Abstract
Introduction:
This study was conducted to assess the difference in biosafety behaviors among nurses working in a levels 2 and 3 hospital unit in Morocco and the need for additional biosafety training before working in a high-containment facility.
Methods:
A prospective cross-sectional and observational study was conducted among new and experienced nurses in the Center of Virology and Tropical Infectious Diseases of the Military Teaching Hospital Mohammed V of Rabat, Morocco, between December 1, 2017 and February 28, 2018 (3 months). We compared behaviors in biosafety practices (donning gloves, handwashing, etc) between the 2 groups, and data were collected by observing workers directly or via camera.
Results:
A total of 31 nurses, 14 newly graduated nurses (45.2%) and 17 experienced and trained (54.8%) nurses, were observed during 89 entries into high-containment hospital rooms. Among the behaviors observed, only donning an inner pair of gloves was observed consistently between the 2 groups. Experienced and trained nurses washed their hands (100%) and observed the antiseptic contact time (71%), whereas recently graduated nurses ignored these activities significantly (79% and 32%, respectively). During handling of the patient, the trained and experienced nurses were significantly better than the new ones in the practices of biosafety and personal protection. Gaps in biosafety were observed more frequently among newly graduated workers, especially donning outer gloves before patient contact, nurses touching their faces, and the act of wearing jewelry while working.
Conclusion:
Safety training should begin early in the training curriculum with theoretical and practical courses. It must be introduced into the general biosafety policy as part of a safety and standard working culture. It is recommended that personnel be required to pass a proficiency exam in critical biosafety tasks before working with high-risk patients.
Introduction
Biosafety is defined as “the discipline addressing the safe handling and containment of infectious microorganisms and hazardous biological materials.” Biosafety is applicable to clinical, diagnostic, teaching, research, or production facilities where work is performed with infectious agents that may cause serious or potentially lethal disease. 1 The practice of safe handling, whether it is pathogenic microorganisms in the biological laboratory, patients in a hospital care unit, or waste management, is accomplished through the application of containment principles and risk assessment. 2
Human error and poor techniques can compromise the best of safeguards to protect the worker. 3 Thus, a safety-conscious staff, well-trained and informed in the recognition and control of hazards, is key to the prevention of acquired infections, incidents, and accidents. For this reason, initial education and continuous in-service training in biosafety measures is essential, 4 and personnel must receive specific training in handling pathogenic and potentially lethal agents and be supervised by scientists competent in handling infectious agents and the associated procedures. 3 In Morocco, laboratory technicians and nurses benefit from minimal training in biosafety, and this training is distributed sparsely throughout the microbiology curriculum.
After assignment to laboratories in which tick-borne pathogens are handled or to hospital high-containment units that require specific knowledge of biosafety practices, new graduates appear insufficiently trained. As a result, there can be serious incidents and accidents. 5 To reduce incidents and laboratory/hospital-acquired infections by workers, additional training is provided at the Laboratory of Virology, Mohammed V Military Teaching Hospital, a high-containment hospital facility, to new nursing graduates. This training consists of active learning experiences with hands-on activities, including donning and doffing personal protective equipment (PPE), using antiseptics, managing waste, and other practices for reducing transmission of infection from patients to workers.
The purpose of this study is to confirm the gaps in knowledge and behavior in biosafety practices of recent nurse graduates compared to trained and experienced nurses and demonstrate the need for additional specialized training for workers in higher containment hospitals.
Material and Methods
This prospective cross-sectional study was performed in the Center of Virology and Tropical Infectious Diseases of the Military Teaching Hospital Mohammed V of Rabat, Morocco, between December 1, 2017 and February 28, 2018 (3 months).
This center receives patients suffering from serious bacterial, viral, or parasitic infections as well as patients suspected of tropical infectious diseases, arbovirus infections, and hemorrhagic fevers. For this purpose, the center has 2 sections of biosafety containment rooms—a section with biosafety Level 2 (BSL-2) rooms and a section of BSL-3 rooms.6 -9 Entry procedures in both room types are identical in requirements of glove usage, changing during patient handling, use of antiseptics, handwashing, and waste management. In Morocco, the primary differences between Level 2 and 3 rooms are the respiratory protection used and the use of anterooms with negative pressure at Level 3. Level 2 rooms resemble conventional hospital rooms with additional protections: use of filtering facepiece respiratory masks (FFP1), specialized clothing, equipment and antiseptic use, but lack an anteroom and are not under negative pressure. These rooms are used to prevent respiratory transmission of meningitis, pneumonia, chickenpox, or other viral diseases. Level 3 rooms meet all BSL-3 standards and are used only in situations where there is a risk of severe disease that can be transmitted by the respiratory route. These rooms are under negative pressure (–30 Pa), supply and exhaust air are HEPA-filtered, and supplies are brought into the rooms using a passthrough box. At Level 3, workers use higher protective FFP respiratory masks (FFP2 or FFP3, depending on the risk assessment). Health care workers enter the patient’s room via 2 tandem anterooms that are under negative pressure and are required to wait 2 minutes between anterooms to allow negative pressure to be maintained. Door openings and pressure are automated using a computer program. In Level 3 rooms, highly infectious patients with infections of multidrug-resistant tuberculosis, pandemic influenza, and MERS-CoV and patients suspected of hemorrhagic fevers are being treated.
