Abstract
This study assessed biosafety performance between 5 selected private-sector hospital medical laboratories in Karachi, Pakistan. A 40-question questionnaire was distributed among 120 technical staff to better understand the practices and awareness level of technical staff in place. To assess the supply of biosafety facilities and equipment to keep laboratory workers and the surrounding environment safe from an outbreak of infectious diseases, a checklist was designed. After securing informed consent, the researcher anonymously interviewed technical staff from all sections of the pathology laboratory, filled out the standard questionnaire, and completed the checklist through personal observations. Biosafety awareness recorded among 5 laboratory technical staff was moderate (72%). Major gaps identified with reference to biosafety measures were in administrative controls, standard microbiological practices, and facility design/secondary barriers. The gaps included the following: the biosafety officer is appointed in only 1 laboratory, standard operating procedures are not in place in 3 laboratories, no training protocol is defined for staff, no immunization record is maintained, and incident reporting and recording is lacking at most laboratories. There is an adequate supply of simple personal protective equipment like gloves, goggles, masks, and lab coats. However, complex equipment, such as eyewash stations and biological safety cabinets, is not available in many laboratories. In Karachi, Pakistan, biosafety performance is compromised in most of the laboratories, but better performance also is demonstrated by laboratories that are accredited and certified.
There is no doubt that a clinical laboratory in a hospital plays an important role to serve the patients. Therefore, an effective use of clinical laboratories will translate into effectiveness and efficiency of the organization. Nevertheless, although many organizations accept this to be true, they fail to realize that as part of their clinical laboratory management practices, there is the need for management to ensure that workers in the clinical laboratories work in a safe and healthy environment that will promote their maximum utilization. Emphases should be made to avoid accidents that are costly to both affected staff and the organization. Consequently, management and staff should make every possible effort to avoid them from happening in the workplace. Workers’ poor knowledge of health and safety measures also hinders clinical laboratories’ effectiveness. Therefore, proper training of staff will be important to examine their effectiveness.
Biosafety is an important issue in worldwide laboratory settings. Workers in clinical laboratories, especially those who are working in microbiology laboratories, are more susceptible to laboratory-acquired infections. 1 Biosafety is also an important element of a quality management system as it is a measuring tool for compliance with accreditation and certification standards. As far as safe handling and containment of infectious microorganisms and hazardous biological material is concerned, biosafety has become the code of practice in microbiological and biomedical laboratories for the past 2 decades.
A growing body of literature has examined biosafety performance around the world. In some regions, biosafety performance has significantly increased, but in developing countries, there is still a need to improve biosafety practices, especially diagnostic laboratories in Africa, where biosafety performance has been compromised because of poor administrative controls and unavailability of biosafety facilities. 2 Another study conducted in clinical laboratories in Nigeria revealed a similar biosafety condition, where there was a strict need for adherence to biosafety policies and practices as the level of biosafety performance was not impressive. 3 Similarly, biosafety precautions adopted by the laboratory workers in Sudan were below the standards with reference to biosafety awareness and practices. 4
Lack of awareness of biosafety measures and practices among clinical laboratory personnel and inadequate supply of biosafety equipment are the main reasons for poor biosafety performance in Pakistan. 5 A biorisk survey conducted among clinical laboratories in Karachi 6 identified that no biological safety cabinets (BSCs) were available in many biosafety level 2 (BSL-2) laboratories. Negligence with respect to all major practices, personal protective equipment (PPE), engineering controls, and risk assessment was observed. Moreover, there was no system of Laboratory associated infections (LAI) reporting in place, and knowledge and training of biosecurity and biosafety were below the standards. The present study attempted to examine the current situation of biosafety performance by assessing the awareness level and practices of technical staff and by inspecting the availability of biosafety facilities and equipment provided for clinical laboratory workers to keep themselves and the environment safe from outbreaks of any occupational infection.
Methods
This study was conducted from July 2016 to December 2016 in Karachi, Pakistan. In this study, the survey analyzed biosafety performance among 5 selected laboratories in Karachi through a structured questionnaire and checklist. This study encompassed 6 months. After securing informed consent, a researcher anonymously interviewed technical staff from all sections of the pathology laboratory, filled out the standard questionnaire, and completed a checklist through personal observations.
The instrument had been prepared after thorough and comprehensive study, discussions with supervisor and seniors, and researcher satisfaction followed by in-depth review of the literature,7 -9 which guided the researcher to compile a comprehensive questionnaire and checklist for the research study. To ensure face validity and content validity, the tool was given to a committeel of 3 experts. After slight modifications suggested by experts, the tool was verified to collect the data.
A comprehensive questionnaire of 40 items was prepared for technical staff and a checklist of 40 items for personal observations. Using the survey questionnaire, the current level of awareness and safety practices/standard microbiological practices of technical staff was measured. The questionnaire contained 3 broad sections: sociodemographics, general biosafety, and standard microbiological practices. In general biosafety, questions were related to the general terms and concepts of biosafety (eg, what BSL stands for, which level of biosafety was staff working in, what kind of PPE was used for eye protection). Moreover, questions related to hand washing, handling of sharps, mouth pipetting, decontamination of work surfaces, and so on were included in standard microbiological practices, and personal observations in the form of a checklist highlighted availability of biosafety facilities and equipment in the form of administrative controls, primary barriers/PPE, and secondary barriers/engineering control. Furthermore, the checklist also contained the major standard microbiological practices to personally monitor practices of technical staff. Close-ended questions in the questionnaire offered a set of alternative answers from which the respondents were asked to choose the one that most closely represents their views.
Sampling Plan
Technical staff from 5 different private-sector hospital medical laboratories were selected for data collection. Hospitals were selected considering feasibility and responses from management. Among those 5 hospitals, 3 were so small that the whole population was included. For the other 2 hospitals, simple random sampling of staff was employed. Excel (Microsoft, Redmond, WA) was used to randomly select 47 technical staff from laboratory A and 30 technical staff from laboratory B.
Study Design
This research is a cross-sectional descriptive study in the form of a survey to analyze biosafety performance through a structured questionnaire and checklist. It made use of both quantitative and qualitative tools in analyzing the data collected through questionnaire, checklist, interviews, and personal observations. The analysis of data collected was done at the end of the data collection. To interpret frequency, graphical presentation was used through Excel, while qualitative analysis of the personal observations for standard microbiological practices was made and the results were described in narrative form.
Sample Size
The sample size for this study was 120 technical staff working in BSL-2 medical laboratories of 5 different private-sector hospitals in Karachi. They included 47 staff from laboratory A, 30 staff from laboratory B, 20 staff from laboratory C, 13 staff from laboratory D, and 10 staff from laboratory E. Laboratory A is large and accredited by the Joint Commission International Accreditation (JCIA), College of American Pathologists (CAP), and International Organization for Standardization (ISO). Laboratory B is medium sized and ISO certified, and laboratories C, D, and E are small sized.
After securing informed consent, all technical staff who voluntarily participated in this survey were interviewed using a structured questionnaire. Personal observations were recorded by the researcher in the form of checklist, and interviews were also conducted for the management staff to check their perspective toward biosafety administrative controls. After collection of data, analysis was performed, based on information provided by respondents. Classified the responses and summarizes based on information provided by respondents.
Results and Discussion
We undertook this study to evaluate the current situation of biosafety performance among 5 different hospital medical laboratories in Karachi, Pakistan, through a structured questionnaire and checklist. Our study revealed a few improvements with regard to the awareness level of technical staff and supply of simple PPE in comparison to previous studies conducted in Karachi. However, major deficiencies were also found in critical areas of biosafety measures, indicating a compromised situation of biosafety in Karachi, Pakistan.
Further details of the survey are presented below.
