Abstract
Theiler’s murine encephalomyelitis virus (TMEV) is a naturally occurring enteric infection, easily passed from mouse to mouse in communal housing. However, TMEV is often inoculated intracerebrally (IC) to produce a mouse model of multiple sclerosis (MS). It has long been accepted that maintaining colonies of IC-infected mice within laboratory animal facilities poses a risk of spreading infection from mouse to mouse via the fecal-oral route as well as contaminated equipment or personnel. Interestingly, the extent of virus shedding from IC-inoculated mice has not been investigated, although several publications have remarked on the lack of virus in the peripheral body of this MS mouse model. Viral shedding, thus infectivity, would require that TMEV escape the central nervous system (CNS) and be found in bodily secretions. We hypothesized that if the virus can escape the CNS, it would be found circulating within blood or other secretions postinjection (PI), after the blood-brain barrier has been experimentally breached. The data presented show no TMEV RNA was found in the serum, saliva, or feces during the acute and chronic infection stages, although all subjects were positive for TMEV RNA in the CNS. These results, in conjunction with published anecdotal evidence, suggest that mice IC-inoculated with TMEV are not contagious, and thus a relaxation of containment methods is warranted. This report is an example of a collaborative effort between biosafety and research professionals to identify and collect scientifically relevant data to inform a risk assessment.
In 1934, Max Theiler described an encephalomyelitis virus in his population of mice that, when introduced via the intracerebral (IC) route with a central nervous system (CNS) homogenate, resulted in an incubation period followed by partial or full paralysis. 1 Since that time, Theiler’s murine encephalitis virus (TMEV), when IC-inoculated, has become a model system for human neurodegenerative diseases such as multiple sclerosis (MS). Importantly, the route of infection is critical to disease progression; for instance, when introduced via the intranasal (IN) route, the infection only resulted in paralysis in a small percentage of mice. 1 Originally, infection via the intraperitoneal (IP) route was not successful; however, years later, Theiler reported successful IP infection with an injection of a highly virulent strain at a concentration 10 million times stronger than that needed for IC inoculation. 2 Theiler described that the natural occurrence rate of paralysis, independent of any experimentation, occurred in approximately 1 out of 2000 Swiss strain mice. 3 Theiler also noted that several mice in his laboratory spontaneously contracted the encephalomyelitis infection during the course of experimentation with other unrelated infectious agents at a rate greater than that of the natural occurrence rate. 3 These observations suggest that the virus rarely travels into the CNS when acquired naturally. These data also suggest that natural occurrences of the infection are more likely to result in encephalomyelitis when the mouse is concurrently infected with parallel pathogens.
Indeed, it has since been found that TMEV is a naturally occurring enteric virus that normally does not cause physically observable symptoms. The disease is spread through the oral-fecal route with virus isolated from feces for up to 53 days postinfection. 4 Corroborative evidence includes studies in 1940, when Olitsky demonstrated that intestine infected with virus, having been contracted naturally through shared housing, could be ground up and IC-injected into a second mouse to produce encephalomyelitis while CNS homogenate did not produce encephalomyelitis. 5 Olitsky’s data suggested that TMEV is naturally an enteric virus that resides in the intestine and does not cross the blood-brain barrier (BBB) on its own. More recent data have corroborated this interpretation; for instance, IP-injected wild-type C3FeLe.B6-a/a mice showed no signs of paralysis in contrast to their SHP1-deficient mutant littermates (a phenotype characterized by blood-borne macrophage invasion of the CNS). 6 These data indicate that a healthy, intact BBB does not allow the virus to cross into the CNS, yet a compromised BBB does.
It is important to note that enteric TMEV is one of a small number of viruses that is considered common to laboratory mice. Pritchet-Corning et al 7 identified TMEV using antibody serology in 0.26% of the 435 772 mice that were tested. Although common, the virus can have a number of adverse effects if it were to spread in a laboratory animal facility. For instance, weaning pups are generally very susceptible to the virus and often die due to encephalitis. 8 In addition, the viral infection typically causes major changes in immune activity,9 -11 which could directly skew results in immunological, microbiological, and cancer-related studies. Last, immunocompromised mice have been found to be highly susceptible to TMEV, which causes paralysis, death, and decreased growth. 8 It is therefore imperative that laboratory animal facilities prevent TMEV infection and spread once identified. For the above reasons, laboratory animal facilities are likely using stringent biosecurity and biosafety precautions when the IC TMEV model is used.
Following IC inoculation, Theiler was not able to detect infectious blood on day 1, 3, 6, or 8 postinjection (PI) or in the spleen, kidney, adrenal, or liver at acute and chronic stages, although he did detect virus in the sciatic nerve. 3 Theiler also noted that spread of infection in the CNS was rapid, detecting infectious virus in the spinal cord within 24 hours of infection, and that virus could remain present and infectious in the CNS for up to 1 year after infection, with longer durations lasting in mice that developed paralysis. 3 Lipton 12 injected brain-passaged DA strain into SJL mice yet could not detect infectious virus in the blood and did not find viral antigen in the intestine, although he found that antibody levels rose from day 7 PI to 2 months PI. Viral RNA was detected by quantitative polymerase chain reaction (qPCR) in the spinal cord 1 year PI, yet none was detected in the lung, heart, liver, kidneys, spleen, mesenteric lymph nodes, small intestine, or colon 4 months PI. 13 This led Trottier et al 13 to suggest that perhaps the BeAn strain of virus remains fully within the CNS after IC inoculation. It is important to note that TMEV has been shown to not cause breakdown of the BBB, 14 and instances of TMEV transmission out of the nervous system into the blood, intestinal tract, saliva, or feces, where it can naturally transmit outside of the body, have not been documented. Therefore, the extent of contagiousness of TMEV following IC inoculation is unknown.
If there is a period of contagiousness during infection with TMEV, it may be more likely to occur during the acute infection, specifically because of the necessary puncture of the BBB to deliver the virus IC. This study used previously established qPCR methods 13 to detect the presence of viral RNA in the blood, saliva, and feces during the acute stage of TMEV infection and at selected periods of time during chronic infection with the goal of identifying and defining the time frame when IC-inoculated mice may be shedding virus and thus spreading TMEV to cage-mates and possibly entire colonies.
