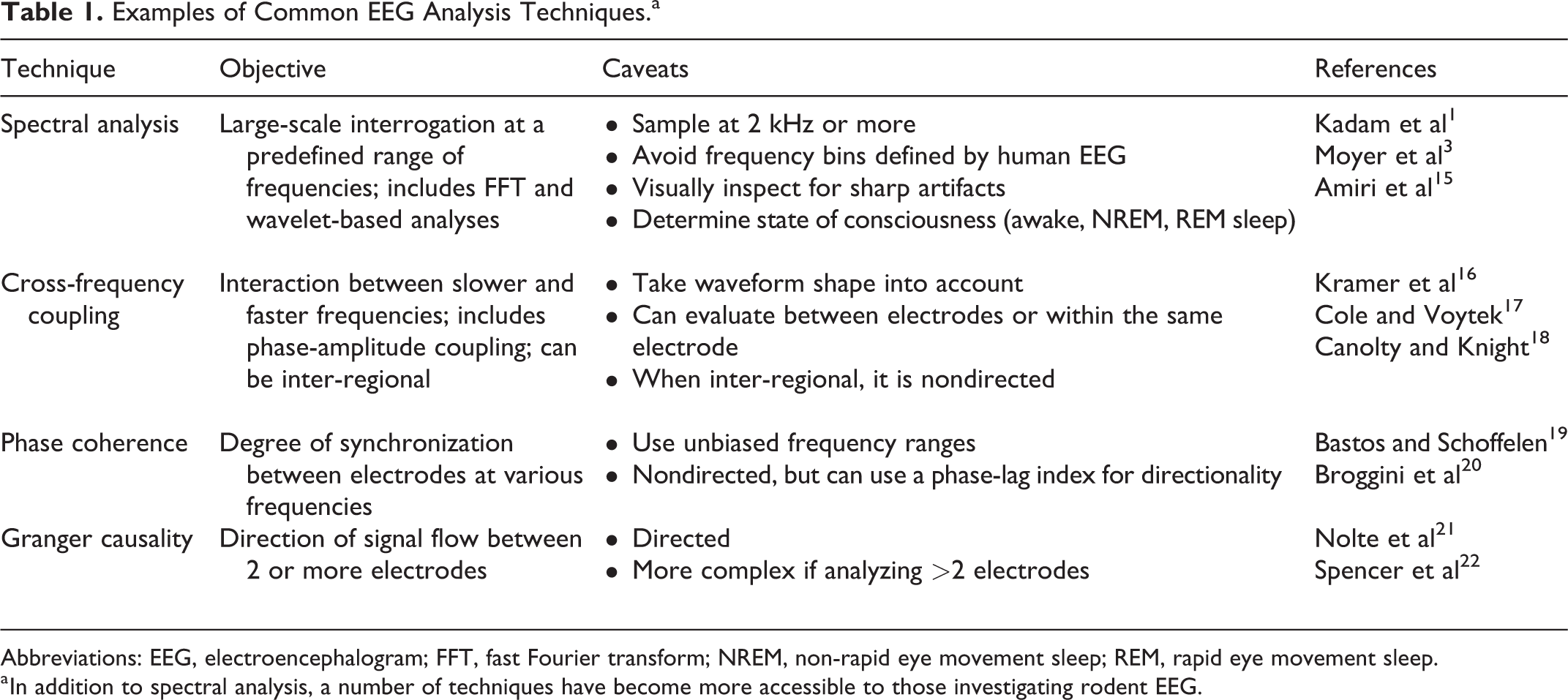
Abbreviations: EEG, electroencephalogram; FFT, fast Fourier transform; NREM, non-rapid eye movement sleep; REM, rapid eye movement sleep.
a In addition to spectral analysis, a number of techniques have become more accessible to those investigating rodent EEG.
Epileptiform EEG analysis must be tailored to the model that is being experimentally tested. For example, the electroclinical expression of an absence seizure is characterized by high-amplitude generalized spike-and-wave discharges associated with behavioral arrest. 23 In contrast, a focal-onset seizure may arise in one region with low-voltage fast activity and then secondarily generalize, evolving clinically in a graded fashion as defined by behavioral metrics such as the Racine scale. 24 Epileptiform activity can be identified using visual inspection and manual scoring, but with longer periods of recording and low seizure frequency, this can be a laborious and time-intensive task. Therefore, a supervised or unsupervised machine learning approach can be employed. 25 These approaches can be applied to counting seizures, spikes, and sharp waves. While promising, a user-friendly, plug-and-play seizure detection algorithm remains a work in progress. With any algorithm, the burden of proof is on the investigator to ensure that the analysis has a high sensitivity and specificity compared to the gold standard of visual inspection.