Among 120 studied laboratory personnel, most of the staff were technologists/scientists (78%), except for laboratories D and E. The proportion of female staff was relatively high (55%) compared to male staff, except for laboratories D and E. Experience of 1 to 2 years was predominant at 36%. Most staff had completed master’s and graduate degrees (53.3% and 29.2%, respectively). About 81% of the studied staff worked the morning shift.
To have a hazard-free working environment, each staff member dealing with the infectious agent or potentially infected material must have knowledge of potential hazards, be trained to follow such practices, and be familiar to deal with such safety devices. 10
Safety awareness is getting worldwide attention. Scholars and experts from all over the world have made different definitions of safety awareness from different points of view. Safety awareness from a personnel safety point of view may be explained as being responsible, not overestimating one’s own self-confidence, and taking a cautious attitude to control one’s behavior. This study revealed that biosafety awareness among private-sector laboratory workers was of a moderate level (72%). However, workers at laboratory A had a relatively high percentage of 79%. On the other hand, a very low percentage of 43% was also observed in laboratory E (Figure 1).
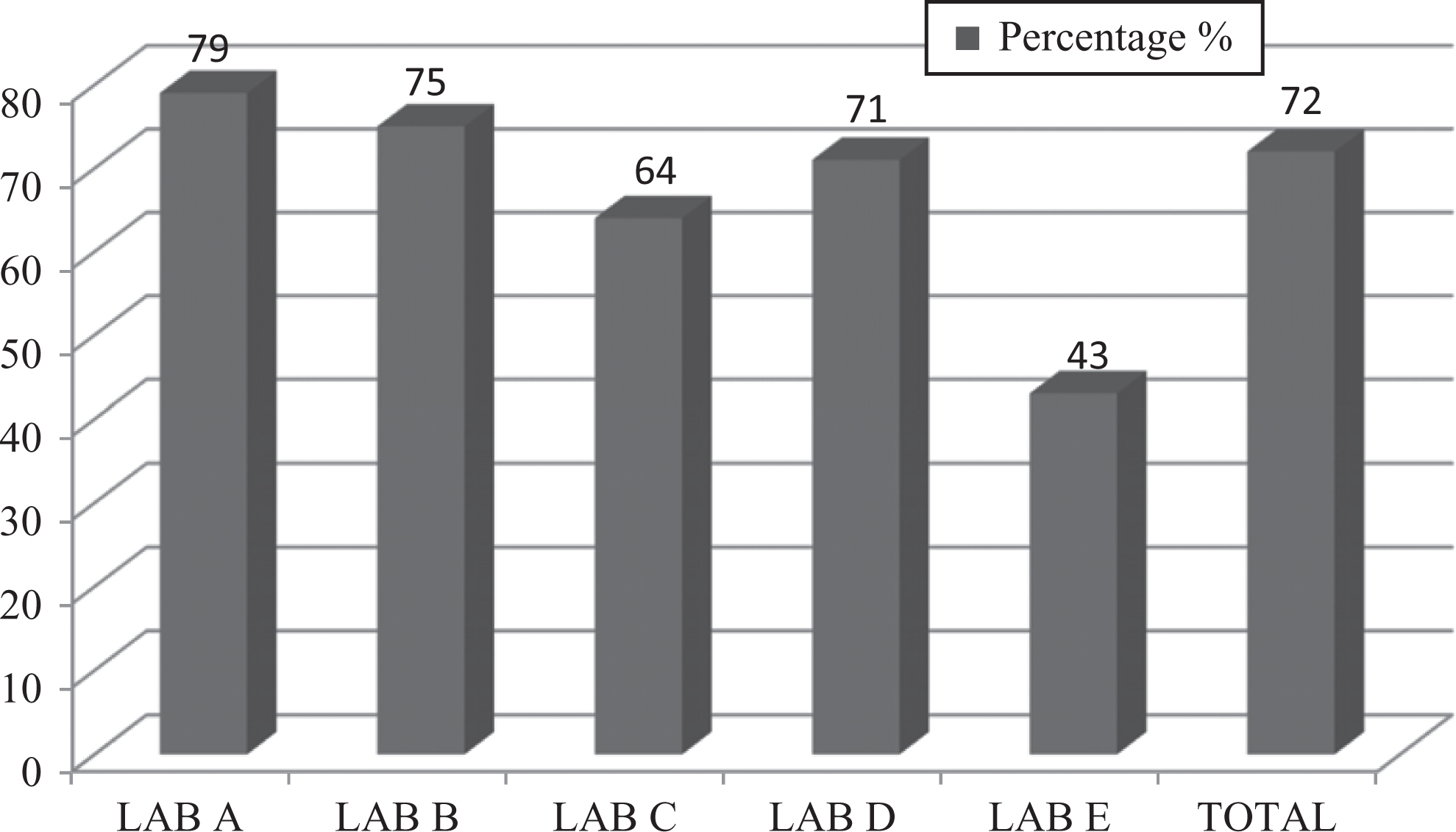
Graph represents percentages of overall biosafety awareness of studied laboratory personnel.
Administrative Controls
To control the spread of infections in the work environment, use of administrative controls is the first priority.11,12 This involves those controls that are taken by management to reduce risks by making policies for workers and by providing appropriate training (eg, training, staff immunization, incident reporting). Our observations indicated that the level of biosafety management in place in 5 of these laboratories was not adequate at 37%.
It was alarming to note that a biosafety officer was appointed in only 1 laboratory (20%). A safety manual or standard operating procedures (SOPs) were not in place in 3 laboratories (60%). There was no formal biosafety training protocol defined for staff. A staff immunization record was not provided in many laboratories. One of the major deficiencies observed was for incident reporting and recordkeeping. Although the staff were aware of incident reporting, no record was maintained. Percentages of biosafety training, staff immunization, and incident reporting in all 5 laboratories are shown in Figure 2.
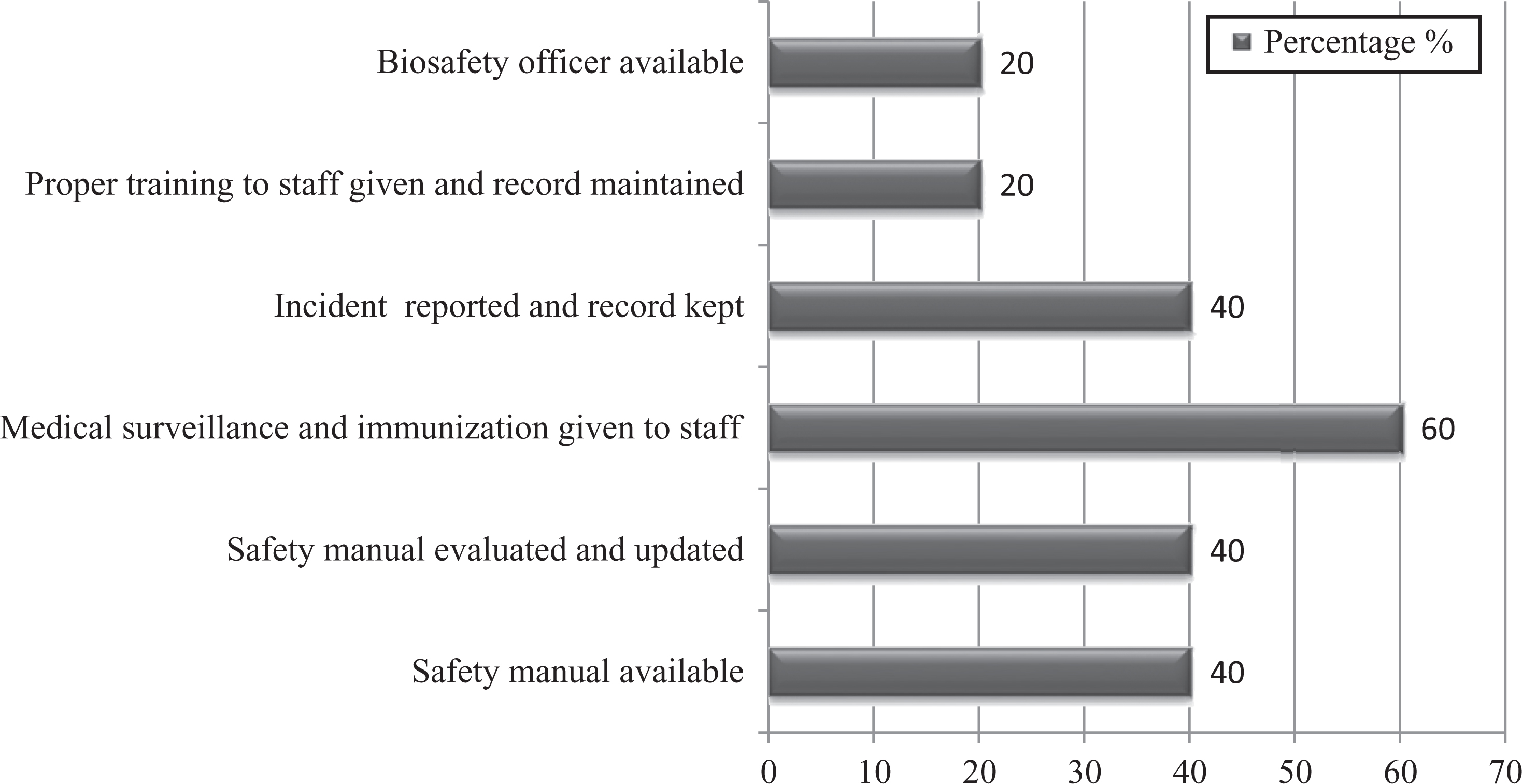
Graph represents percentages of administrative controls of studied laboratories.
