Abstract
Introduction:
The performance of 2 disinfectant chemicals, peracetic acid (PAA) and hypochlorous acid (HOCl), was evaluated using a Venturi-nozzle-based light decontamination system (LDS) for delivery. The atomization equipment combined low-pressure air and disinfectant via a handheld lance, producing a fine, dense aerosol. A range of microorganisms, including Bacillus cereus and Bacillus anthracis (Vollum) spores, were used as test challenges to evaluate chemicals and equipment.
Methods:
The tests undertaken included assessments over fixed and variable exposure times, use of multiple surface materials, and a live agent challenge.
Results:
Over a fixed-time exposure of 60 minutes, aerosolized PAA gave 7- to 8-log reductions of all test challenges, but HOCl was less effective. Material tests showed extensive kill on most surfaces using PAA (≥6-log kill), but HOCl showed more variation (4- to 6-log). Testing using B. anthracis showed measurable PAA induced spore kill inside 5 minutes and >6-log kill at 5 minutes or over. HOCl was less effective.
Discussion:
The results demonstrate the importance of testing decontamination systems against a range of relevant microbiological challenges. Disinfectant efficacy may vary depending on product choice, types of challenge microorganisms, and their position in a treated area. The most effective disinfectants demonstrate biocidal efficacy despite these factors.
Conclusion:
The data confirmed PAA as an effective disinfectant capable of rapidly killing a range of microorganisms, including spores. HOCl was less effective. The LDS system successfully delivered PAA and HOCl over a wide area and could be suitable for a range of frontline biosecurity applications.
Introduction
Surface and air decontamination take many forms across different settings and may require the removal of microorganisms and/or chemicals residues. Within the built environment, the challenges faced by frontline decontamination operatives may be considerable and are influenced by the location of the contaminants, accessibility for those tasked with remediation, the types of surfaces affected, and the levels at which the contaminant is present in the environment.1,2
In the past, automated decontamination systems, designed primarily for room or interior vehicle use, have been widely reported.3 -5 Evaluations of systems designed for wider area use have been less commonly described, with the focus usually placed on chemical and radiological decontamination and with knowledge gaps remaining for microbiological applications. 6
The aim of the current study was to assess the biocidal performance of 2 readily available chemicals with known disinfectant qualities, peracetic acid (PAA) and hypochlorous acid (HOCl). A backpack-type delivery system was evaluated for chemical delivery and comprised a Venturi-based handheld lance powered by a compressed air cylinder. This allowed up to 4 L of liquid disinfectant to be delivered as a fine mist within 2 minutes.
To use this equipment safely and effectively in a simulated or real-life setting, the operator must be fully protected against both the aerosolized disinfectant and any environmental hazard being treated. The operator’s need for appropriate personal protective equipment (PPE) was therefore an integral part of this testing, and these requirements are also described.
The disinfectant testing used a range of microbiological challenges and materials to assess the efficacy of the disinfectants and the performance of the delivery system. The 2 aerosolized chemicals had not been tested previously in a side-by-side fashion against such a range of challenge microorganisms or using this type of delivery approach.
The testing strategy therefore sought to achieve the following:
Establish the biocidal efficacy of 2 aerosolized chemicals against a range of microbiological challenges, initially over an exposure period of 1 hour. Further assess the 2 chemicals over various timed exposure periods, using a single bacterial spore type. This included an additional 2-step (tandem) delivery approach to determine an optimal effective contact time. Assess the 2 chemicals against a range of coupon materials to determine whether material composition influences disinfectant performance Determine the efficacy of the test chemicals against live agent Bacillus anthracis str. Vollum within a Containment Level 3 (CL-3) laboratory to draw comparisons between live agent data and other data from Bacillus spp.
Methods
Disinfectant Chemicals Under Test
Earlier UK government-funded testing with PAA had assessed PAA treatment concentrations of 0.5%, 1%, 1.5%, and 2% (data not shown) and indicated that a 2% preparation should be used for aerosolized delivery rather than a lower concentration. 7 The 2% PAA was made up from a stock solution of ∼39% to 40% PAA (Sigma-Aldrich), diluted with deionized water immediately prior to each test. Four liters of 2% PAA was sufficient to fill the reservoir of the backpack spray system.
The HOCl was a commercial product supplied ready to use for surface disinfection at 0.5% (Biomimetics Health Industries UK Ltd). The product was regarded by the supplier as suitable for the type of testing planned and was poured directly, undiluted into the backpack reservoir immediately prior to testing.
Disinfectant Delivery Equipment
The light decontamination system (LDS) can use a range of liquid chemical decontaminants for both airborne and surface treatments. 8 The 4 L of product per load is atomized via a pressurized, handheld lance. The system is primarily designed for emergency first-responder personnel to work in confined and remote environments where the deployment of automated decontamination systems may be impractical.
The directional control, air turbulence effects, and a claimed 1- to 2-µm droplet size produced by the handheld lance are intended by the manufacturer to keep the droplets in the air long enough for consistent disinfectant surface coverage of the target surfaces. Manufacturer claims extended to a spray projection distance of up to 30 m. The lance system was operated manually, relying on operator action to determine the width of delivery to ensure that target surfaces were effectively treated. The LDS system is designed to allow treatment of non-line-of-sight surfaces, a claim assessed within the test approach used here.
Personal Protective Equipment for the Operator
The operator’s PPE included a PolyProtect 12 CBRN Suit and SARATOGA® Gloves (Blucher, Erkrath, Germany). The suit is a lightweight 1-piece coverall, developed for first responders and designed for use in all climatic conditions. The suit offers protection against chemical and biological contamination.
The SARATOGA® gloves offer the wearer dexterity with chemical vapor and liquid protection. Several layers of sacrificial gloves (nitrile) were placed over the top of each SARATOGA® glove during live agent testing. This prevented any cross-contamination of active biological agents, for example, during lid replacement of sample tubes during multiple timed sample recoveries.
Respiratory protection was provided by a General Service Respirator–Evolution Specialist (GSReS) mask (3M™-Scott™). The GSReS (Figure 1) has self-sealing valves to minimize the risk of contamination during filter canister changes and can be used with any standard (STANAG 4155/ DIN 40 mm) threaded filters. The respirator can also be used with positive pressure breathing apparatus (SCBA or airline supplied) to function as a modular respiratory protection system (MRPS); incorporating modular side fittings and an auto switchable facility between filter use and SCBA.

Respiratory protection: the General Service Respirator Evolution Specialist (GSReS) mask (3M™-Scott™).
For testing carried out using B. anthracis str. Vollum, the GSReS system was configured in MRPS mode using a CBRN 40 mm filter (3M™ Scott™ Propak®) and a SCBA system (379 bar; 9.5-L cylinder; 75-minute use duration) to accommodate the length of the timed experiments. The SCBA was the primary system in use with the filter as a backup.
