Abstract
Introduction:
National Institutes of Health (NIH) defines gene therapy as an experimental technique that uses genes to treat or prevent disease. Although gene therapy is a promising treatment option for a number of diseases (including inherited disorders, some types of cancer, and certain viral infections), the technique remains risky and is still under study to make sure that it will be effective and safe.
Methods:
Applications of viral vectors and nonviral gene delivery systems have found an encouraging new beginning in gene therapy in recent years. Although several viral vectors and nonviral gene delivery systems have been developed in the past 3 decades, no one delivery system can be applied in gene therapy to all cell types in vitro and in vivo. Furthermore, the use of viral vector systems (both in vitro and in vivo) present unique occupational health and safety challenges. In this review article, we discuss the biosafety challenges and the current framework of risk assessment for working with the viral vector systems.
Discussion:
The recent advances in the field of gene therapy is exciting, but it is important for scientists, institutional biosafety committees, and biosafety officers to safeguard public trust in the use of this technology in clinical trials and make conscious efforts to engage the public through ongoing forums and discussions.
Introduction
Gene therapy involves altering the genes inside your body’s cells in an attempt to treat or stop disease. 1 Genes contain your DNA that code for much of your body’s form and function, from making you grow taller to regulating your body systems. 2 Genes that do not work properly can cause disease. Gene therapy replaces a faulty gene or adds a new gene in an attempt to cure disease or improve your body’s ability to fight disease.3 -5 This powerful technology is currently used to treat or improve the health condition of the patient by modifying the patient’s cells genetically.6,7 It provides a unique approach to address both inherited and acquired diseases by delivering a therapeutic gene material and its associated regulatory elements into the nucleus to correct the loss of function caused by mutation or express the deficient gene product at physiologic levels.8 -12 The use of this technology requires assessing health and environmental risks and further stresses the importance of biosafety governance in risk assessment and mitigation.5,13 -18
In the present day, both preclinical and clinical settings have used both viral and nonviral vectors in innovative modifications and applications.1,19 These vector systems offer the potential for efficient gene delivery or can act as a vital tool or vehicle to treat genetic disease.2,20,21 As of 2019, almost 2600 gene therapy clinical trial studies have been completed, are ongoing, or have been approved worldwide.22,23 Furthermore, 232 gene and cell therapy trials have been approved worldwide, and 4 gene therapy products that were approved by the Food and Drug Administration (FDA) are available in the United States.15,22,23 The majority of these gene therapy trials, almost 70%, are based on viral vectors.15,24 Although transfer of genetic material into cells may be accompanied through nonviral vector-based delivery systems, we will be evaluating only the most common biosafety issues associated with viral-based vector systems in this review.
Viral vectors have generally proved efficient tools for gene delivery to target cells/tissue, a critical aspect of achieving therapeutic efficacy. 25 Furthermore, viral vector expression cassettes can be engineered with elements to enhance target specificity and increase transgene expression.12,26 These features have been explored and exploited to develop efficient methods for the delivery of genes of interest into mammalian cells. However, a vast repertoire of viral vectors exists with profoundly different properties. To date, 5 main classes of viral vectors have been tested for clinical applications.25,27 These include retroviruses (RV), adenoviruses (AV), adeno-associated viruses (AAV), lentiviruses (LV), and herpes simplex viruses (HSV).8,28 Although many viral vectors are safe and effective delivery vehicles for clinical gene therapy, some viral vectors are considered risky for potential oncogenesis.3,29 Furthermore, some viral vectors (eg, Adenovirus vector, Herpesviral vector) are toxic and can be immunogenic. 22
Using viral vector systems for gene therapy are promising treatment options for several diseases. However, these methods remain risky and are still under study to ensure safety and efficacy during clinical trials. Additionally, there are limited resources available to institutional biosafety committees (IBCs) and health care providers when it comes to risk assessment and developing procedures to minimize risk and exposure in a research/clinical setting. This review article focuses on the viral vector systems utilized for gene therapy and some of the biosafety challenges associated with each given that there are no consensus guidelines for working with them in a research/clinical setting because it is dictated by risk assessment.