Standard Microbiological Practices
The level of biosafety practices of technical staff in the 5 studied laboratories was moderate at 68%. However, it was also found that biosafety practices of laboratory A workers were much better (80%) in comparison to rest of the 4 laboratories (Figure 3).
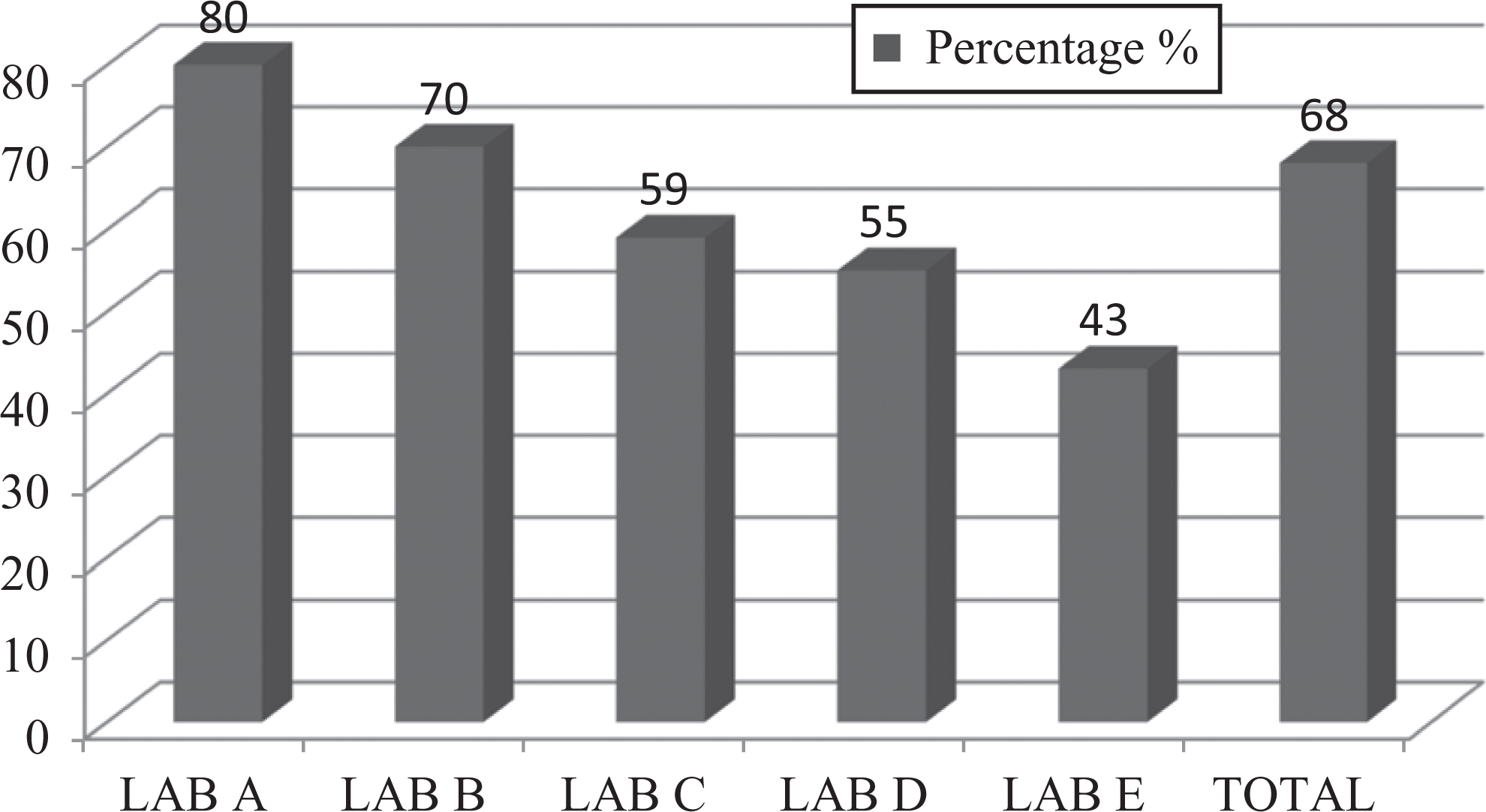
Graph represents percentages of standard microbiological practices of studied laboratory personnel.
This survey revealed that 78% of staff washed their hands after removing gloves and before leaving the laboratory; 88% of staff did not eat, drink, smoke, apply cosmetics, or handle contact lenses while working in a BSL-2 laboratory; and 77% of workers practiced transportation of biological materials from one lab to the other in a leakproof, shatter-resistant secondary container. Inside a BSL-2 laboratory, storage of food items is not allowed. This study found that only 63% of staff stored food items outside the laboratory (Figure 4). The practice of mouth pipetting is considered obsolete and forbidden in the laboratory in any circumstances. 8 It is evident from the study that most staff did not follow mouth pipetting (83%), but 8.3% of staff still practiced it in some areas (Figure 5). As per Occupational Safety and Health Administration (OSHA) standards, contaminated needles and other sharps should not be bent, recapped, or removed manually. 13 This survey report revealed 70% compliance with this (Figure 6), indicating that there is still a need for improvement to avoid any exposure to blood-borne pathogens as needles and sharps are the sole source of exposure to infectious agents among laboratory workers. When a sharps container became full, 72% of staff got new container and sent it for incineration, indicating that there is enough supply of separate sharps containers for disposal of sharps.