Challenge Microorganisms Used in Each Test
Fixed duration testing (1 hour)
B. cereus ATCC 14579 was grown at 30°C on manganese-enriched nutrient agar for 5 days, and spores were harvested into phosphate buffered saline (PBS) and washed twice to remove any agar. The preparation was resuspended in PBS containing 10% bovine serum albumin (BSA) and freeze dried (Labylo freeze dryer, Severn Sales, UK). To prepare the stainless-steel test coupons (Figure 2), an aliquot of the freeze-dried preparation was resuspended in sterile distilled water at approximately 2 × 109 cfu.ml-1, and 50-µl aliquots were pipetted onto prepared coupons and dried under ambient conditions within a Class I microbiological safety cabinet (MSC). The preparation conditions gave a final level of BSA in the test product of >5%.

Stainless-steel test coupons presented to allow horizontal, vertical, or inverted positioning.
B. thuringiensis HD-1 Cry-
This spore-forming test organism (commonly referred to as Bt Cry-) was developed and supplied by the Defence Science and Technology Laboratory, Porton Down, UK. Bt Cry- is reported to have the most similar spore structure to B. anthracis of any surrogate spore but is low hazard. 9 Spore preparations were prepared in the same way as for B. cereus spores, with identical levels of BSA.
Geobacillus stearothermophilus
A standard test organism; this commercial test coupon was purchased as a 106 spore preparation on steel coupons (Mesa Labs, Inc., France).
MS2 bacteriophage ATCC 15597-B1 (MS2) has been used by others for decontamination testing work, including fumigation and PAA-based approaches.10,11 MS2 stock was quantified via serial dilution using nutrient broth as the diluent. Each serial dilution (100 µl) was added to 300 µl of a 4-hour Escherichia coli ATCC 15597 culture. The viral-bacterial suspensions were each added to 3 ml of semisolid nutrient agar before being poured over a prewarmed nutrient agar plate. The overlay plates were left to dry for 20 minutes before incubating them at 37°C overnight. The numbers of plaque-forming units in the E. coli lawn were counted, and MS2 virus stock concentration was calculated. A 50-µl aliquot of quantified MS2 stock was pipetted on to prepared coupons.
Mycobacterium fortuitum NCTC 8573 offers a faster growth rate than other mycobacterial species and can be enumerated accurately on agar plates. It has been widely used for UK government disinfectant testing. 12 A broth culture was prepared by growth in Middlebrook 7H9 broth containing 1% Tween 80 and 10% ADC enrichment supplement for 3 days at 37°C. The culture was resuspended in PBS containing 0.3% BSA at approximately 2 × 109 cfu.ml-1, and 50-µl aliquots were pipetted on to prepared coupons. The culture was not freeze dried.
Timed duration testing
For this test, B. cereus and B. thuringiensis HD-1 Cry- were used in the same spore preparations as for fixed duration testing but across a wider range of time periods.
Multiple material testing
This test employed B. thuringiensis HD-1 Cry- only, with spore preparation as described previously. Coupons were prepared in square 2 × 2 cm sections from carpet, glass, finished (painted) plaster, and unplasticized poly vinyl chloride (uPVC). Concrete surfaces were prepared using preautoclaved concrete common bricks with a single brick face marked with 2-cm diameter circles, 4 cm apart. Each concrete brick therefore provided space for a triplicate of samples, which were prepared in 50-µl aliquots as for other materials and loaded and dried on the concrete surface prior to testing.
Live agent testing
This testing was carried out using B. anthracis str. Vollum. The bacterium was cultivated in the presence of manganese salts to produce a spore preparation on solid agar. Surface colonies were harvested, resuspended, and dried down on to steel coupons by air drying. Freeze drying of B. anthracis was not used due to the aerosol exposure risk involved. Organic material was present in the spore preparation as residues from the agar medium, due to the harvesting method used. Although not quantified, this organic residue was deemed equivalent to the BSA soilant present in the other Bacillus spore preparations. A 50-µl aliquot was pipetted on to prepared coupons.
Coupon Placement and Test Facilities
Fixed duration, timed duration, and multiple material testing
All testing, apart from live agent work, was carried out in a semienclosed tented area (3 m × 6 m), located inside a much larger unheated hangar that had front and rear doors open, allowing free movement of outdoor air through the facility. Tests were carried out during the UK winter months of November to February when average outdoor air temperature was between 3°C and 8°C; average humidity for the period was between 84% and 89% (www.worldweatheronline.com). Microbiological challenges, mounted on petri dishes, were placed on impervious plastic shelving located at one end of the tent (Figure 3a). Samples were placed at floor level (on the bottom shelf), at 1.3 m (on the middle shelf), and at 2.1 m (on the top shelf) to provide a stratified component to the test. The Venturi lance-delivering chemical spray was operated from the opposite end of the tent (Figure 3b). The tent was closed with VELCRO® seals during spraying and for the duration of the exposure time. At the end of the appropriate exposure time, microbiological samples were removed from the tent for processing in the laboratory.

(a) Tented structure showing shelving position. (b) Tented structure showing operator position.
The fixed duration test was the first test performed to provide baseline data to assist other testing approaches. The 5 test microorganisms—which for this test did not include B. anthracis—were prepared on 1.5-cm diameter stainless steel coupons. Triplicate coupons of each organism type were placed in 3 orientations (horizontal, vertical, and horizontal upside down) in the 3 locations (floor level, 1.3 m, and 2.1 m). Two percent PAA and 0.5% HOCl (4 L each) were delivered from the backpack system over a period of 2.5 to 3 minutes, and samples were then left in the tent for a dwell period of 1 hour. Three separate test runs were carried out for each chemical.
For the timed duration/tandem delivery testing, B. cereus and B. thuringiensis HD-1 Cry- were prepared on 1.5-cm diameter stainless steel discs. Triplicate coupons of each organism were placed in a horizontal orientation only at a height of 1.3 m. The additional samples required in this test for timed removal made the use of additional positions impractical for a single test. PAA and HOCl (4 L) were delivered from the backpack system, and based on information obtained from earlier fixed duration test results, coupons were exposed to different dwell periods for the 2 chemicals. This was because early data suggested that only PAA were effective enough to work over periods of <1 hour. Sample exposure times were as follows:
PAA: samples removed after 1, 5, 15, 30, and 60 minutes; HOCl: samples removed after 60 minutes.
In addition, for both PAA and HOCl, triplicate samples were exposed to a second (tandem) delivery of 4 L of the same chemical and left for a further 1 hour before processing. In summary, 3 individual test runs were carried out for each chemical and for each time point/chemical delivery schedule.
For multiple materials testing, B. thuringiensis HD-1 Cry- spores were prepared as for steel but on a variety of surfaces and placed at agreed locations (Table 1). Three test runs were again carried out for each chemical.
Material Types and Locations for Material Compatibility Testing Work.