The most straightforward technique to quantitatively interrogate epileptiform EEG is spectral analysis. A modern unbiased approach considers the spectrum of frequencies from ultradian and multidian oscillations (<1 Hz) to high-frequency oscillations (HFOs, >80 Hz), with a focus on the range that is applicable to the time period and hypothesis being tested. 26,27 Spectral analysis is commonly performed using a fast Fourier transform (FFT) or a wavelet-based analysis. The FFT provides a global snapshot of power across frequencies, but its ability to compute power over short time window is limited. In contrast, a wavelet-based analysis is most appropriate for evaluating changes in a wide range of frequencies over time, but its main limitation is that it is more computationally taxing. Importantly, spectral analyses can be confounded by any sharp components or waveforms that are not symmetric in their rising and falling phases. 16,17 For example, interictal spike-wave discharges in rodent models of temporal lobe epilepsy can occur with or without associated HFOs. 28 Due to the sharp nature of a spike, spectral analysis can inadvertently transform a spike into HFOs even when they do not exist. 15 Therefore, visual inspection to confirm the presence of true HFOs is recommended. With these considerations in mind, rodent models of temporal lobe epilepsy have played an important role in understanding the relationship between HFOs and the epileptiform EEG. For example, ripples (80-200 Hz) have been associated with seizures with low-voltage fast onset and fast ripples (250-500 Hz) have been associated with seizures with hypersynchronous onset, which align with specific underlying pathologies in each seizure type. 28 More advanced ictal EEG analyses such as Granger causality (see below) are now also accessible in open source software such as EEGLab and Brainstorm and can be used to examine the temporal spread of seizures on a time scale of milliseconds. 29,30 Absence seizures of childhood absence epilepsy (CAE), for example, have long been characterized as having a “generalized onset” because the onset appears diffusely throughout the EEG. Granger causality measures in a rodent model of absence epilepsy, however, have shown that these seizures most commonly originate in the perioral region of the somatosensory cortex and rapidly spread to other regions in the brain. 30,31 These findings have led the ILAE to update the electrographic description of absence seizures as “originating at some point within, and rapidly engaging, bilaterally distributed networks.” 32
Just as “epilepsy is more than just seizures,” the background EEG contains a rich array of data beyond the epileptiform activity associated with seizures. Spectral analysis provides an important starting point, but important findings can be missed if analyses are restricted to predefined frequency bins across a limited range of frequencies (eg, 1-30 Hz divided somewhat arbitrarily into the delta, theta, alpha, and beta bands defined by human EEG). At the higher end of the frequency spectrum, for example, HFO analysis in rodent models of temporal lobe epilepsy have revealed that fast ripples (250-500 Hz) are a potential biomarker for epileptogenesis, frequently appearing prior to the development of seizures. 28 In addition to changes in baseline spectral profiles, response of the background EEG to antiseizure drugs can also be quantified. In 2 models of absence epilepsy, changes in normalized gamma power inversely correlated with response to various antiepileptic drugs. 33 Of note, since EEG power has a nonlinear relationship with frequency (P = 1/f), differences within and between groups are more readily detected after a log-transformation of the raw data. 34
Using the GUIs mentioned above, more advanced background EEG analyses are accessible without needing a great deal of computational expertise. One such analysis is cross-frequency coupling (CFC), the degree to which slower frequencies interact with faster frequencies. The most commonly studied type of CFC is phase-amplitude coupling (PAC), where the phase of slower frequencies is coupled to the amplitude of faster frequencies. 18,35 Gamma amplitude coupled to theta phase in the hippocampus, for example, reflects normal memory processes in both rodents and humans. 36 -38 Consistent with these findings, aberrant PAC in the hippocampus and neocortex in rodent models of mesial temporal lobe epilepsy (MTLE) has been linked to poor performance on memory tasks. 39,40 Theta–gamma and theta HFO coupling have also been described in the parietal cortex of wild-type mice, with a strong dependence on sleep state. 41 Aberrant parietal PAC has been identified in the awake background of mouse models of absence epilepsy, even after seizures have been pharmacologically treated. 42 Whether this aberrant PAC also correlates with abnormal behavior in rodent models of non-MTLE remains to be seen.