Materials and Methods
Mice and Viral Administration
Female SJL mice were purchased from Envigo (formerly Harlan Laboratories). Groups of at least 3 replicates were tested, as was adequate in a similar qPCR study. 13 Four replicates were planned to still have 3 remaining replicates in the event of anesthesia-related death during the IC injection procedure. SJL mice were chosen due to their susceptibility to chronic infection. TMEV infection initially causes an acute state of encephalomyelitis in mice immediately after IC injection of TMEV, and recovery begins within several weeks in nonsusceptible mice such as C57. However, in SJL and other susceptible strains, the infection gives way to a progressive demyelinating course starting at about 35 days PI. 15 Genetic variations in the major histocompatibility complex Class I molecules have been linked to susceptibility to chronic TMEV infection, 16 and the H-2D region, in particular, is thought to be involved in SJL susceptibility. 17
All institutional policies regarding animal care and biosafety were followed (Institutional Animal Care and Use Committee protocol NEU02123Y; Animal Biosafety Level 2 [ABSL-2]). Mice were housed and samples were obtained in quarantined rooms within the animal facility. Housing cages were enclosed with filter-top lids. Personnel protective equipment (PPE) on personnel entering the quarantined rooms included gowns, booties, bouffant caps, gloves, and N95 masks. PPE was removed upon exit from the quarantined rooms. All used bedding, and PPE was autoclaved prior to disposal by the animal facility personnel. All other waste accumulated in the quarantined rooms was disposed of as regulated medical waste by the laboratory personnel. Any materials or samples removed from the room and brought to the laboratory bench were double-contained and the primary container was wiped with chlorine dioxide solution.
A sample of BeAn 8386 strain, BHK-21 cell-purified working stock 20A, 18 was generously gifted from Dr Howard Lipton, University of Illinois, Chicago, in December 2013. Virus culture and titration services were provided by the Genomics/shRNA Shared Resource at Roswell Park Cancer Institute (RPCI) in Buffalo, New York (funded by National Cancer Institute P30CA16056 and RPCI Core Grant). Methods followed were as per previously published protocols. 19 Subjects were anesthetized with 50 mg/kg ketamine and 2 mg/kg xylazine IP with a 26-gauge needle, and virus was IC-injected with 3 × 106 plaque-forming units (PFUs) of TMEV by a 0.5-mL tuberculin syringe with attached 27-gauge 0.5-inch needle (BD) into the right cerebrum at 6 to 7 weeks of age.
Live Biomaterials Collection
To obtain a fecal sample, a previously published procedure was followed. 20 Subjects from shared cages were removed from their cages and separated so as to distinguish from which mouse feces came. One to 3 fresh fecal pellets were collected from each mouse at each time point. Then, 1× phosphate-buffered saline (PBS) (Invitrogen) was added to the fecal sample at 10 times the volume of feces, supplemented with 50 μg/mL gentamicin (Sigma). The sample was vortexed until mixed and centrifuged at 900 g for 20 minutes at 4°C. The supernatant was removed from the pellet and passed through a 0.45-μm syringe filter and then either immediately used or stored at –80°C. To obtain a saliva sample, a previously published procedure was followed, modified for a smaller strain of mouse. 21 A 10-μL pipet was used to release 10 μL water into the mouth of the subject and quickly reaspirated into the same pipet tip. Then, 10 μL 1× PBS, supplemented with 50 μg/mL gentamicin, was added to bring the sample to an approximate volume of 15 μL, and the sample was either immediately used or stored at –80°C. Serum was collected by collecting 2 to 3 drops of blood from the temporal facial vein and allowing it to clot by sitting undisturbed at 25°C for 15 to 30 minutes. It was centrifuged at 16 000 g for 10 minutes, and the supernatant was removed and then either immediately used or stored at –80°C.
CNS Tissue Collection
Subjects were sacrificed by IP injection of 100 mg/kg sodium pentobarbital. Following cessation of breathing, the head was decapitated and the brain was removed from the skull and flash frozen in <–20°C dimethylbutane. The spinal column was then removed from the body, and the rostral and caudal ends were cut open. A 10-mL syringe with an 18-gauge needle filled with 1× PBS was inserted into the rostral end and used to flush the spinal cord out through the caudal end into a petri dish filled with 1× PBS. The spinal cord was also flash frozen, and both the brain and spinal cord were stored at –80°C. Homogenization was performed by bead-beating with 0.5-mm glass disruptor beads (Scientific Industries) and Mini Beadbeater MBB-8 (Biospec Products).
RNA Isolation, Complementary DNA Synthesis, and qPCR
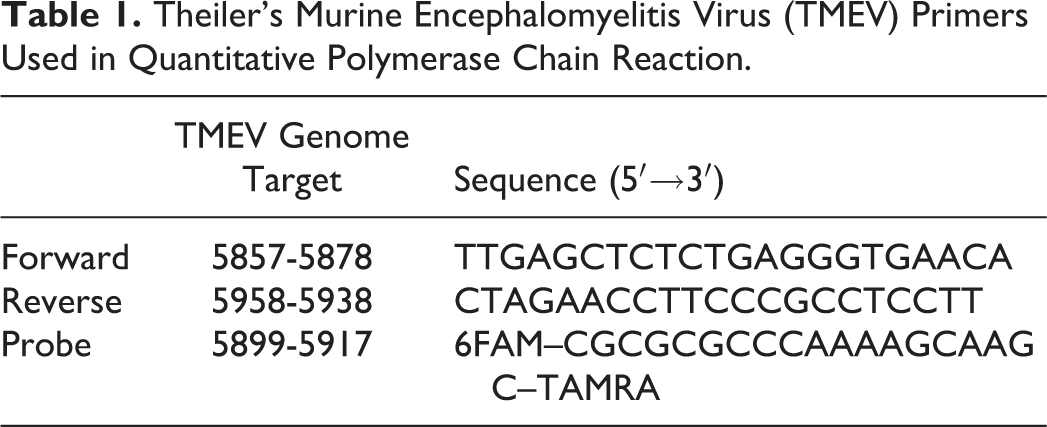
RNA was isolated with the aid of EZNA Total RNA Kit I (Omega Biotek). Work surfaces and equipment were cleaned prior to procedure start with RNase Away (Sigma). RNA concentration was quantified by a Nanodrop 2000c spectrophotometer (Thermo Scientific), and samples were either used immediately or stored at –80°C. RNA samples were diluted in DEPC water (Invitrogen) to standardize all reactions to one concentration in 10 μL. Complementary DNA (cDNA) was synthesized with the aid of the SuperScript III First-Strand Synthesis System (Invitrogen). cDNA samples were either used immediately or stored at –20°C. Forward primer, reverse primer, and probe were ordered from Integrated DNA Technologies following the design published for “Primer 1,” “Primer 2,” and “Probe” as previously published 13 (Table 1). Quantitative PCR was performed with the aid of the AmpliTaq Gold 360 PCR Kit (Applied Biosystems). Two separate master mixes were made to conduct the qPCR in duplicate, consisting of 1 μL 5 pmol/μL forward primer, 1 μL 5 pmol/μL reverse primer, 1 μL 3 pmol/μL probe, 0.5 μL Enhancer (Applied Biosystems), 12.5 μL AmpliTaq Master Mix (Applied Biosystems), and 4 μL DEPC water. The concentration of cDNA added to each reaction was controlled by adding equal volumes of product synthesized by controlled quantities of starting RNA. The qPCR was conducted in a CFX thermocycler (Bio-Rad). The incubation program followed the previously published design 13 : 50°C for 2 minutes, 95°C for 10 minutes, and 40 cycles of 95°C for 15 seconds followed by 60°C for 1 minute, with plate reading fluorescence occurring at the terminus of the 60°C incubation at the end of each cycle. Cycle threshold (Ct) values for each subject were calculated using CFX Manager software, version 3.1 (Bio-Rad).