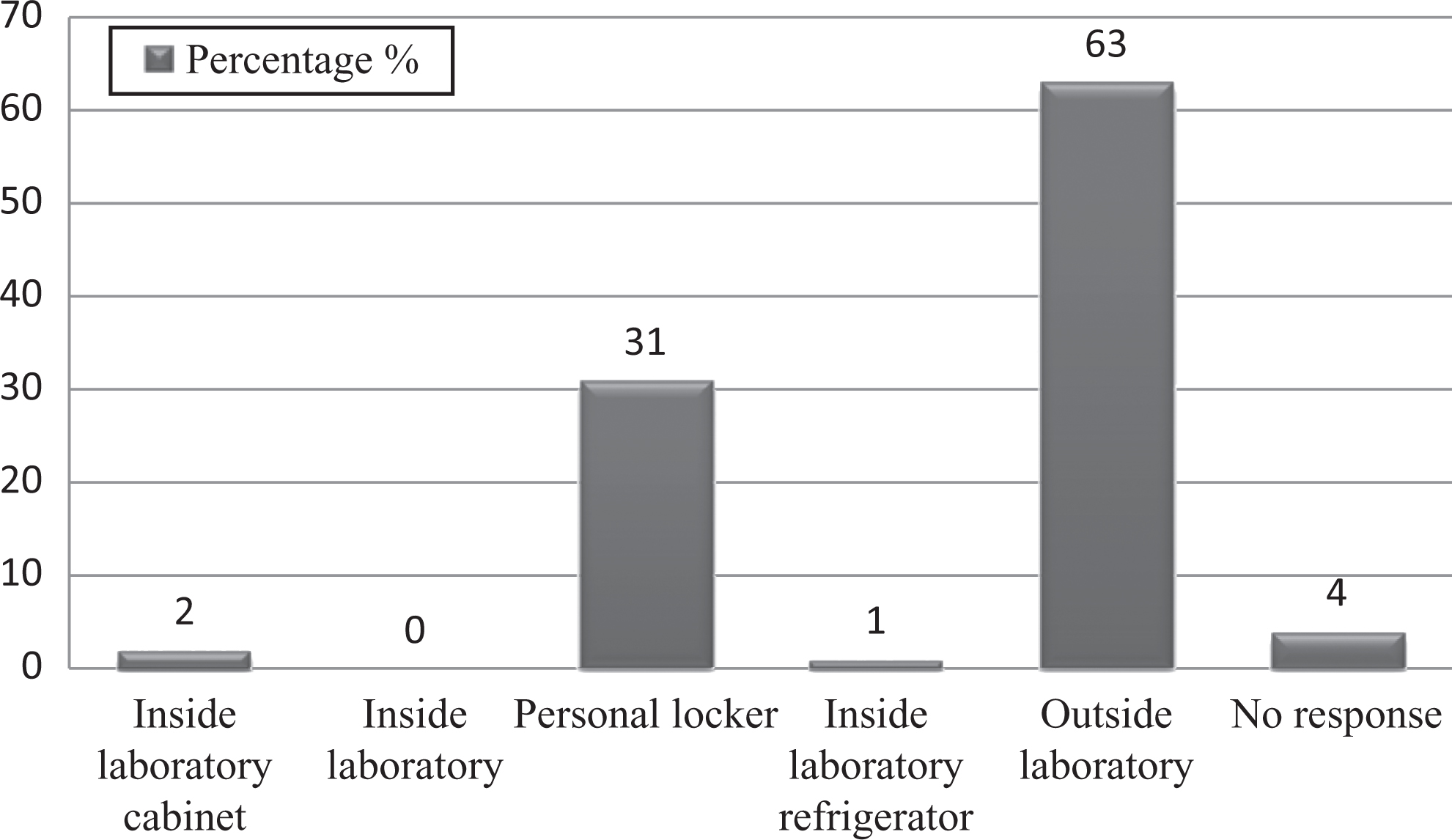
Graph represents percentages of studied laboratory personnel practices for food storage inside the laboratory.
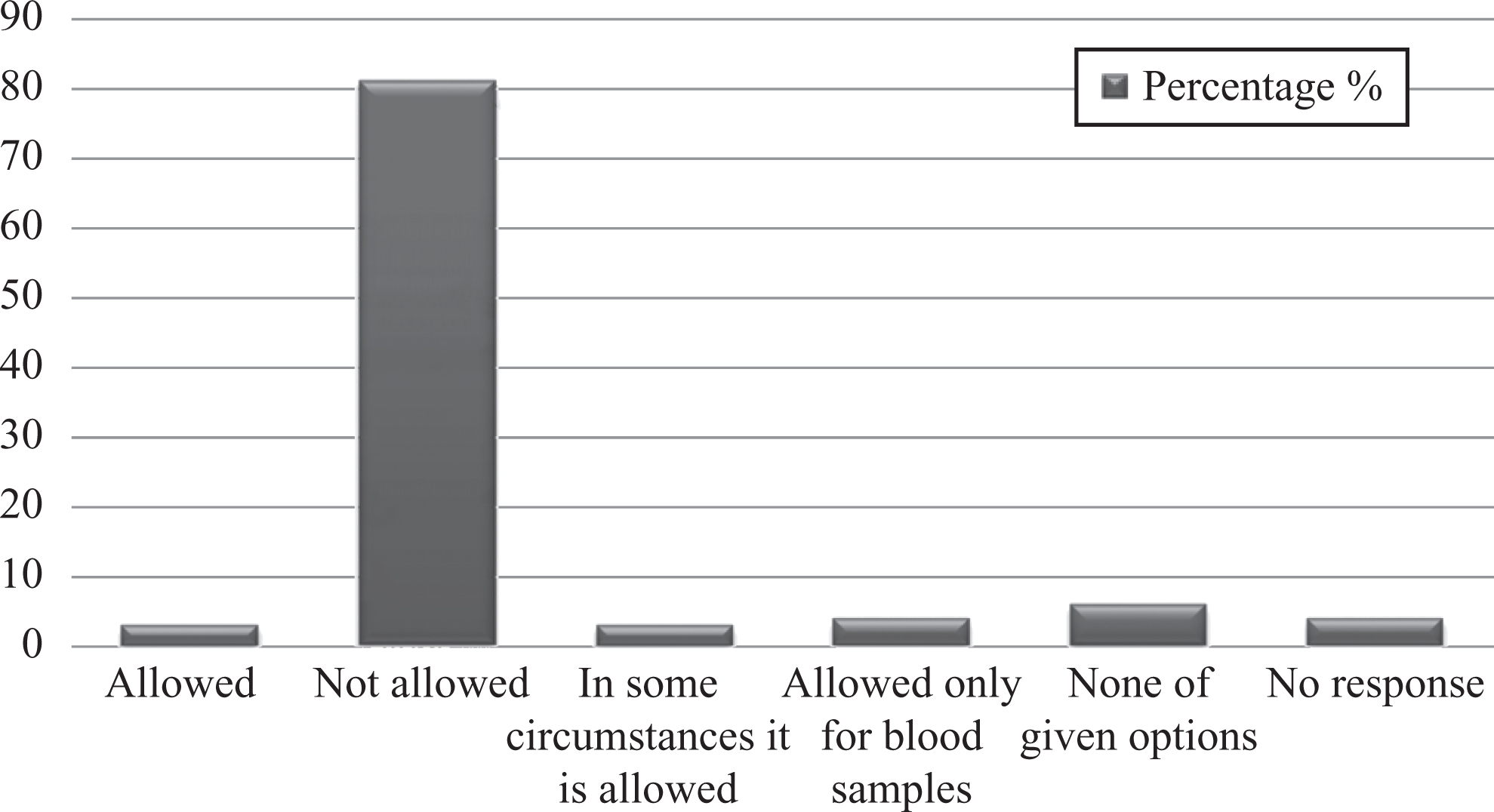
Graph represents percentages of studied laboratory personnel for practice of mouth pipetting.