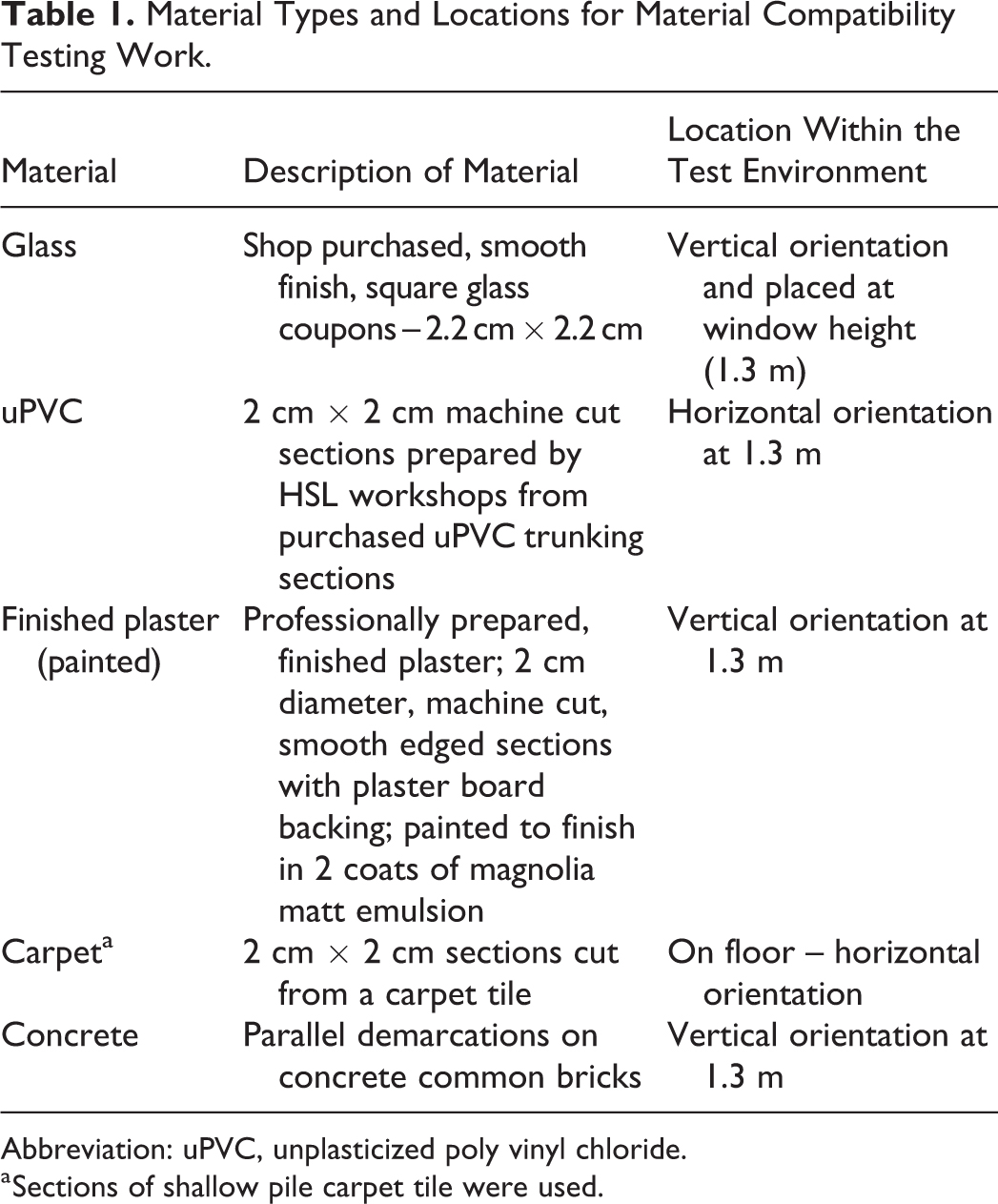
Abbreviation: uPVC, unplasticized poly vinyl chloride.
Sections of shallow pile carpet tile were used.
Live agent testing
Live agent testing was carried out in a 115 m3 CL-3 facility; equivalent to US/European BSL-3. B. anthracis coupons were set up in triplicate in each of the 3 locations in the MSC, 20 cm from the left, 20 cm from the right, and at a central position, all at 20 cm from the back of the MSC. At each location, metal racks held 3 sets of 3 Falcon tubes containing Stop Solution (PBS with 9.5 g.l-1 lecithin, 5 g.l-1 sodium thiosulphate, and 150 ml.l-1 Tween 80) each with lids on but loosened to facilitate easy removal. These sets of tubes were sufficient to allow 3 time-resolved sample collections. A corresponding number of coupons with live agent immobilized on them were placed in front of the Falcon tubes in upturned Falcon tube lids. After chemical fog exposure of required duration, lids were removed from Falcon tubes and Falcon tube lids holding coupons inverted onto the tubes, thus dropping the coupons into “Stop Solution” without any need for direct handling of the coupons. In this way, coupons could be exposed to disinfectant for defined time periods.
This was the only work undertaken in a CL-3 facility and required a more directional delivery of the sprayed disinfectants. This was because for risk assessment purposes, all challenge coupons were positioned in a small area within a Class I MSC rather than on open benches. The power of the spray delivery system was such that the operator stood 4 m away from the coupons to avoid overdelivery of disinfectant chemicals. A short, circular poly-tunnel was constructed—1.5 m diameter, bottom edge 0.8 m above floor level, opening 0.5 m from the operator’s position, and terminating 0.5 m from the front of the MSC. This was used to focus the spray delivery out of practicality, to avoid wider chemical contamination of the CL-3 facility with airborne chemicals. In all other aspects, the Venturi backpack system was used as for other tests.
Processing of Bacterial Test Coupons
Following chemical treatment, microorganisms were recovered from challenge coupons to determine the efficacy of the chemical treatment. In all cases, the numbers of live microorganisms recovered from exposed coupons were compared with those recovered from the unexposed controls and any log reduction calculated. With mixed material types, it is difficult to achieve full recovery of all seeded microorganisms, but using a direct comparison of recovered microorganisms from treated samples and untreated controls, prepared on the same material types, ensured that any measured log-reduction for the treated samples was an accurate representation of microbiological kill due to chemical treatment. Test and control samples were otherwise processed identically.
For the fixed duration and multiple material testing, each control or test coupon was recovered in to 10-ml PBS containing 2 sterile glass beads. For concrete samples, the sample surface was swabbed with a sterile rayon-tipped swab (Sterilin), premoistened with PBS, and the swab was placed in to 10-ml PBS containing 2 sterile glass beads. For the timed duration and live agent testing, coupons were recovered immediately into Stop Solution (PBS containing 9.5 g.l-1 lecithin, 5 g.l-1 sodium thiosulphate, and 150 ml.l-1 Tween 80), with 2 sterile glass beads. Samples were vortex mixed for 1 minute and left to stand at room temperature for 30 minutes (timed duration) or 5 minutes (live agent). Samples were again mixed and immediately serially diluted in PBS and 100-µl aliquots plated on to appropriate duplicate agar plates and incubated as follows:
B. cereus: Nutrient Agar, 30°C, 18 hours; B. thuringiensis HD-1 Cry-: Tryptone Soya Agar, 25°C, 18 hours; G. stearothermophilus: Nutrient Agar, 55°C, 18 hours; M. fortuitum: Middlebrook 7H10 Agar, 37°C, 3 days; B. anthracis: Nutrient Agar, 37°C, 18 hours.