These techniques can be expanded further to examine the functional connectivity between regions, divided broadly into nondirected or directed analyses. 19 Nondirected analyses are more computationally straightforward, but directed analyses have the advantage of revealing the direction of information flow. One type of nondirected analysis that can be applied to 2 different electrodes is inter-regional PAC, which evaluates the coupling between the phase of slow frequencies in one electrode and the amplitude of fast frequencies in another electrode. Strong coupling indicates that one region may be strongly influencing the activity in another region, but directionality of that coupling should not be assumed. Another nondirected interelectrode measure is “phase coherence,” which is the degree to which 2 regions are synchronized in the frequency domain. Greater phase coherence indicates a greater degree of functional connectivity between the 2 leads but, again, does not by itself establish the direction of information flow. For example, in a rat model of temporal lobe epilepsy, nondirected analyses of the 30 seconds prior to seizure onset revealed strong inter-regional PAC between the phase of hippocampal theta oscillations and the amplitude of medial prefrontal cortex (mPFC) gamma oscillations as well as strong theta phase coherence between these 2 regions. 20 However, in order to infer directionality between the 2 regions, Granger causality was required to show that hippocampal theta phase directed mPFC gamma activity and not vice versa. 20 Directionality can also be inferred using a “phase slope index”, which quantifies the consistency of the phase lag between 2 sites that have phase synchrony. 21 These functional connectivity measures are relatively straightforward examples of how signals from distinct regions can be analyzed, but more refined approaches are also available. 19,22
What do these changes to the background EEG represent? As the cell type-specific correlates of EEG activity become better elucidated, changes in specific frequency ranges may shed light onto underlying cellular and circuit-level defects. 43 For example, gamma power is associated with the function of fast-spiking, parvalbumin-expressing interneurons 44 and theta power with the function of both parvalbumin- and somatostatin-expressing interneurons. 45 There is also emerging evidence supporting the role of both somatostatin- and parvalbumin-expressing interneurons in generating hippocampal theta–gamma PAC. 46 While dysfunction in circuits involving these cell types may be expected to alter the power of specific frequency ranges, developmental and compensatory mechanisms should also be taken into consideration. In addition, computational modeling has predicted that changes in connectivity between different cell types should alter the expression of CFC, 47 but further work is necessary to dissect these interactions in rodent models of epilepsy.
When attempting to translate findings from bench to bedside, there are key differences between rodent and human EEG that should be recognized. First, rodent “theta” (6-10 Hz) is an overlapping but overall faster frequency range than human theta (4-7 Hz). 48 Similarly, rodent models of absence epilepsy are most frequently characterized by generalized spike-and-wave discharges occurring at a rate of 5 to 9 Hz, much faster than the 3 Hz discharges characteristic of patients with CAE. 23 In addition, theta rhythms in the hippocampus are nearly continuous in rodents but more intermittent during an exploration task in humans. 48 The EEG in sleep also has some interspecies differences, with sleep largely concentrated during daylight hours and in a more fragmented manner in rodents than in humans. In addition, human sleep spindles in NREM sleep are divided into fast centroparietal (12-15 Hz) and slower frontal (9-12 Hz) spindles. Rats have an overall faster frequency (∼21 Hz posteriorly and ∼16 Hz anteriorly) than in humans, 49 while mice have 9 to 12 Hz spindles globally. 50 As high-density EEG recording in rodents become widely utilized, further differences between rodent and human EEG are likely to be illuminated. 51,52
In sum, with the increased accessibility of rodent EEG acquisition and analysis, researchers are approaching an exciting frontier in epilepsy and disorders comorbid with epileptic phenotypes. In 2014, the National Institute for Neurologic Disorders and Stroke established the latest benchmarks for epilepsy research, including understanding the causes of epilepsy and epilepsy-related neurologic, psychiatric, and somatic conditions (area 1). 53 The EEG biomarkers have shown significant promise toward advancing this goal. With the ongoing standardization of parameters set forth by the ILAE/AES Translational Task Force, epilepsy researchers are universally well positioned to make unprecedented progress in uncovering the deepest mysteries buried within the rodent EEG.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health NINDS K08 NS096029 and the NARSAD Young Investigator Grant #27523.