Viral Vectors
The 5 main classes of viral vectors can be categorized in 2 groups according to whether their genomes integrate into host cellular chromatin (oncoretroviruses and LV) or persist in the cell nucleus predominantly as extrachromosomal episomes (AAV, AV, and herpes viruses).6,30 The choice of viral vectors for clinical use depends on the efficiency of transgene expression, ease of production, safety, toxicity, and stability. 31 Furthermore, the types of vectors are represented by both RNA and DNA viruses with either single-stranded (ss) or double-stranded (ds) genomes. 8 The National Institutes of Health (NIH) Recombinant DNA Guidelines and the World Health Organization (WHO) Laboratory Biosafety Manual categorize infectious agents into risk groups for laboratory research (Risk Groups 1-4). 23 The risk group informs about the level of containment needed to minimize risk during the handling of these infectious agents as designated by its biosafety level. 23 The Centers for Disease Control and Prevention (CDC), WHO, and NIH have published recommendations for work at different biosafety levels (BSL-1 to BSL-4) for wild-type virus and viral vector handling in a laboratory research setting. When viral vector systems are used in gene therapy experiments in a clinical setting, these recommendations are adapted to do a comprehensive risk assessment and for developing a mitigation plan.19,23 As with risk group classification, biosafety levels differ by viral vector systems. Historically, IBCs have been responsible for reviewing any risks associated with viral shedding and insertional mutagenesis during the protocol review process. 32 The main groups of viral vectors applied for gene therapy and their recommended risk group/biosafety level are summarized in Table 1.
Viral Vectors Commonly Used in Gene Therapy and Their Recommended Risk Group/Biosafety Level.a
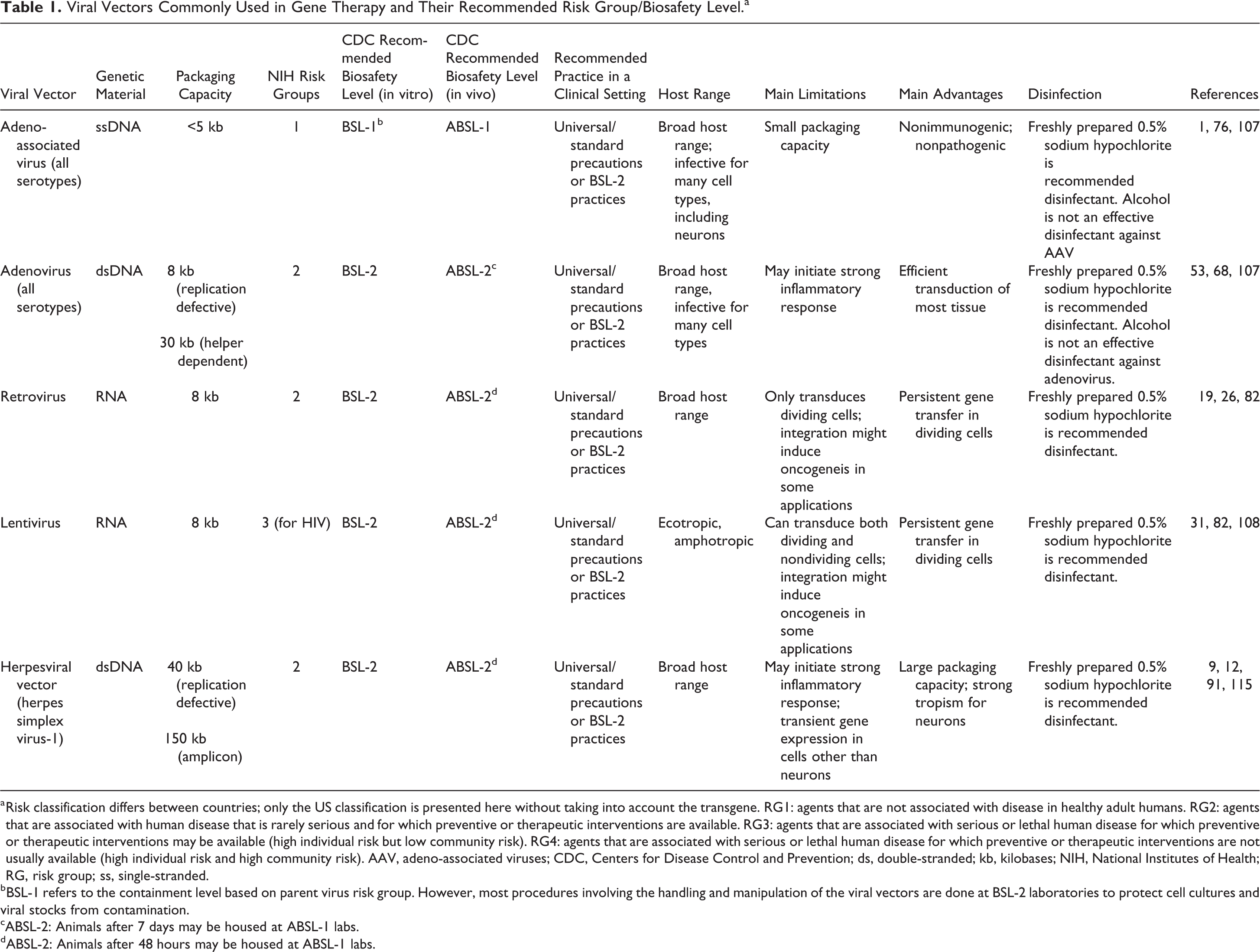
Risk classification differs between countries; only the US classification is presented here without taking into account the transgene. RG1: agents that are not associated with disease in healthy adult humans. RG2: agents that are associated with human disease that is rarely serious and for which preventive or therapeutic interventions are available. RG3: agents that are associated with serious or lethal human disease for which preventive or therapeutic interventions may be available (high individual risk but low community risk). RG4: agents that are associated with serious or lethal human disease for which preventive or therapeutic interventions are not usually available (high individual risk and high community risk). AAV, adeno-associated viruses; CDC, Centers for Disease Control and Prevention; ds, double-stranded; kb, kilobases; NIH, National Institutes of Health; RG, risk group; ss, single-stranded.
BSL-1 refers to the containment level based on parent virus risk group. However, most procedures involving the handling and manipulation of the viral vectors are done at BSL-2 laboratories to protect cell cultures and viral stocks from contamination.
ABSL-2: Animals after 7 days may be housed at ABSL-1 labs.
ABSL-2: Animals after 48 hours may be housed at ABSL-1 labs.
Adeno-Associated Virus Vectors
Adeno-associated virus vectors are nonpathogenic parvoviruses, consisting of a linear single-stranded DNA (ssDNA) genome of approximately 4.7 kilobases (kb), with two 145 nucleotide-long inverted terminal repeats (ITRs) at the termini.33,34 The virus does not encode a polymerase and therefore relies on cellular polymerases for genome replication.
19
The AAV genome consists of 3, open-reading frames (ORF) flanked by ITRs.