As only a proportion of the total sample was plated out, the limit of detection (LOD) was 50 cfu per test coupon.
Processing of MS2 Bacteriophage Test Coupons
Following chemical treatment, each coupon was recovered into 10 ml nutrient broth containing 2 sterile glass beads. The samples were vortex mixed for 1 minute and left to stand at room temperature for 30 minutes. Each sample was again vortex mixed and immediately serially diluted, and 100-µl aliquots of each dilution were mixed with 300-µl aliquots of a 4-hour culture of the E. coli host. The bacterial-viral suspensions were mixed with 3 ml of semi-solid Nutrient Agar and poured on to Nutrient Agar plates. The plates were left to dry for 20 minutes before incubation overnight at 37°C. Plaque-forming units were counted and virus concentration calculated from the samples.
Statistical Analysis
Log reduction was calculated by taking the mean of the triplicate exposed samples and comparing this to the mean of the triplicate unexposed controls. Due to some variability in numbers of microorganisms on each coupon, it was possible that should efficacy of the fogging agent be low, the mean of the exposed samples could have a bacterial count that was greater than the mean of the unexposed controls. This would result in a negative log reduction. In these situations, the study team reported that there had been “no kill,” and a zero log reduction assumed.
Log reduction was not normally distributed and so was summarized using the median, minimum, and maximum observed values. Presented data display the observed log reduction, and a reference line at 6-logs was added as a value of particular interest. Tobit regression was used to compare log reduction between the factors of interest, with the lower limit for censoring fixed at zero. This incorporates the uncertainty associated with the no kill values, which were assumed to be zero. Two-way interactions between the factors of interest were entered one at a time and tested using the joint Wald test. Only the statistically significant 2-way interactions were kept in the final models. All models were adjusted for the unexposed cfu/challenge coupon unless otherwise specified. Model residuals deviated from the normal distribution, and so robust variance estimation was used throughout. The joint Wald test was used to test the overall statistical significance (at P < .05) of variables and interactions. When making pairwise comparisons, P-values were adjusted for multiple comparisons using Sidak’s procedure. Adjusted log reductions plus 95% confidence intervals (95% CIs) were estimated from the models to enable comparison to the 6-log reference value.
Results
Fixed Duration Testing
The 5 microorganisms (B. cereus, Bt Cry-, Geobacillus, MS2, and M. fortuitum) were each treated with each of the 2 disinfectants (PAA and HOCl) in 3 replicate runs.
Tables 2 and 3 show the number of log reduction values, the number that achieved full kill, and a summary of log reduction when disinfected with HOCl and PAA, respectively. Eighty-seven percent of the results achieved full kill when PAA was used (Table 3), whereas just 29% achieved full kill when HOCl was used (Table 2), which was a statistically significant difference, χ2(1, N = 270) = 92.4, P < .001. The range in the log reductions achieved was similar for both disinfectants. There was evidence that the proportion of results achieving full kill depended on the type of microorganism whether the disinfectant was HOCl or PAA (both Fisher’s exact P < .001). When disinfected with HOCl, B. cereus, Bt Cry-, and MS2 all had a low number of results achieving full kill (Table 2). When disinfected with PAA, B. cereus again generated the lowest number of results with full kill (n = 17; 63%), but here Bt Cry- and MS2 achieved full kill for all results (Table 3). There was no evidence that the proportion of results achieving full kill depended on the placement of the samples for either of the disinfectants (both Fisher’s exact P > .10).
Fixed Duration Spray Testing Results: Number of Log Reduction Values and the Level of Reduction Observed by Microorganism and Sample Placement When Disinfected with Hypochlorous Acid.a
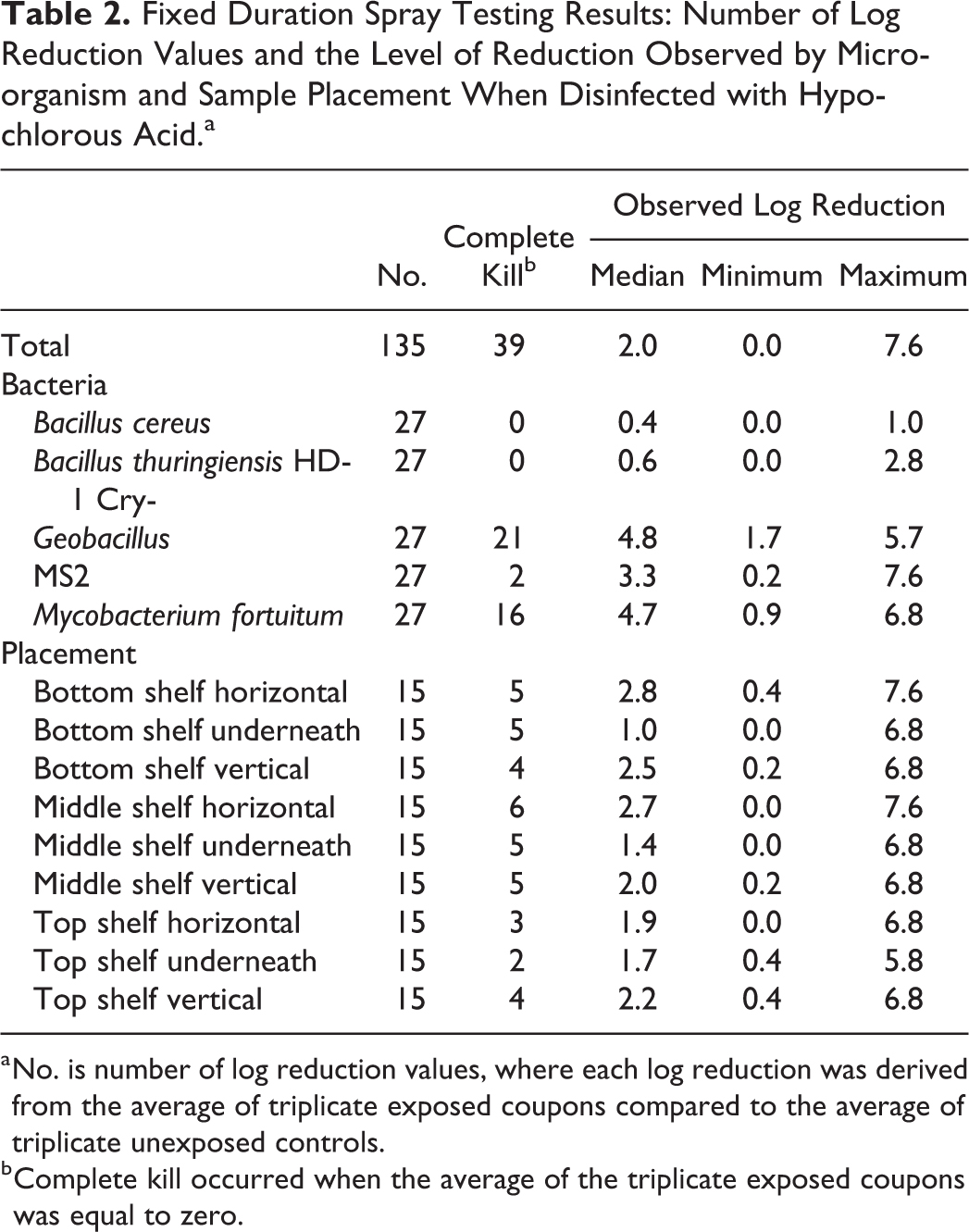
No. is number of log reduction values, where each log reduction was derived from the average of triplicate exposed coupons compared to the average of triplicate unexposed controls.