Theiler’s Murine Encephalomyelitis Virus (TMEV) Primers Used in Quantitative Polymerase Chain Reaction.
Results
Detection of Viral RNA in Acute Infection
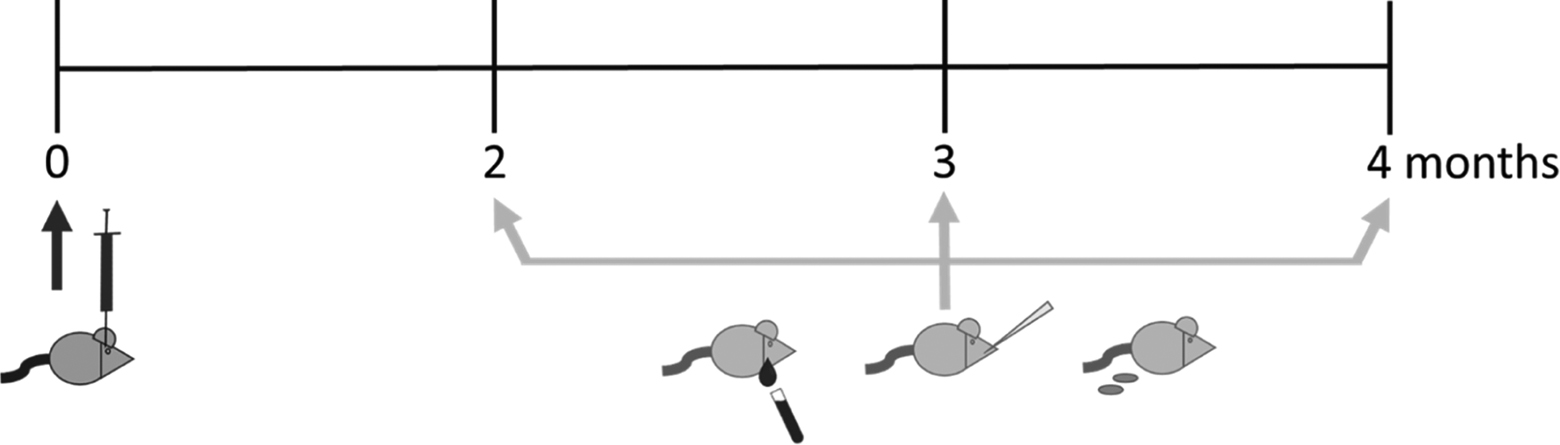
Four TMEV-infected subjects and 3 saline-injected subjects were followed over a 21-day period (Figure 1). RNA was isolated from freshly collected blood, saliva, and feces at 1, 3, 5, 7, 14, and 21 days PI. At 21 days PI, brains and spinal cords were extracted and flash-frozen, then later thawed for RNA isolation. Complementary DNA was reverse-transcribed for all samples from each material during the same reaction along with a positive control viral supernatant and a negative control. Quantitative PCR was performed in duplicate, with all cDNA from each material, the positive control, the reverse transcription negative control, and another negative qPCR control on a single plate, targeting the TMEV virus via the FAM primer-probe system over 40 cycles. Neither of the negative controls showed any amplification on any qPCR plates.
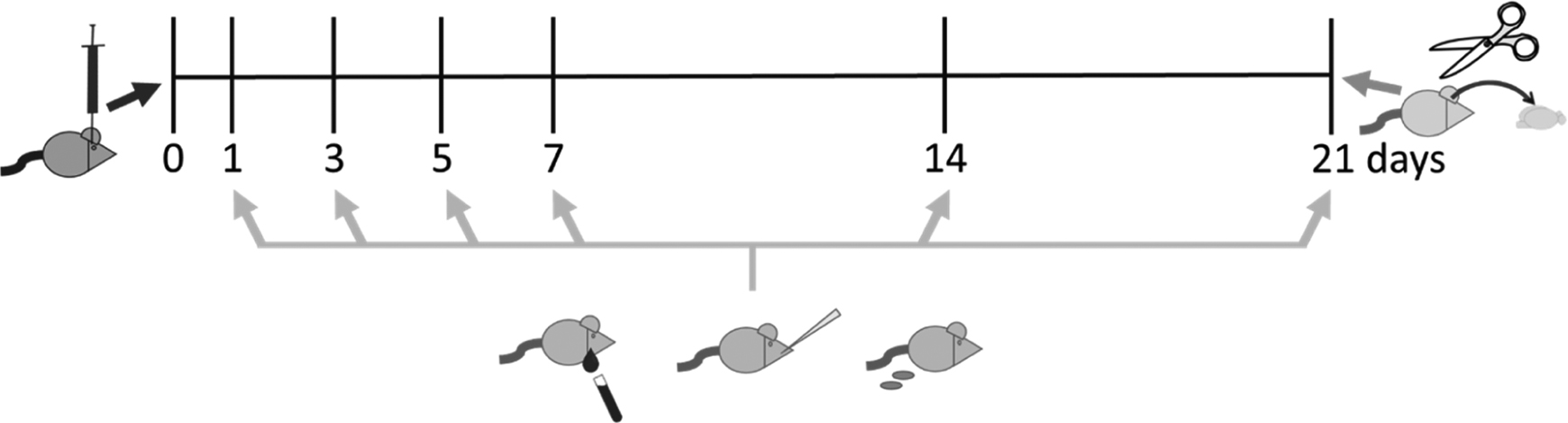
Timeline acute infection collections.
Target amplification could not be detected in serum, saliva, or feces at any of the time points (Table 2). One sample amplified after the 39th cycle, but melt curve analysis indicated that the product was not the TMEV target (Figure 2), thus constituting nonspecific amplification. In contrast, viral amplification was found in all infected brains and 3 of the 4 infected spinal cords (Figure 3).