35
The ITRs function as the viral origin of replication (
AAV is one of the most common vectors used in gene therapy. 19 One of the primary reasons for using AAV is that AAV has a long-term and efficient transgene expression in various cell types in many tissues such as liver, muscle, retina, and the central nervous system (CNS).43 -45 However, the application of AAV has some associated disadvantages. The preexisting immunity to human AAV vectors is comparable to the AV, and the integration into the host genome (if it occurs at all) is random, which can lead to accidental activation or inhibition of endogenous gene expression. 46 Another substantial limitation of AAV vectors is their small packaging capacity that is generally considered to be <5 kb. 19 This limitation makes AAV vector design for diseases involving larger genes challenging or impossible.38,47 Additionally, various AAV serotypes have exhibited remarkably different expression patterns due to differences in cell entry and intracellular activities. 48 For instance, AAV1 is suitable for expression in skeletal muscle and the retina,49,50 whereas AAV5 transduces neuronal and lung cells efficiently.51,52 By contrast, since AAV2 has long-term, albeit poor, expression levels, the discovery of novel rhesus monkey serotypes AAV7 and AAV8 might be of interest for human gene therapy applications.53,54 In particular, AAV8 demonstrated higher levels (up to 100-fold higher) of factor IX expression in liver cells than for any other AAV serotype tested. 55 Moreover, preimmunization with different AAV serotypes did not compromise the expression levels. 55
AAV vectors have an excellent safety record in clinical trials and preclinical animal studies.2,56 AAV is generally handled at BSL-1. 57 In a clinical setting, universal precautions or standard precautions are utilized while working with AAV vectors. Exceptions include synthetic or recombinant AAV constructs created in the presence of a helper virus and those that contain a potentially harmful transgene. 58 While AAV can infect humans and other primate species, AAV is not known to cause disease. 19 Approximately 80% of humans are seropositive to AAV strains. 40 However, repeated administrations, which can trigger an immune response, may be a limiting factor. 59 Applying a different AAV serotype for each readministration typically alleviates this reaction. 60 Moreover, several gene clinical trials have revealed that AAV vectors do not stimulate a heightened immune response. 60 In some animal models, the integration of recombinant AAV increased the incidence of tumor formation, but this association has not been observed in humans. 61 Although ineffective in replication as an isolated agent, AAV has the potential for replication competence in the presence of a helper plasmid or virus, and stock solutions should be tested for the presence of other agents. 62
Adenovirus Vectors
Adenoviruses are a class of DNA viruses with a double-stranded 34 kb to 43 kb genome, which employs alternative splicing to encode genes in both the sense and antisense orientations. 63 The AV genome is flanked by 2 ITRs and contains 8 transcription units. 64 The early regions (E1A, E1B, E2, E3, and E4) are the first regions transcribed and encode proteins involved in activating transcription of other viral regions and altering the cellular environment to promote viral production. 64 The late regions (L1-L5) are transcribed from an alternatively spliced transcript. 65 Similar to recombinant AAV vectors, the AV genome remains in an extrachromosomal form following infection. 