Abstract
The objective of this study was to evaluate various procedures for decontamination of vehicles and equipment during outbreaks of notifiable animal diseases in cold weather. The evaluation was done in 4 field trials held in outdoor operational settings in Canada, at ambient temperatures from –2°C to 11°C. Procedures included various combinations of dry cleaning, wet cleaning, disinfection, and final rinsing. Geobacillus stearothermophilus spores and infectious bursal disease virus (IBDV) were used as surrogates for bacterial and viral pathogens, particularly Bacillus anthracis spores and foot-and-mouth disease virus. Spores and viruses were suspended in a light organic soil preparation, inoculated onto stainless-steel disks, and covered with a heavy soil preparation. Inoculated disks were attached to various surfaces of farm vehicles and equipment. In all field trials, spore and IBDV reduction was greater (P < .05) on disks where the soil was completely removed, as compared with only partially removed. Greater (P < .05) spore and IBDV reduction was seen when disinfection and final rinse steps were included than when not included after dry and wet cleaning in 3 of 3 and 1 of 3 field trials, respectively. A wet-cleaning step before application of a disinfectant increased (P < .05) spore and IBDV reduction versus no wet cleaning. The results provide evidence that vehicle and equipment decontamination in cold weather could be significantly improved by thorough removal of organic matter to enhance disinfection and elimination of disease agents.
Keywords
Large-scale outbreaks of most notifiable animal diseases, such as highly pathogenic avian influenza, foot-and-mouth disease, and anthrax, 1 can cause high losses of farm animals, challenge public and animal health, and lead to tremendous economic damage. It is critical to apply mitigation measures to control or eradicate such diseases. These measures or activities include establishing disease control zones, controlling animal movement, culling infected animals, and disposing of contaminated carcasses and materials, followed by cleaning and disinfection of infected premises. Vehicles, tools, and equipment used for these activities must be decontaminated before moving off the affected site. Furthermore, vehicle and equipment decontamination is one of the permit requirements for transportation between control zones, as vehicles and equipment traffic have been identified as a risk factor in the spread of diseases.2,3 In addition, agricultural biosecurity measures, such as entry requirement for vehicles and equipment, are generally augmented by producers on susceptible species premises and associated facilities to prevent the entry of disease-causing agents, which also calls for increased decontamination of vehicles and equipment.
Equipment decontamination in agricultural settings is a multistep technical process, including all stages of cleaning and disinfection to reduce biological contamination to a harmless level. 4 In general, the process includes dry cleaning, wet cleaning, disinfection, and final rinsing.5,6 Dry cleaning is to remove soil, dirt, and other organic matter from surfaces by scraping, sweeping, and/or vacuuming. Wet cleaning is to further remove debris and dirt until surfaces are visibly clean, and it involves soaking, pressure washing, and/or scrubbing with soapy water and/or detergents. 6 Disinfection is treatment with disinfectants that are delivered by spraying or fogging or as foams.7,8 The effectiveness of disinfection is influenced by temperature, concentration of disinfectant solutions, contact time of disinfectant with target agents, and the presence of organic matter.9 -11 Final rinsing is to remove disinfectants to prevent corrosion of vehicles and equipment. Each decontamination step contributes to the overall removal and/or killing of disease agents. Although general rules for decontamination following an animal disease outbreak are well known, 12 information on the impact of each step on the effectiveness of decontamination process is limited, especially under cold weather conditions.
The objective of this study was to identify key steps to improve decontamination processes for vehicles and equipment through evaluating various procedures used in the field at temperatures close to the freezing point. These procedures included various combinations of dry cleaning, wet cleaning, disinfection, and final rinsing. The evaluation was conducted in outdoor operational settings in the spring and fall in Prince Edward Island (PEI), Ontario, and Manitoba, which represent various field conditions in Canada. These trials were conducted in partnership with Canadian military personnel, who require information on biological decontamination for cold weather operations. The partnership, in part, drove the selection of disease agents and disinfectants tested. Prior to the evaluation, laboratory studies were performed following the Health Canada guidance document for disinfectant drugs 13 to assess the combinations of concentrations and contact times of 3 disinfectants—Virkon, bleach, and surface decontamination foam (SDF)—for inactivation of animal disease viruses and bacterial spores.9,10 Virkon and bleach have been widely used to kill infectious agents in various environments, and SDF was developed for inactivating chemical and biological agents. 14 Building on the results of the initial laboratory studies, infectious bursal disease virus (IBDV) and Geobacillus stearothermophilus spores were used as surrogates for foot-and-mouth disease virus and anthrax spores, respectively, for the evaluation of the field decontamination procedures.
Materials and Methods
Zoning and Staging of Cleaning and Disinfectant Activities
The Canadian Food Inspection Agency’s disease control operations on infected premises involve the use of a 3-zone containment system (Figure 1) to prevent the inadvertent spread of disease agents. The hot zone is the area that is considered to be contaminated or potentially contaminated with disease agents. The warm zone is a transitional area that is set up as the pathway from the hot zone to the cold zone for contamination reduction. The cold zone is a clean and controlled area of support functions for operations on the quarantined premises. Cleaning occurs in the hot zone, disinfection in the warm zone, and rinsing of disinfectants in the cold zone.
