Abstract
Introduction:
The safe and compliant transportation of infectious substances and biological specimens is critically essential for public health, scientific research, and disease diagnostics. With its global implications, this study aims to evaluate the International Transportation Association (IATA) regulations for shipping infectious substances and biological specimens in Pakistan. It explores the current level of awareness of IATA regulations and identifies existing gaps and challenges.
Objectives:
Develop and implement training modules that comply with IATA regulations and the Global Biorisk Management Curriculum (GBRMC).
Methods:
To promote the adoption of IATA regulations in Pakistan, the Association for Biorisk Management and Health Security Partners collaborated with experts, including certified IATA developed a project in partnership with experts and certified IATA professionals in Pakistan to start a series of workshops in Pakistan focused on IATA regulations. The project comprised two initial phases: a survey to assess the gaps in the existing regulations and their implementation for shipping infectious substances and a series of workshops based on the training of trainers concept. The project was spread over 2 years.
Results:
Only 1.29% of institutions surveyed offer training in this domain. There is a pressing need to enhance the IATA and local regulations governing the shipment of infectious substances from laboratories. Drawing on international regulations and experiences, we can bolster Pakistan’s capacity to manage the shipping of infectious agents based on national requirements and priorities.
Keywords
Introduction
The safe and secure transport of infectious substances and biological specimens is essential for global public health, scientific research, and disease diagnostics. The International Air Transport Association (IATA) has strict regulations for shipping these materials. 1 Observing these guidelines is vital for reducing the potential risks of transporting hazardous biological substances by air across international borders; the principles can also be utilized to improve the safety of terrestrial transportation, including domestically. However, training effectiveness in IATA regulations may vary based on the different characteristics and requirements of the groups involved in the shipping process. There is an urgent and growing need for customized training programs to handle infectious substances and biological specimens in Pakistan. Given the inefficient training provided, this safety and security aspect must be emphasized more extensively, encompassing life and health sciences.
These regulations apply to any material capable of causing an unreasonable risk to health, safety, and property during transport. The classification system encompasses category A (high-risk) and category B (lower risk), which govern the stipulated proper shipping names and identification numbers for each category. Emphasizing the importance of triple-packaging patient specimens and compliance with the requisite labeling is crucial. In the Exempt specimens’ category, the triple packaging only with the proper shipping name as “Exempt Specimen” is significant in qualifying for an exemption from other category A and category B regulatory requirements. An infectious substance is regulated as hazardous under the U.S. Department of Transportation (DOT) Hazardous Materials Regulations (HMR), 49 CFR Parts 171–180. Pakistan has not yet developed any specific rules in this context, but the international laws, including those of the U.S. DOT and IATA regulations, are still followed. The HMR applies to any material DOT determines is capable of posing an unreasonable risk to health, safety, and property when transported in commerce. An infectious substance must conform to all applicable HMR requirements when transported or offered for transportation by air, highway, rail, or water. 2 The overarching principles governing the international air transport of dangerous goods are delineated in Annex 18 to the Convention on International Civil Aviation Organization (ICAO) to the Safe and Secure Transport of Dangerous Goods by Air. Adherence to specific principles is pivotal in ensuring the safe carriage of dangerous goods via air transport. These principles have informed the development of the Technical Instructions to facilitate transport while upholding safety standards, thus enabling the transportation of hazardous goods without jeopardizing the aircraft’s or its occupants’ safety, provided all mandated prerequisites are met3,4
This study aims to evaluate the impact of tailored training on professionals’ understanding and application of IATA regulations for shipping infectious substances and biological specimens in their institutes in Pakistan. It investigates the current status of implementing IATA regulations in Pakistan and identifies gaps and challenges. It also proposes a specialized approach to improving the effectiveness of such training. This study aims to significantly enhance compliance with international standards and ensure safe and efficient transportation of biological materials. This approach is significant in improving public health and safety but also substantially contributes to global health security efforts, research progress, and the country’s overall health infrastructure, highlighting the colossal implications of this work. 5
Training Requirements
Individuals must undergo training before shipping functions such as classifying, identifying, packing, marking, labeling, or documentation. Furthermore, this training must be renewed every 2 years according to IATA regulations and 3 years with U.S. DOT regulations. However, the IATA time frame for the training schedule is followed internationally. Training dates should be specified by month and year, and any regulatory changes affecting the individual’s duties require additional training. Maintenance of comprehensive training records, including a course certificate, is crucial. It should include the individual’s name, the month of the most recent training completion, a description or reference to the training materials used, the name and address of the training organization, and proof of completed tests. 1 Employers are responsible for ascertaining that their employees are competent and proficient in carrying out functions related to shipping hazardous goods. This responsibility is not just a formality but a demonstration of the employer’s unwavering commitment to their employee’s safety and well-being, and it underscores each employee’s integral role in safely shipping dangerous materials. 1
WHO offers practical guidance for compliance with international regulations governing the transportation of infectious substances across all modes of transport. This guidance, which includes the revisions effective from January 1, 2021, offers comprehensive information on the classification, identification, packaging, marking, labeling, documentation, and refrigeration of infectious substances for transportation. By following this guidance, the secure delivery of infectious substances can be ensured.6,7 The strategic sequencing and optimization of service routes inside the institute and outside are pivotal in maximizing productivity while curtailing costs. Methodical route sequencing reduces costs and fosters a dependable, secure, and compliant work environment for teams and partners transporting hazardous substances at local levels. 8
The Hazardous Materials Regulations under the designated authority U.S. DOT, encompassing parts 171 through 180, delineate the applicability of packaging qualified for transporting hazardous materials in commerce and the pre-transportation and transportation functions. 9 The need for heightened awareness of IATA regulations and guidelines in Pakistan, particularly in comparison to other developed nations, underscores the importance of aligning with the ICAO charter and UN directives. This biphasic project for developing the Training of Trainers program for IATA training on its rules and its implementation at the national level in life and health sciences institutes phase where it will stand alone as contributing to the One Health objective. The alignment of Pakistan’s practices with IATA, ICAO, and UN regulations is not just a matter of regulatory compliance but a vital step toward ensuring public safety, environmental protection, and national security. The sensitivity of this issue lies in the high stakes involved, making it imperative for Pakistan to prioritize education, training, and systemic reforms to bridge the existing gaps. This will enhance safety and strengthen Pakistan’s position in the global aviation and trade community. This alignment is not just a suggestion but a necessity for safely transporting dangerous goods. This should be done while concurrently addressing the lacunae prevalent in the public and private systems dealing with this issue. 10
Methods
This project was initiated in October 2023 by a team of experts from the Association for Biorisk Management (ABM) and Health Security Partners (HSP), with input from International Air Transport Association (IATA) certified professionals on its regulations and guidelines. This IATA training on safe and secure transportation of infectious substances is recommended to be regularly included in the protocols of Pakistan’s life, health, and veterinary sciences institutes, which conduct research and diagnostics work.