Study Population
We compared behaviors in biosafety practices between 2 groups:
Group I: 17 trained and experienced nurses in biosafety, having accumulated greater than 6 months experience. This group was composed of nurses who have received on-the-job training, consisting of at least 2 cycles of theoretical and practical biosafety training. Each cycle consists of 20 hours of training including biosafety simulation exercises where they learn about PPE donning and doffing techniques, use of antiseptics and disinfectants, sorting, and waste disposal. Group II: 14 new nursing graduates, having less than 6 months working experience and lacking training at the center. This group is composed of graduates from the National Nursing Institute that have joined the Mohammed V Military Teaching Hospital in Rabat, Morocco. The 3-year training cycle for nursing students does not include specific hours for biosafety education, and biosafety information is scattered throughout the course of microbiology. During a lesson about a virus, for example, the teacher will inform the students about the class of pathogens to which the virus belongs and the collective and individual ways of preventing the disease that it causes without, however, discussing risk management and mitigation for handling the virus.
Study Design
We considered that for this study, the most significant actions to observe were biosafety practices of nurses when entering and leaving a patient’s room and during contact with the patient. These acts were easily observable and measurable. The 2 groups of nurses were compared by measuring the differences in compliance with established biosafety practices as described in the hospital exposure control plan following the guidelines of the World Health Organization (WHO; Practical Guidelines for Infection Control in Health Care Facilities) and the Institut de veille sanitaire (IVS) of France.