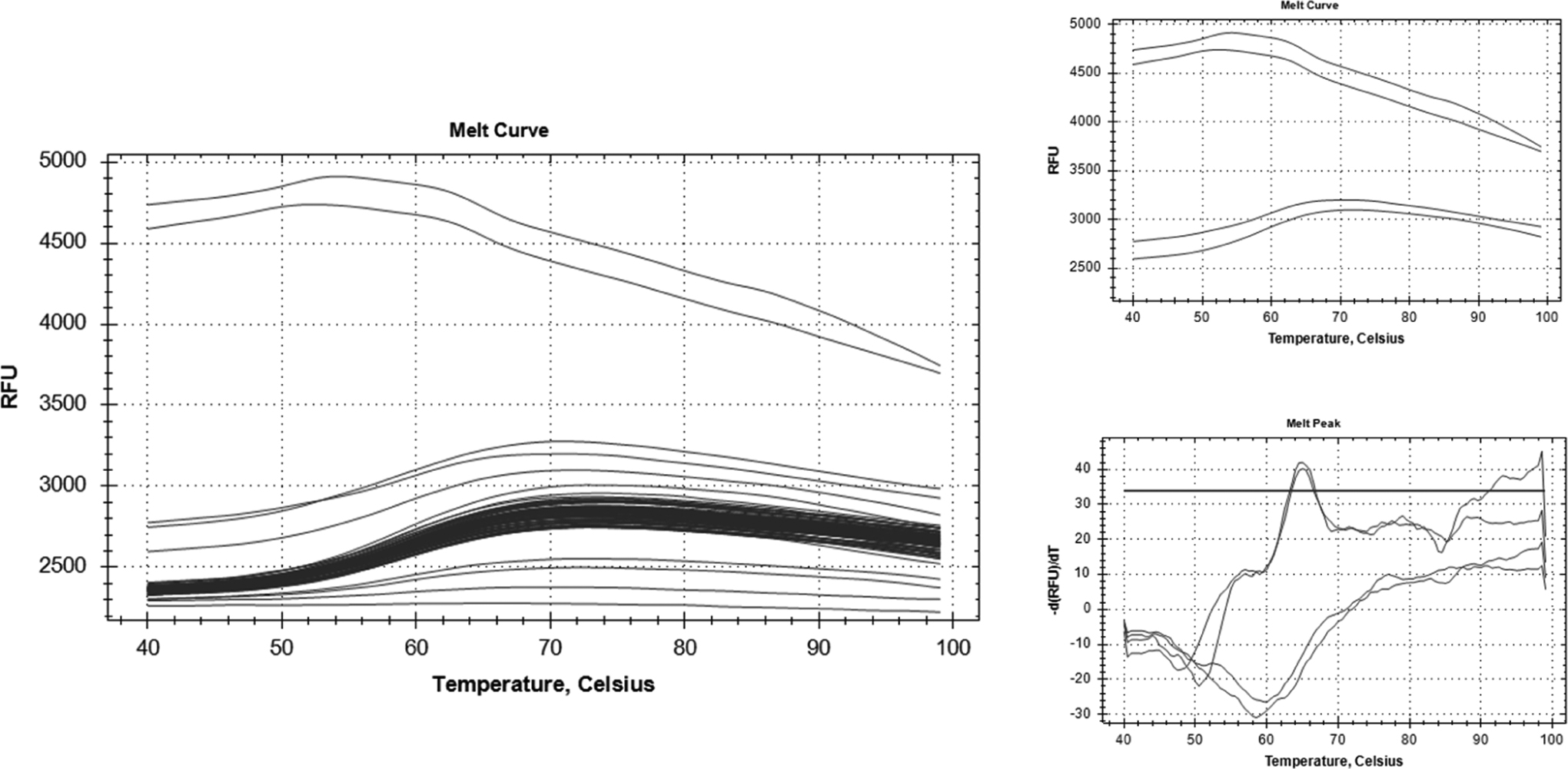
Nontarget amplification. Left: representative melt curve showing samples including “TMEV2 feces 5 days PI” (bottom) and viral supernatant (top). Right: melt curve graphs showing contrast between “TMEV2 feces 5 days PI” (bottom) and viral supernatant (top). PI, postinjection; RFU, relative fluorescence units.
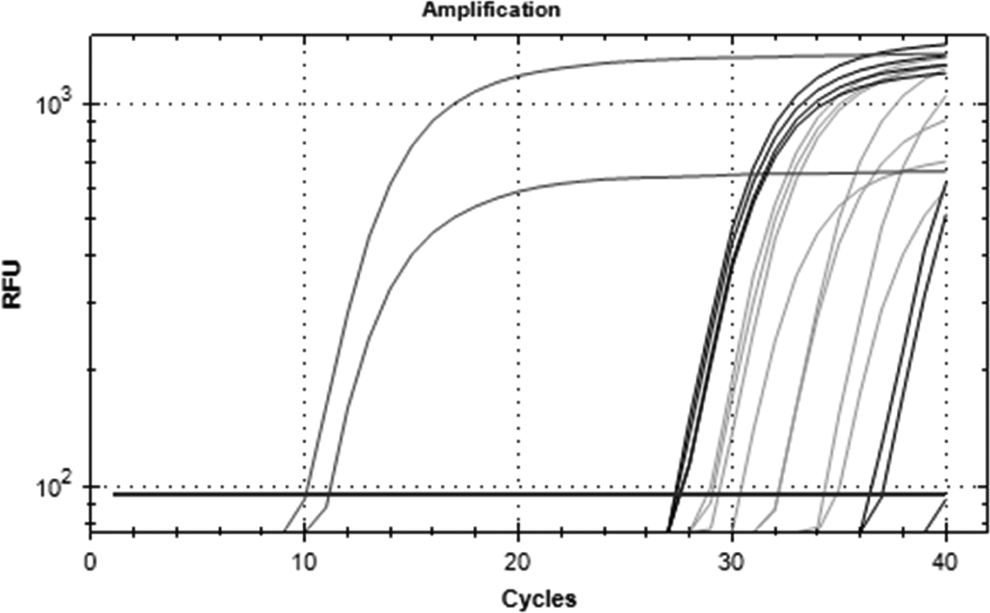
Viral RNA amplification in the central nervous system. Dark gray, viral supernatant (far left curves); black, infected spinal cords; light gray, infected brains. Uninfected spinal cords and brains exhibited no amplification. RFU, relative fluorescence units.