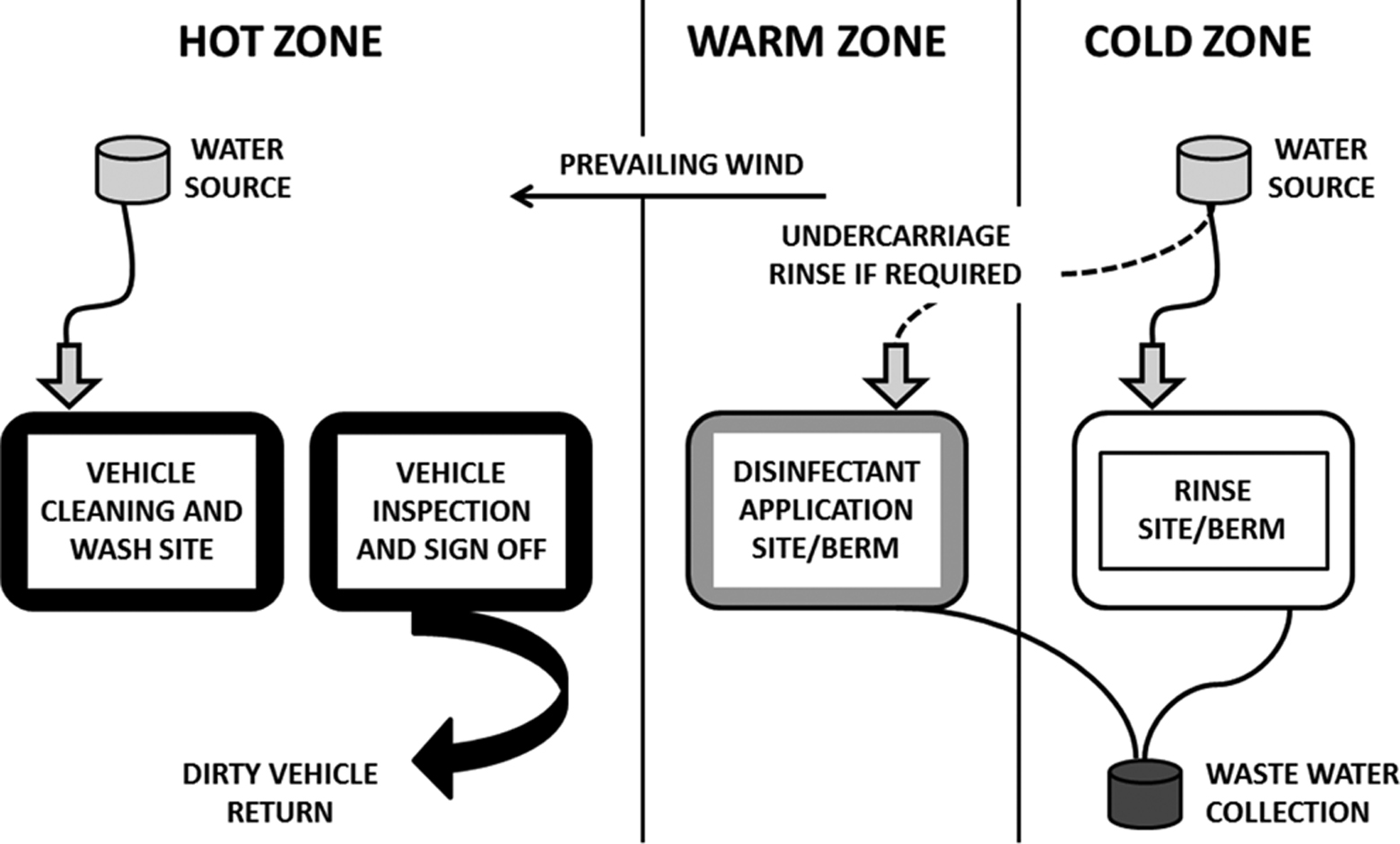
Diagram showing the 3-zone containment system.
To collect disinfectant runoff from the warm and cold zones, berms were constructed according to materials on hand at the site. In the Ottawa trials, a commercially available berm (Insta-Berm; West Coast Spill Supplies, Saanichton, Canada) was used (Figure 2). In the Manitoba trial, a shallow divot was made and lined with tarpaulin, which was secured with steel rods (5-9 m in length). In the PEI trial, the berm was built on hard-standing fence posts, with the tarp edges rolled over the posts to secure the edges. Runoff was collected from the berms via a sump pump to waste tanks. Disposal of the runoff or effluent was driven by local regulations and risk assessment, including but not limited to water table conditions, disease susceptibility of nearby species, and biodegradability of disinfectants.

Photograph showing a decontamination crew member in full personnel protective equipment spaying a tractor trailer in a berm. Arrows indicate the 3 sites from the exterior of a tractor trailer: 1 = open area on the side of the trailer, 2 = wheel well of the tractor, and 3 = center area of the undercarriage.
Personnel Protective Equipment
Personnel carrying out decontamination tasks were required to wear full personnel protective equipment (PPE; Figure 2) within the decontamination zones. They donned and doffed PPE in a dedicated area separated from the decontamination zones. Full PPE included a privacy layer (light cotton or thermal underclothes), a primary layer (cotton coveralls with elastic waist and zippered closure), a secondary layer (ChemMAX 1 disposable coveralls or equivalent, with respirator hood, elastic back/wrists/ankles, and sealed seams; Lakeland, Ronkonkoma, New York), glove liners, nitrile gloves with extended cuffs, 7600 series full facepiece air-purifying respirator with multicontaminant cartridge and P100 filter (Honeywell Safety Products, Smithfield, Rhode Island), and chemical-resistant boots.
Location, Schedule, and Temperature of Field Trials
The first 2 trials were held in Ottawa, Ontario, in November 2010 and March 2011; the third trial in Brandon, Manitoba, October 2011; and the fourth trial in Harrington, PEI, November 2011. The ambient temperatures were 9ºC∼11ºC and 3ºC∼5ºC in the first and second trials held in Ottawa, respectively, and –2ºC∼0ºC and –1ºC∼2ºC in the trials held in Manitoba and PEI, respectively.