10
The frequency of errors and risky behaviors during entry and exit from a room whose patient had (or was suspected to have) an infectious disease caused by a Risk Group 2 or 3 pathogen were counted. Each nurse was observed during several entry-exits. The following data were collected:
Training and experience of the worker (< or > 6 months) Time (in months) of experience in the care unit
Handwashing before entering the patient room (response: yes or no) Adequate time for handwashing and action time for use of the antiseptic (correct if >2 minutes) Donning gloves (inner gloves) before the entry (response: yes or no) Donning outer gloves before patient contact (response: yes or no) Changing gloves after patient contact (response: yes or no)
Doffing outer pair and donning new pair of gloves before exiting the patient room (response: yes or no) Discarding outer gloves after exiting the room (response: yes or no) Removing inner gloves after exiting the room (response: yes or no) Use of antiseptic gel after exiting the room (response: yes or no)
Wearing jewelry or watch (response: yes or no) Touching face and or surfaces (response: yes or no) Putting gloved hands in pockets (response: yes or no).
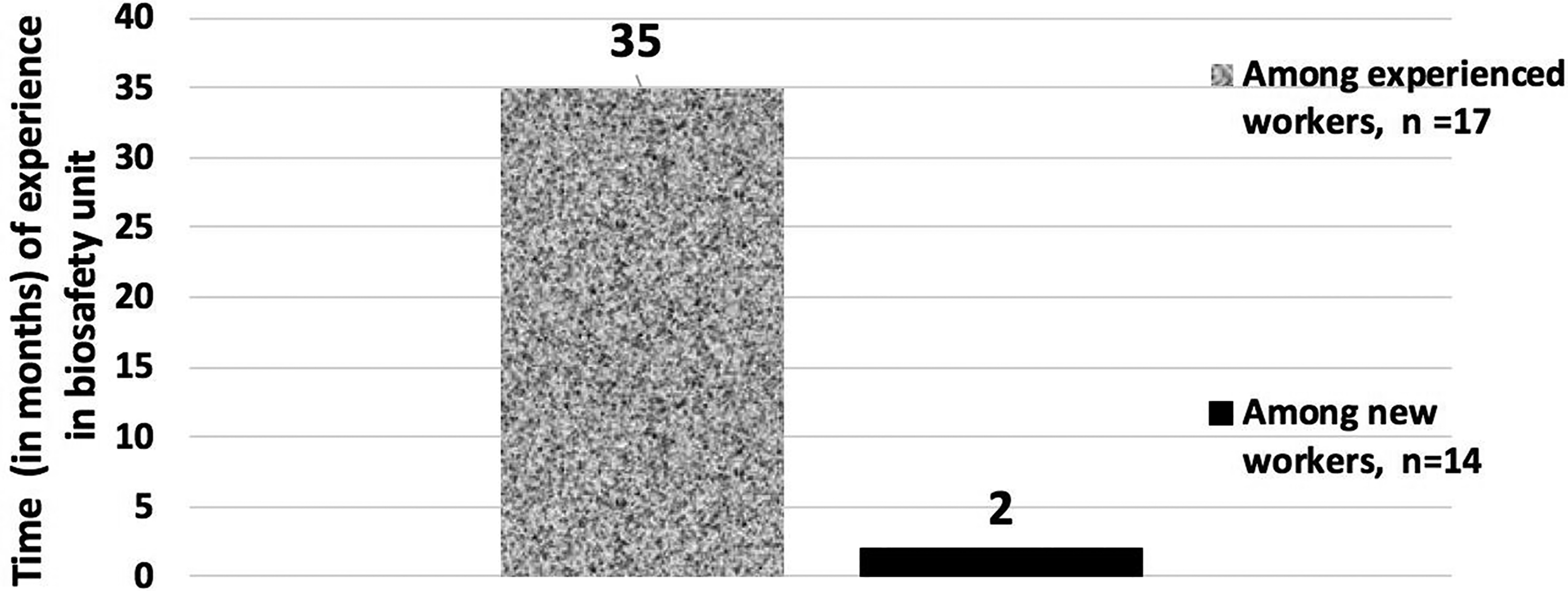
Average duration of experience in a high-containment hospital.
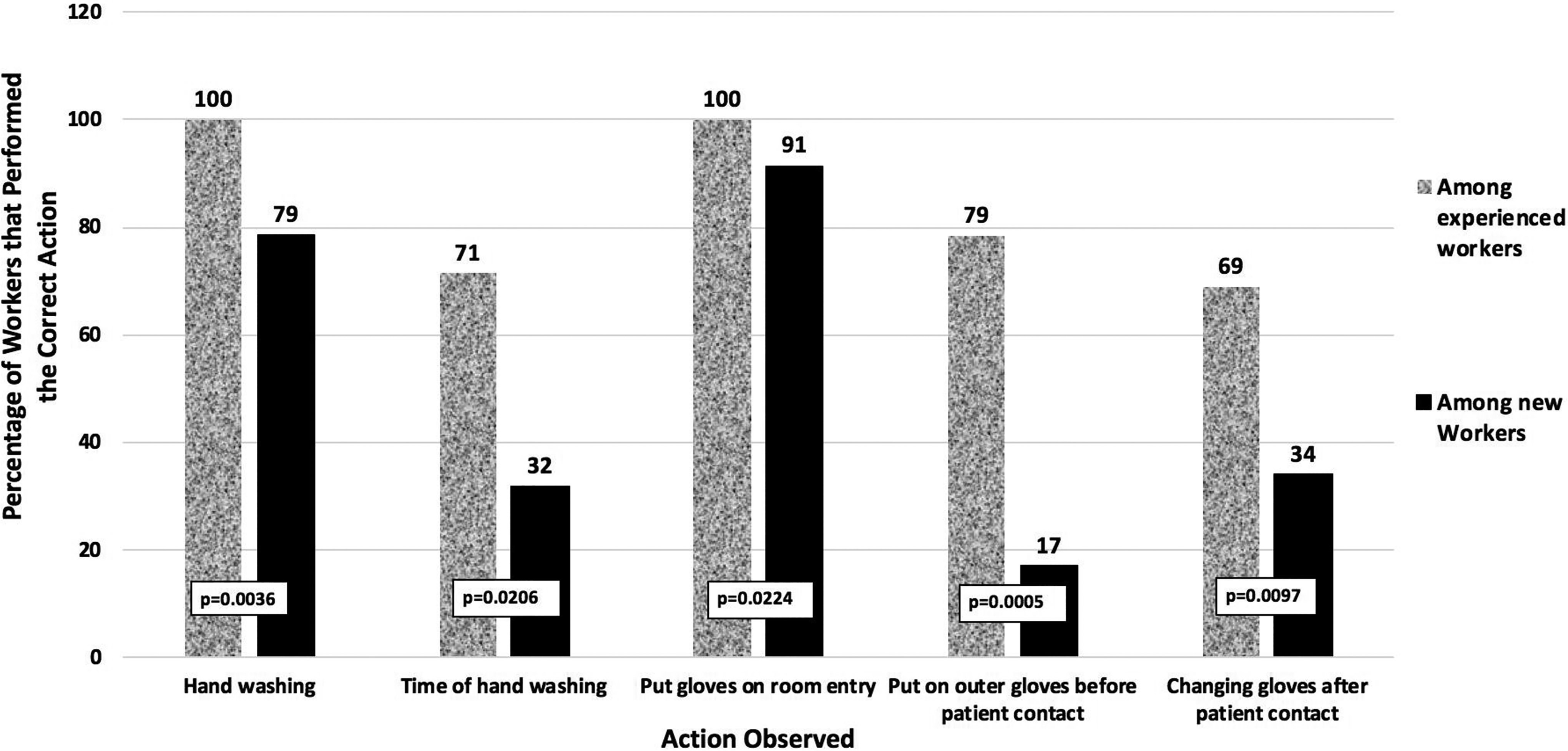
Comparison of percentage of experienced and newly graduated nurses that correctly performed handwashing, gloving, and degloving activities on room entry and patient contact.