Ct for TMEV-Infected and Saline-Injected Biomaterials During Acute Infection.a
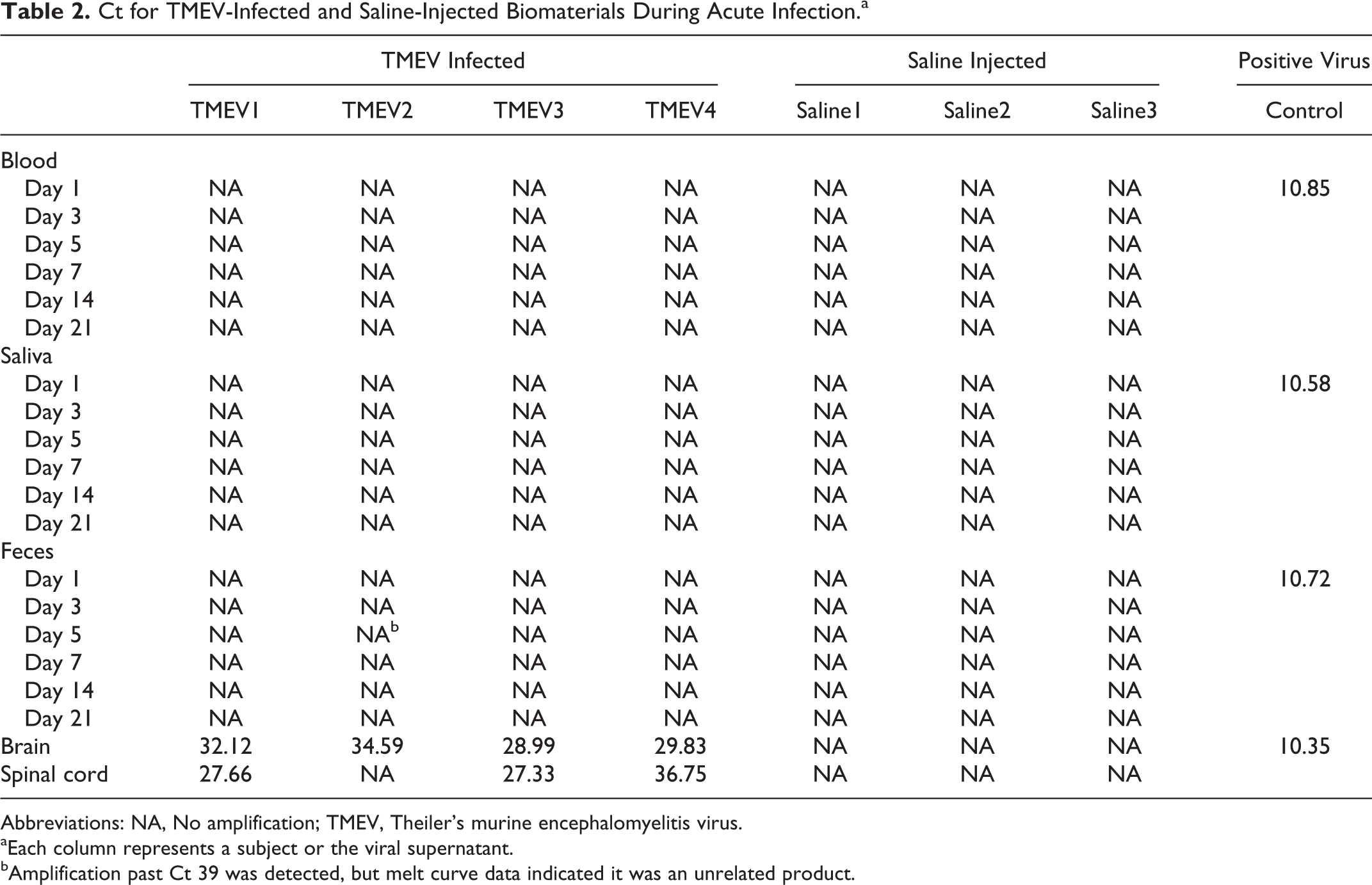
Abbreviations: NA, No amplification; TMEV, Theiler’s murine encephalomyelitis virus.
Each column represents a subject or the viral supernatant.
Amplification past Ct 39 was detected, but melt curve data indicated it was an unrelated product.
Detection of Viral RNA in Chronic Infection
To investigate contagiousness through viral shedding during chronic infection (Figure 4), blood, saliva, and feces were collected from 3 infected subjects at 2 months, 3 months, and 4 months PI (65, 91, and 120 days PI). Saliva and feces were not collected from these 3 subjects at 3 months PI, but collections from an additional 2 mice at 2 months PI (61 days PI) were analyzed instead (Figure 5). Each sample, plus viral supernatant positive control and water negative control, was reversed transcribed to produce cDNA, and qPCR was performed as described above. Of infected subjects examined 2, 3, and 4 months PI, none of the materials showed any amplification of the TMEV target (Table 3).

Chronic TMEV-infected mouse. Top: example of a fully progressed TMEV-infected adult SJL mouse at about 1 year PI, when difficulty righting itself becomes visually apparent. Bottom: example of a saline-injected adult SJL mouse of comparable age standing upright at rest. PI, postinjection; TMEV, Theiler’s murine encephalomyelitis virus.
Timeline for chronic infection collections.