66 Humans have 51 different serotypes of AV; 45% to 80% of the population harbors neutralizing antibodies against Ad5, the most prevalent, due to natural infections, which typically date back to infancy. 67 Standard recombinant adenoviral vectors have the E1 unit deleted, but the removal of E3 can create vectors capable of packaging up to 7.5 kb of foreign DNA. 68 Vectors with both E1 and E4 units deleted result in lower immune responses. 69 Finally, in helper-dependent, or gutless, adenoviral vectors, all adenoviral genes are deleted, and a helper virus provides adenoviral genes for production. 70 Although these adenoviral genes have a carrying capacity of more than 30 kb, they are currently challenging to manufacture. 71 Another concern with adenoviral vectors, in general, is that the particles themselves elicit cellular inflammatory responses as opposed to relatively inert AAV virions. 72
Compared with other viral gene delivery systems, adenoviral vectors offer significant advantages. First, the AV is the most effective means of delivering genes in vivo as most human cells express the primary AV receptor and the secondary integrin receptors. 73 Thus, human cells are easily infected with AV vectors and consequently yield high levels of the transgene expression. Second, the development of gutless adenoviral vectors allows us to circumvent antiadenoviral vector immunity. 63 The AV vector now represents 1 of the 3 major viral vector categories in the gene therapy toolbox, along with AAV and lenti/retrovirus vector systems.19,22 Interestingly, some of the inherited shortcomings of AV, such as immunity evoked against the AV capsid and low-level expression of AV genes, may now prove beneficial for the development of anticancer immunotherapies, where inducing immunity against cancer or directly killing the cancer cell is the goal.74,75 The combined immunity against the AV and the short time of expression may prove beneficial for using AV to develop potential vaccines.76,77
Work with AV vectors is done using BSL-2 practices. 78 In a clinical setting, universal precautions or standard precautions are utilized while working with AV vectors. 23 Although AV cause mild respiratory disease or conjunctivitis in healthy adults, they are notoriously immunogenic. 6 Several episodes of inflammatory responses to AV vectors in clinical trials have been documented, including 1 death after direct injection of an adenoviral vector into the hepatic artery. 79 Some first-generation adenoviral vectors, which retain a considerable proportion of the genome, have initiated dose-dependent apoptosis of various target cells in vitro and in multiple mouse models in vivo, indicating a potential for direct cytotoxicity.24,80 The widespread distribution and tropism of AV support the potential for intracellular recombination or complementation of the vector with previously acquired wild-type AV in either packaging cell lines in vivo, thus making the adenoviral vector replication competent. 81 This risk can be reduced substantially by using cell lines with decreased or no homology between vector and helper sequences. Another risk-reduction strategy is regular quality control checks of viral vector stocks for possible low levels of contamination with the replication-competent virus. Currently, researchers have determined these threshold levels arbitrarily because no standards govern thresholds to date. Further improvements regarding reactions with the immune system, viral longevity, vector packaging capacity, and contamination will be pursued as we continue making breakthroughs using adenoviral vectors in gene therapy.