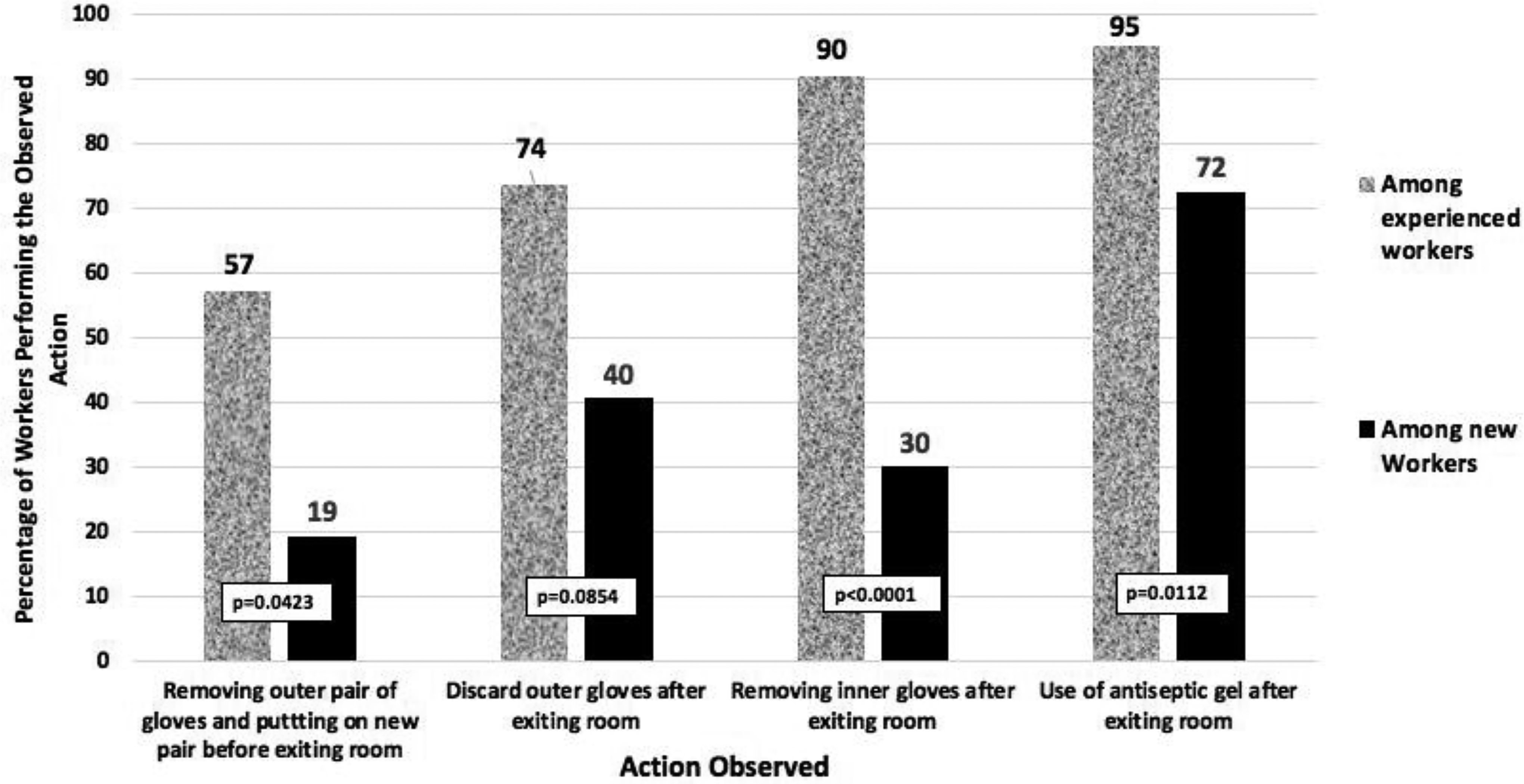
Comparison of percentage of experienced versus newly graduated nurses that follow proper biosafety procedures when exiting a patient room.