Ct for TMEV-Infected Biomaterials During Chronic Infection.a
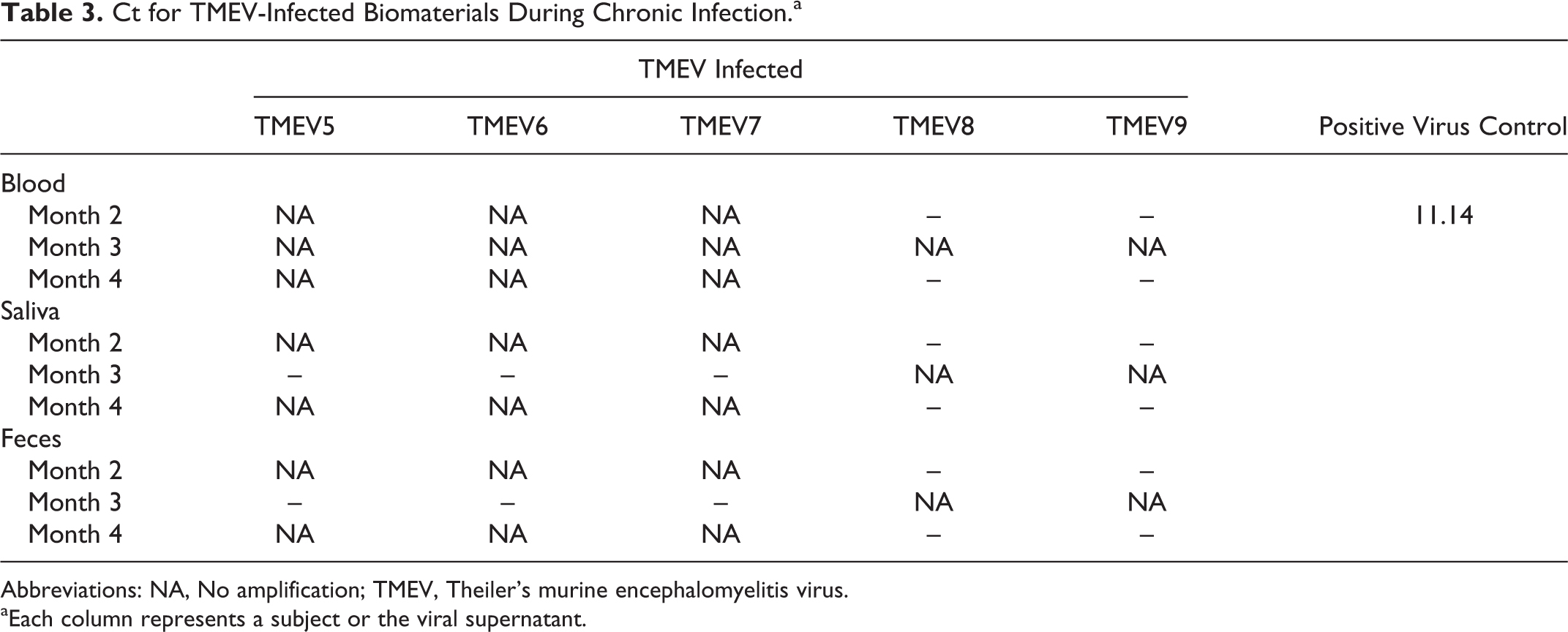
Abbreviations: NA, No amplification; TMEV, Theiler’s murine encephalomyelitis virus.
Each column represents a subject or the viral supernatant.
Quantification of Viral RNA in the CNS
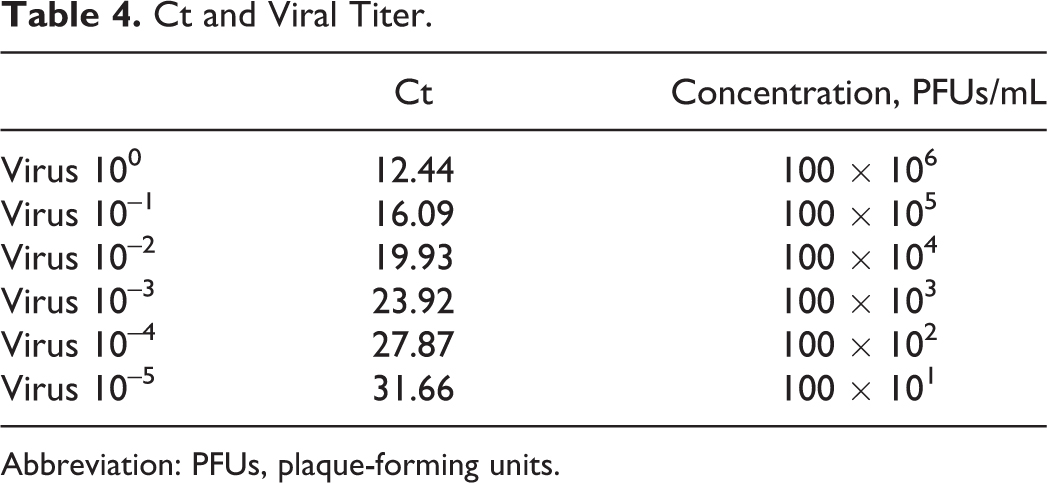
The approximate PFUs per milliliter of virus in the brain and spinal cord was calculated using the known concentration of the viral stock. Quantitative PCR was performed with five 10-fold serial dilutions on the previously synthesized cDNA of the brains, spinal cords, and viral supernatant (Figure 6). Ct values and their corresponding known concentrations from the virus dilutions are listed in Table 4. Known concentrations and Cts were graphed for the virus dilutions to produce a standard curve (Figure 7) and to obtain the following formula for calculating concentration from Ct: y = (2 × 1011)(e-0.595x).
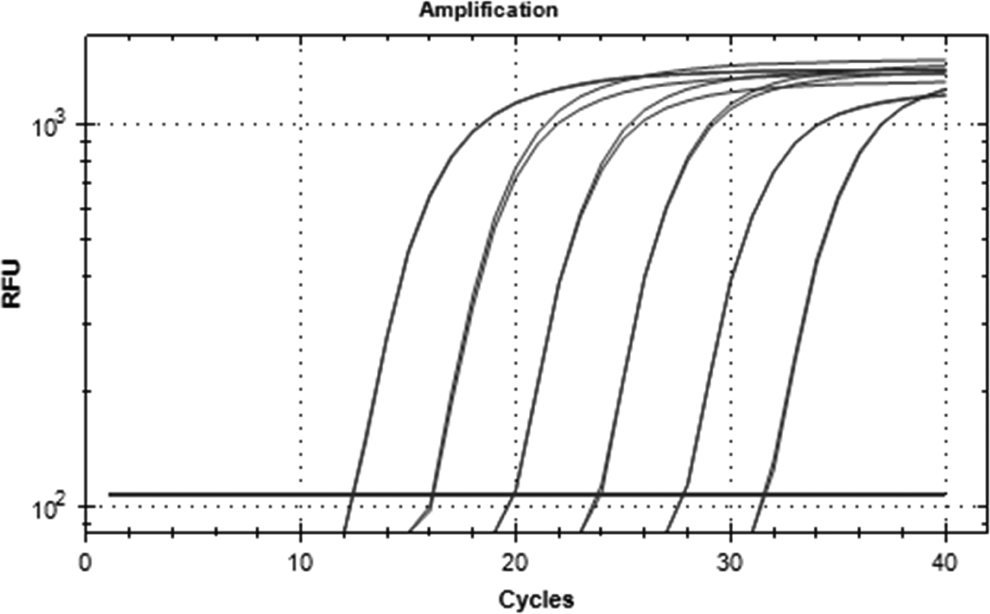
Viral supernatant and dilution amplification. Paired curves from left to right: stock virus followed by 10–1, 10–2, 10–3, 10–4, and 10–5 dilutions. RFU, relative fluorescence units.