Vehicles and Equipment
Typical farm vehicles and equipment were selected from available items at the research stations at the time of the trials and included pickup trucks, tractors, trailers, and manure spreaders. These vehicles and equipment had been through at least 1 week of rural use with no cleaning prior to the trials.
Decontamination Procedures
Table 1 shows the procedures for the various combinations of the following decontamination steps that were evaluated in the 4 field trials.
Decontamination Procedures Evaluated in the Various Field Trials.

Abbreviations: C, cleaning; D, disinfection; PEI, Prince Edward Island.
Dry Cleaning
An initial cleaning step was used to remove organic material from the vehicles and equipment prior to washing. All surfaces of the vehicles and equipment were lightly sprayed with water to reduce dust and then brushed and scraped from top to bottom to remove as much mud, manure, plant material, and dirt as possible and to minimize recontamination by the falling dirt. The cab of the vehicles was swept and the floor mats and other removable items removed.
Wet Cleaning
A pressure washer was used to apply hot water (50ºC ± 2ºC, only in the first Ottawa trial) or cold water (10ºC ± 2ºC) with household detergent to the equipment exterior, starting at the top, working down the sides, and finishing with the wheel arches, mudguards, wheels, and undercarriage. Brushes and scrapers were used to remove dirt where required. Stubborn areas were rinsed and washed several times until visibly clean. The impervious surfaces of cab interiors (including pedals and levers) were washed with detergent and hot water with clean cotton hand towels.
Disinfection
Three disinfectants were used in the studies for disinfection of the exterior of vehicles and equipment. SDF (Allen-Vanguard Corporation, Ottawa, Canada) was mixed per the manufacturer’s instructions: 8.4 kg of GP 2100 decontaminant and 132.5 L of water were mixed for 4 to 5 minutes in the UCS Foam Dolly System; then, 4.75 L of GCE 2000 was added and the solution mixed for 10 seconds. Common household bleach (The Clorox Company, Oakville, California) containing about 5.25% sodium hypochlorite (available chlorine, 52 500 ppm) was diluted 10-fold with tap water to obtain a chlorine concentration of 5250 ppm. The 2% Virkon solution (Antec, Suffolk, United Kingdom) was prepared by dissolving 20 g of Virkon tablets per liter of water. Disinfection solutions were prepared at room temperature (∼20ºC) and applied within 1 hour thereafter.
The cleaned vehicles and equipment were then moved into the warm zone for disinfectant application. When moved into the warm zone, the tire treads were sprayed with water to prevent transfer of dirt and to keep wheels clean for disinfection. To maximize contact, disinfectant was applied from the bottom up, starting with the undercarriage, wheels, and wheel wells and moving upward over the body of the vehicle and then the top. Once the entire piece was covered with disinfectant, the measurement of the contact time was started. One hour of contact time was used for SDF and bleach and 20 minutes for Virkon (a virucide). Disinfectant was reapplied to maintain a wet surface during the contact time.
Lysol wipes were used for disinfection of the interior cab of vehicles according to the manufacturer’s instruction.
Final Rinse
After the predetermined contact time for disinfection, the vehicles and equipment were moved into the cold zone. The pressure washer and cold water were used to rinse off the disinfectants.
Evaluation with Spores and Viruses
To evaluate the decontamination procedures, stainless-steel disks with spore or virus inoculum were mounted onto the vehicles and equipment prior to the initial cleaning step and were recovered after certain procedures (Table 1) for determination of number of spores or viruses on the disks. The evaluations were based on the reduction of spores or viruses on the disks.
Spore and Virus Inocula
Spore and virus inocula were prepared as described previously,9,10 with minor modification. For the spores, 1.0 mL of a suspension of G. stearothermophilus American Type Culture Collection strain 7953 spores (Steris, Mississauga, Canada) containing approximately 108 spores was mixed with 9.0 mL of a light soil preparation, which contained 0.35% weight/volume (w/v) tryptone, 0.25% w/v bovine serum albumin, and 0.04% w/v mucin in 0.01M phosphate buffered saline (PBS), pH 7.2, and the mixture heated at 100°C for 1 hour with constant stirring. For the virus, 10 mL of the light soil preparation was added to 1 vial of lyophilized IBDV CLONEVAC D78 (Intervet, Kirkland, Canada), containing approximately 108 50% tissue culture infectious dose (TCID50) of IBDV, and the suspension was mixed.
Inoculation of Carrier Disks
Stainless-steel disks (1 cm in diameter, 0.75 mm thick; Muzeen and Blythe, Winnipeg, Canada) for use at selected test sites on the exterior of the vehicles were prepared as sets. For each set, 4 foam double-sided removable mounting squares (2.54 × 2.54 cm, catalogue no. 108, Scotch; 3M, USA) attached to a backing sheet were placed in a sterile glass petri dish lined with filter paper. The protective film was removed from the upper surface of each of the 4 squares, and 4 degreased, prewashed, autoclaved steel disks were attached to each square (Figure 3). One disk in each square was labeled with a unique identifier number. Ten microliters of the spore inoculum, sonicated for 5 minutes immediately before use, was pipetted onto the surface of the 3 unnumbered disks in 1 square and allowed to dry. Ten microliters of the IBDV inoculum was applied to the surface of the 3 unnumbered disks in another square and allowed to dry. No inoculum was placed on the 3 unnumbered disks in each of the other 2 squares. Subsequently, 50 μL of a heavy soil preparation (light soil preparation with 5% w/v garden soil) was pipetted over the dried inoculum on all disks with spore or virus inoculum and onto the surface of all unnumbered disks with no inoculum and allowed to dry. A glass lid was placed on the top of the petri dish and the dish sealed with paraffin film. Sealed petri dishes were placed in jars and stored at –80°C until used. Synthetic rubber discs (1.1 cm in diameter and 1.5 mm thick; product 27351, Sigma, Oakville, Canada), with or without the spore or virus inoculum but without heavy soil overlay were prepared as described for evaluation of the disinfection in the cabs of the vehicles with Lysol wipes. Extra steel and rubber disks with the spore or virus inoculum and with or without the heavy soil overlay were prepared for use as controls.