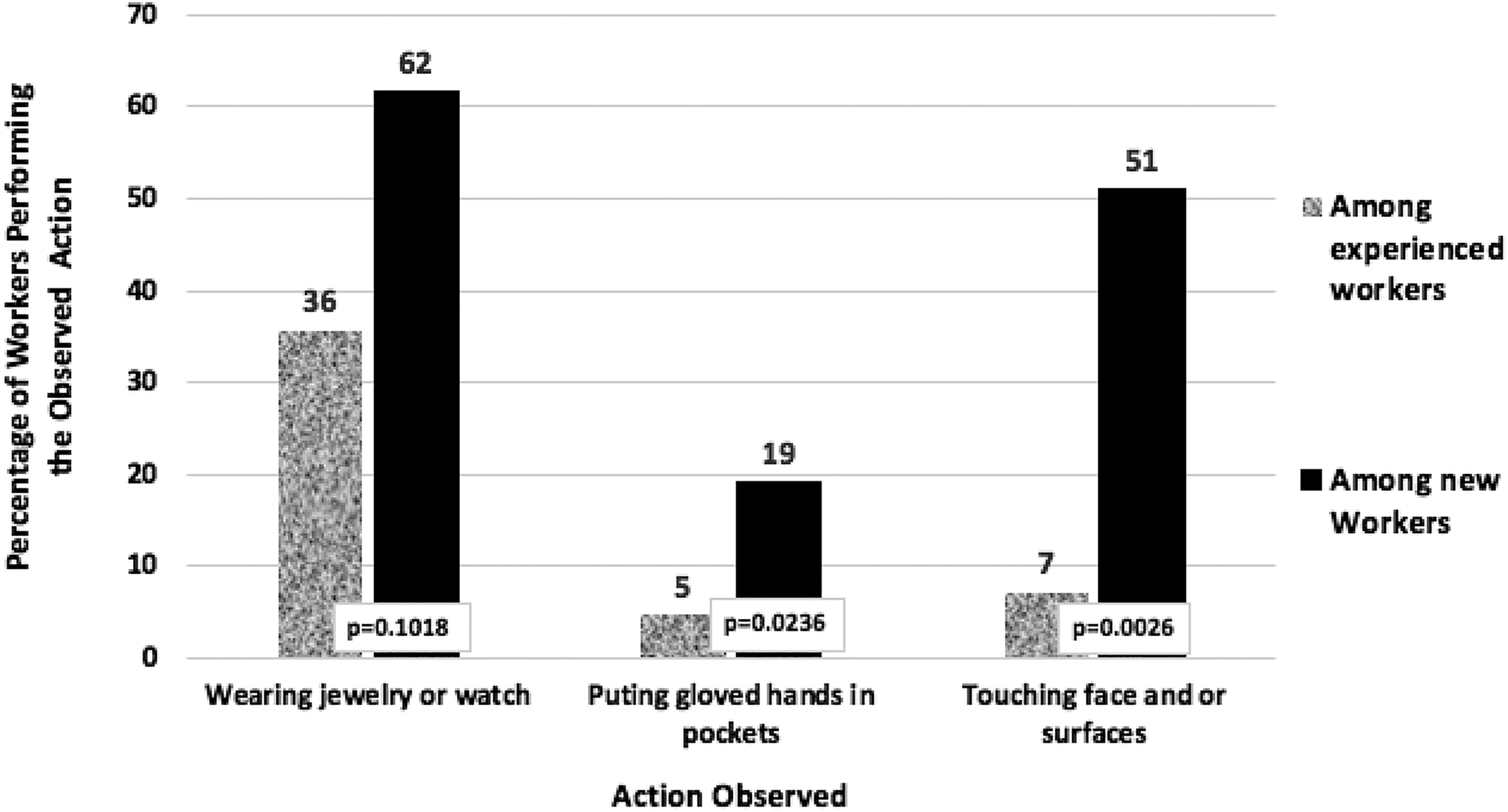
Comparison of percentage of experienced versus newly graduated nurses in compliance with general biosafety policies.