Phase I
The study design was cross-sectional, descriptive, and observational. It involved quantitative and qualitative data collection methods to assess respondents’ knowledge and practices regarding transporting infectious substances and adherence to IATA Dangerous Goods Regulations. A survey was created and conducted to assess the effectiveness of the Global Biorisk Management Curriculum (GBRMC) from Sandia National Laboratory NM USA-based training program in identifying knowledge gaps in the ABM Cohort and efficiently implementing the training program. It was designed to gather quantitative and qualitative data from the respondents, focusing on issues and practices related to transporting Infectious Substances and compliance with IATA Dangerous Goods Regulations.
The survey, developed on Google Forms, addressed the main themes of IATA regulations. It comprised eight sections covering demographic information, training and awareness, risk assessment procedures, packaging and labeling, transportation logistics, documentation and record keeping, collaboration and communication, and continuous improvement. The questionnaire comprised 22 statements covering these topics. One hundred 80 professionals from the Association of Biorisk Management and its associated groups, representing the provinces of Sindh, Punjab, Balochistan, and KP in Pakistan, responded to the survey.
Respondent feedback was analyzed and evaluated using Microsoft Excel and IBM-SPSS version 23. Different variables were compared using descriptive statistics and hypothesis testing, including the chi-square, and presented in tables and graphs. The level of significance was determined using a 95% confidence interval.
Based on the survey results and the lack of regulations and information, a customized training program was created for different groups, including veterinary and agriculture science professionals, laboratory personnel, shipping staff, and animal healthcare providers affiliated with the ABM. This was the first step in developing a structured program for shipping infectious substances per IATA regulations after performing a risk assessment and need assessment to raise awareness among biosafety professionals about this important but overlooked specialty in Pakistan.
The program was designed to meet the specific needs of various groups, including veterinary and agriculture science professionals, laboratory personnel, shipping staff, and animal healthcare providers affiliated with the ABM.
The training content was carefully crafted to cover all relevant sections of IATA regulations and best practices for shipping infectious substances and biological specimens.
The GBRMC was closely followed.5,11 The IATA-certified facilitator was identified with the proper credentials in Biosafety and Biosecurity, including RBP, CBSP ABSA International (ABSA), and IFBA certifications, which were essential requirements for the trainer to conduct training.
A 3-day workshop was organized for the targeted cohort of 20 professionals, selected after assessing their eligibility and the extent to which their roles and responsibilities were involved in shipping infectious materials. The workshop represented ABM with the support of HSP in compliance with Section 1.5 of the IATA regulations for transporting Infectious substances. The third day was allocated for biosafety training to complete the process per IATA recommendations. The biosafety training was focused on best practices for handling infectious substances with criteria of handling, processing, and transporting high-consequence pathogens.
On day 2, a tailored multiple-choice test with 50 questions was developed in compliance with IATA guidelines and the GBRMC curriculum to test the participant’s ability to manage different categories of infectious substances.1,5 The pass mark for the test was 85%; individuals who achieved this score were awarded a certificate. An additional pre- and posttest dedicated to the third day of biosafety and biosecurity training was developed and implemented to measure participants’ improvements in knowledge and comprehension levels in this respect.
12
Participants feedback form created on the Likert scale and was dependent on the following factors:
Workshop content Quality of materials Speaker expertise Effectiveness of presentation style Facilitator’s ability to engage and interact with the audience Efficient management of logistics and organization Networking opportunities Quality of audio and video during presentations Effectiveness of interactive sessions and activities Participation in Q&A sessions and discussions Hands-on workshop value Constructive feedback and improvement recommendations
Participant’s feedback was recorded and documented with an overall rating on a Likert scale from 1 to 5, i.e., from strongly disagree (1) to strongly agree (5). SPSS version 23, applying the Cronbach alpha test, was used to assess the reliability of the workshop.
Based on the overall findings of this research, it will be able to develop a set of best practices, recommendations, and guidelines for the infrastructure of transporting infectious substances in compliance with IATA regulations at the national level. By addressing these objectives, the research, based on training for the target cohort, aims to enhance compliance with IATA regulations for shipping infectious substances and biological specimens, thereby improving safety, efficiency, and regulatory adherence in the shipping process.
Phase II
After the in-person training and the workshop’s success for IATA training, phase II will focus on further refining the materials and identifying the areas for improvement in the training program based on feedback and evaluation results. The resource training materials with the targeted curriculum will be used for a broader audience for capacity building to ensure the widespread adoption of IATA rules. The training program will be rolled out nationally, and the institutes involved in shipping infectious agents by air, road, rail, or post have already been identified. An online support system will be established for the targeted audience during and after the workshops. During the workshops, an online support system will provide real-time assistance to participants during the training sessions. This could include access to digital resources, live Q&A sessions, and technical support. After Workshops, the online system will serve as a continuous learning platform, offering resources such as updated guidelines, FAQs, and forums for discussion. This ensures that participants can revisit the material and stay updated on changes to IATA regulations. The online support system enhances the effectiveness of the training program by providing ongoing assistance and fostering a community of practice among participants. 13
A series of workshops have been designed and will be implemented by prioritizing the importance and need assessment basis for each province and institute initiative to receive this training, considering the One Health objectives. Integrating One Health objectives into the design and implementation of these workshops underscores the importance of a holistic, collaborative, and proactive approach to the safe transport of infectious substances. By prioritizing human, animal, and environmental health interconnectedness, the training program addresses immediate safety concerns and contributes to long-term health security and sustainability. This alignment with One Health principles ensures that the workshops are impactful, relevant, and aligned with global best practices. The team of experts will be identified and deployed based on their expertise and IATA certification for upcoming workshops under the ABM management to develop a safe and secure BioRisk Management system for shipping infectious agents in their institute and provide a basic level of professionalism. This strategy will help implement the work practices for following IATA guidelines better across the country, expanding its reach. This will help develop a national network of certified trainers who can provide ongoing training and support. This will help create an enhanced national capacity to comply with and enforce IATA guidelines/regulations, resulting in a sustainable framework for continuous training and further implementation. It will focus on integrating the IATA regulations and practices into national policies and ensuring long-term compliance. 14
Results
A total of 180 professionals from the ABM Cohort participated in the survey. The respondents represented various provinces, with (n = 33) from Balochistan, (n = 45) from KP, (n = 78) from Punjab, and (n = 24) from Sindh (Table 1 and Figure 1).
