Abstract
Institutional health and safety programs are responsible for minimizing personnel risk in working with animals that have been administered hazardous chemicals. Regulations and associated guidance are lacking in specific processes for managing these animals. A chemical control banding system categorizes chemicals into bands where each band level is associated with specific control practices. This article describes a general approach to the engineering, administrative, and personal protective equipment practices for developing an animal chemical control banding system. An internal committee should be responsible for conducting the risk assessments to assign chemicals used in animals into band levels, with many factors and resources included to facilitate in this process. The authors provide examples from their home institution where an animal chemical banding system was implemented. Institutions can use this information when designing their own programs, which will likely be unique in consideration of their specific needs and resources.
Chemicals are frequently administered to animals as part of a research protocol to evaluate in vivo effects of the compound or elicit pathology to establish a disease model. Some of these chemicals are known to be hazardous agents, while the toxic effects of some compounds have not been thoroughly evaluated. Many institutions face challenges associated with these protocols, but limited guidance is available for managing the animals and associated waste once a compound has been administered.
This article is intended to serve as a resource for research organizations developing a program to assess and mitigate risk when working with research animals that have been administered potentially hazardous compounds. We describe a general approach to an animal chemical control banding methodology, including some examples from the authors’ institution. Our program includes a risk assessment process performed by a designated committee to assign a compound to a particular band level. This committee also provides a hazard assessment summary to the researchers, which includes the band level and required practices. The control banding described addresses the handling of animals after chemical compounds have been administered, as the general use of chemicals in an animal facility has been previously discussed and is covered by the same regulatory standards as in a research lab.1,2 The primary focus of this article is small rodents, but the general principles can be extrapolated to larger species. Each research institution will require a unique system based upon their circumstances and resources.
Regulatory Requirements and Guidance
Regulatory and accrediting agencies overseeing animal research expect institutions to have processes that minimize the risk for personnel working with animals administered hazardous chemicals, but specific requirements are lacking. The Guide for the Care and Use of Laboratory Animals, a standard to qualify for Public Health Service funding and Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International accreditation, recognizes the institutional responsibility for addressing occupational health and safety and that efforts should be made to isolate animals and their waste after the administration of hazardous chemicals. 3 The Occupational Health and Safety of in the Care and Use of Research Animals, a reference also used by AAALAC International, includes that institutions must have a plan for handling known and potential chemical hazards, from working with the chemical to disposal of the animal waste. 4 The Occupational Safety and Health Administration (OSHA) standard Occupational Exposure to Chemicals in Laboratories (also known as the Lab Standard, 29 CFR 1910.1450) is a performance-based regulation and provides guidance for the nonspecific nature of laboratory work. The OSHA Hazard Communication standard (29 CFR 1910.1200) is integrated into the Lab Standard and dictates that potentially exposed employees must be adequately informed of hazards and basic protective measures. Prudent Practices in the Laboratory, Handling and Disposal of Chemicals, published by the National Research Council, includes extensive detail on handling of the chemicals. 5 Although these documents are important for the risk assessment process, they do not provide procedures for working with animals after they have been administered the chemical.
Environmental Protection Agency (EPA) regulations will determine the potential environmental hazard posed by the chemical. The EPA Resource Conservation and Recovery Act (RCRA) regulates the handling of hazardous wastes and includes many chemotherapeutic drugs administered to research animals (40 CFR 261-262). Occasionally, investigators submit protocols that request the use of pesticides, such as organochlorine compounds. The EPA regulates pesticide use, including defining banned and restricted use pesticides, under the Federal Insecticide, Fungicide and Rodenticide Act (40 CFR 150-180). The EPA Toxic Substances Control Act covers many chemicals but excludes some categories such as pesticides and drugs and is less likely to be applicable. The EPA requirements most directly affect waste handling requirements, but a chemical that warrants EPA regulation likely has the potential to affect human health.
Animal Chemical Control Banding System
Control banding is the general concept of grouping chemical hazards into categories, or bands, and subsequently assigning control measures to each band.6,7 The basis for the band categories could be similar hazards (eg, toxicity, irritant, allergen), level of risk, or other characteristic as relevant for the institution. Bands have different nomenclature, such as categories or levels, but the central principle is that the same control measures can be effective across a range of hazards assigned to a particular band. Although the chemical hazards in any animal program are numerous, the control measures needed to manage exposure risk are far fewer and can be consolidated into limited options. Therefore, a chemical control banding system for an animal facility simplifies the process of managing the variety of chemicals used in animals while allowing for a graded risk management approach and strategic use of resources.