Data Collection and Statistical Analysis
A staff explanation meeting on the steps, objectives, and outcomes of the study was arranged to provide an opportunity to inform participants and obtain their written consent. The data were collected independently by 3 different scorers, either directly (by visual observation) or by viewing the videos recorded by the surveillance system. Before starting the observations, the 3 scorers agreed on which actions should be observed, what outcome constituted a positive or null score, and how the data would be collected. In addition, the observers agreed that each observer would score results of 2 observations, then ignore 1, and repeat this process to enhance randomness of the observations that were being scored. All observations were performed by observers without informing the nurses that they were being observed and their actions recorded.
After predetermining the scoring system, a preliminary experiment was performed in which the 3 scorers observed 10 nurses separately during their work and compared the results of their observations to ensure that the scorers were coding the data the same way. These preliminary results allowed harmonization of the methods for observations and validation of data collection.
Cameras are installed in the corridors of the whole establishment. Since there is no camera in the patient’s room, the observation of the biosafety practices was performed by the authors or a trained biosafety nurse (major of the service) who did not take part in the study (ie, was not observed).
Data were analyzed with a Wilcoxon ranked sum, 2-sample test using JMP software.
11
Analysis was performed on the data comparing biosafety actions (dependent variable) of newly graduated nurses and the more experienced nurses with more extensive biosafety training (extent of prior training; independent variable).
Validation of the Method and Duration of the Observations
The participants were a heterogeneous population made up of new graduates with minimal biosafety training during their educational process and experienced workers that had received more extensive biosafety training after graduation on the job. A duration of 6 months of experience was chosen since trained, experienced nurses had more than 6 months experience, as opposed to the new nurse graduates who had less than 6 months experience. Typically, in our experience, this is the shortest period of time for a newly graduated nurse to acquire a minimum knowledge in biosafety and improve their behavior and actions. It was observed that over time, more experienced nurses would correct the actions of less experienced nurses. In addition, this time period corresponded to the cycle of graduation of new nurses and assignment of new graduates to institutions in Morocco. The experimental time frame and choice of biosafety actions to be observed were critiqued by the authors, who have expertise in biosafety, and was approved by the biosafety professionals present in the MENA VI Biosafety meeting organized by SANDIA laboratories in Rabat, Morocco, November 7-10, 2017.
Results
Rate of Participation and Characteristics of the Participants
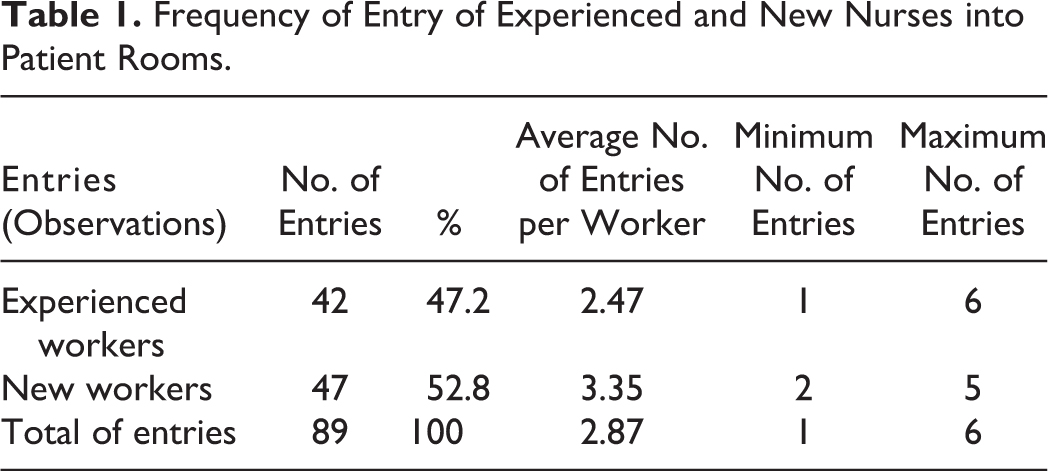
During the period of the study, 31 nurses (14 new graduates [45.2%; <6 months experience] and 17 experienced and trained [54.8%; >6 months experience]) were observed. In addition to time of work experience, new graduates and experienced nurses differed in the extent of formal biosafety training that they received since experienced nurses received additional training after graduation from the National Institute of Nurses. The average duration of experience in a biosafety unit ranged from 0.5 to 52 months. This duration was greater among experienced nurses (35 months, range = 16-52 months) compared to newly graduated nurses (2 months, range = 0.5-5 months) (Figure 1). Observation of the staff in action, conducted at random, accounted for 89 entries. The frequencies and other characteristics of these room entries and exits are described in Table 1.
Frequency of Entry of Experienced and New Nurses into Patient Rooms.
Agent Hazards Encountered During Routine Work in the Biosafety Unit
During the study, most of the patients were hospitalized for suspicion of MERS-CoV (12 patients, 46%) or for an infectious disease under investigation. Table 2 presents the distribution of confirmed or suspected infectious diseases among hospitalized patients during the study period.