Complete kill occurred when the average of the triplicate exposed coupons was equal to zero.
Fixed Duration Spray Testing Results: Number of Log Reduction Values and the Level of Reduction Observed by Microorganism and Sample Placement When Disinfected with peracetic acid.a
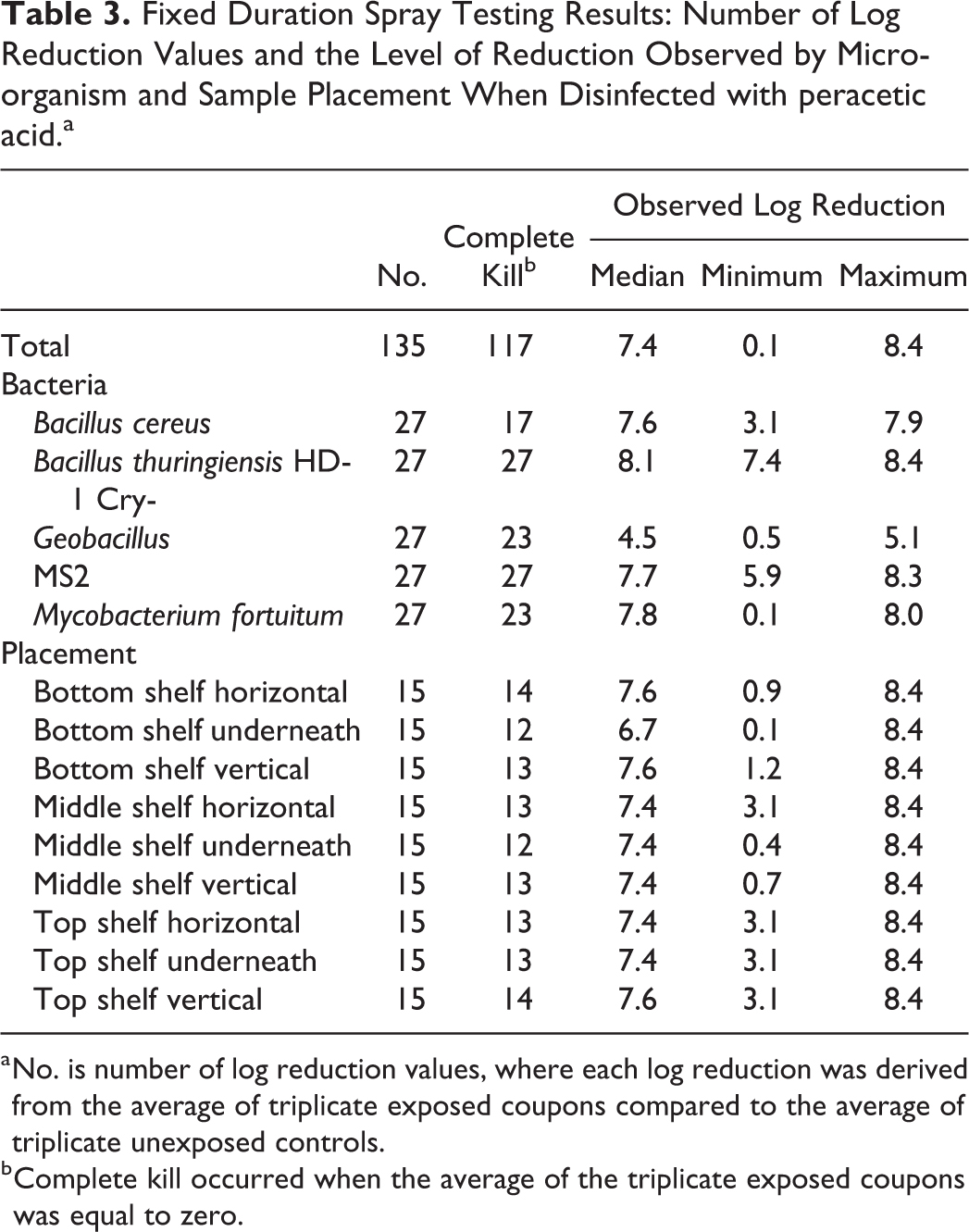
No. is number of log reduction values, where each log reduction was derived from the average of triplicate exposed coupons compared to the average of triplicate unexposed controls.
Complete kill occurred when the average of the triplicate exposed coupons was equal to zero.
Figure 4 shows the observed log reduction by sample placement, microorganism, and disinfectant. Tobit regression was used to investigate differences in log reduction by these factors. There was no evidence of differences in the achieved log reduction at the various locations (P > .10), and this result did not differ by disinfectant type (interaction P > .10). Samples placed underneath shelves tended to have lower log reductions than the other locations, but these differences were less than 1-log and were not statistically significant (all adjusted P > .10). There was a statistically significant interaction between disinfectant and bacteria (P < .001), providing strong evidence that the effectiveness of the disinfectant on the microbiological challenges depended on the type of disinfectant. For B. cereus, Bt Cry-, and MS2, the log reduction achieved by the HOCl disinfectant was statistically significantly lower than that achieved by the PAA disinfectant (adjusted P < .001). For M. fortuitum, the log reduction achieved by HOCl was still lower than that for PAA, but the difference was not statistically significant (adjusted P > .10). There was no evidence of a difference in the effectiveness of the 2 disinfectants for Geobacillus (adjusted P > .10). Only Bt Cry- and MS2, when disinfected with PAA, achieved a log reduction that was statistically significantly greater than 6-logs (P < .05). When disinfected with HOCl, the log reduction for MS2 and M. fortuitum was statistically significantly less than 6-logs (P < .05), and that for B. cereus and Bt Cry- was statistically significantly less than 1-log (P < .05).
Fixed-time spray testing results (all microorganisms): log reduction by sample placement, microorganism, and disinfectant.
Timed Duration Testing
B. cereus and Bt Cry- were each treated with the 2 disinfectants in 3 replicate runs, with samples removed at agreed time points. A second tandem delivery of disinfectant was also carried out and samples left for a further 1 hour.
Table 4 shows the number of log reduction values, the number that achieved full kill, and summarized log reduction. Twenty-five percent of the results achieved full kill when the disinfectant was HOCl, and 71% achieved full kill when the disinfectant was PAA. There was no evidence that the proportion of results achieving full kill depended on the extraction time of the sample for HOCl or PAA (both Fisher’s exact test P > .10).