Photograph showing a set of triplicate stainless-steel disks with or without inoculum of Geobacillus stearothermophilus (GST) spores or infectious bursal disease virus (IBDV) that were overlaid with soil. Four disks with unique identifier numbers, but without inoculum or soil, were also included.
Attachment of disks to selected vehicle sites and removal after treatment
On the day of the trial, the petri dishes containing the mounting squares and disks were removed from the freezer, allowed to come to ambient temperature, and taken to the trial site. Four squares (1 set of disks) were removed from the backing sheet and attached to a selected site on each vehicle. Three sites from the exterior of each vehicle were selected according to their accessibility. The open area on the side or bed of the vehicle or equipment represented easy access; the wheel rim or well, medium access; and the center area of the undercarriage, difficult access (Figure 2). Rubber disks were attached to the dash, door, and floor of the vehicle cab.
Control disks were placed away from the vehicles and exposed to the same environmental conditions as the vehicles but received no washing or disinfectant treatment. In addition, in the PEI trial, control steel disks with no inoculum were placed on the vehicles at sites remote from the test disk sites, and these disks received either the wash treatment or the wash-and-disinfectant treatment.
After treatment, the squares with the disks attached were removed from the vehicles, placed into sterile plastic petri dishes, and taken to the laboratory. At the laboratory, the 3 unnumbered disks from the squares with the spore inoculum and the 3 unnumbered disks without inoculum from 1 square in each set were each placed into a sterile 15-mL numbered tube. The 3 unnumbered disks from the squares with the virus inoculum and 3 unnumbered disks without inoculum from 1 square in each set were placed into numbered tubes containing 3 mL of universal transport medium (Alere Canada, Stittsville, Canada). All disks were stored at –80°C until analyzed.
Analysis of Spore Disks
The procedure described previously 9 was essentially used. The tubes containing the disks were removed from the freezer and brought to room temperature. Sterile PBS (1 mL) containing 0.1% Tween 80 was added to each tube and the tubes vortexed. The tubes and contents were then heated at 100°C for 45 to 60 minutes to kill the vegetative cells of G. stearothermophilus and environmental bacteria. A 10-fold serial dilution of the suspension in each tube was made in sterile water, and each dilution was passed through a 0.2-μm membrane filter with a magnetic filter holder. The filter unit was subsequently rinsed 3 times with 10 mL of PBS. Each membrane filter was placed on the surface of a tryptic soy agar plate and the plates incubated at 56°C, with relative humidity approximately 60%. Colonies were counted after 2 and 4 days of incubation.
Analysis of IBDV Disks
The procedure used previously 10 was followed with minor modification. Briefly, the tubes containing the disks and transport medium were removed from the freezer, thawed, and vortexed. Ten-fold serial dilutions of the suspensions were made in growth medium (minimal essential medium with 10% inactivated fetal bovine serum, 1% antibiotic-antimycotic, 1% sodium pyruvate; all components from Invitrogen, Burlington, Canada). Fifty microliters of each dilution was inoculated into 6 wells of a plate containing an overnight culture of Vero cells in growth medium. Plates were incubated at 37°C, 5% CO2, for 5 to 6 days. IBDV infection was detected with an immunofluorescence assay. In brief, cells were washed, fixed with 30% acetone, washed with PBS, and incubated with a 1:250 dilution of a chicken IBDV antiserum (Charles River Laboratories International, Wilmington, Massachusetts). Cells were washed with PBS + 1% Tween 20, incubated with a 1:150 dilution of DyLight 488 conjugated goat anti-chicken immunoglobulin G antibody (Jackson ImmunoResearch Laboratories, West Grove, Pennsylvania), washed again with PBS + 1% Tween 20, and visualized with a fluorescent microscope. Samples were considered positive for IBDV when immunofluorescence positive cells were detected. Virus titer expressed as TCID50 was determined by the Reed and Muench 15 method.
Data Analysis
The difference in recovery of live spores or IBDV from control and test disks was recorded as spore or virus reduction (log10), respectively. The data presented are the means of spore or virus reduction from 9 to 79 observations under specified decontamination treatments, as shown in the tables. Student’s t test was used to determine statistical significance (P < .05) in difference in log10 spores or virus recovered from disks.
Results
Reduction of G. stearothermophilus Spores on Inoculated Steel Disks
The effect of various decontamination procedures on the reduction of G. stearothermophilus spores is shown in Table 2. In all 4 trials, spore reduction was significantly greater (P < .05) when the soil overlay was completely removed, as compared with only partly removed, from the disks after decontamination (4.1-5.6 vs 0.7-1.3 mean log10, respectively). Spore reduction was also significantly greater (P < .05) when a procedure with a disinfectant and subsequent rinse (4-step procedure), as opposed to no disinfectant and no additional rinse (2-step procedure), was used (3.3 vs 2.1, 3.8 vs 2.9, and 4.7 vs 3.4 mean log10, respectively), and the reduction was highest with bleach or SDF and lowest with Virkon. The wet-cleaning step significantly increased (P < .05) spore reduction as compared with skipping this step (4- vs 3-step procedure) before application of a disinfectant (4.3 vs 2.2 mean log10, respectively). The highest reduction of spores was seen on disks attached to the vehicle side or bed and wheel rim or well and lowest on disks attached to the undercarriage. No difference in spore reduction was observed with the use of hot or cold water for wet cleaning vehicles.