Distribution of Confirmed or Suspected Infectious Diseases Among Hospitalized Patients During the Study Period.

Observations of Biosafety Practices Among Newly Graduated Nurses and Experienced Nurses
All nurses were observed for biosafety practices upon entry to the patient room (handwashing and proper handwashing technique, donning gloves on patient entry, donning a second pair of gloves before patient contact, and changing gloves after patient contact). When entering the patient’s room, all of the experienced and trained nurses washed their hands and followed the required antiseptic action time (Figure 2).12,13 Newly graduated nurses often washed their hands (79%); however, they often did not follow the guidelines on antiseptic contact time. Further, the new graduates often donned gloves on entry; however, they were noticeably less likely to don outer gloves before patient contact or change their gloves after patient contact as compared with experienced nurses.
After handling the patient and when leaving the room, the trained, experienced nurses were much more likely than the new nurses to use correct practices of biosafety and personal protection (Figure 3). These practices included changing their gloves after patient contact, removing both inner and outer gloves upon exit, and the proper use of antiseptic gel. The methods used indicated that, as might be expected, the more experienced nurses showed greater maturity and know-how when performing these operations.
Additional practices that were observed included wearing of watches and jewelry, putting gloved hands in pockets, and touching surfaces and faces. All of these practices can increase the likelihood of worker contamination and infection. These practices were observed more frequently among recently graduated workers, especially with respect to wearing jewelry (Figure 4). Two-thirds of newly graduated individuals made this mistake while only one-third of experienced nurses erred in this practice. On the other hand, regarding touching of faces and surfaces, less than 1 out of every 10 experienced nurses did so, while half of the newly graduated nurses made this error.
Discussion
Among health workers, nursing professionals are the ones who face higher risks of exposure to biological material, due to their professional routine and contact with patients.14 -17 Nursing is a profession that requires clinical expertise to perform assistance effectively and safely; therefore, an in-depth knowledge of biosafety standards is essential. 18 During student training, it is necessary to strengthen their knowledge and skills in these practices. Education of nurses must cover a range of biosafety and biosecurity principles and practices. 19 Thus, this study first aims to confirm the difference between nurses with minimal training and those that are more experienced and have had more extensive and specific biosafety and biosecurity training. The biosafety practices of nurses in a high-containment unit, requiring advanced biosafety and biosafety skills, are compared between new graduates versus experienced and trained nurses. Although this study inevitably presents biases, such as the fact that the new nurses copy the practices of the experienced ones over time, and it is difficult to differentiate between know-how due to experience and that due to the training, the differences in behavior are important to study in order to address these gaps.
It is now accepted that values are acquired; this means the nurses learn either directly or indirectly by observing others’ behavior. 20 Following academic education, development of professional values in nurses is mainly influenced by experts in the profession, colleagues, patient care situations, and organizational values. 21 Therefore, the work practices of experienced nurses, the expectations of supervisors, the biosafety/biosecurity culture, and training of new nurse graduates greatly impacts both safety practices of the nurses and health of patients in high-containment hospitals.
Prior to this study, there was no published literature on the impact of including biosafety/biosecurity education in the curriculum of nursing students and the effects of further education of new nursing graduates during their assignment in hospitals receiving patients with diseases due to Class 2 or 3 pathogens.
This study revealed that the majority of new Moroccan graduate nurses have modest general biosafety knowledge and practices and that their level of biosafety performance evolves favorably with training and experience. For example, some practices were performed similarly between the 2 groups (donning gloves on room entry and handwashing). The new nurses made 2 to 3 times more mistakes and adhered 4 to 5 times less to biosafety practices as compared with the more experienced nurses. Again, these observations reveal that it is crucial to worker, patient, and public health to include biosafety/biosecurity training in the university nursing curriculum prior to graduation.