Timed Extraction Experiment Results: Number of Log Reduction Values and the Level of Reduction Observed by Disinfectant, Bacteria and Sample Extraction Time.a
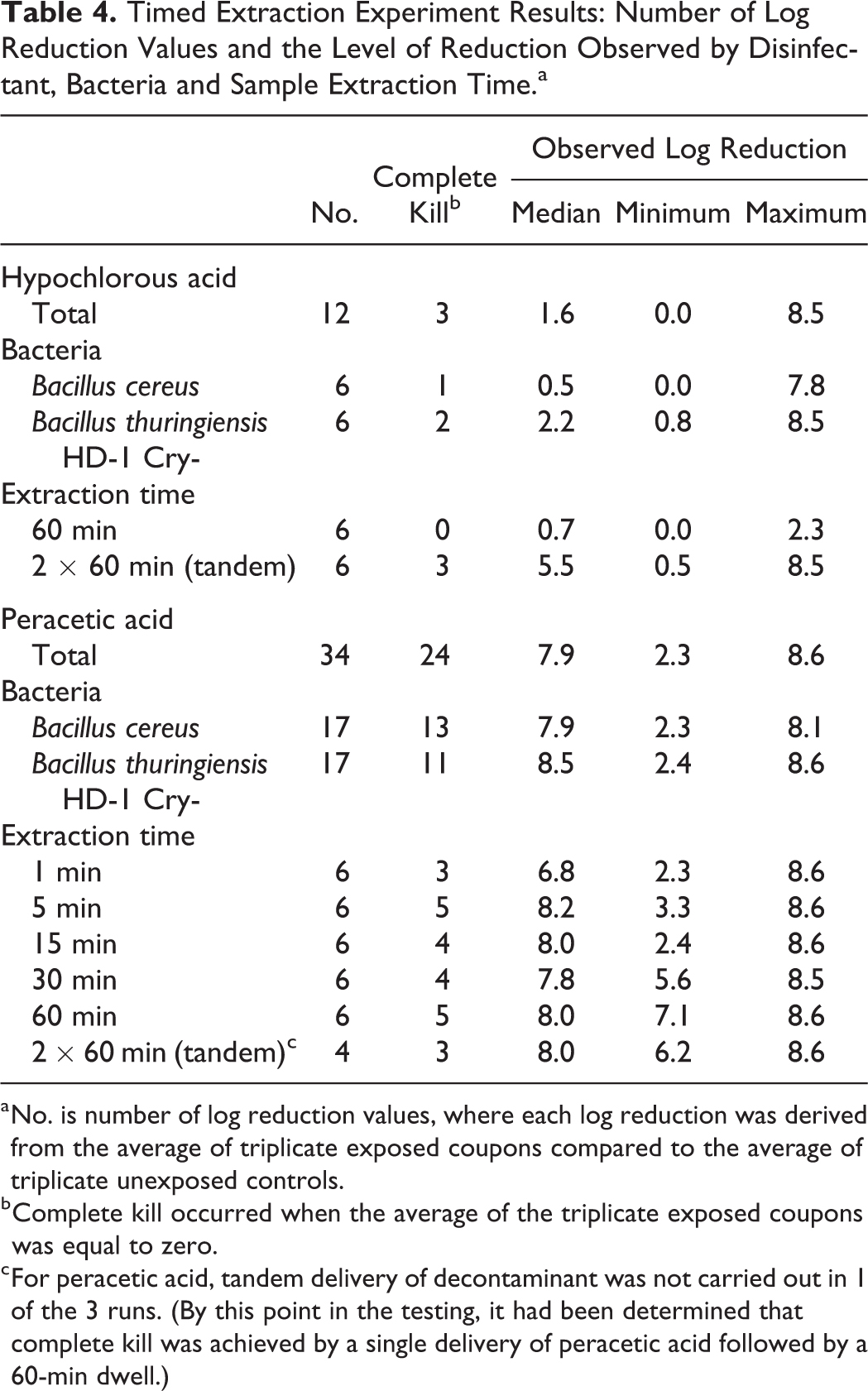
No. is number of log reduction values, where each log reduction was derived from the average of triplicate exposed coupons compared to the average of triplicate unexposed controls.
Complete kill occurred when the average of the triplicate exposed coupons was equal to zero.
For peracetic acid, tandem delivery of decontaminant was not carried out in 1 of the 3 runs. (By this point in the testing, it had been determined that complete kill was achieved by a single delivery of peracetic acid followed by a 60-min dwell.)
Figure 5 shows the observed log reduction by sample extraction time and microorganism for each disinfectant. Separate Tobit regression models were used to investigate differences in log reduction by these factors for each disinfectant. When disinfected with HOCl, tandem samples (ie, those treated twice) achieved a log reduction that was statistically significantly greater than samples analyzed after a single 60-minute treatment. This difference was estimated to be around 4.6 logs, although it was subject to large uncertainty (estimated difference = 4.6, 95% CI = 1.7-7.4, P = .005). For samples analyzed 60 minutes after disinfection with HOCl, the adjusted log reduction was statistically significantly less than 6-log (P < .05). For samples disinfected with HOCl, there was a statistically significant difference between the log reductions for B. cereus and for Bt Cry- (P = .032).
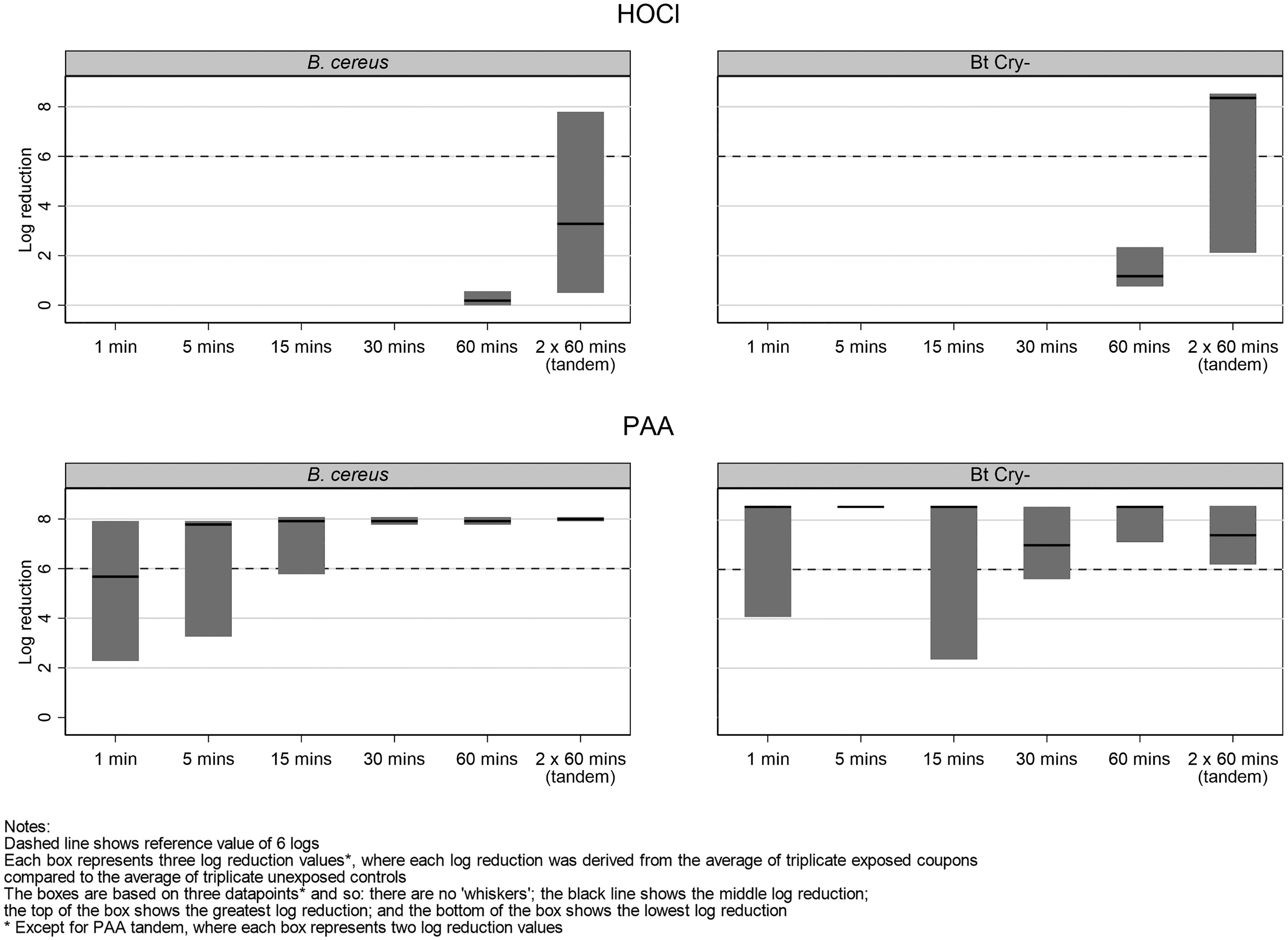
Timed extraction experiment results (2 challenge microorganisms and both disinfectants): log reduction by extraction time and microorganism for each disinfectant.