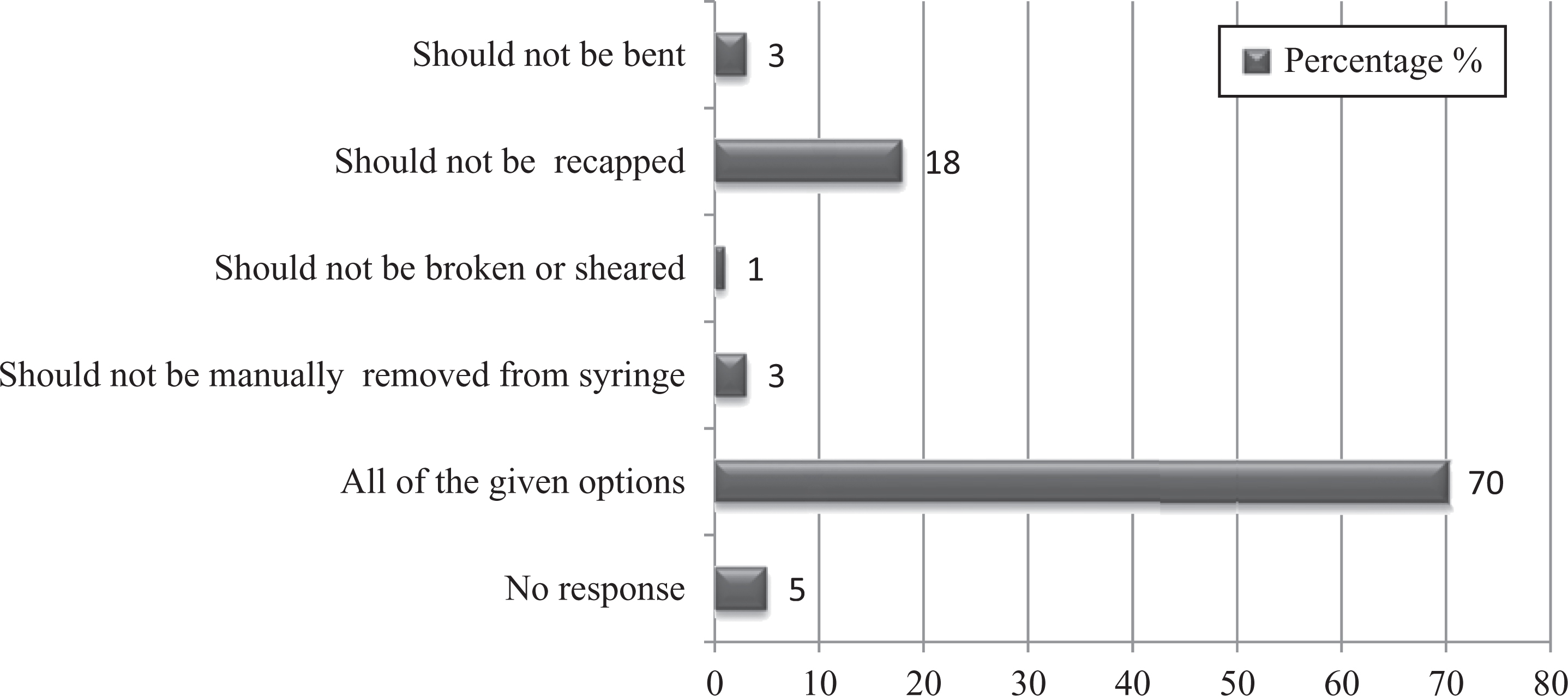
Graph represents percentages of studied laboratory personnel practice for handling and discarding needles.
Decontamination of working benches and other surfaces should be done daily at the start and after completion of work. This survey revealed that most respondents (84%) followed this practice (Figure 7). Moreover, it was also noted that 41% of workers followed proper decontamination of equipment. In case of biological spills, 63% of staff referred to the safety manual for spill cleanup, 16% called the safety committee, 5% waited for housekeeping, and 5% left laboratory.
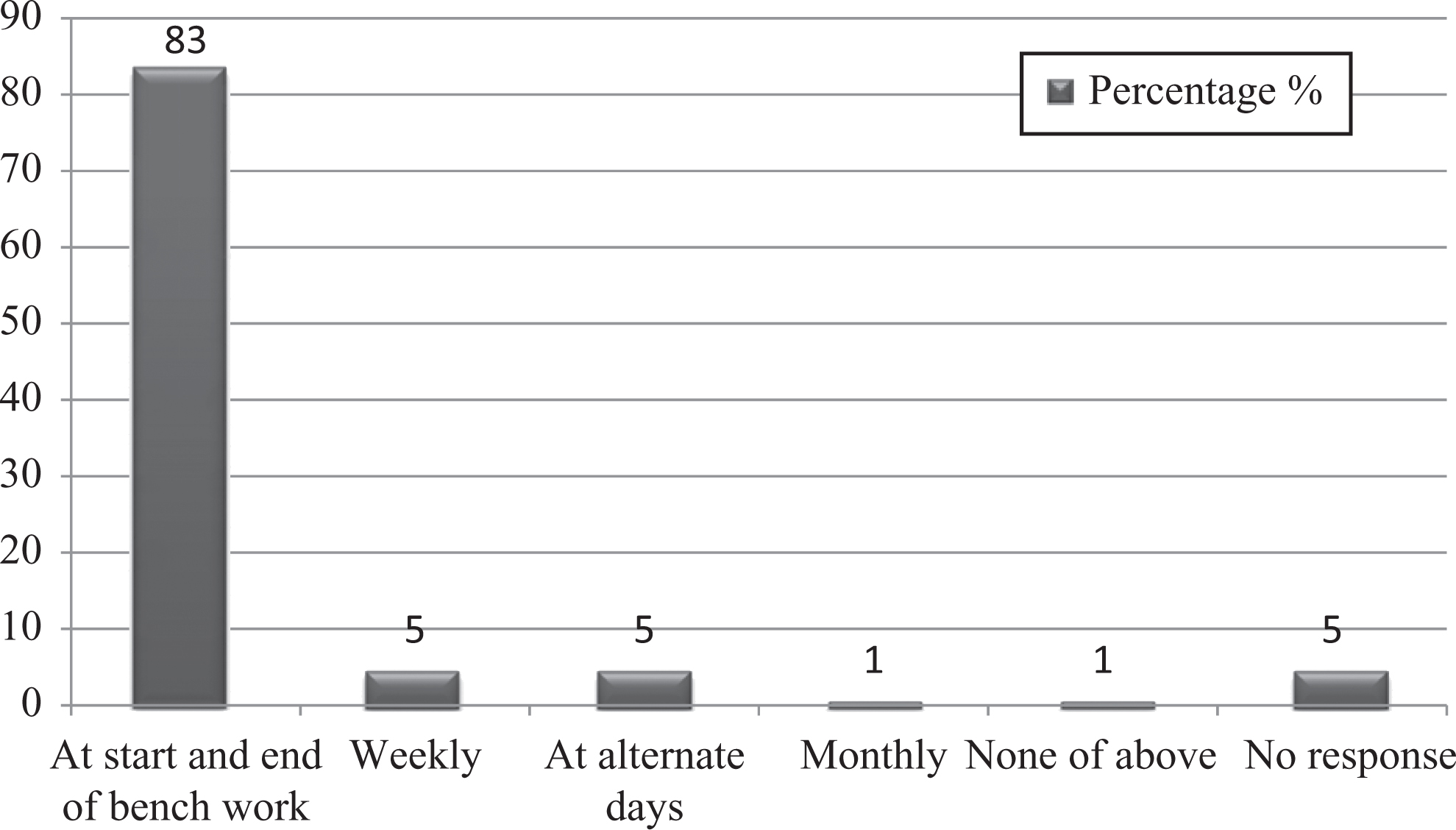
Graph represents percentages of studied laboratory personnel for practice of decontamination of bench.