Retrovirus Vectors
Retroviruses are relatively complex enveloped RNA viruses with a diploid ssRNA genome and consist of at least 4 genes,
Retroviral vectors have been extensively used to deliver therapeutic genes in the context of gene therapy, clinical applications for monogenic disorders, cancer, and infectious diseases, providing a stable and efficient expression of the transgene to patients.30,89 -91 Retroviruses have numerous advantages over other vectors. The most significant advantage that retroviral vectors offer is their ability to transform their ssRNA genome into a dsDNA molecule that stably integrates into the target cell genome. 85 This feature enables the retroviral vectors to modify the host cell nuclear genome permanently. Currently, retroviral-vector-mediated gene transfer has been reinvigorated with the development of a new retroviral vectors class derived from LV. 8 The LV have the unique ability among RV to infect noncycling cells. 92 Vectors derived from LV have provided a substantial leap in gene-editing technology and present new avenues to achieve significant levels of gene transfer in vivo. 19
Containment levels for the use of retroviruses are determined based on the cell types they infect. BSL-1 is appropriate for RV that do not infect human cells. BSL-2 is necessary if they are used to infect human cells. 56 The primary safety risk with the use of RV arises from their ability to integrate into the host cell chromosome, which raises the possibility of insertional mutagenesis and oncogene activation. 93 Generation of replication-competent RV in target cells or tissues is the primary risk related to the use of retroviral vectors. 94 Assessment of this risk is critical in determining the safety associated with the use of retroviral vector systems.
Furthermore, the target cell range of the vector is also a safety issue. Incorporation of a virus envelope that can infect cells from multiple species increases both the risk of generating replication-competent retroviruses and the potential danger of any resulting virus, which could spread from one species to another.30,94 Future studies that utilize retroviral vectors in gene therapy experiments must explore safer strategies that focus on the risks associated with in vivo recombination, generation of mosaic RV, and long-term storage of viral genetic information.
Lentivirus Vectors
Lentiviruses, a type of RV, consist of a single-stranded positive-sense RNA sequence that is transcribed into a DNA and integrated into the host genome, causing persistent infection.
95
Most lentiviral vectors (LVVs) are derived from HIV-1 and retain the capability to integrate into the genome of infected cells.
96
Wild-type HIV genome with all of its genes and regulatory elements provides the backbone for LVVs.
96
In the first-generation LVVs, the envelope protein and the
Lentiviral vectors have become very popular for clinical applications due to their ability to more efficiently transduce nonproliferating or slowly proliferating cells, such as CD34+ stem cells. 99 Gene transfer using LVVs into CD34+ HSCs has been used to treat several genetic diseases, including β-thalassemia, 100 X-linked adrenoleukodystrophy, 101 metachromatic leukodystrophy, 102 and Wiskott-Aldrich Syndrome. 103 Recently, multiple clinical trials used third-generation, self-inactivating LVVs to introduce genes into hematopoietic stem cells to correct primary immunodeficiencies and hemoglobinopathies. 104 The LVVs have been used to alter T cells by introducing genes to generate immunity to fight cancer through the delivery of chimeric antigen receptors (CARs) or cloned T-cell receptors. 105 CAR T-cell therapies developed using LVVs have been successful in the clinical setting in patients with B-cell malignancies. This has resulted in the regulatory approval of the first genetically engineered cellular therapy using these vector systems. 106
The use of LVVs in research is still associated with potential risks, and the long-term safety of these clinical interventions is still being evaluated. While lentiviral systems are derived from HIV, their organization across numerous plasmids and the deletion of many HIV proteins lowers the likelihood of generating replication-capable virus.85,107 These vector systems are handled at BSL-2. The limitations of using LVVs in clinical trials today are mainly due to insufficient methods for the production of high-titer virus stocks and the safety concerns related to their origin from HIV, despite the engineering of packaging cell lines and deletions of genes required for viral replication.90,108 One approach to address safety issues has been to develop LVVs incapable of replication in human cells. Although LVVs are less associated with insertional mutagenesis than other RV, these vectors still provide evidence of off-target effects. 109
Herpesviral Vectors