When disinfected with PAA, there was no evidence of a difference in log reduction with extraction time (P > .10). However, the variability in the observed log reduction decreased with increasing extraction time (Figure 5). The log reductions for extractions after 30 minutes, 60 minutes, and tandem samples were all statistically significantly greater than 6 logs (all P < .05). There was some indication of increasing log reduction with increasing extraction time for PAA disinfections. However, this was of borderline statistical significance and was not a large increase at 0.2-logs per additional 10 minutes before extraction (estimated increase per 10 minutes = 0.2 log, 95% CI = –0.004 to 0.5, P = .054). There was no evidence that the expected log reductions were different for the 2 bacteria after the application of PAA (P > .10).
Multiple Materials Testing
Only Bt Cry- was tested here on a range of surfaces in 3 replicate runs using each of the 2 disinfectants.
Table 5 shows the number of log reduction values, the number that achieved full kill, and summarized log reduction. Around 33% of the results achieved full kill when the disinfectant was HOCl, and 87% achieved full kill when the disinfectant was PAA. One result on uPVC that was disinfected with HOCl gave no kill. There were 2 results that did not achieve full kill when disinfected with PAA, on concrete. There was no evidence that the proportion of results achieving full kill depended on the coupon type for either of the disinfectants (both Fisher’s exact P > .10).
Different Surface Testing Results (Bt Cry-): Number of Log Reduction Values and the Level of Reduction Observed by Disinfectant and Material Type.a
No. is number of log reduction values, where each log reduction was derived from the average of triplicate exposed coupons compared to the average of triplicate unexposed controls. HOCl, hypochlorous acid; PAA, peracetic acid; uPVC, unplasticized poly vinyl chloride.
Complete kill occurred when the average of the triplicate exposed coupons was equal to zero.
Figure 6 shows the observed log reduction by coupon type for each disinfectant. Separate Tobit regression models were used to investigate differences in log reduction by coupon type for each disinfectant. Samples from all tests were compared to the same control samples (for that disinfectant and material type) to calculate the log reduction. Therefore, it was not possible to adjust for the control concentration in the Tobit regression models. There were statistically significant differences between material types for both disinfectants (both P < .01). When disinfected with HOCl, the greatest log reductions tended to be for samples on concrete coupons, and this was the only material type that achieved a log reduction that was statistically significantly greater than 6-logs (adjusted log reduction = 6.5, 95% CI = 6.5-6.5). The adjusted log reductions for samples placed on uPVC and carpet were both statistically significantly less than 6-logs, at 0.8-logs (95% CI = –0.2 to 1.7) and 2.2 logs (95% CI = 1.3-3.1), respectively. There was large variability in the observed log reductions for glass and plaster surfaces (Figure 6). When disinfected with PAA, all materials except the concrete achieved full kill and an adjusted log reduction that was statistically significantly greater than 6-logs. The adjusted log reduction for the concrete surface was 4.9-logs (95% CI = 3.6-logs to 6.2-logs) when disinfected with PAA, and this was statistically significantly less than that achieved for other material types. However, the control concentration was lower for concrete coupons when disinfected with PAA, and so this may at least partially explain some of the difference.
Multiple material testing: log reduction by tile type for each disinfectant (all data are for Bt Cry-).
Live Agent Timed Extraction Testing
B. anthracis was treated in 3 replicate runs with each of the 2 disinfectants. In each test, approximately 2 L of disinfectant was delivered from the backpack system via the Venturi nozzle. However, several hundred milliliters of this deposited inside the plastic poly-tunnel during each test, and therefore less than 2 L was actually delivered to the target MSC surfaces.
Table 6 shows the number of log reductions, the number that achieved full kill, and summarized log reductions. None of the results achieved full kill when HOCl was used, and 1 result at each extraction time gave no kill. However, 59% of the results achieved full kill when PAA was used, and none observed zero kill.
Live Agent Timed Extraction Results (Bacillus anthracis): Number of Log Reduction Values and the Level of Reduction Observed by Disinfectant, Extraction Time, and Sample Position.a
No. is number of log reduction values, where each log reduction was derived from the average of triplicate exposed coupons compared to the average of triplicate unexposed controls. HOCl, hypochlorous acid; PAA, peracetic acid.
Complete kill occurred when the average of the triplicate exposed coupons was equal to zero.
Figure 7 shows the observed log reduction by sample extraction time and sample position for each disinfectant. There were fewer extraction times for the HOCl disinfectant, and so the 2 disinfectants were analyzed using separate Tobit regression models. There was no evidence of a difference in log reduction with different extraction times when disinfected with HOCl (P > .10 for the categorical variable and the continuous variable). All 3 extraction times achieved a log reduction that was statistically significantly less than 2-logs. There was some indication of increasing log reduction with increasing extraction time for PAA disinfections. However, this was of borderline statistical significance and was not a large increase at around 0.2-logs per additional 10 minutes before extraction (estimated increase per 10 minutes = 0.2-log, 95% CI = –0.03 to 0.3, P = .092). The adjusted log reductions were statistically significantly greater than 6-logs for extraction times of 15 minutes, 30 minutes, and 60 minutes after disinfection with PAA (all P < .05).
Live agent timed extraction results: log reduction by extraction time (minutes) and sample position for HOCl (all B. anthracis).