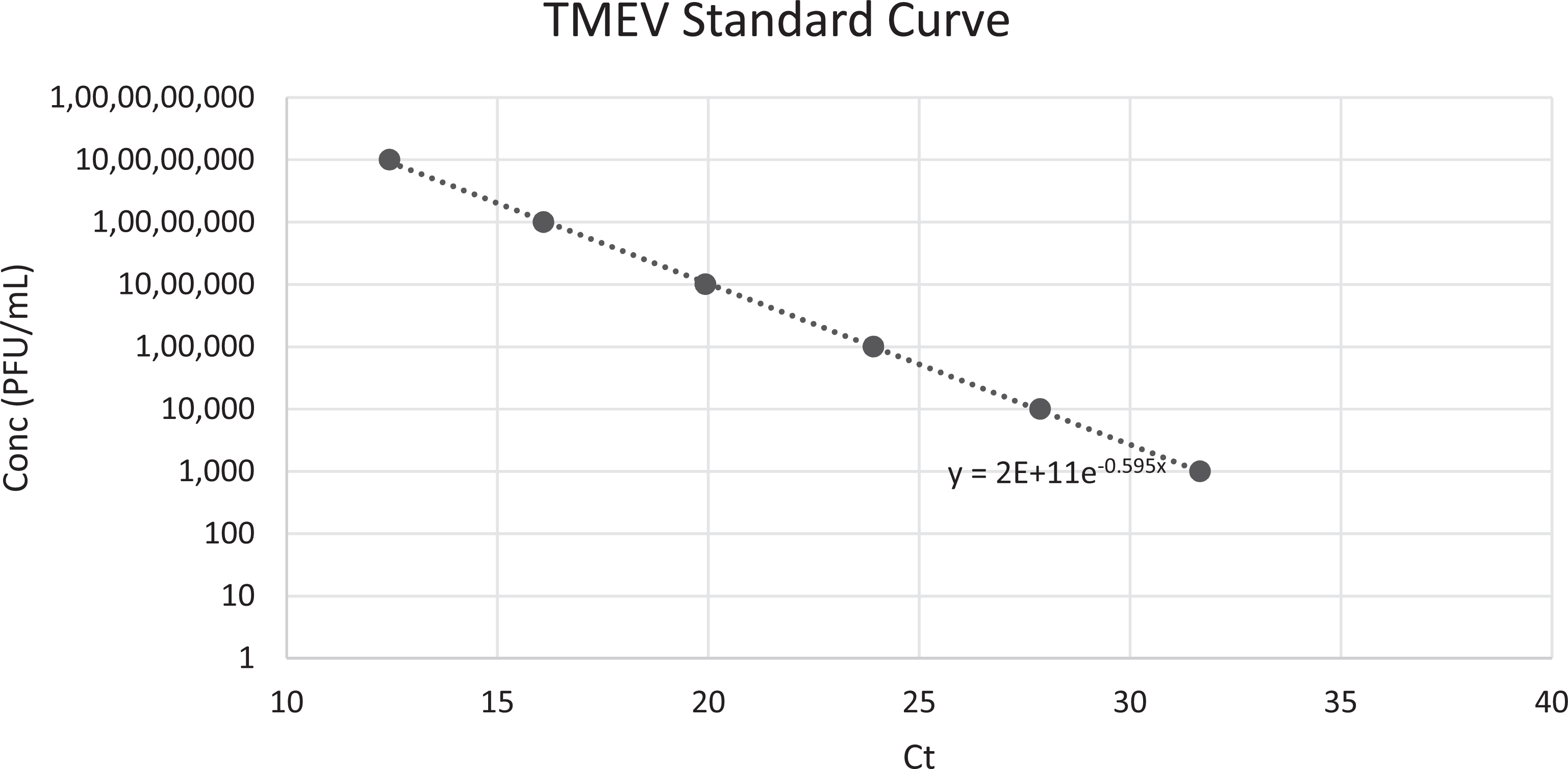
Theiler’s murine encephalomyelitis virus (TMEV) amplification standard curve. Known virus dilution concentrations plotted against obtained Cts to produce a formula from which to identify approximate concentration from Ct. Conc, concentration in plaque-forming units/mL.
Ct and Viral Titer.
Abbreviation: PFUs, plaque-forming units.
Finally, the formula was applied to Cts obtained from the same qPCR plate for cDNA corresponding to brain and spinal cord samples (Table 5) to calculate the approximate viral concentration for each sample. Viral concentration in the subject brains ranged from 8.8 × 101 to 1.5 × 103 PFUs/mL. Viral concentration in the subject spinal cords ranged from 1.7 × 101 to 4.2 × 103 PFUs/mL. The differences between viral distributions in the spinal cord compared with the cerebrum ranged from none to over 10-fold greater in these data at 21 days PI. Trottier et al 13 found the disparity in viral distribution to range from 10-fold more in the spinal cord compared with the brainstem/cerebellum and 100-fold more in the spinal cord compared with the cerebrum at 46 to 124 days PI. 13 This may indicate that the virus gradually favors distribution in the spinal cord as disease progresses. However, while the brains were extracted whole and intact, the spinal cord extraction occasionally broke, resulting in loss of tissue. While every attempt was made to retrieve the entire spinal cord, some portions may have been lost. This factor may be relevant due to the potential for uneven viral density throughout the CNS, and thus portions of the spinal cord with differing viral concentration may have been sampled, potentially altering the calculated viral concentration calculated in Table 5.
Ct and Viral Concentration in the Central Nervous System.a
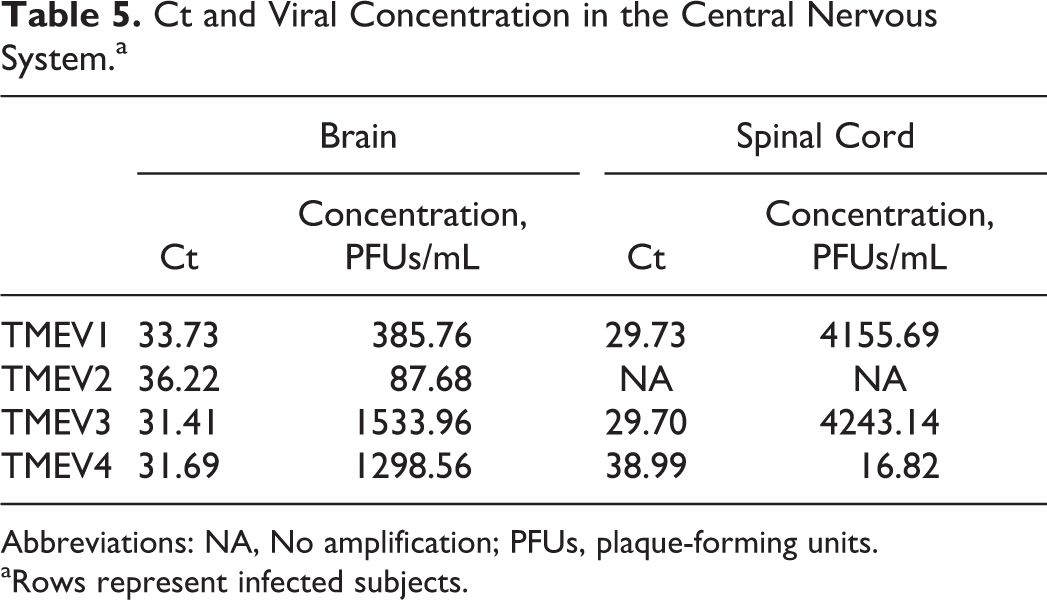
Abbreviations: NA, No amplification; PFUs, plaque-forming units.