Respondents institute jurisdiction
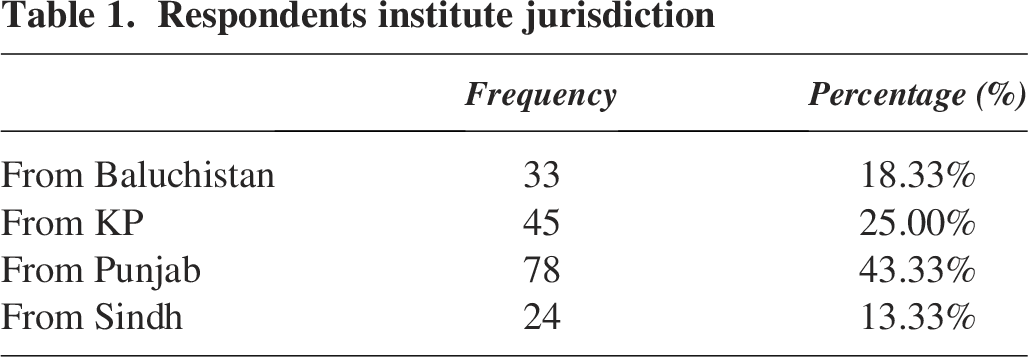
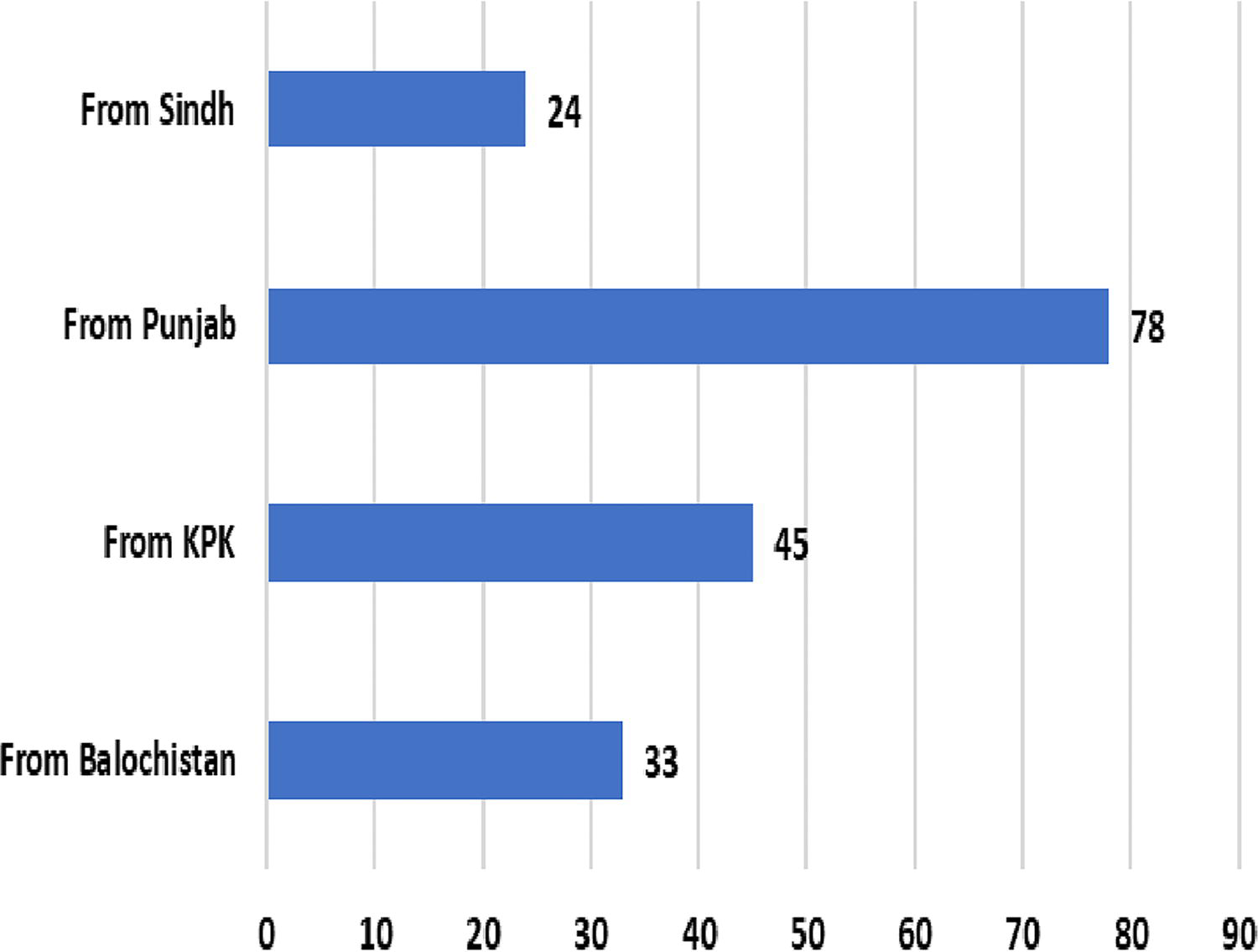
Respondents’ institute jurisdiction.
The survey included a diverse group of respondents from different institutes, with (n = 113) from research institutes, (n = 93) from diagnostic institutes, and (n = 10) from other institutes in Pakistan associated with the ABM and HSP. Most respondents were from the veterinary sciences field, including professors, associate and assistant professors, Doctor of Veterinary Medicine, and laboratory technologists working in research laboratories and universities. These findings are visually represented in Figure 2.