A uniform standard for chemical banding has not been developed as has been done for biohazards, 8 but the general concepts of the animal biosafety levels can be applied to animals that have been administered chemicals. The band levels should be primarily defined by the protective measures put in place, which are determined after assessing the types of risks encountered and the standard institutional practices. An assigned committee should be responsible for determining the band levels for compounds within the context of a given protocol.
Institutions will vary in the number of band levels created depending on the complexity and variability of standard practices and available control measures. The following controls should be considered for each band level: engineering controls (housing system and conditions, work environment, waste disposal), administrative controls (written operating procedures, hazard communication, training, occupational health screening), and personal protective equipment (PPE). The authors’ institution uses 4 band levels. Table 1 summarizes the control practices for each level, although the risk of each protocol is assessed and modifications to a particular level could be made as deemed necessary for certain protocols. For example, regulatory requirements might dictate particular disposal standards, or a unique method of administering the chemical might create unanticipated challenges. The authors’ institutions use the lowest level, Animal Control Level (ACL) 1, for animals that receive potentially or known hazardous chemicals but for which standard animal handling practices are deemed sufficient to control the risk. Other institutions might choose alternate acronyms as ACL also refers to the containment of infectious arthropods. This scenario most frequently occurs because a compound is metabolized very quickly or into nonhazardous compounds. ACL 1 is used to distinguish hazardous chemicals for which no additional precautions are needed from nonhazardous agents (eg, standard of care medications) that do not need to be reviewed by the designated committee. The number of band levels for other institutions could be 2 or more, while realizing that the higher the number of levels, the greater the complexity of the program.
Animal Chemical Band Level Control Measures at the Authors’ Institution.a
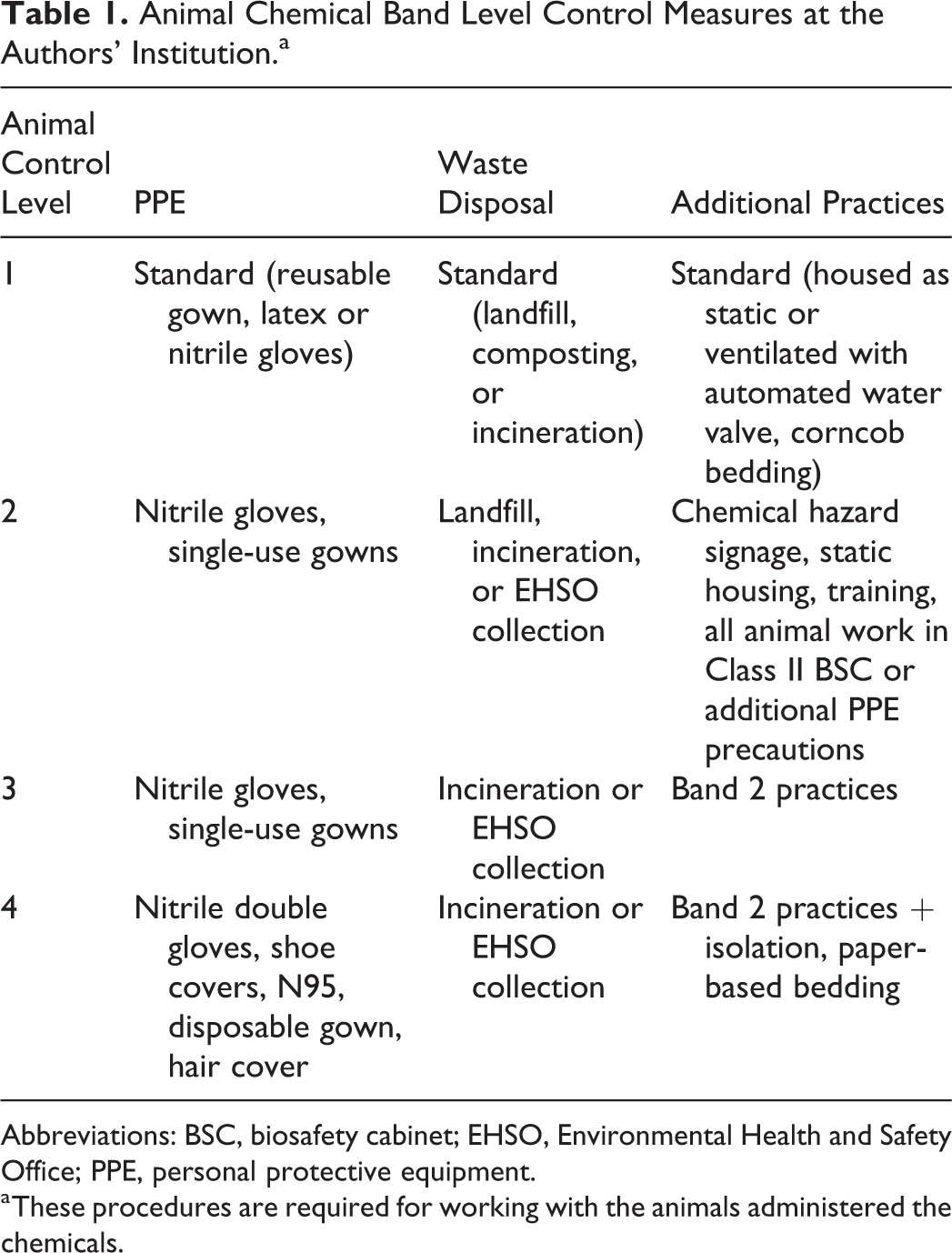
Abbreviations: BSC, biosafety cabinet; EHSO, Environmental Health and Safety Office; PPE, personal protective equipment.