Training plays an important role to get employees aware of hazards and to avoid any work-related infections. Training should transmit knowledge and skills so staff can identify hazards, adopt safe behaviors, and change attitudes to reduce exposure to these hazards. In removing or controlling hazards, an empowerment perspective prioritizes the workers’ role. To achieve this goal, health and safety training must be conducted and designed in ways that train workers how to identify hazards as well as how to engage effectively as a team to move the organization to eradicate workplace hazards. The present study demonstrated that 38% of staff got training on a monthly basis, 19% annually, and 18% biannually (Figure 8). This survey indicated that accidents/injuries were reported to the supervisor/safety officer by 88% of staff, but unfortunately, the record was not maintained for it.
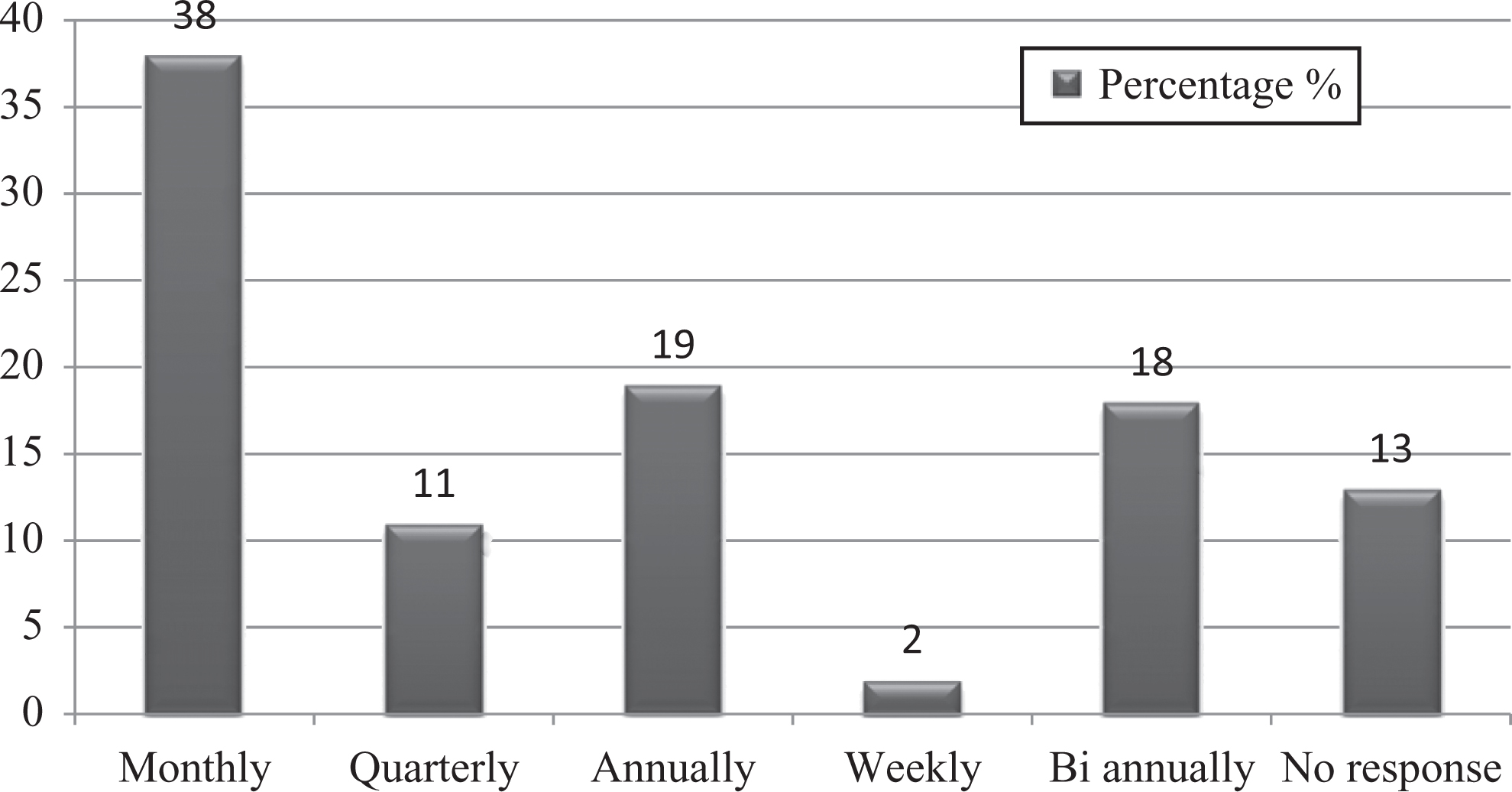
Graph represents percentages of studied laboratory personnel for biosafety training courses.
Personal observations recorded are as follows.
Staff and management were aware of not eating and drinking in the laboratory, but water coolers were discovered inside 2 laboratories, even though there was food storage inside the laboratory. Although staff has knowledge not to work without lab coats and gloves, most of the staff were found working without gloves and lab coats, especially in the evening shift when management was gone. Awareness regarding close-toed footwear was good, but most of the staff were found working with sandals. Spill cleanup procedure was not available and followed in 4 of the laboratories, and in these laboratories, there was no system for decontamination of waste prior to disposal.
PPE/Primary Barriers
Personal protective equipment, also known as PPE, includes equipment that protects workers against hazards of infectious material while handling them. It includes gloves, lab coats, masks, and so on. The present study showed that there was adequate knowledge regarding simple PPE. For instance, 98% respondents had knowledge of using a mask for respiratory protection. Gloves should be inspected for leakage before use; worn to completely cover the hands and wrists; replaced if torn, damaged, or contaminated; and removed if soiled before handling the telephone and keyboard. Our survey revealed that 82% employees were aware of these measures. On the other hand, knowledge regarding removal of gloves while answering the phone, opening the laboratory doors, and using a keyboard was 72%. Most (87%) staff were familiar with the abbreviation of PPE. In addition, 73% were aware that shoes should be comfortable, have nonslip soles, and be fluid impermeable and close-toed (Figure 9). Knowledge regarding usage of goggles for eye protection was 89%.
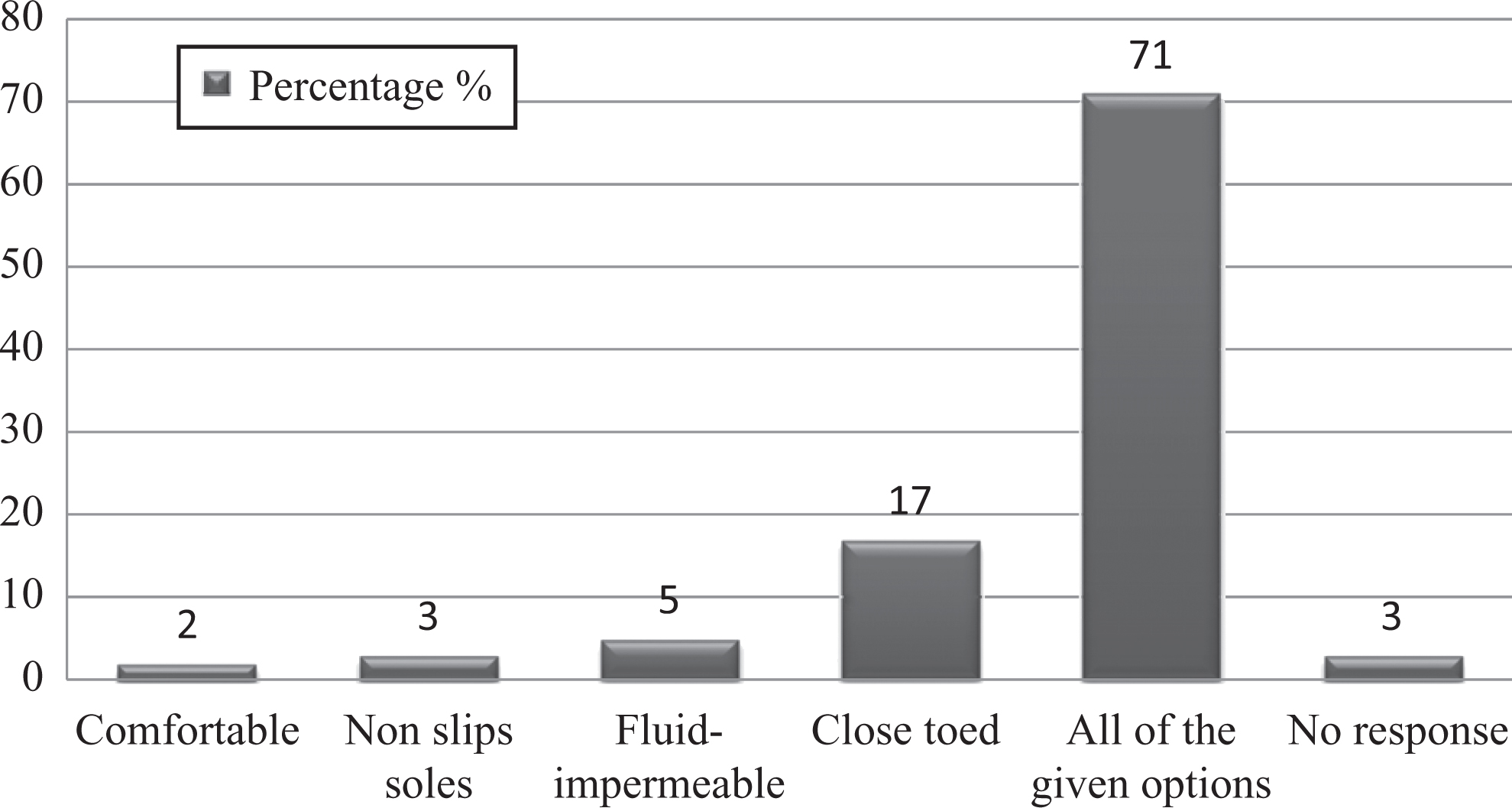
Graph represents percentages of studied laboratory personnel awareness regarding kind of footwear permissible in the laboratory.