Reduction of Geobacillus stearothermophilus Spores on Inoculated Steel Disks After Decontamination.
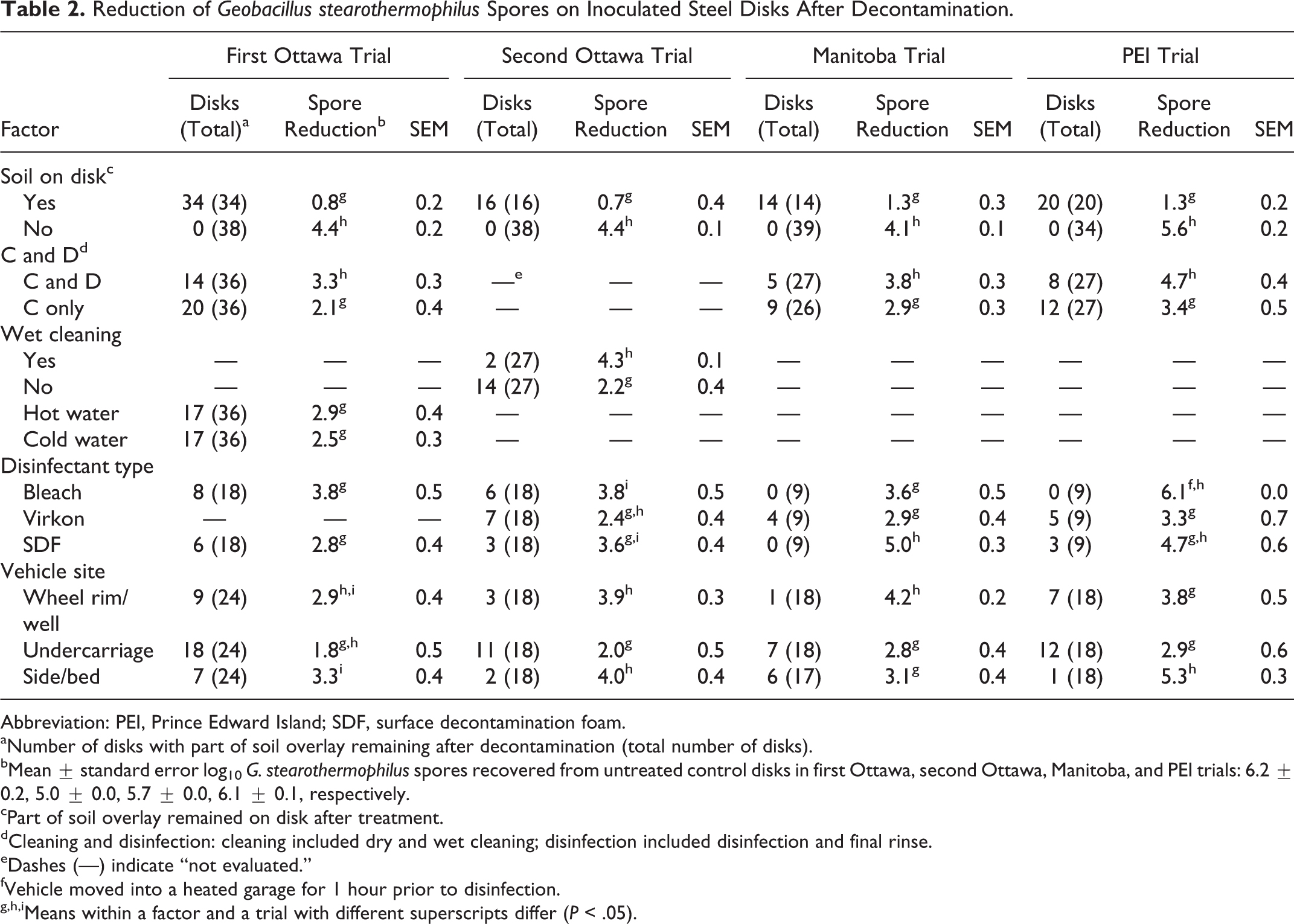
Abbreviation: PEI, Prince Edward Island; SDF, surface decontamination foam.
Number of disks with part of soil overlay remaining after decontamination (total number of disks).
Mean ± standard error log10 G. stearothermophilus spores recovered from untreated control disks in first Ottawa, second Ottawa, Manitoba, and PEI trials: 6.2 ± 0.2, 5.0 ± 0.0, 5.7 ± 0.0, 6.1 ± 0.1, respectively.
Part of soil overlay remained on disk after treatment.
Cleaning and disinfection: cleaning included dry and wet cleaning; disinfection included disinfection and final rinse.
Dashes (—) indicate “not evaluated.”
Vehicle moved into a heated garage for 1 hour prior to disinfection.
Means within a factor and a trial with different superscripts differ (P < .05).
Recovery of G. stearothermophilus Spores from Uninoculated Steel Disks
The transfer and survival after decontamination of G. stearothermophilus spores from inoculated to uninoculated disks, located directly adjacent to the inoculated disks, were very low (data not shown). The highest mean recovery of spores after decontamination from any of the uninoculated disks in the 4 trials was 43.4 (1.6 log10) colony-forming units, as compared with a mean inoculum of 631 000 (5.8 log10) colony-forming units on the inoculated disks prior to decontamination.