These procedures are required for working with the animals administered the chemicals.
Engineering Controls
Housing systems
The chosen housing system will depend on the risks of contaminating the environment, personnel, and other animals, as well as the standard and available options within the animal facility. The main variables to consider include the protection provided at the cage top level, intracage ventilation and exhaust, and the water delivery mechanism. Filter top cages are available for mice, rats, guinea pigs, and other small rodents but might not be the standard used in some facilities, particularly for the larger rodents.9 -12 These filters prevent dispersion of airborne particulates but must allow adequate gas exchange and therefore could allow the transmission of some chemical vapors.13 -16 Contaminated particulates will likely be a greater concern than vapors after animals have received chemicals. Filter tops should be a minimum precaution for at least some animal chemical band levels if possible, to reduce the additional protective measures that would be required otherwise. 12
Individually ventilated caging systems have grown in use due to their ability to reduce gaseous waste products such as ammonia. 16 Air from individually ventilated cages is then HEPA (high efficiency particulate air) filtered and exhausted from the system without exposure to other animal cages. Optimally, the air would be exhausted out of the building to reduce thermal load and the potential for contamination of the macroenvironment, although the air could be returned to the room if adequately filtered.3,12 A ventilated housing system would be preferred for chemicals excreted by the respiratory route, assuming that the air is adequately captured from the cage and the rack exhaust then removed from personnel areas.
Rodents are provided water via a water bottle or automated water systems. Automated systems provide water from a central source that is delivered to each cage via a drinking valve. Some valve models are easily removable and others fixed to the frame of the system. The effluent is removed such that the water from one cage would not be provided to another. However, the valve itself could serve as a risk for cross-contamination between animals. Water bottles would be a prudent choice for chemically contaminated animals, or a reliable system for decontamination between animals is required.
The banding system procedures should also incorporate procedures to minimize cage change frequency. For example, placing animals in a clean cage immediately before administering chemicals and choosing a housing process that requires less frequent cage changes would reduce exposure for animal care personnel.
Work area
Rodents are handled in work environments that differ in levels of personnel protection, based on the species, institution, and the type of procedure performed. Routine procedures and cage change for mice are typically done within a cabinet that offers some personnel protection and possibly for larger rodents as well. 9 Although chemical fume hoods are ideal when working directly with hazardous chemicals, they will not offer the level of biosecurity for the animal that an animal resources program will likely require when working with mice in a housing room as they offer no product protection. Chemical fume hoods could be of use within individual laboratories where product protection is less of a concern or when larger amounts of volatile chemicals necessitate its use. The amount of chemical excreted from the animal will typically be much lower than when working with the chemical itself, and therefore the controls required for working with animal might not be as stringent as for the chemical handling.
Biological safety cabinets (BSCs) are frequently used when working with animals, with a type of Class II BSC likely the most readily available. 12 A HEPA filter will not be effective at removing volatile chemicals but will eliminate contaminated particulates and nanoparticles. 17 A Class II B2 is designed for chemical containment because no air is recirculated within the cabinet and the exhaust is removed from the building. 8 However, other biosafety cabinets are likely sufficient for working with chemically contaminated animals because the chemicals are often excreted in minute amounts and/or as nonvolatile compounds. Any Class II would be acceptable for handling nonvolatile toxic chemicals, although amounts should be minute for an A1. 8 Even for limited amounts of volatile chemicals, a Class II Type A2 or B1 would be adequate if the A2 has a canopy connection so that exhaust is removed from the building. 8
Many animal facilities use animal transfer stations instead of biosafety cabinets for some procedures, particularly for cage changes. 12 Animal transfer stations vary widely in their protective characteristics, with some primarily designed to protect the animal and some approaching the personnel protection provided by a Class II BSC.12,18 These transfer stations would need to be evaluated on an individual basis to determine if they are sufficient for handling procedures for chemically contaminated animals.