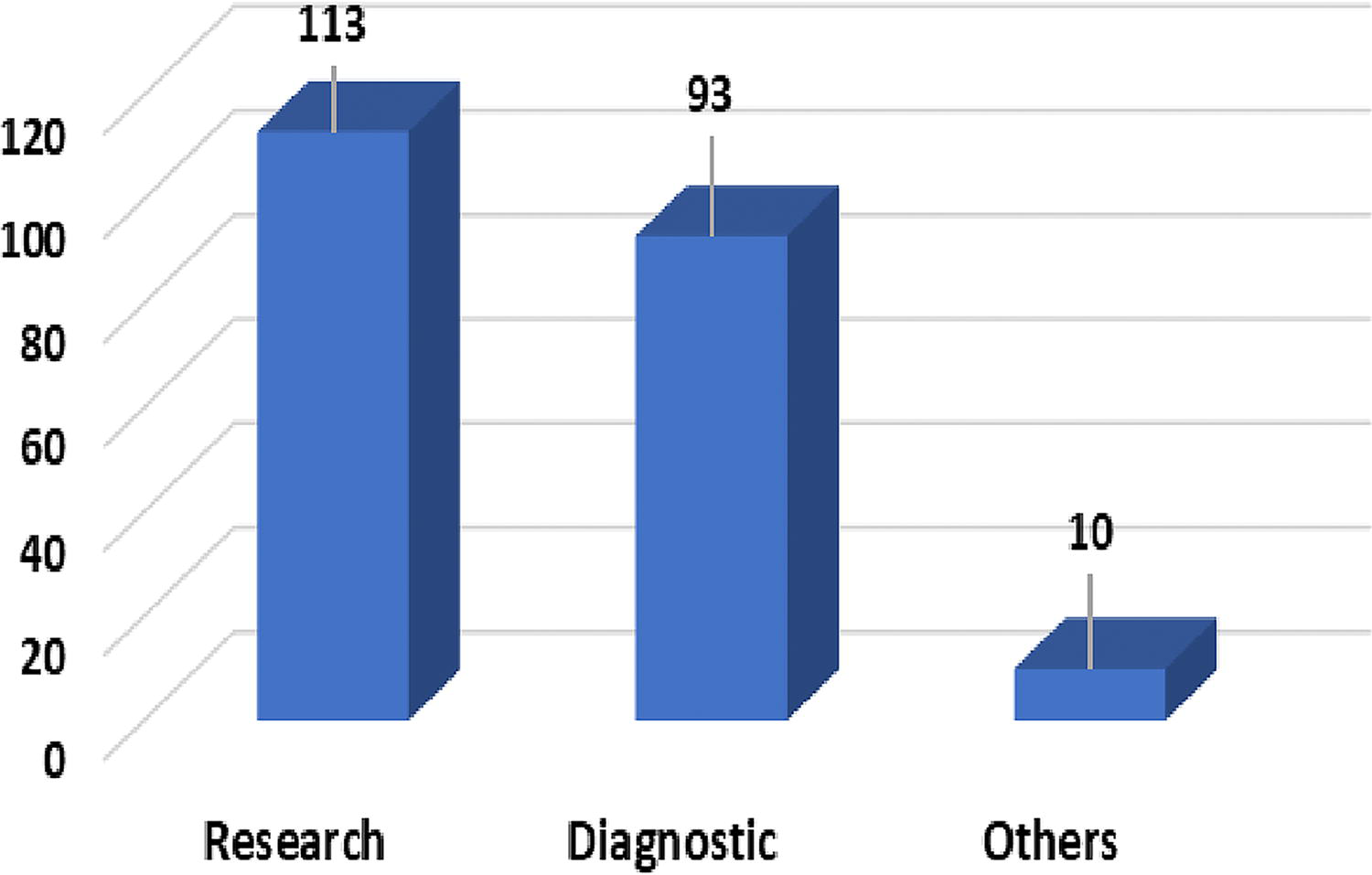
Primary focus of your institute activities.
More than two-thirds of respondents (67.2%) mentioned that they have not been trained in IATA regulations, while 65% have received prior training in Biosafety and Biosecurity.
In addition to demographic information, the questionnaire survey comprised 22 statements encompassing Training and Awareness, Risk Assessment procedures, Packaging and Labeling, Transportation Logistics, Documentation and Record Keeping, Collaboration and Communication, and Continuous Improvement.
The survey findings revealed that 11.1% of respondents were unaware of IATA regulations, and 48.9% responded negatively when asked about them. This indicates that 60% needed to become more familiar with IATA regulations governing the transportation of dangerous goods. These results underscore a clear imperative for enhanced awareness and education regarding these critical regulations (Table 2).
Are you aware of the International Transportation Association regulations for the transport of dangerous goods?

The survey revealed that institutes need to implement risk assessment strategies more effectively. A total of 71.6% respondents expressed uncertainty about risk assessment strategies for shipping infectious substances.
Almost 1 in 10 (88.9%) institutions need more facilities for conducting training and awareness sessions on the transportation of infectious substances. Furthermore, 67.2% of lab workers involved in transporting biological substances were unsure whether they had received recent training on safety protocols and procedures (data not shown).
Moreover, when probed about packaging and labeling procedures, respondents demonstrated a profound lack of knowledge, and when asked about the frequency of reviews and updates, they showed profound negligence. Inconceivably, 17.8% expressed uncertainty, while 19.4% used only primary packaging and 20% used secondary, and 42.8% reported using triple packaging for shipping infectious substances, uncertain whether it met UN specifications and requirements. An alarming 42.2% also admitted to the absence of a systematic process for packaging and labeling packages containing biohazardous substances with appropriate hazard symbols and information (Table 3).
Which type of packaging listed below is used for transporting biological substances compliant with international standards?

In this study, 75% of respondents stated that specific transportation routes for Dangerous Goods to minimize risks have yet to be established. The study aimed to determine whether institutions maintain comprehensive records of all biohazardous substance shipments, including the types and quantities of materials transported. The results showed that only 55% of respondents knew this requirement. Expressly, 21.1% indicated a lack of awareness, while 23.9% stated no records are being maintained. This underscores the need to implement strategies to improve understanding and record-keeping regarding biohazardous substances.