This survey report revealed that there was enough supply of simple PPE (eg, lab coats, gloves, and masks) for the employees to work in BSL-2 laboratories during routine work (83%). However, complex safety equipment (ie, BSCs) was available in only 2 laboratories (40%) (Figure 10).
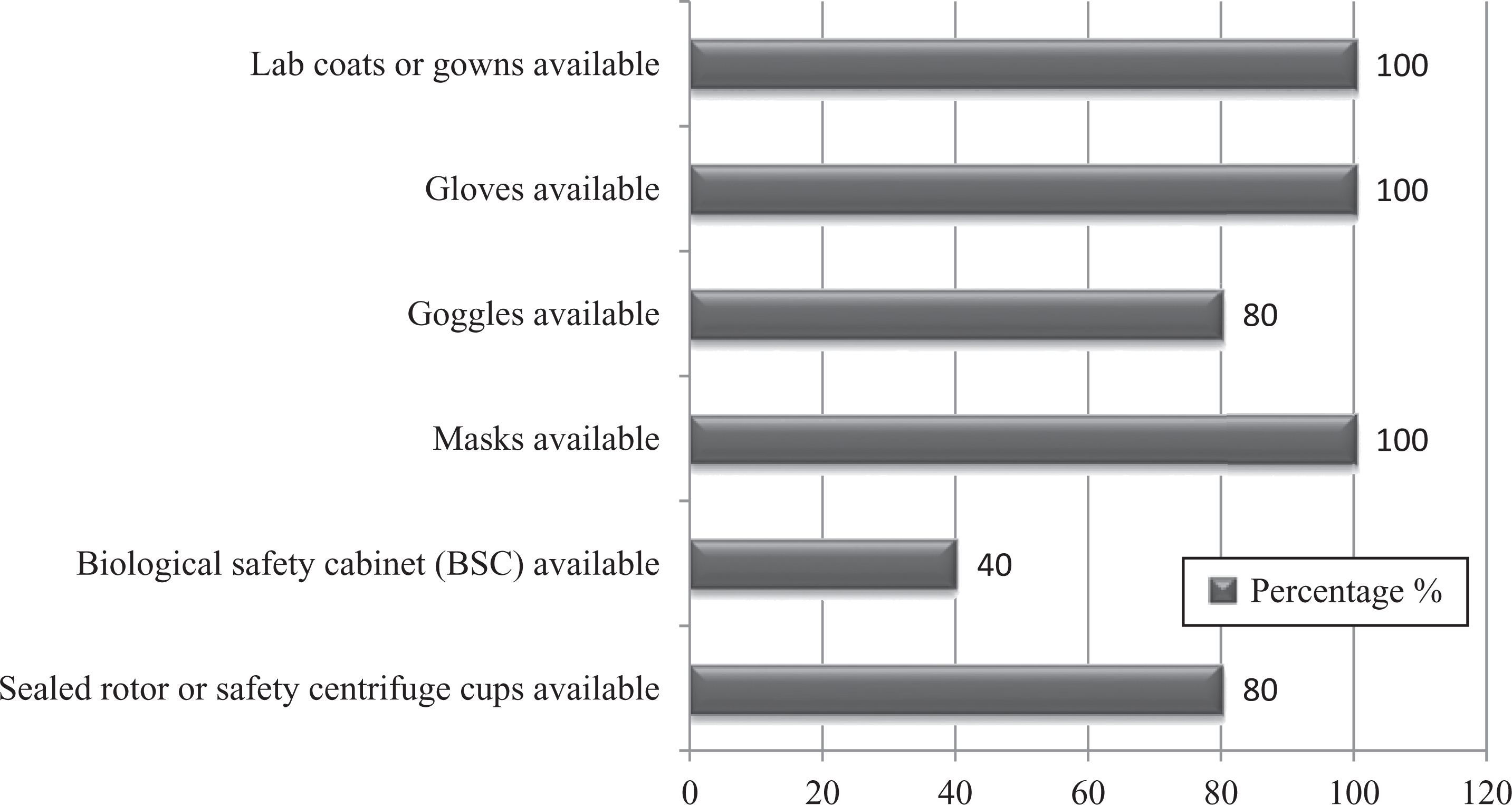
Graph represents percentages of availability of personal protective equipment/primary barriers for studied laboratory personnel.
Facility Design/Secondary Barriers
Those biosafety measures that reduce the hazards of the working environment are categorized as engineering controls or facility design. These types of controls are permanent and important in comparison to the other 3 controls, as these do not depend on the employee. Our survey report identified that the overall percentage of biosafety toward facility design of 5 laboratories was 63%. It was alarming to note that there was no biohazard sign posted at the entrance of all the laboratories. An eyewash station was available in only 3 of the laboratories (60%). In most laboratories, there was no adequate space for storage. Surprisingly, a hand-washing sink was not found in one of the laboratories (Figure 11).