Personnel might need to work with animals outside of a cabinet. Larger species such as rats and guinea pigs are more likely than mice to have their cages changed on an open counter due to practical constraints. Researchers might also need to perform procedures on a bench top, relying more heavily on PPE for protection. The macroenvironment in these scenarios could either be a laboratory that would operate under the general laboratory biosafety level 2 (BSL-2) standards or an animal facility meeting the criteria of OSHA guidance for working with laboratory animals and the Guide for the Care and Use of Laboratory Animals.3,19 The Guide dictates that facilities in which animal hazards are present should have sealed surfaces to reduce dust accumulation and facilitate sanitation, much like standard housing room. The general Guide recommendation is for 10 to 15 air changes per hour in the macroenvironment, but it emphasizes a performance standard considering many factors such as heat load and species. The Guide also mentions that increased air exchange rates might be needed to reduce and contain hazards. Standards are available for reducing contaminants within the macroenvironment and should largely be considered in the construction phase.20 -23 The method for transporting animals and associated equipment out of the housing room if needed and methods for handling spills of those materials should also be determined.
Waste disposal
The committee should determine the appropriate mechanism for collection and disposal of waste. The waste will include bedding, enrichment items, food, water, PPE, and research materials. Low-dust bedding might be preferable when airborne particulate must be minimized. Most commercially available mouse beddings produce very low levels of dust, but levels do vary and hardwood materials might produce more dust.24,25 Larger species might be housed on wood shavings or other materials that can vary more in dust production.25,26 Likewise, enrichment items like cotton nesting material might produce more particulates than shredded paper for nesting material, and the risk might be great enough to warrant solid enrichment such as plastic hiding places and wheels. Waste materials besides bedding and enrichment items are less fungible and are unlikely to contribute to particulate and therefore are unlikely to differ between band levels.
Waste products may require collection by multiple types of personnel, including Environmental Health and Safety Office (EHSO) staff, animal resources staff, or the researcher, and proper disposal mechanisms must be available for all of them. Chemicals administered to the animals via food or water would need to be collected by EHSO if they are categorized as EPA hazardous wastes (40 CFR 261-262). Researchers must be educated that a biohazard container is not appropriate for RCRA hazardous waste supplies, and they might need yellow chemical disposal containers if they will create RCRA empty (also often referred to as “trace”) containers. If chemicals are not defined as EPA hazardous waste, the normal waste streams would likely be appropriate.
After animals are administered chemicals, a conservative approach would be to collect the animal-associated waste, such as bedding, for incineration. A yellow hazardous chemical container or biohazard container could be used for this collection, provided that the latter is designated for incineration and not autoclaving alone. Carcasses are typically collected for incineration, and additional measures to collect those are likely unnecessary. The contaminated bedding must be collected with a method to reduce airborne particulates. A variety of bedding disposal stations are available that have a downdraft system for this purpose, and some can be fitted with HEPA, carbon, and other filters to reduce contaminants into the environment. The institution might also determine that the animal waste associated with some chemicals could be directed for landfill instead of incineration. The permitted waste streams are one of the band level distinguishing features at the authors’ institution.
Administrative Controls
Training based on written standard operating procedures will be the foundation of administrative controls. OSHA training required for working directly with hazardous chemicals will likely be provided to researchers as part of the standard EHSO laboratory safety training. Training for animal users should focus on the unique procedures and precautions for working with animals given hazardous chemicals. Training comprehension should be assessed, which can be accomplished with a test on the material. 3 Visual observations of personnel performing the work to assess comprehension might also be beneficial, especially in high-risk situations. Continuing education is required and provides an opportunity to remind infrequent users of the requirements and update all relevant personnel on changes to the programs. 3 The course developed for continuing education could be aligned with an annual or triennial protocol renewal process for convenience and ease of tracking.
Hazard communication for animals administered chemicals should be based upon an institution’s current signage system, as consistent formats assist with personnel quickly identifying hazards. Programs could use the animal biosafety signage for housing rooms as a template for room and/or housing rack signs for chemical usage. At a minimum, the posted information should include the chemicals, associated hazards, required PPE, and emergency contacts. Unlike the standard for biohazards, chemical hazards do not have a single hazard symbol. The Hazard Communication Pictograms could be used to indicate the specific chemical hazards, although they are not required because the signs reference animals instead of a primary chemical container. The location of the signage depends on how the institution chooses to segregate the cages: at the room, rack, or location on the rack. If contaminated cages are in a standard housing room, the individual cage would need identification as well. Many institutions have a form of cage-level identification for communication and possibly even biohazard labeling, and the chemical signage should be consistent with these labeling systems if possible. When developing signage, the institution should be prepared for protocols that use many more chemicals than are often seen on biohazard protocols. For simplicity and clarity, the institution might choose to group drugs that have the same type of hazard into one category on the room or rack signage. For example, indicating that a certain class of chemotherapeutics is used instead of listing each individual compound. The specific chemical could still be at the cage level, without making the room sign unwieldy.