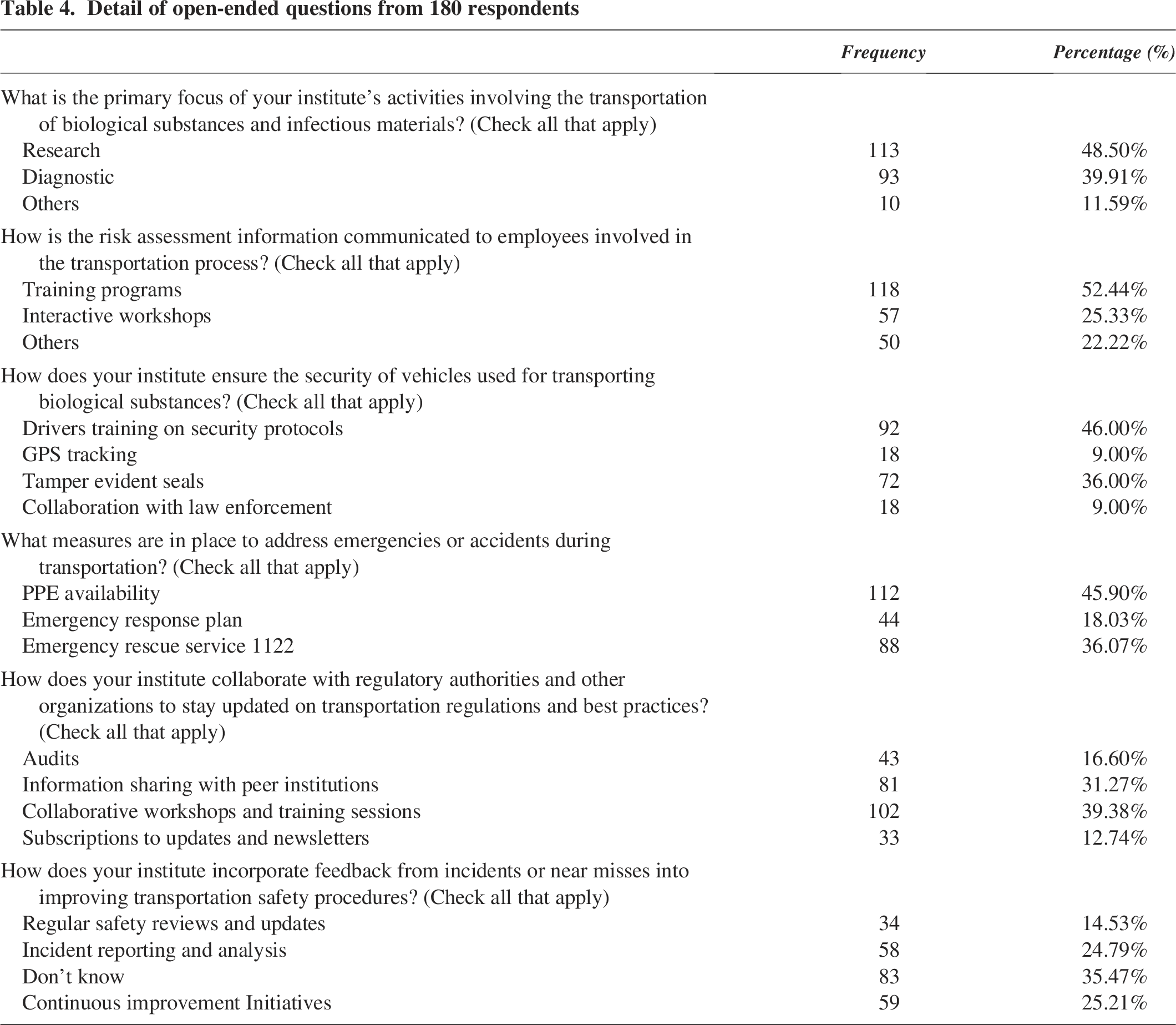
Furthermore, 62.2% of the respondents were unaware of a standardized reporting system for incident reporting or deviation from established procedures during transportation. The research findings also revealed no established protocol for communication and collaboration with regulatory authorities. Most respondents strongly agree that they need more knowledge about the process. Additionally, the study discovered that no established process is adopted in response to continuous improvement initiatives. About 35.47% of respondents reported having specific initiatives in their institutes but could not identify them. Detailed responses from open-ended questions are provided in Table 4.
Detail of open-ended questions from 180 respondents
Following a thorough analysis of survey findings and identified gaps, the IATA-certified professional developed the workshop curriculum and shared it with subject matter experts in HSP to refine it further, following the GBRMC Assessment, Mitigation, Performance (AMP model). After finalizing the curriculum, a cohort of 20 professionals from diverse institutes operating under the ABM framework was purposively selected to participate in a research-oriented workshop. Out of the 20 participants, 13 lacked prior training in IATA regulations for transporting infectious substances, while seven individuals had previously undergone training and expressed a desire for a refresher course.
The workshop included participants receiving course materials and training conducted by an IATA-certified trainer from ABSA. The 2-day total of 16 h of training followed the GBRMC AMP model and covered IATA regulations on the safe and secure transportation of infectious substances. The attendees sat around round tables to foster communication across all groups. The facilitator suggested that attendees sit next to people they did not know to reduce the hindrance of openly sharing during the small-group activities. After the facilitator completed the 2-day interactive IATA workshop (65 min), time was allotted to complete a multiple-choice question (MCQ) on a hard copy test explicitly developed for the workshop. The test comprised 50 multiple-choice questions on this topic, with a passing score of 85%. A score of two was given for each correct answer, and a score of zero was given for each incorrect answer for the MCQ test. The results were verified by another expert specializing in IATA regulations for shipping dangerous goods. Out of 20 participants, 18 passed the test, making it a successful endeavor with a success rate of 90%.
On the third day of the workshop, an 8 h comprehensive biosafety training session was conducted, accompanied by a pre-test and post-test questionnaire prepared by the RBP and CBSP-certified professionals within ABM and HSP experts designed to assess participants’ understanding of biosafety and biosecurity protocols. The impact of awareness was assessed through meticulous pre- and post-training assessment sessions. The results indicated a substantial increase in awareness among the participants, with the pre-test assessment yielding a score of 71.0% and the post-test assessment demonstrating a score of 96.0%. This reflects a 25.0% escalation in awareness of “Transport of Infectious Substances & Adherence to IATA Dangerous Goods Regulations.” Furthermore, a thorough paired t-test analysis unveiled a highly significant p value of 0.003 Table 5.
Paired samples test
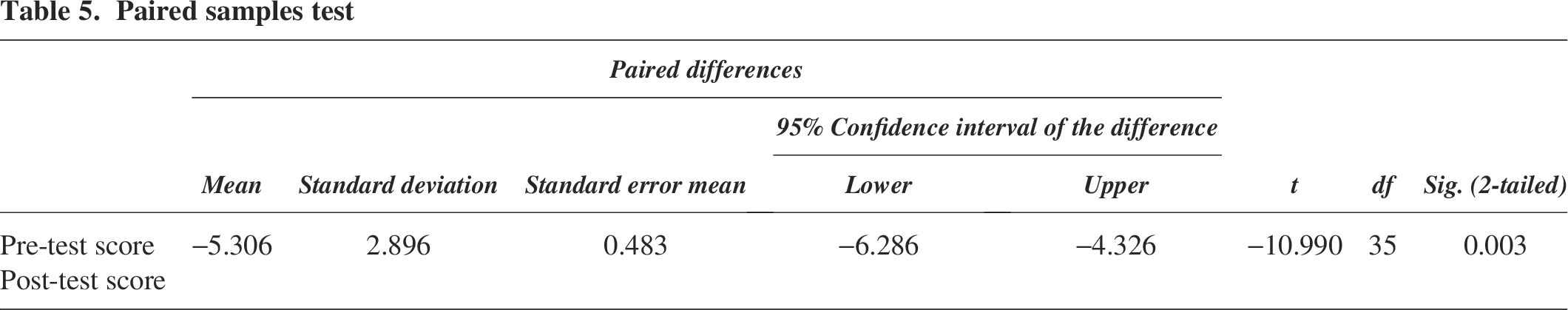
These results demonstrate the effectiveness of the training program in enhancing participants’ understanding of biosafety and biosecurity protocols.