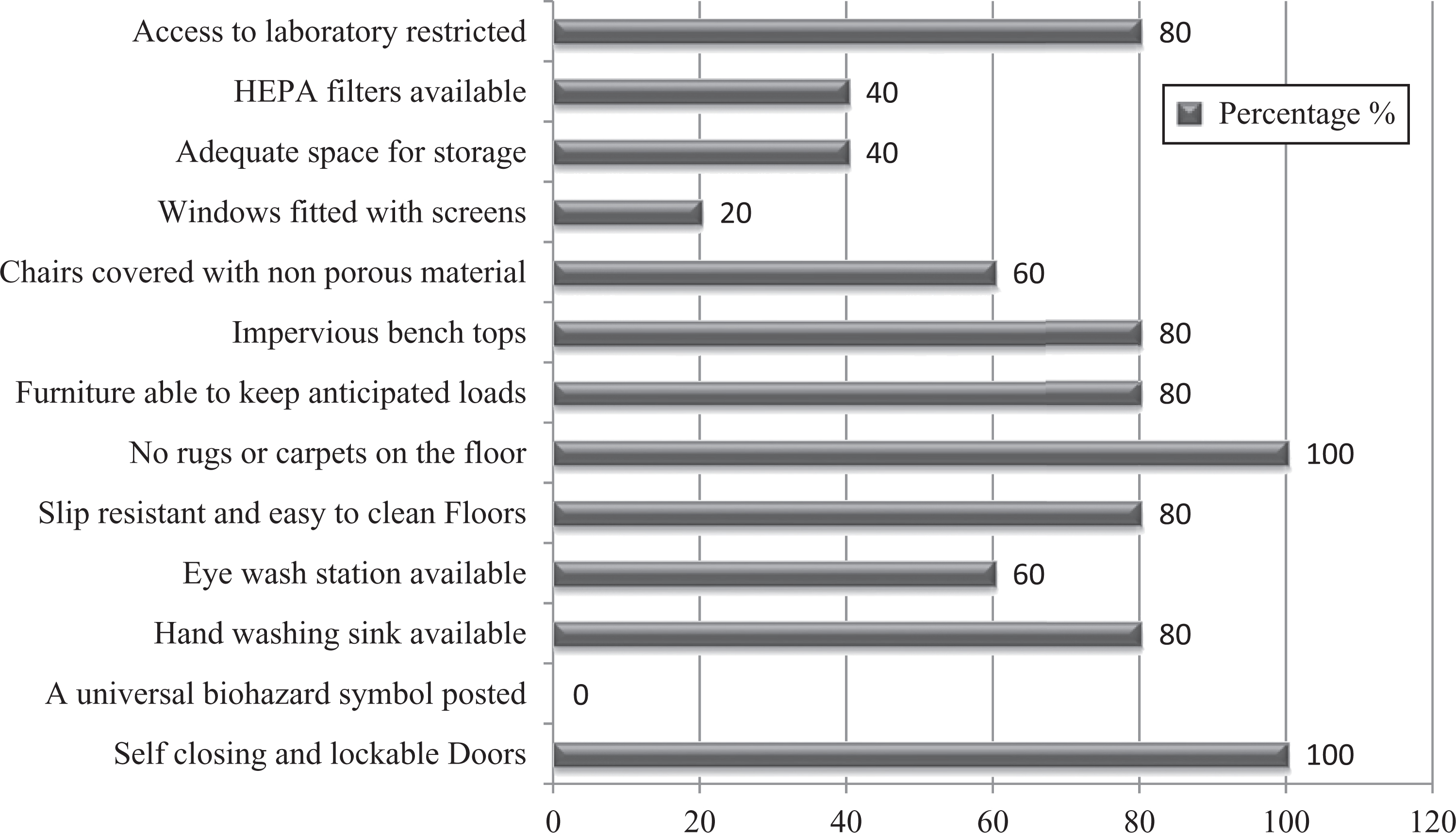
Graph represents percentages of facility design/secondary barriers of studied laboratories.
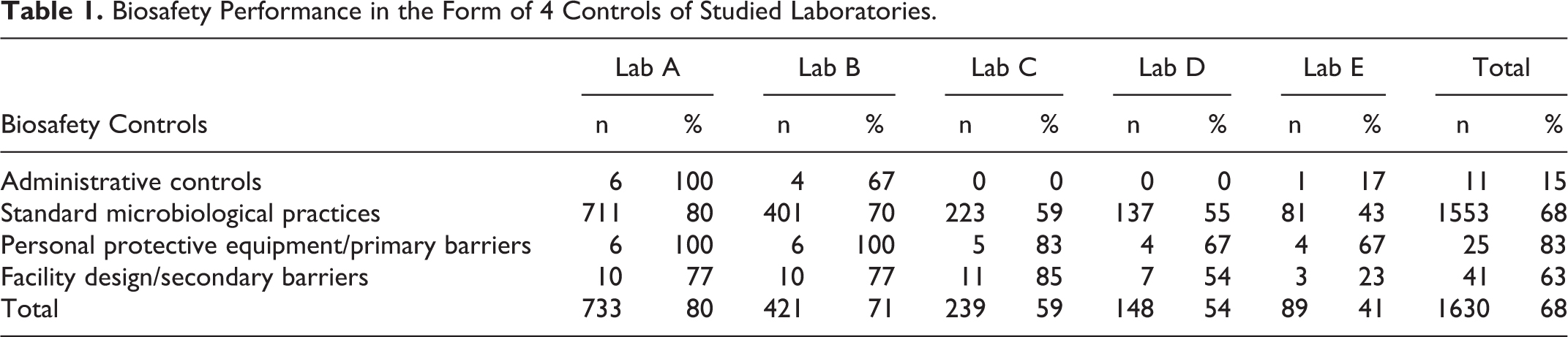
This study found that the percentage of biosafety measures toward administrative control, standard microbiological practices, PPE/primary barriers, and facility design/secondary barriers was 80% in laboratory A and 71% in laboratory B. The percentages were 59% and 54% for laboratories C and D, respectively, indicating a moderate level of biosafety measures in the form of 4 controls of the 5 medical laboratories in Karachi (Table 1). However, a much more compromised situation was demonstrated by laboratory E with a total score of 41%.
Biosafety Performance in the Form of 4 Controls of Studied Laboratories.
Conclusion
This study attempted to reexamine the current situation of biosafety performance among 5 selected hospital medical laboratories in Karachi, Pakistan. On the whole, the situation is not very promising with reference to all critical areas of biosafety. On the other hand, better performance also was demonstrated by laboratories that are accredited and certified. Major gaps identified with reference to biosafety measures were in administrative controls, standard microbiological practices, and facility design/secondary barriers. The gaps included the following: a biosafety officer is appointed in only 1 laboratory, SOPs are not in place in 3 laboratories, no training protocol is defined for staff, no immunization record is maintained, and incident reporting and recording is lacking in most laboratories, indicating carelessness of management staff toward biosafety measures. In addition, there is an adequate supply of simple PPE (eg, gloves, goggles, masks, and lab coats) for technical staff. However, complex equipment, such as eyewash stations and BSCs, is not available in many laboratories.
Recommendations
Based on this study, there is a strict need to overcome deficiencies identified in the biosafety performance of the clinical laboratories in Karachi, Pakistan. We suggest the following remedies to improve biosafety performance:
Frequent training with respect to adherence to biosafety practices is very important. Therefore, there should be a defined training protocol at the institutional level. There should be a trained biosafety officer who is familiar with modern biosafety training aspects, especially a behavioral expert who not only trains staff according to a safety manual but also is able to change staff behavior toward biosafety, as well as be able to look after all biosafety-related issues of laboratories in collaboration with management. Better performance of biosafety needs to be observed in hospital laboratories that are CAP, ISO, and JCIA certified, indicating accreditation and certification are important factors for good performance. There should be a regulatory body at the national level that will be responsible for all biosafety issues, define rules for biosafety, and have authority to punish those not complying with these laws. There should be management commitment toward biosafety in order to have a working environment that is hazard free. It is important for management to have a serious attitude toward biosafety, records should be maintained for an incident or accident, proper safety manuals should be maintained, and policies should be in place and implemented. There should be a system to check that a sufficient number of staff are working in the laboratory and enough workload has been provided for employees, as heavy workload is one of the important factors for ignoring biosafety rules among laboratory workers. Pocket-size biosafety handbooks or cards should be given to staff so that in case of a spill or accident, staff can follow these rules easily. Spill kits and a written spill management plan should be in place at the national and institutional levels for all laboratories to handle biological spills.
Footnotes
Acknowledgments
We thank the BSAP (Biological Safety Association of Pakistan) and all medical laboratory personnel in the 5 selected hospitals for their kind support and cooperation in conducting this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