Chemical hazards in animals are unique from most biohazards in that many animals are often maintained in the housing room long after they no longer pose a hazard. Institutions that designate some viral vectors as temporary animal biosafety level 2 (ABSL-2) might already have a process for communicating short-term hazards, but otherwise, a new sign for identifying when animals are no longer contaminated will need to be developed. At the author’s institution, cage stickers that dissolve in cage wash are placed directly on the cage with the date that animals are no longer considered contaminated. These stickers alert animal care both that the animals no longer need signage and that the cage materials still need to be handled as contaminated (Figure 1).

A cage sticker that will dissolve in cage wash can be used to alert animal resources staff when the animals are no longer chemically contaminated.
The fluidity of chemical status for any particular room and cage poses a risk for inadequate signage at any given time, and a system that allows for easy replacement and updating of signage will facilitate accuracy. The researchers are the only personnel who know the current status, so giving them easy access to the resources will encourage compliance if they are relied upon to place the signage. For example, room or rack signage could be provided to them whenever they submit or amend their chemical protocol, so that they can easily place it in a sign holder when the hazard is present.
Administrative controls to minimize and oversee personnel exposure should be incorporated into the overall program and at the level of each protocol. Personnel working with the animals must be listed on the chemical use application and will already be enrolled in the occupational health program because of their work with animals. Only these research personnel and animal resources staff who have undergone the required training should have access to the animals. If an occupational health professional does not sit on the committee, the committee must inform the institutional occupational medicine program of what compounds are in use so that appropriate resources such as antidotes can be provided in the case of exposure.
Personal Protective Equipment
The PPE required for routine animal handling varies by institution and might be sufficient or require minor modifications when working with most chemically contaminated animals. OSHA requires a risk assessment to determine appropriate PPE but does not dictate specific requirements for working with animals (29 CFR Part 1910 subpart I). The animal and associated waste will likely qualify as incidental contact with the chemical and not require the increased protection necessary for extended contact with the chemical itself. Specific glove brands should be rated as appropriate for its intended use because glove material permeability varies with the thickness and type of chemical. For example, gloves used for hazardous drugs could be made of different materials but should meet the American Society for Testing and Materials (ASTM) standard for chemotherapy use.17,27 The standard nitrile and latex gloves used within an animal facility are likely adequate for incidental exposure to most of the chemicals used in animal models.28 -30 Powder-free gloves would be preferred to remove the risk of the powder absorbing the chemical, although most facilities have likely eliminated powdered gloves because the Food and Drug Administration (FDA) ban on the use of the use of powdered gloves includes veterinary care (21 CFR 895). 27 Nitrile might be preferred over latex when working with chemically contaminated animals because personnel can more easily observe punctures in nitrile gloves and nitrile might be more resistant to some hazardous drugs.29,30 Regardless of the material, double gloving will provide additional protection by increasing the thickness and reducing the impacts of glove defects and tears.28 -30 Double gloves can also facilitate workflow as replacing the outer pair is faster than washing hands when trying to reduce the risk of contaminant transfer between animals. The benefits of double gloving must be balanced with the possible loss of dexterity it creates.
Protective clothing is usually provided as part of standard PPE when working with animals, and additional head, face, or shoe coverings could be included for each band level as deemed necessary by the risk assessment. Coverings such as lab coats and gowns should be either disposable or dedicated to the individual user so as not to potentially expose uninformed personnel. Coverings made of repellant material, such as Tyvek, are more expensive than permeable cloths and could be used for higher-risk exposure parts of the body, such as arms, or with more hazardous protocols. Additional PPE might be unnecessary when working with animals within a biosafety cabinet, but the research or species might require work outside of a biosafety cabinet. A surgical mask or N95 respirator, hair cover, safety goggles, and/or face shield should be considered when particulates might be generated. Shoe covers might reduce the tracking of particulates out of the room. Dedicated clothing and/or shoes might be necessary to eliminate the risk of trace amounts leaving the facility in particularly high-risk scenarios.
Personal respiratory protection is more likely necessary during administration of chemicals than when working with the animals but might be required in situations where a biosafety cabinet is unrealistic or insufficient protection. If animals are administered highly hazardous agents but chemical vapors from the animal are not a concern, a particulate respirator such as an N95 should be sufficient to minimize the risk of exposure to even a fine particulate. A particulate respirator might also be indicated where hazardous agents are administered via a dust-producing food. In the event aerosol exposure is a risk, respiratory protection for chemical vapors would be required. This would more likely be necessary in larger animals or with respiratory delivery or high-volume respiratory excretion of a drug.