The post-workshop evaluation included feedback from attendees, with ratings ranging from “Strongly Agree (score = 5) to Strongly Disagree (score = 1)”. The evaluations cover various aspects, such as the overall implementation of the workshop, the creation of a comfortable space, and the expertise of the facilitators, among other questions on the evaluation form. The general trend for each survey item indicates a high level of agreement among the attendees.
In the evaluation form provided at the workshop’s conclusion, most respondents “Strongly Agreed” with all 17 statements. The percentage breakdown of responses was 69.4% for “Strongly Agree,” 21.5% for “Agree,” and 9.1% for “Neutral.” The average score was 4.60 ± 0.379, which aligns closely with “Strongly Agree.” None of the respondents selected “Strongly Disagree” or “Disagree” for any of the questions in the evaluation form (Table 6).
Evaluation form analysis based on the Likert scale
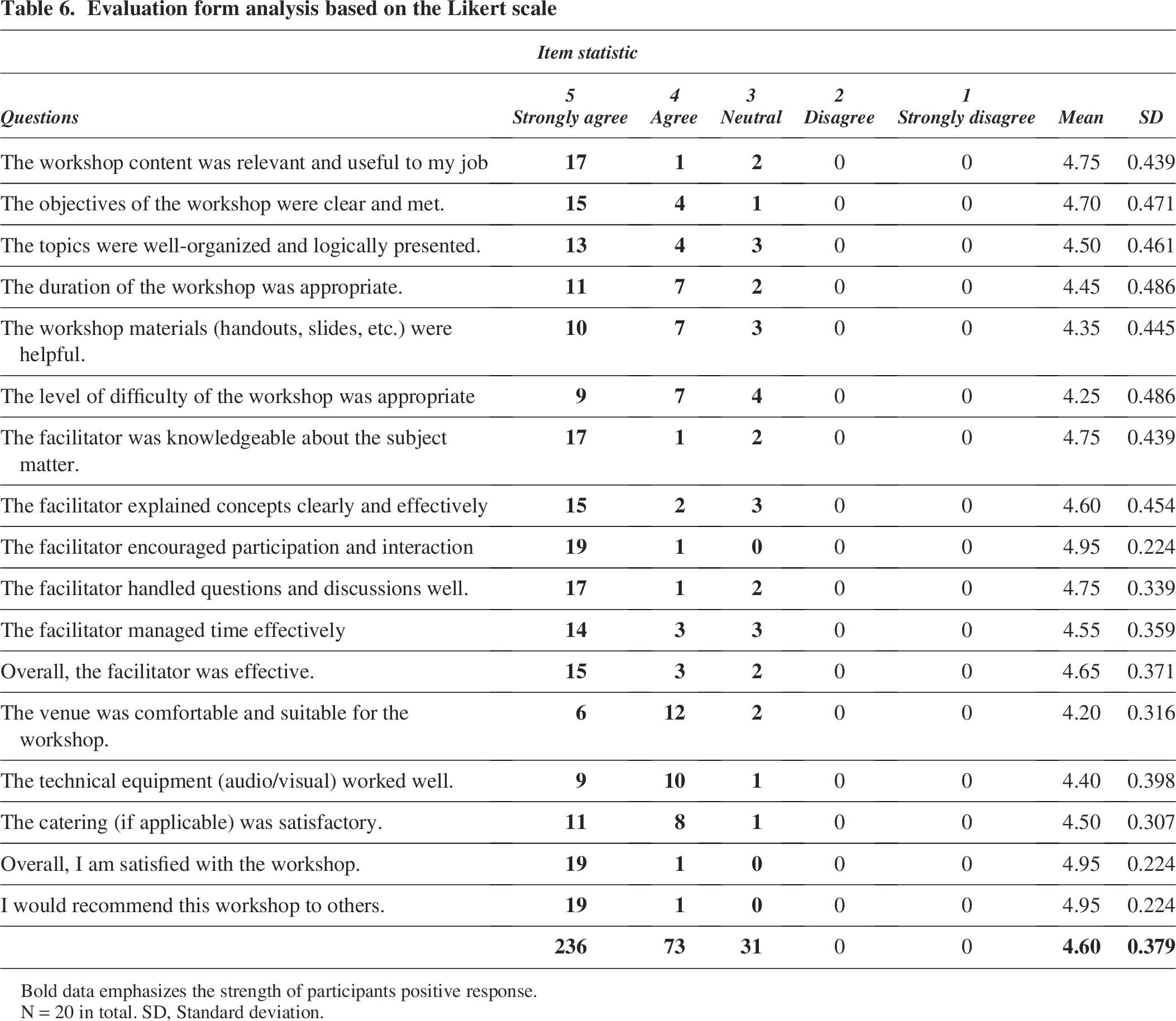
Bold data emphasizes the strength of participants positive response.
N = 20 in total. SD, Standard deviation.
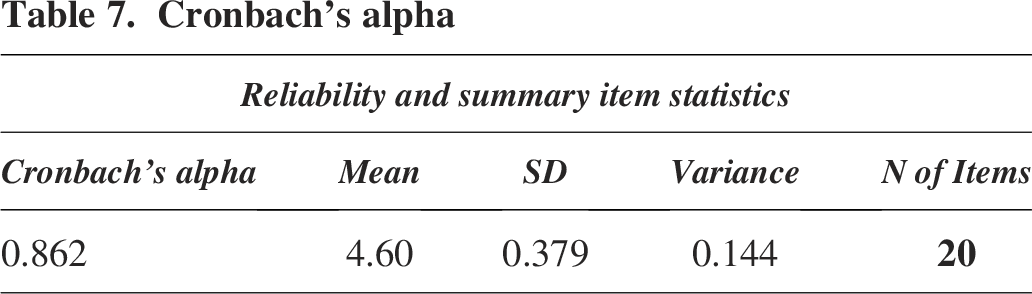
Cronbach’s alpha (α) is a statistical measure used to assess the internal consistency of a set of items, indicating how closely related they are as a whole. It is commonly employed to measure the reliability of a scale to assess the evaluation forms. Typically, a Cronbach’s α value falling between 0.6 and 0.7 indicates an acceptable level of reliability. In this case, the calculated Cronbach’s α based on the Likert scale evaluation is 0.862, surpassing the 0.6–0.7 threshold. This figure confidently asserts high reliability and consistency in the workshop, focusing on the IATA guidelines for safe and secure shipping of infectious substances (Table 7). In conclusion, the workshop’s structure exhibits a robust framework and reliable approach to safely transporting potentially hazardous materials (Figure 3).