Reduction of IBDV on Inoculated Steel Disks
In 3 of the 4 trials, reduction of IBDV was significantly greater (P < .05) on disks where the soil overlay was completely removed than on disks where part of the soil overlay remained after decontamination (3.5 vs 1.4, 3.3 vs 1.4, and 2.5 vs 1.9 mean log10 TCID50
Reduction of IBDV on Inoculated Steel Disks After Decontamination.
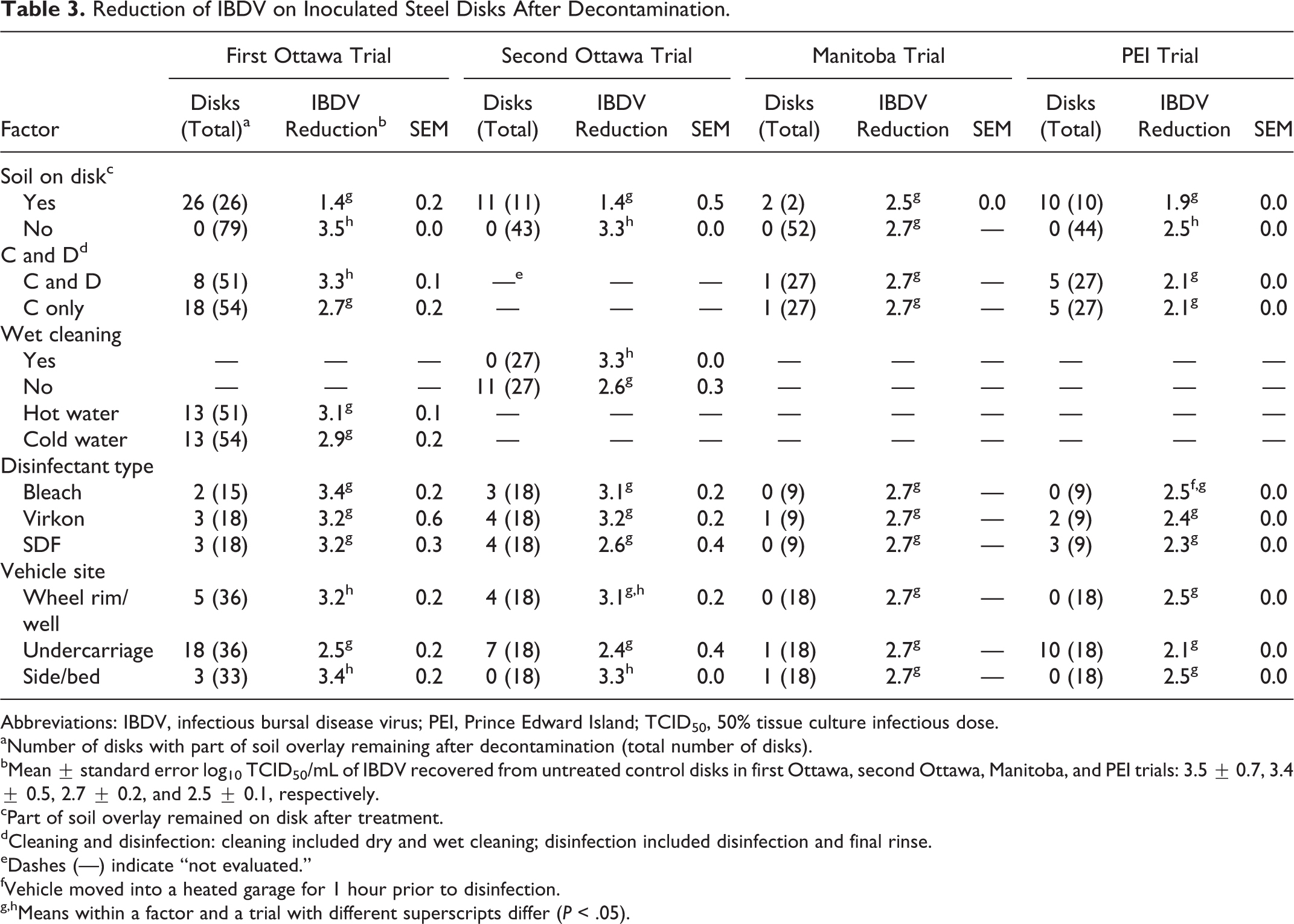
Abbreviations: IBDV, infectious bursal disease virus; PEI, Prince Edward Island; TCID50, 50% tissue culture infectious dose.
Number of disks with part of soil overlay remaining after decontamination (total number of disks).
Mean ± standard error log10 TCID50/mL of IBDV recovered from untreated control disks in first Ottawa, second Ottawa, Manitoba, and PEI trials: 3.5 ± 0.7, 3.4 ± 0.5, 2.7 ± 0.2, and 2.5 ± 0.1, respectively.
Part of soil overlay remained on disk after treatment.
Cleaning and disinfection: cleaning included dry and wet cleaning; disinfection included disinfection and final rinse.
Dashes (—) indicate “not evaluated.”
Vehicle moved into a heated garage for 1 hour prior to disinfection.
Means within a factor and a trial with different superscripts differ (P < .05).
Recovery of IBDV from Uninoculated Steel Disks
IBDV was not recovered from any of the uninoculated disks following decontamination in any of the trials (results not shown).
Removal of Soil Overlay from Steel Disks
Part of the soil overlay remained after decontamination on a higher proportion (P < .05) of disks inoculated with spores than on disks inoculated with IBDV (36.1% vs 18.4% respectively) and on a higher proportion (P < .05) of disks inoculated with either spores or IBDV than on uninoculated disks (>18% vs <5%, respectively; Table 4).