Discussion
The 2 chemicals used for fogging were already known to be effective for killing microorganisms. In general, the LDS backpack decontamination system and handheld lance worked effectively and reliably to deliver the liquid disinfectants. Although a technical issue was encountered with the trigger on the lance, due to the extreme cooling effects on the air outlet valve of the equipment, this was promptly rectified by the manufacturer, and the problem did not recur.
Initial testing with inert coupons exposed over 1 hour used microbiological samples at different heights and orientations. Although there was no statistical evidence that the effectiveness of the disinfectant depended on placement, samples positioned underneath shelves had slightly lower log reductions, but these differences were small. This showed that the spray delivery system reached samples not in the direct line of sight as well as more exposed challenge materials. Immediately after this and other treatments, droplet condensation could be observed on some surfaces, but these were not drenched in liquid.
Fixed duration testing indicated that the effectiveness of the 2 chemicals was different, depending on the microbiological challenge. HOCl was less effective than PAA against B. cereus, Bt Cry-, and MS2. It also performed worse for M. fortuitum, but this was not a statistically significant difference. There was no evidence of a difference in performance of the 2 disinfectants for G. stearothermophilus. Using PAA, only Bt Cry- and MS2 achieved a log reduction that tended to be greater than 6-log, although for the remaining microorganisms, the reduction was close to this. Complete kill was observed in almost all cases with PAA, and where survival of microorganisms was observed, it was generally at a low level. For HOCl, the log reduction for B. cereus and Bt Cry- tended to be less than 1-log, with efficacy against MS2, G. stearothermophilus, and M. fortuitum higher, although only MS2 achieved greater than a 6-log reduction.
A second, tandem delivery step was incorporated into the testing regime with B. cereus and Bt Cry-. This was largely to determine any potential benefits for the use of HOCl since PAA had already shown high levels of microbiological kill after a single treatment. Previous work demonstrated that 2 deliveries of disinfectant, with a dwell period in between, can increase levels of efficacy compared with a single delivery. 5 For HOCl, tandem delivery gave increased bacterial kill over a range of reductions between 2.8-log to 7.5-log.
A tandem delivery approach was also used with PAA, but there was no evidence that tandem disinfections increased the log reduction, mainly because a complete kill was typically obtained following a single delivery of this chemical. Based on the data, samples exposed to PAA were then removed after shorter dwell periods to determine whether a greater than 6-log kill of B. cereus and Bt Cry- could be achieved in less than an hour. Exposure times of 30 minutes or more (including tandem disinfections) achieved log reductions that were consistently greater than 6-log for both species.
Additional testing was carried out on various surfaces to investigate any influence of surfaces and disinfectant performance. There was evidence that different material types gave different levels of kill for Bt Cry- spores when using HOCl. Concrete gave reductions greater than 6-log, higher than on steel, suggesting that hypochlorous acid surface interaction with the concrete gave improved efficacy, in contrast with observations for PAA. However, HOCl was less consistent and less efficacious on carpet, uPVC, glass, and plaster, suggesting that material type could influence treatment outcome.
When disinfected with PAA, complete kill of Bt Cry- was observed on all material types except for concrete, with log reductions above 6-log for carpet, glass, plaster, and uPVC. The observed log reduction for concrete samples was around 4.9-log. However, the recovered level of Bacillus spores for concrete untreated controls was just over 6-log, so also lower than for other coupon types, probably due to it being more difficult to recover the bacteria from this more porous surface. This factor may partially explain the proportionally lower observed log reduction for concrete. In addition, organic acids are known to react with the calcium in concrete and may cause actual structural damage as a result. No observed surface damage was noted on concrete material, but any such chemical reactivity might impair microbiological kill on this surface. Further investigation of any possible PAA-concrete interaction was outside the scope of the project and, interestingly, was not reflected by the plaster samples (also rich in calcium).
The different coupon types were placed at different locations within the tented structure, and it is possible that this could be responsible for some of the differences observed between materials. However, the fixed duration testing found little evidence of sample location affecting the achievable log reduction. It is therefore unlikely that location could entirely explain any differences in observed bacterial reduction, and it is more likely to be due to properties of the materials themselves.
Most whole room and outdoor decontamination testing is carried out with surrogate microorganisms due to the health and safety concerns of using disease-causing organisms outside of the laboratory. However, in this study, some limited live agent testing was completed in a CL-3 facility using B. anthracis. The method of preparation of the spores and the disinfectant delivery was aligned as closely as possible to the other work carried out with the Bacillus surrogates, such that while not identical, reasonable comparisons could be made.
For B. anthracis treated with the HOCl, log reduction for all treatments was low (< 2-log), with no evidence that log reduction increased with extended contact time. For B. anthracis treated with PAA, there was some indication of a greater reduction for a 15-minute contact time or more, compared to a 5-minute contact, but these were not statistically significant effects. In fact, contact times of 5 minutes, 15 minutes, 30 minutes, and 60 minutes all achieved log reductions that tended to be greater than 6-log.
The data obtained for B. anthracis with both disinfectants was broadly in agreement with the data obtained for the surrogates B. cereus and Bt Cry-. While B. cereus and B. thuringiensis have been used for many years as surrogates of B. anthracis, the development of the Bt Cry- strain is a more recent advancement. 9 The results here show that Bt Cry- is a suitable surrogate organism for B. anthracis decontamination testing, offering comparable data from closely aligned biocidal treatments.
The PPE ensemble used for this work was critically important as it protected the operator of the decontamination equipment throughout the various periods of treatment. The PPE and RPE worked effectively in this respect and allowed targeted delivery of the airborne disinfectants within both test areas. Importantly, the RPE system enabled the operator to conduct safe, extended duration testing within the CL-3 facility, without the need for changing cylinders, with the SCBA set providing an adequate supply of clean air for 75 minutes.
Conclusion
The results of this work demonstrate the importance of evaluating decontaminant delivery equipment and disinfectants against a range of relevant microbiological challenges and in appropriate settings. Our results confirm that the biocidal efficacy of a treatment can vary significantly depending on the types of microorganisms used and where they are positioned in the test area. Overall, it is evident that while HOCl is an effective disinfectant against certain microbiological challenges, it has more limited efficacy against spores when applied as a fine mist. In contrast, PAA consistently gave higher levels of kill against all microbiological challenges, including Bacillus spores.
The most effective disinfectants provide a high level of biocidal efficacy regardless of these challenges, and 2% PAA appears to fall into this category. Results from the semienclosed tented area tests—where airflow was present through the surrounding building—suggest that treatment of outdoor spaces may be possible with this approach. However, further work would be required to fully demonstrate the combined capability of the LDS and PAA for true outdoor situations.
Footnotes
Ethical Approval Statement
Not applicable to this study.
Statement of Human and Animal Rights
Not applicable to this study.
Statement of Informed Consent
Not applicable to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclaimer
The contents of this research paper, including any opinions and/or conclusions expressed or recommendations made, do not supersede current Health and Safety Executive policy or guidance.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Scott Safety, part of the 3M group. 3M and Scott are trademarks of 3M Company.