Herpes simplex virus is an enveloped virus with a dsDNA genome more than 150 kb in length, which is divided into long and short unique segments (UL and US, respectively) and flanked by inverted repeated sequences (TRL/IRL and TRS/IRS). 110 The HSV genome encodes approximately 90 genes, almost half of which are nonessential and can be eliminated in recombinant vectors. 111 Currently, gene delivery and gene therapy applications use 3 types of HSV vectors. 111 First, amplicons are gene delivery systems packaged through transfection into producer cells of plasmids containing the HSV origin of DNA replication, HSV cleavage-packaging sequences, and transgene(s) of interest. 47 The plasmids form head-to-tail linked concatemers that are packaged into the viral particles, which can, in general, accommodate large fragments of foreign DNA. 47 In a second system, the deletion of genes necessary for the lytic cycle of HSV can create less toxic, replication-defective vectors that elicit a smaller immune response than wild-type HSV vectors. 112 Deleting various combinations of the immediate, early genes that otherwise lead to the expression of the HSV infected cell proteins (ICP0, ICP4, ICP22, ICP27, and ICP47) creates several versions of replication-defective HSV vectors. 112 Finally, replication-competent HSV vectors are attenuated versions of HSV that contain the genes required for replication in vitro but lack the genes necessary for replication in vivo. 113 Replication-competent vectors serve as oncolytic therapies and vaccines, where the set of deleted genes must lead to the appropriate balance of replicative attenuation for safety and lytic activity for tumor toxicity or vaccine immunogenicity, respectively. 113
HSV-1 is a neurotropic virus that displays numerous necessary adaptations to the nervous system. With regard to the neurological application, the design of gene therapy vectors can exploit each adaptation rationally. 114 HSV vectors containing latency-active promoters LAP1 and LAP2 have been engineered for long-term expression (up to 300 days) in the peripheral nervous system. 114 HSV vectors have also found applications in the CNS. 115 However, because wild-type HSV causes encephalitis, developing nontoxic replication-defective vectors with deletions in the IE genes (ICP4, ICP22, ICP27) 114 is essential. Furthermore, recent breakthroughs in the field of HSV-1 technology authorize and support the use of HSV-1 as vaccine vectors for the delivery of foreign antigens. 116 In particular, HSV vectors exhibit several advantages for prophylaxis against viral infections. 115
HSV is transmitted by direct contact and mucous membrane exposure to body excretions and respiratory droplets. 113 Although molecular alterations to the viral genome have dramatically reduced virus-associated risks, HSV remains a known human pathogen with inflammatory and immunomodulatory effects. BSL-2 is appropriate for many constructs of HSV. 19 Despite high seroprevalence of both HSV1 and HSV2 in the United States (approximately 53% and 16%, respectively), few robust data that support in vivo recombination or complementation are available. 70 However, fatal meningoencephalitis can occur, especially in immunocompromised persons. 113
Recommendations for Policies and Procedure Development for Comprehensive Risk Assessment for Gene Therapy Research Utilizing Viral Vector Systems
Safe use of viral vector systems in human gene therapy research is a major concern for most IBCs in a research/clinical setting because there is no single document that summarizes the guidelines of working with them. The NIH Recombinant DNA guidelines and the fifth edition of
The IRB review is necessary to protect the rights and welfare of research subjects in clinical investigations. 117 The IRB requires a comprehensive review and approval for any research with human subjects supported by the U.S. Department of Health and Human Services (HHS) or regulated by FDA.118,119 The IRB review is necessary for research supported or conducted by any of the federal agencies subscribing to the Common Rule, for research on products regulated by FDA, and for research conducted by investigators at any institution giving Federalwide Assurance (eg, universities assuring the federal government that all of their research will conform to federal rules on human subjects research).120,121 An IRB is also responsible for approving amendments to a study and review a progress report at least yearly. 122 An IRB has the authority to approve, require modifications to (as a condition of approval), or deny approval to gene therapy research and informed-consent documents. 119 Additionally, an IRB has the authority to terminate or suspend any study for noncompliance. 123 The revised NIH Guidelines (April 2019) eliminate the RAC review, protocol registration, and reporting requirements associated with gene therapy research. 124 In addition, a frequently asked question blog on the NIH website outlines the separation of the IBC with the IRB during the human gene therapy protocol review process. 125 The revised NIH Guidelines highlight the critical role of IBCs in reviewing gene therapy research studies. 124 IBCs are now mandated by the NIH Guidelines, Section III-C, to undertake a review of safety risks at the level of the investigators’ institutions (eg, universities and research centers) for all forms of human gene therapy research. 124 This highlights the need for IRBs to collaborate with institutional IBCs during the protocol risk assessment review process. As IBCs play a critical role in providing oversight of gene therapy research, it is recommended that institutional compliance committees (eg, IRB, IBCs) play a significant role in ensuring compliance with education, training, risk assessment, and policy development of research involving human gene therapy.