Rows represent infected subjects.
Discussion
Risk assessments are an essential tool when determining biosafety and biosecurity precautions while using infectious organisms. In the case described, TMEV is an infectious virus whose tropism is restricted to mice. Naturally occurring TMEV causes an enteric infection in mice and is transmitted via the fecal-oral route. Importantly, TMEV is used by a number of investigators as a model for neurodegenerative diseases such as MS. Typical precautions taken at animal facilities include housing these animals in dedicated, negatively pressured rooms and using dedicated cages and equipment for fear of disease spread throughout the facility. While performing a risk assessment for the use of TMEV, the authors discovered that critical information was lacking. Specifically, the missing information was the potential for IC-inoculated mice to shed TMEV. Anecdotal evidence suggested that the virus remained in the CNS and was not spread peripherally, a requirement for viral shedding. We therefore designed an experiment where we could quickly acquire the missing data required for a complete and thorough risk assessment. The hypothesis of contagiousness through TMEV shedding in the IC-inoculated MS mouse model was investigated using the BeAn8386 strain in SJL mice and found to be unsupported.
Originally, due to the perceived risk associated with TMEV to other mouse colonies, the laboratory animal facility required ABSL-2 procedures, including negative air pressure to prevent contaminated air from escaping the quarantined rooms and exiting into the corridor. Mice were housed in filter-topped cages, although it was necessary to behaviorally test the mice on an open benchtop since the behavioral testing apparatus could not fit into a biosafety cabinet. This testing was performed in a separate negatively pressured, quarantine room directly adjacent to the housing room. Mice were transported between rooms in their filter-topped cages. PPE consisted of a gown, booties, bouffant cap, N95 mask, and gloves, all of which had to be disposed of prior to exiting the quarantined animal housing or testing rooms.
Decontamination procedures as established by the laboratory animal facility for any object that had entered the quarantined rooms or come into contact with a quarantined mouse were saturation with a chlorine dioxide solution or through gaseous chlorine dioxide decontamination. With regard to administrative tools, laboratory testing equipment, and furniture, these quarantined rooms were unidirectional containment spaces, meaning any object brought into these rooms could not be removed and would have to be devoted to TMEV studies in these quarantined rooms. Note paper could not be removed from the room, so laboratory books had to be photocopied and a digital scanner had to be installed to scan and export notes for later printout and paste in laboratory notebooks and files. Costly experimental tools, such as specially designed behavioral testing apparatus, cameras, magnetic resonance imaging holding beds, surgical heating pads, and peristaltic pumps, could not be decontaminated as defined by the facility as, due to their electrical components, polymer composition, or size limitation, they could not be submerged in chlorine dioxide solution or treated in chambers with chlorine dioxide gas.
Due to the measures instituted, the research group using TMEV approached the biosafety office to perform a risk assessment. The biosafety office has developed an in-house Biological Risk Assessment & Mitigation Tool to be used by investigators and the biosafety office to establish the most appropriate biosafety and biosecurity measures for a project. Importantly, while performing the risk assessment, the team found no evidence to support an airborne mouse-to-mouse infection model for the TMEV virus, making filter-top cages an unnecessary expense. In addition, there is no evidence to support the possibility that the TMEV virus can infect humans, thus making the use of N95 masks unnecessary. With the added lack of evidence that IC-inoculated mice shed TMEV as discussed in this report, single-use masks and bouffant caps appear to be an unnecessary expense. Following the risk assessment process including the data discussed above, we have concluded that the separation of TMEV-infected housing in a separate or barrier room is a worthwhile precaution, but other ABSL-2 measures such as filter-topped cages in an already negatively pressured, quarantined room may be excessively costly for the risk at hand. To prevent potential but unlikely transfer of viral particles from room to room on the surface of human skin or clothes that may come in contact with mice, we have concluded that reasonable PPE requirements for handling TMEV-infected mice should include gloves, disposable shoe covers, and a reusable gown donned upon entry into the room and removed prior to exit. The lack of evidence to suggest IC-inoculated mice shed TMEV, along with its classification as a Picornaviridae, has allowed for a revised, less stringent decontamination procedure for equipment, note paper, and other objects. The new procedure allows for wiping all surfaces with a decontaminant suitable for use with other small, nonenveloped viruses. These decontaminants include sodium hypochlorite, ethylene oxide, and hydrogen peroxide. Equipment treated with any of these decontaminants can be removed from the treatment or holding room and reused.
The results described in this report were used to better inform a complete risk assessment for TMEV use in SUNY at Buffalo’s laboratory animal facilities. Importantly, the informed risk assessment allows for less restricted use of TMEV in neurodegenerative research within the laboratory animal facilities without the fear of pathogenic spread between cages or colonies. In particular, these results allow for the expanded use of equipment used to test behavioral change and outcome due to disease. Basic precautions should still be followed to prevent the possible spread of virus in the event that it does find its way out of the CNS and past the intestinal lining, where it is known to naturally spread out of the body. However, CNS to periphery spread has yet to be documented, and the high-level protections and expensive precautions associated with IC-inoculated TMEV models appear to be an unnecessary use of valuable resources.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R. Zivadinov received personal compensation from Teva Pharmaceuticals, Biogen Idec, EMD Serono, Genzyme-Sanofi, Claret Medical, IMS Health, and Novartis for speaking and consultant fees. He received financial support for research activities from Teva Pharmaceuticals, Genzyme-Sanofi, Novartis, Claret Medical, Intekrin, and IMS Health.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Study partially funded by UUP Professional Development Award 889303-68 awarded to David R. Pawlowski.