Hazard Review Process
The hazard review process should be a collaborative effort conducted in a committee structure within the office responsible for institutional safety. The members should include EHSO personnel and animal resources veterinarians at a minimum. Membership or input from a faculty toxicologist, animal care operations, and an occupational health physician are also highly valuable. At the author’s institution, the Chemical Safety Committee is a subcommittee of the Institutional Research Health and Safety Committee (RHSC). The RHSC is convened with the same members as the Institutional Biosafety Committee (IBC) and serves similar functions as an IBC at many other institutions. If an institution chooses to have its IBC review these protocols, similar expertise should be represented.
The institution should determine a process for identifying the use of chemical in animals. At the authors’ institution, the intent to administer a chemical to experimental animals is disclosed during the Institutional Animal Care and Use Committee (IACUC) protocol review process. An EHSO representative or veterinarian refers the principle investigator (PI) to the institutional Chemical Safety Committee if the compound requires review. The PI submits a Notice of Intent (NOI) that provides critical information to the Chemical Safety Committee so that a risk evaluation can be conducted effectively. This NOI is evaluated using a banding process discussed earlier that ultimately results in the assignment of an ACL from 1 to 4 depending on the qualitative risk to humans, with ACL 4 designated for the greatest potential to affect humans. The Chemical Safety Committee then creates a hazard assessment for the PI that includes not only the band level but also the laboratory and animal facility precautions as relevant for the protocol. The band level control measures only apply to animal work, so the hazard assessment is an opportunity to address hazard mitigation that might not be covered by band level practices, such as administration procedures. IACUC will not issue an approval until the institutional Chemical Safety Committee approval process is complete. Each protocol should be considered unique and is evaluated as such because of the diverse scope of chemicals and procedures.
A committee will likely need to consult multiple sources to adequately evaluate risk. Table 2 lists some relevant references, and they will have varying applicability depending on the compound. The National Institute for Occupational Health and Safety (NIOSH) incorporated guidance from the American Society of Health System Pharmacists (ASHP, formerly American Society of Hospital Pharmacists) in a report on hazardous drugs in 2004, and the list has been regularly updated with the most recent changes proposed in 2018.31,32 The NIOSH Registry of Toxic Effects of Chemical Substances summarizes the literature available on the hazardous properties on many chemicals but was last updated in 1997 and is difficult to search. A literature search, and specifically TOXNET, will be invaluable in determining current information on chemical compounds. American Conference of Governmental Industrial Hygiene (ACGIH) Threshold Limit Values (TLV) or OSHA Permissible Exposure Limits (PEL) are not generally useful for animal model experimentation because testing for the compounds in animal waste is generally not feasible. The committee should also use the expertise and experience of other institutions that have protocols for managing particular compounds when available. The committee will always need to apply its professional judgment, particularly for novel compounds.