Cronbach’s alpha
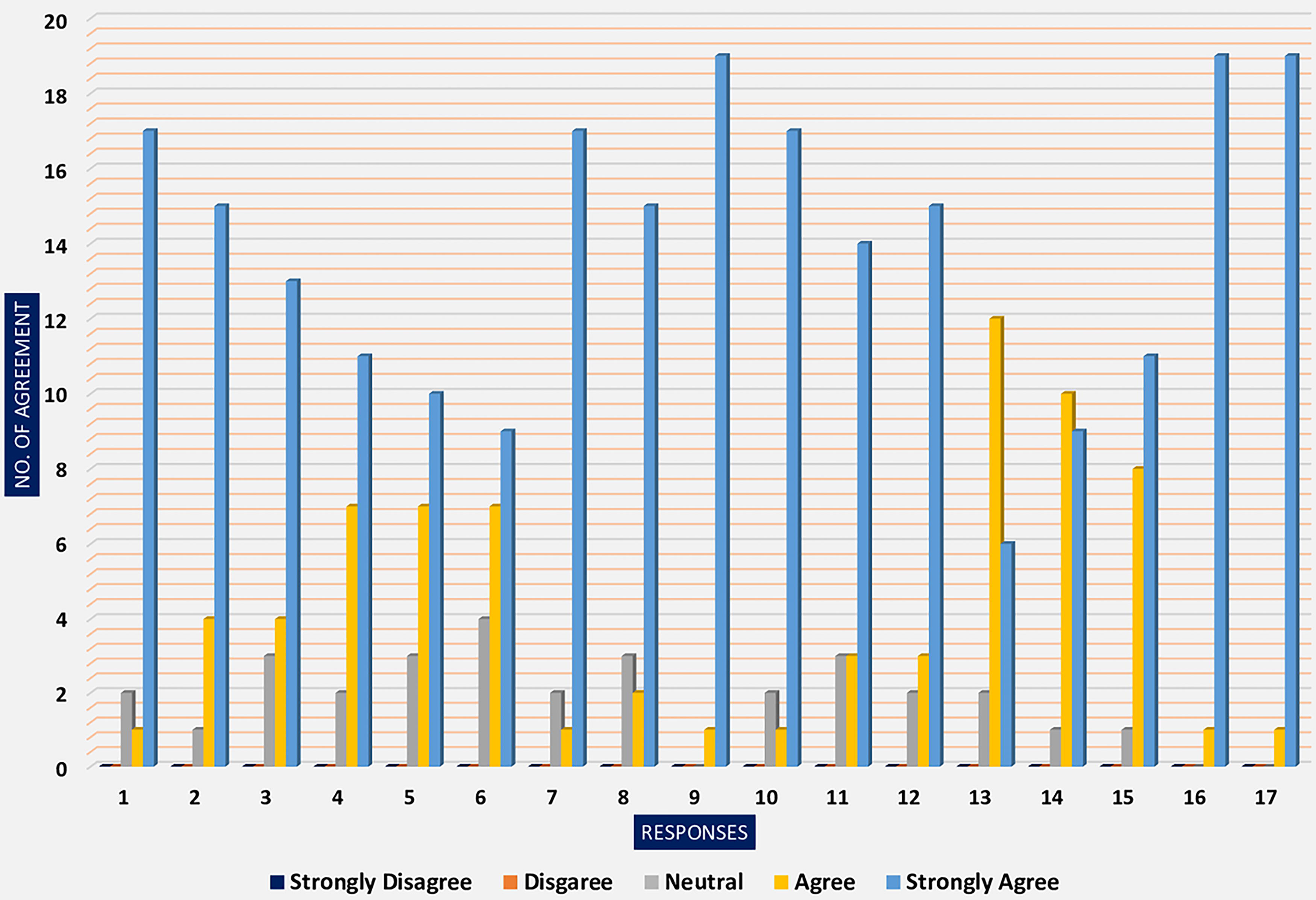
Evaluation form analysis based on the Likert scale.
In the comment section of the evaluation form, 18 participants provided additional comments. Some feedback included positive remarks such as, “17 participants highly appreciated the facilitator’s knowledge and attitude. It was a great workshop,” as mentioned by 15 participants. Regarding suggestions for improvement, 8 participants pointed out that the duration of the workshop should be extended. Additionally, 18 participants recommended that this workshop be developed into a country-wide initiative, which is already part of our next phase for its implementation. One participant also mentioned that while the workshop was a good idea, the content should focus more on local conditions lacking in our culture and how we can bring about positive change in this context.
Discussion
The transportation of infectious substances and biological specimens plays a crucial role in global health, research, and pharmaceutical industries. It’s vital to ensure the safe and compliant shipment of these materials to prevent public health risks and uphold the integrity of the specimens. The IATA establishes regulations to govern these shipments, but compliance can be challenging due to the complexity of the guidelines. This underscores the necessity of tailored training programs for specific groups involved in the shipping process. The IATA Dangerous Goods Regulations (DGR) provide a comprehensive framework for safely transporting hazardous materials, including infectious substances and biological specimens. These regulations cover various aspects, such as classification, packaging, labeling, and documentation. Strict adherence to these regulations is crucial, given the potential risks of mishandling these substances. Despite clear guidelines, several challenges hinder compliance. The detailed and technical nature of the IATA DGR can be overwhelming, especially for individuals not regularly involved in the shipping process.
Most stakeholders, including researchers, laboratory personnel, and shipping staff, must thoroughly understand the latest regulation updates and changes. However, the different groups involved in the shipping process have varying levels of knowledge and experience. This inconsistency in knowledge and expertise leads to unreliable compliance practices, which lead to risky practices and procedures when shipping infectious substances.
These challenges prompt a robust discussion of best practices for ensuring compliance with IATA regulations. It is crucial to explore strategies to address the guidelines’ complexity, raise stakeholders’ awareness, and provide targeted training programs to enhance compliance and ultimately contribute to global health and safety. 15
Tailored training programs can address these challenges effectively. Organizations can enhance understanding and compliance with IATA regulations by customizing the training content to meet the specific needs of different cohorts. Critical components of such training programs include:
Targeted Content: Develop training materials specific to the roles and responsibilities of different cohorts. For instance, laboratory personnel might need detailed guidance on packaging and labeling, while shipping staff might focus more on documentation and handling procedures. Practical Demonstrations: Incorporating hands-on training sessions that simulate real-world scenarios. This can help participants gain practical experience and confidence in handling infectious substances and biological specimens. Regular Updates and Refresher Courses: Training programs should be regularly updated to reflect the latest changes in IATA regulations. Offering refresher courses can help reinforce knowledge and inform all stakeholders about new requirements.