During the protocol review of human gene therapy research, the IBCs play a critical role in reviewing risks associated with viral shedding. Shedding in the field of gene therapy means dissemination of the gene therapy product through the excreta of the treated patient or subject. 126 Despite the substantial number of clinical gene therapy trials that have been conducted worldwide, there is currently no summary of actual shedding data available for different viral vector systems. There have been studies that have shown that some of the vectors used in gene therapy research can be excreted from the body for a least a week after injection. 32 This shedding of the vector can be found in the blood, urine, saliva, and stool for up to a week following injection. 127 However, there are limited peer-reviewed publications that outline shedding data of different viral vector systems when administered to human research subjects. Most of the Investigator Brochures and also the product insert of ZOLGENSMA recommend 30-day universal or standard precautions.126,128 In general, most health care facilities recommend universal/standard precautions with patient material between 14 and 30 days after administration for both health care staff and direct family members, dependent on viral shedding analysis. 128 Additionally, instructions are provided to family members and caregivers to practice good hand hygiene for a minimum of 2 weeks after the injection. 128 This requires washing hands with soap regularly and using appropriate protective gloves if coming into direct contact with patients’ bodily fluids and waste. 128 This provides an insight into the occurrence of shedding of vectors in clinical practice and can offer support for environmental risk assessment in future clinical gene therapy applications using different viral vector systems so that a consensus criteria can be developed. Some recommendations for the Risk Assessment process for IBCs for gene therapy research using Viral Vector Systems is summarized in Figure 1.
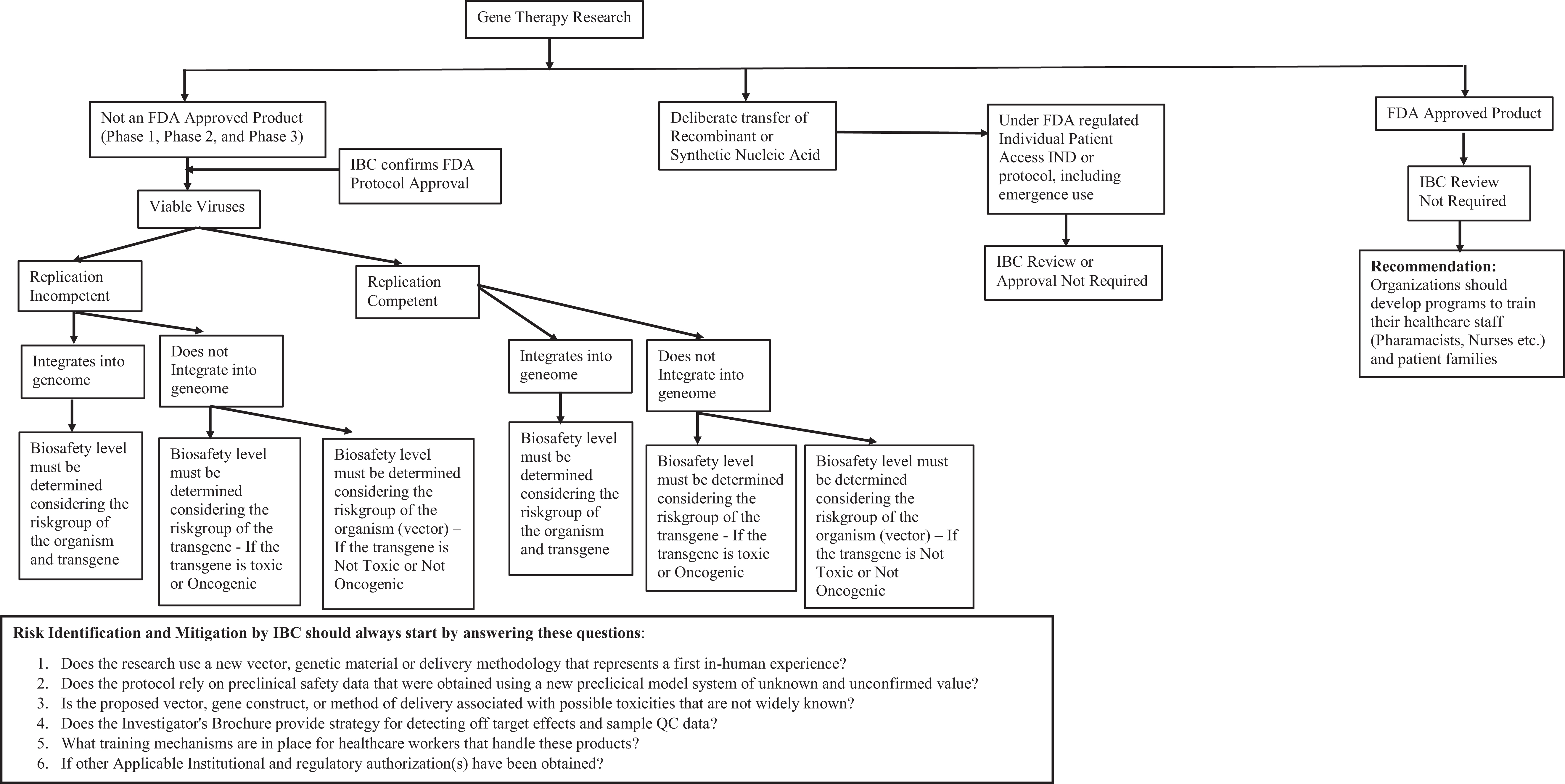
Risk assessment process for institutional biosafety committees for gene therapy research using viral vector systems.