Information Resources to Assist in Assigning a Band Level for Chemicals Used in Animals.
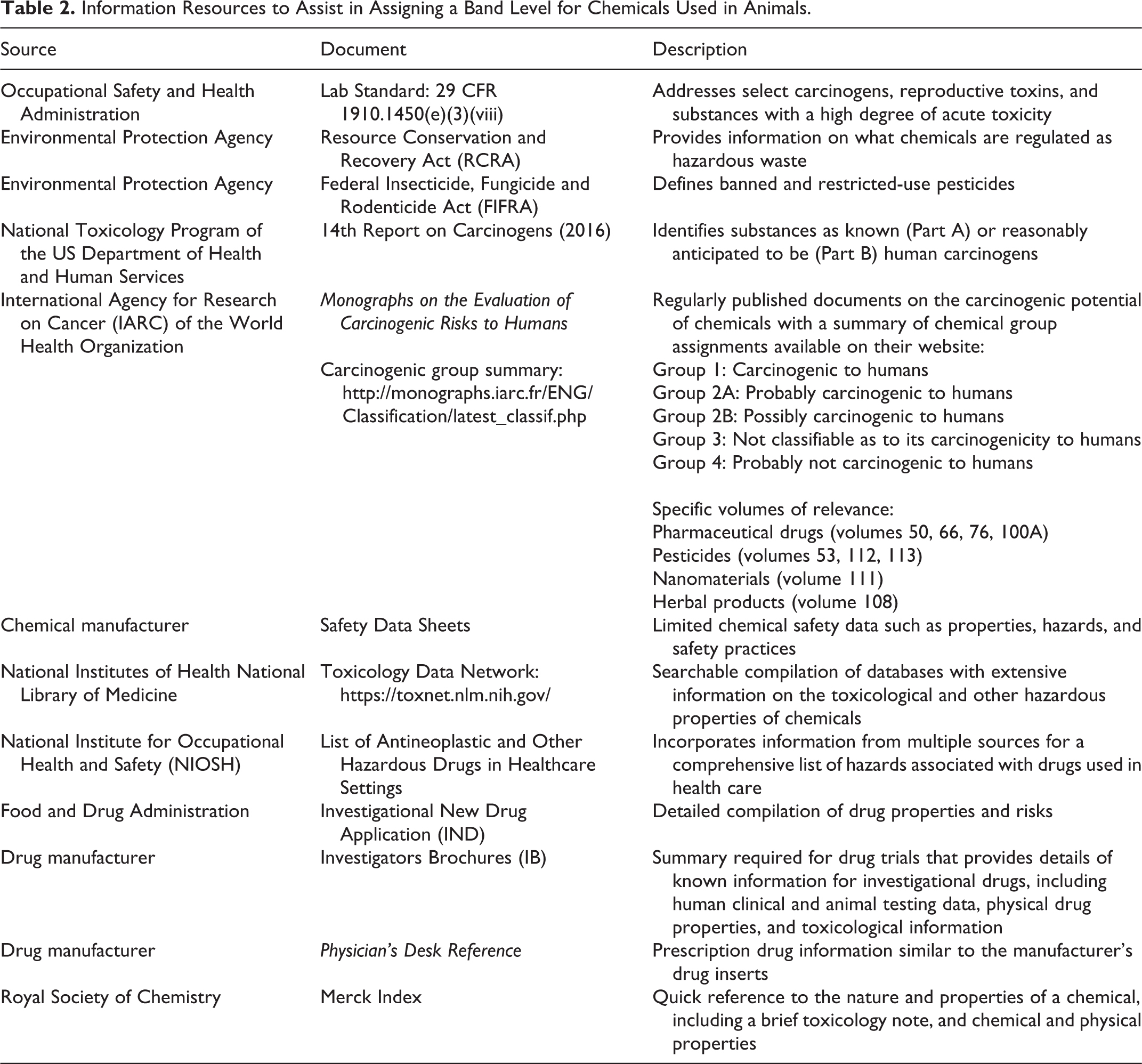
The authors’ institution streamlines the NOI process by limiting review of unnecessary compounds or those managed through other mechanisms. Drugs are excluded from the NOI process if they pose a minimal hazard risk or are not administered to the animal, including standard of care drugs (eg, analgesics, antibiotics), fixatives, and cleaning compounds. Anesthesia drugs are not evaluated in the NOI with the exception of urethane, which is considered a carcinogen and requires case-by-case evaluation. EHSO provides oversight of inhalant anesthetic use and other potential hazards that do not result in chemically contaminated animals through their policies and inspections.
Qualitative Assessment Criteria
Criteria for a qualitative assessment of risk include evaluation of compound properties (hazard category, severity, and physical properties), pharmacokinetics (metabolic rate and products), dose administered, route of administration and excretion, species, and protocol-specific information that might alter the band level or practices.
Hazard category and severity
A detailed description of the types of hazards posed by chemicals can be found elsewhere, 5 but generally, the committee will need to determine and address the following potential hazards posed by the chemicals: teratogen, acute or chronic toxicant, mutagen, carcinogen, irritant, sensitizer, immune modulating, endocrine disruptors, and environmental hazards. The category could affect the band level and will be necessary for the hazard communication.
Lethal dose, or LD 50, is a basic value to determine the toxicity of a chemical. If an LD 50 is unavailable, assessments could be made based on similar compounds. Many pharmaceutical compounds are potent, and appropriate risk reduction procedures must be considered. Compounds should be assumed to affect animals as they do people if alternative information is not available while recognizing this will not always be true.
Physical properties
While often not a prevalent risk in this type of evaluation, physical properties of a chemical should be considered in the risk assessment. The more important physical properties evaluated include flammability and volatility. For example, studies of the hepatic system occasionally use carbon tetrachloride, which is toxic and volatile, and ventilated housing systems might be warranted in these experiments.
Pharmacokinetics
The metabolic rate and by-products provide essential information to determine risks and potential routes of excretion. Compounds with a short half-life might present a negligible excretion hazard. The metabolites could have more, less, or similar toxicity compared with the parent compound.