Regulations
The Moroccan Code of Work contains the general principles of prevention such as evaluating and avoiding risks, adapting the work to the employee (workstations, work equipment, methods of work, etc). The code recommends the replacement of what is dangerous by what is not dangerous or, at the very least, less dangerous and the application of a plan for prevention (integrating technology, work organization, conditions of work, social relationships, and environmental factors).22,23
Despite these general safety regulations, Morocco currently has no national biosafety or biosecurity specific legislation, regulation, and guidelines. 24 However, Moroccan law prohibits the employer from allowing employees to use products or substances or apparatus or machinery recognized by the competent authority as being likely to harm their health or to compromise their security.22,23 It is the same with the GPEA (Guide of Good Practice of the Execution of Biological Analyzes), which stipulates in chapter 1, paragraph 5, that the personnel of the laboratory must be qualified, trained, and informed of the tasks performed. 25 It is in this context that public and private institutions are obliged to train all employees likely to be exposed to a microbiological work environment and give them sufficient training and knowledge of the biological agents present in the activity. 22
Biosafety Curricula
Safety training should begin early in the training curriculum with theoretical and practical courses and awareness sessions. Biosafety and biosecurity must be introduced into the general safety policy and become part of the work culture.26,27 Safety and biosafety at the workplace should be integrated into and modeled in undergraduate and graduate courses. Instilling these principles into future biosafety professionals and scientists before they enter the workforce would help establish an integrated culture of safety in containment facilities. 28 Ideally, professional biosafety associations, working with institutes and universities, would design and offer formal biosafety modules as a proactive way to shift from passive training to active instruction for the next generation of nurses. Also, they would create and implement a biosafety and biosecurity curriculum that culminates in a nationally or internationally recognized diploma. 29 This approach would reach students at a formative period in their professional training and help ensure that biosafety best practices become part of the backdrop of these individuals’ future work routines.
Training
The WHO, various other government agencies, and a few private-sector organizations provide biosafety and biosecurity support and training throughout the world. Their efforts would be exponentially enhanced if the training materials developed for these efforts were consolidated and more widely used through the national, regional, and international biosafety associations.
In its report of 2016, the WHO focused on the lack of biosafety and biosecurity training of professional health care workers in Morocco. This quantitative and qualitative deficiency is due to certain problems inherent in training programs, teaching methods, and the location of internships. 30
In Morocco, biosafety training is carried out at some institutes that initiate an accreditation or qualification process. 24 But it should also be integrated into national conferences, seminars, audiovisual presentations, laboratory exercises, and other aspects of the academic experience.
Indeed, rigorous biosafety learning is required to update new graduates, and the seriousness of biosafety and the learning strategy should be discussed and enforced at all levels.3,31 The results of this study suggest that new graduates should only work in lower containment areas of the institution until they are trained and qualified in biosafety practices.
The WHO report of 2016 recommended that the Moroccan government establish or enhance a national training program in basic and continuous biosafety and biosecurity practices and review competencies on a regular basis. Two national associations of biosafety and biosecurity exist in Morocco: the Moroccan Biosafety Association (MOBSA) and the Moroccan Association of Biosafety (AMBS). Both are members of the International Federation of Biosafety Associations (IFBA). 32 Every year, each association organizes training workshops in the kingdom’s facilities and universities for the benefit of students in medicine, pharmacy, veterinary medicine, dental surgery, and biology.
Conclusion
The study shows gaps in the biosafety behaviors of new graduates of nursing schools in the daily practice of their profession. As a result of this study, it is recommended that new nursing graduates should be required to accumulate 3 months of work in conventional units before being assigned to BSL-2 or BSL-3 units, receive specialized hands-on training to prepare them for working in Level 2 or 3 areas, and be required to pass a proficiency exam in critical biosafety tasks prior to working with high-risk patients.
Footnotes
Acknowledgments
The authors thank the participants in the MENA VI Biosafety meeting organized by Sandia National Laboratories and International Biological Threat Reduction between November 7 and November 10, 2017, for the discussion and the validation of the study: Eric Cook Neil, William J. Pinard, Karen L. Hopkins, Anita Romero O'Brien of Global and Biological Security, Sandia National Laboratories and the US Department of State's Biosecurity Engagement Program; Betsy Matos, Iowa State University, Dave Harbourt, United States Army Medical Research Institute of Infectious Diseases, USA; Heather Blair, Colorado State University, USA; Luis Alberto Ochoa Carrera, Institute for Epidemiological Diagnosis and Reference, Ministry of Health, Mexico; Marian Downing, Biological Safety Project Manager for Alliance Biosciences and ABSA Past-President (2015); Melissa Morland, University of Maryland, Baltimore, USA; and Patrick Condreay, pc Biosafety Consulting Services, LLC, Carrboro, NC, USA.
Ethical Approval Statement
The ethics committee of the Mohammed 5 Military Hospital gave its approval for the performance of this study.
Statement of Human and Animal Rights
Authors certificate that the procedures and the experiments done respect the ethical standards in the Helsinki Declaration of 1975, as revised in 2000 and 2008, as well as the national law.
Statement of Informed Consent
All participants in this study have provided written informed consent.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The studies reported in this publication were supported by a grant from US Department of States Biosecurity Engagement Program International Twinning Project.
Prior Presentation
This work was presented as an oral platform presentation at the 61st Annual Biological Safety Conference, Charleston, South Carolina, October 2018.