Luxturna (Spark Therapeutics), the first virally delivered gene therapy, was approved in December 2017. 129 In August 2017, the FDA also approved the use of Tisagenlecleucel, marketed as Kymriah (Novartis), which is a treatment for B-cell acute lymphoblastic leukemia. 130 Additionally, in October 2017, Yescarta (Kite Pharma Inc/Gilead) became the first CAR T cell therapy approved by the FDA for use in adult patients. 23 Most recently in May 2019, the FDA approved Onasemnogene abeparvovec, sold under the trade name Zolgensma, a gene therapy medication to treat spinal muscular atrophy in children. 131 As more gene therapy products are getting approved by the FDA, future research should focus on developing biosafety recommendations and policies for health care personnel (physicians, nurses, fellows, pharmacists, etc) handing gene therapy products that should encompass the following: storage, cleaning, preparation, dispensing, accidental spillage/exposure, personal protective clothing, equipment (eg, biosafety cabinets-type), disinfection and decontamination procedures, transport, and disposal of waste. Additionally, a framework for training health care workers and families of patients using these products should be developed.
Conclusions
In this review, we highlight some of the biosafety challenges associated with the current viral vector systems that have applications in gene therapy research. Current advancements in the field of gene therapy have opened the door to cures at a molecular level for many genetic diseases. The design of new experimental viral vectors and emerging technologies highlight the critical role of biosafety professionals in managing gene therapy research protocols, identifying risk level, and developing acceptable policies predominantly in the absence of consensus framework for the risk assessment process.
Footnotes
Acknowledgment
We thank Melody Davis at The Abigail Wexner Research Institute at Nationwide Children's Hospital for critical reading of the manuscript.
Ethical Approval Statement
Not applicable to this study.
Statement of Human and Animal Rights
Not applicable to this study.
Statement of Informed Consent
Not applicable to this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclaimer
The views presented in this publication are those of the authors and do not necessarily reflect the position of the associated institutions.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