Clearance time of the compound will assist the committee in determining the time period for which animals are considered hazardous after they have been administered the chemical. If animals are maintained permanently as contaminated, the population of contaminated animals will become quite sizable and largely occupied by a group that does not actually pose a hazard. Conversely, assigning a unique contamination time frame to each protocol could create a complicated system leading to error and confusion. A more consolidated system of one or a few standards for contamination days based on a conservative estimate of excretion period for most protocols could be developed, and exceptions could be made of unique circumstances where warranted. The authors’ institution uses a standard of 3 days after administration of a chemical for which an animal is considered contaminated, which is used for most protocols in rodents. Animals revert to standard housing conditions after this 3-day period. Alternatively, an institution could use the time periods for contamination as differentiating control measures in the banding system.
Dose administered
The amount of chemical administered is important in determining relative toxicity and the potential for human exposure. Generally, the more compound administered, the higher potential for human exposure, which could lead the committee to assign the same compound to different band levels depending on the dose or species.
Route of administration and excretion
The common routes of administration for chemical compounds include oral (food, water or gavage), intraperitoneal (IP), intravenous (IV), intracranial, intraocular, and dermal. The most likely exposure to personnel is during administration of the compound to the animal. Risks include self-inoculation or contact with the compound from administration site leakage. Sedation of the animal during administration would reduce risks to personnel. Drugs administered via the food or water could be provided by a vendor or made by the researcher, and the higher the hazard, the less likely the researcher is to find a vendor to make it. Risks during the drug-handling and administration process might not factor directly into the band level assigned but should be addressed in a risk assessment summary provided to the researcher.
The route of administration along with pharmacokinetics will determine potential routes of exposure after the chemical has been administered to the animal. Urine, feces, and dermal and respiratory routes are all possibilities to address. An intracranial or intraocular administration uses a very small dose and is unlikely to leave the location in appreciable amounts for general body distribution.
Species
Species differences in species, size, behavior, and toxicological response change risk management methods. For example, mice can require a 10× dose of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) to elicit the same response compared to a primate. 33 Rodents require much less intense husbandry techniques and are easier to contain than nonhuman primates.
Protocol specifics
A common alteration to standard band levels at the authors’ institution is applied when chemicals are used in terminal procedures. Compounds that might normally be above ACL 1 but are used terminally are typically assigned to ACL 1 because the animal will not be housed after the procedure, with special provisions in the hazard assessment for drug administration if needed. Conversely, the severity of hazard might be increased if multiple chemicals are administered to the same animal.
Conclusion
The band level designations and hazard assessment summary will provide a PI with clear instructions on how to conduct their procedures. Challenging scenarios will inevitably arise due to the inherently novel and complex nature of a research environment. The considerations and process described above for reducing risk will serve as a solid foundation to help the institution approach these situations. Some possible challenges to consider include the following:
Nanomaterials are a largely unknown risk after they have been administered to the animals. NIOSH continues to publish updated recommendations for handling nanomaterials, and the National Research Council guidance, Prudent Practices in the Laboratory: Handling and Management of Chemical Hazards,
5
also includes a chapter on nanomaterials. At the authors’ institution, all nanomaterial use in animals is reviewed by the Chemical Safety Committee. Band level designation primarily relies on the hazard of the parent compound or associated compounds in combination with the risk that a nanomaterial could be excreted from the animal because nanomaterials could be more hazardous than the parent compound. Experimental compounds can have poorly documented properties. In this situation, the control level can be established based on chemical structure and properties of similar compounds. A conservative approach of assigning a higher level when the risk is unknown is reasonable, and the committee should be willing to modify band assignments when new information is available. Companies might be reluctant to disclose information on proprietary compounds. A member of the committee might need to sign a confidentiality agreement to obtain the information for determining a band level. Biological toxins often bridge between biological and chemical safety. Usually the IBC would determine if a toxin should be on an IBC or chemical safety protocol. Larger species such as nonhuman primates, sheep, pigs, and others present a significant challenge in management of food, bedding, and waste. Often the housing facility is considered a single cage, and the entire room is managed accordingly. Noncompliance with the required procedures can be mitigated with postapproval monitoring (PAM). PAM can include EHSO inspection, IACUC inspection, and visual inspections by animal resources staff to ensure compliance with signage, protective practices, and training requirements.
A chemical safety committee has a responsibility to both facilitate research and ensure the safety of personnel. The committee should be sensitive to the institution-wide burden placed on researchers and strive to implement safety processes of value and with sensitivity to researcher goals and deadlines. Enhanced controls incur additional animal husbandry costs, husbandry staff time, and research staff effort. The graded approach of an animal chemical control banding system allows for adequate but appropriate controls for efficient use of resources and time.
Footnotes
Acknowledgment
We thank Dr. Michael Huerkamp for his contributions to the development of this program.
Author Contribution
Portions of this material were presented in a webinar for the Southeastern Biological Safety Association in 2017.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