A study done in Pakistan also stressed the importance of assessment and certification. The focus can be on implementing assessment mechanisms to evaluate participants’ understanding and competence. Providing certification upon successful completion of the training can serve as motivation and formal recognition of compliance readiness. 16
The implementation of tailored training programs offers several benefits:
Enhanced Compliance: By providing targeted and relevant information, stakeholders are more likely to understand and adhere to IATA regulations. Reduced Risk: Proper training minimizes the risk of mishandling infectious substances, thereby protecting public health and ensuring the safety of transportation personnel. Operational Efficiency: Well-trained staff can streamline the shipping process, reducing delays and errors associated with non-compliance. Reputation and Trust: Organizations involved in the shipment of infectious substances and biological specimens can enhance their reputation by demonstrating a commitment to safety and compliance.
One book published by ASM Clinical Microbiology Procedures Handbook section 15 emphasized biohazards and safety and safety-related aspects, emphasizing the importance of this scientific discipline and its integration into safe and secure transportation cannot be ignored. A third day of training focused on the prevention and safety aspects of transporting category A, category B, and exempt human specimens was performed to fill the gap in the local scenario based on IATA regulations and experiences of carriers in shipping infectious substances.1,17
WHO Global Consultative Meeting on the Safe Shipment of Infectious Substances addresses the gaps in the knowledge of all the related stakeholders involved in the transportation of dangerous goods, e.g., the impact of transport regulations on countries and international organizations, partnerships and education of pilots to manage the risk of disease transmission in air travel and problems and possible solutions to facilitate shipment of animal pathogens, controlling the response to outbreaks and importance of training. 18 This training addressed the gaps, considering all the related issues that still needed to be discussed in the past.
In the wake of the COVID-19 pandemic, additional measures were adopted to address the challenges of this emergent infectious disease globally. Therefore, a risk-based approach was adapted to the local epidemiological context, and capacities can substantially mitigate the risk but cannot achieve “zero risk.” The report also emphasized the need for additional training to reduce the transmission of SARS-CoV-2, which causes COVID-19, during international transportation of Infectious substances. 19
A book by Mohamed Moussif 2024 “Modernizing Global Health Security to Prevent, Detect, and Respond,” provides a vision for a more protected and safer global public health future to prevent, detect, and respond to (re)emerging threats has been introduced. It aims to chart a way forward with the understanding that future pandemics must and can be prevented. From a public health perspective, which includes global health security, the growing concept of One Health; epidemic and pandemic prevention, detection, and response; reviews of past (e.g., Ebola, MERS-CoV, Zika, and COVID-19) public health emergencies of international concern; roles of information and communication technology; transportation of infectious substances by air and other means, humanmade public health threats; and legal and ethical issues (e.g., viral sovereignty, trust, and transparency) is gaining attention. 20 It provides the academic substance and quality for researchers, lab workers, and practitioners to deeply understand the why of health emergencies and, most importantly—what we can and should do now to prepare. It stresses the importance of training individuals in every sector to handle infectious substances at any level. Our study focuses on this aspect and complies with this study. Another publication on Good Clinical Laboratory Practices in Pakistan, chapter 17 on the Shipping/Transport of Specimens, provides in-depth knowledge on this subject and stresses its importance in One Health. 21
The transportation of infectious substances and biological specimens plays a vital role in the healthcare, research, and diagnostics sectors. It is imperative to adhere to the IATA regulations when shipping these materials, as non-compliance can result in legal ramifications, safety hazards, and delays in critical shipments. Addressing this issue requires tailored training programs to enhance comprehension and compliance with these regulations. Existing training programs may need to be customized to cater to the specific needs of different groups involved in the shipping process of infectious substances, thereby minimizing compliance gaps and potential risks. Ensuring comprehensive adherence to IATA regulations for transporting infectious substances by various groups is essential. This could be attributed to the necessity for realistic implementation of readily available rules and regulations by the Civil Aviation Authority and higher authorities, including the Ministry of Commerce and the Health Ministry in Pakistan. Additionally, it may stem from a need for greater awareness and engagement in both the public and private sectors, coupled with limited resources allocated to this activity. Considering the national resources in this context, there is a clear requirement for more information regarding the transportation of infectious substances. 22
Conclusion/Recommendations
In conclusion, enhancing compliance with IATA regulations for shipping infectious substances and biological specimens requires a strategic approach to training. In this context, the following approach can be adapted to meet this existing huge gap in the present scenario:
Tailored training programs designed to meet the specific needs of different cohorts can significantly improve understanding and adherence to these critical guidelines. By investing in such training initiatives, organizations can ensure the safe and efficient transportation of these materials, ultimately contributing to global health and safety. This IATA training on the safe and secure transportation of infectious substances should be regularly included in the protocols of Pakistan’s life, health, and veterinary sciences institutes, which conduct research and diagnostics work. From Pakistan’s perspective, facilitating the provision of crucial information and training resources by forming partnerships with non-governmental organizations and the public sector involved in shipping infectious substances is essential. IATA regulations, which hold international recognition and comply with ICAO standards, should guide this collaboration.
Authors’ Contributions
S.A.Q.: Conceptualization (lead), methodology (lead), formal analysis lead), writing—original draft (lead), review and editing (equal). J.K.: Project administration (lead), validation, review and editing (equal). C.M.G.: Resources (lead), supervision (lead), review and editing (equal). A.A.: Review and editing AI (lead). P.K.: Supervision (supporting) and review (equal).
Footnotes
Acknowledgments
The authors thank Health Security Partners (HSP) for supporting this innovative IATA workshop project focused on Pakistan. The authors also thank Cecelia Madsen Guinevere, HSP’s program manager, for initiating this project with the help of the Association of Biorisk Management (ABM). The authors are thankful to Muhammad Azheruddin for his continuous technical support. The authors also thank their mentors, Prasad Kuduvalli and Jason A Rao, HSP, for facilitating the project and Eric Cook for giving Shamsul Arfin Qasmi the initiative to write this project. Grammar has been checked by Grammarly.
Author Disclosure Statement
The authors declare that there is no conflict of interest regarding the publication of this article. No financial, professional, or personal relationships have influenced the research, writing, or publication process. The authors have no affiliations or involvements with any organization or entity with a direct financial or non-financial interest in the subject matter or materials discussed in this article.
Funding Information
No funding was received for this article.