Number of Steel Disks with Soil Remaining After Decontamination vs Total Number of Steel Disks.

Abbreviations: G. stearothermophilus, Geobacillus stearothermophilus; IBDV, infectious bursal disease virus.
Number of disks with part of soil overlay remaining after decontamination (percentage of total).
Total number of disks.
Values within a row with a different superscripts differ (P < .05).
Values within a column with different superscripts differ (P < .05).
Recovery of G. stearothermophilus Spores and IBDV from Rubber Disks
After wiping with Lysol, the mean recovery of spores from disks inoculated with an average of 5.6 log10 spores and from uninoculated disks attached to the inside of a truck cab was 2.7 and 2.4 log10, respectively, for the 2 trials (Table 5). IBDV was not recovered from disks inoculated with an average of 4.0 log10 TCID50/mL IBDV or from uninoculated disks after Lysol treatment in either trial.
Recovery of G. stearothermophilus Spores and IBDV from Inoculated and Uninoculated Rubber Disks on the Inside of a Truck Cab After Wiping with Lysol.
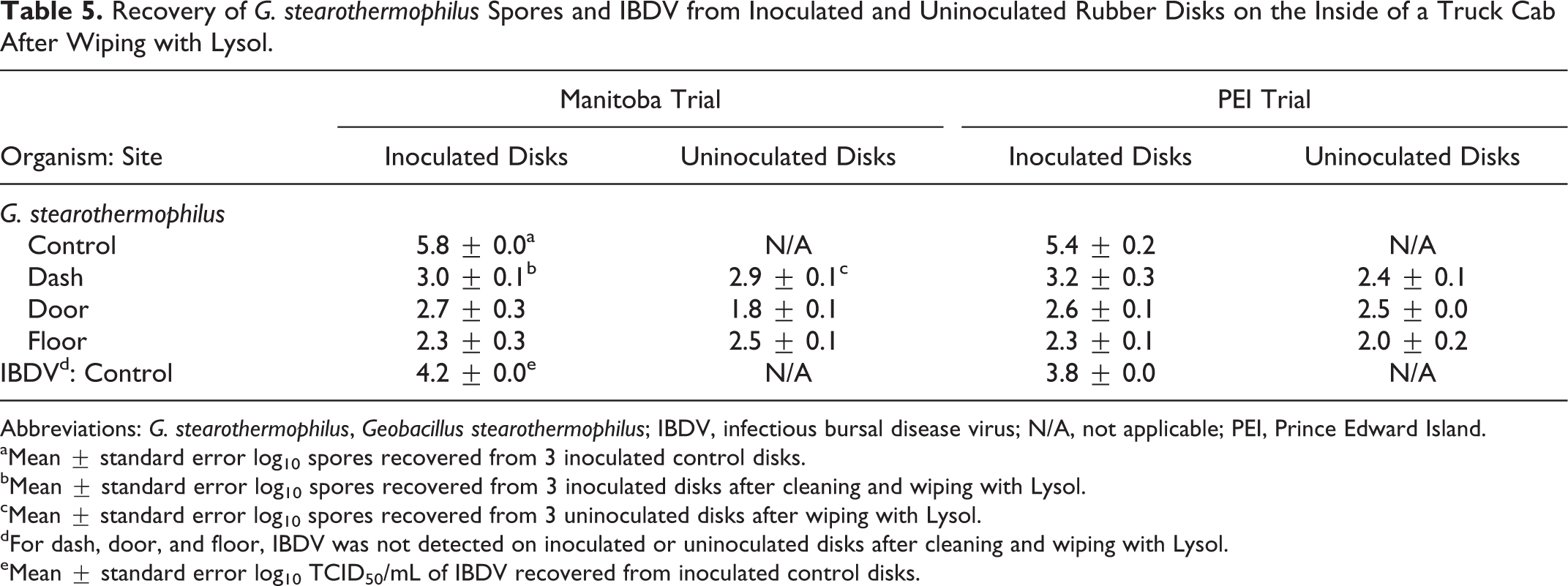
Abbreviations: G. stearothermophilus, Geobacillus stearothermophilus; IBDV, infectious bursal disease virus; N/A, not applicable; PEI, Prince Edward Island.
Mean ± standard error log10 spores recovered from 3 inoculated control disks.
Mean ± standard error log10 spores recovered from 3 inoculated disks after cleaning and wiping with Lysol.
Mean ± standard error log10 spores recovered from 3 uninoculated disks after wiping with Lysol.
For dash, door, and floor, IBDV was not detected on inoculated or uninoculated disks after cleaning and wiping with Lysol.
Mean ± standard error log10 TCID50/mL of IBDV recovered from inoculated control disks.
Discussion
G. stearothermophilus spores and IBDV were used as surrogates for anthrax spores and foot-and-mouth disease virus, respectively, in the present study, as have been used in earlier studies.9,10,16,17 Herein, a mixture of spores or virus and a light soil preparation was applied to steel disks and then overlaid with a heavy soil preparation to simulate the organic load that may be encountered in the field on the exterior surfaces of vehicles, such as tractors and trucks, and on other equipment. Reduction of G. stearothermophilus spores was greatest in all 4 trials on disks where no soil remained after cleaning and disinfection. Similarly, in 3 of the 4 trials, IBDV reduction was greatest on disks where soil was completely removed during cleaning and disinfection. As indicated in other studies, organic matter should be removed prior to disinfection,5,8,18 since it may readily interact with bleach, Virkon, and other disinfectants and thus lower the concentration of active ingredients and form a physical barrier that could reduce or delay contact between the disinfectant and the microorganisms.9,18 -20 It is worth noting that the percentage of disks with soil remaining after cleaning and disinfection was greater on disks that were inoculated with spores or virus in the light soil preparations and overlaid with the heavy soil preparations (36.1% or 18.4%) than on those overlaid with the heavy soil preparations only (4.6% or 2.8%). Likely, the light soil preparations that simulated animal secretions would facilitate the adherence of organic matter, such as soil, manure, or bedding material, to the surface of vehicles and equipment and challenge the cleaning and disinfection process.
Furthermore, when the 4-step procedure (dry and wet cleaning, disinfection, and final rinse) was applied, reduction of spores was higher and the number of disks with soil remaining was lower in the trials, as compared with the 3-step (dry cleaning, disinfection, and final rinse) or 2-step (dry and wet cleaning) procedure. Generally, greater spore reduction was associated with bleach and SDF than with Virkon (no sporicidal claim in product description), but effective disinfection significantly relied on the soil removal. Similarly, the 4-step procedure produced greater IBDV reduction than the 3- or 2-step procedure in the Ottawa trials. However, in the Manitoba and PEI trials, there was no difference in reduction of virus or in the number of disks with soil remaining, with either the 2- or 4-step procedure. Obviously, learning from the Ottawa trials, the cleaning procedure received more attention in the Manitoba and PEI trials. However, environmental factors, such as humidity and ultraviolet radiation in sunlight, might contribute to IBDV inactivation, and the numbers of IBDV recovered from the environment control disks in the Manitoba and PEI trials (2.7 and 2.5 log10 TCID50) were lower than those in the Ottawa trials (3.5 and 3.4 log10 TCID50). In all 4 trials, no difference among the disinfectants in reduction of the virus was observed. Overall, the highest number of inoculated disks with soil remaining was from the vehicle undercarriage, and this may be due to the relative difficulty in accessing this area for cleaning and disinfection.
Inoculation of rubber disks with spores or virus in the light soil preparations, with no overlaying of heavy soil preparation, was done to simulate the relatively clean conditions in the interior of vehicles. Wiping the rubber disks at different sites in truck cabs with Lysol (benzalkonium chloride) wipes reduced the spores from approximately 5.6 to 2.4 log10 and reduced 4.0 log10 IBDV to undetectable levels. These findings are in agreement with previous studies showing that benzalkonium chloride can inactivate various viruses, 21 but its effect on killing bacterial spores is limited. 22 With the wiping, approximately 2.4 log10 spores were transferred from inoculated to uninoculated disks, but no transfer of IBDV to uninoculated disks was detected. This indicated the risk of spreading pathogens by applying inappropriate disinfectants.
Temperature, in addition to organic matter, influences disinfectant efficacy, with the effectiveness generally decreasing as temperature decreases,5,6,23 and a longer contact time is required to achieve equivalent microbial reduction.9,10 During the 4 trials, decontamination of vehicles was performed outdoors at temperatures from –2°C to 11°C, and the disinfection contact time for the exterior surface of vehicles was limited to ≤1 hour. The only exception was 1 vehicle in the PEI trial that was held in a heated (∼20ºC) garage for 1 hour prior to the application of the disinfectant (bleach), and the highest spore reduction (6.1 log10, a complete decontamination) was observed with this vehicle. Thus, the combination of low ambient temperature, the presence of organic matter, the limited contact time, and the resilient nature of the target microorganisms may explain why soil removal was more significant than disinfection for spore and virus reduction in the trials. In the first Ottawa trial, where the effect of rinse water temperature was evaluated, the reduction of spores or viruses with hot water (50ºC) was not significantly greater than that with cold water (10ºC). This result is consistent with a previous finding that cleaning with warm or hot water (40ºC or 60ºC) is slightly more effective than with cold water (10ºC) for bacterial reduction. 6 However, less time was used to achieve visible cleanliness with hot water than cold water. Alternatively, thermo-assisted drying (71ºC, 30 minutes) following wet cleaning was successfully applied to eliminate porcine reproductive and respiratory syndrome virus from contaminated transport vehicles. 23 Although such an approach may not be suitable for disinfection of spores or hardy nonenveloped viruses, elevating the temperature of contaminated vehicles and equipment would certainly benefit the overall decontamination process.
In conclusion, the findings herein suggest that decontamination of vehicles and equipment during outbreaks of notifiable animal diseases in cold weather could be significantly improved by combining (1) physical removal of the disease agents associated with organic matter and (2) enhancing disinfection at elevated vehicle temperatures.
Footnotes
Acknowledgments
We gratefully acknowledge the assistance of Stephen Obsniuk, Robert Poirier, and Robert Lukshis of Allen-Vanguard; WO Eric Winkles and Capt Jeffrey George of the Canadian Forces Fire and CBRN Academy; Nathan Birnbaum (US Department of Agriculture) and Jeff Kempter (Environmental Protection Agency); staff and management of Agriculture and Agri-Food Canada’s Brandon Research and Development Centre and Harrington Research Farm; Canadian Food Inspection Agency’s Cleaning and Disinfection and Biocontainment Response Forces, and the field trial organizers Tim McQuaid, Suzanne Nadeau, Megan Bergman, Shari Reitmier, Wayne Clayton, and Kevin Miller.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was part of a project entitled “Verification of Decontamination Processes in the AgriFood Context,” funded by the Canadian Chemical, Biological, Radiological, and Nuclear Research and Technology Initiative (CRTI 08-0122TD).
