Abstract
Respiratory protection is a key component of the biosafety program for the Regional Biocontainment Laboratory at Duke University. Two types of loose-fitting powered air-purifying respirators (PAPRs) equipped with high-efficiency particulate air (HEPA) filters have been approved for use in the facility. Although both respirator systems were previously certified by the National Institute of Occupational Safety and Health for use in occupational environments that pose a risk of respiratory exposure to infectious agents, there are currently no regulatory requirements for routine field-testing of units in situ. This report describes a method for conducting on-site total leak testing for each HEPA-filtered PAPR used in a biocontainment setting. This testing evaluates the integrity of the filter itself and whether it is seated properly in the filter housing. On-site routine performance testing such as this provides an enhancement to the safety procedures governing research activities assigned to biosafety level 3 or animal biosafety level 3. The outlined test method is currently used in the Regional Biocontainment Laboratory at Duke University to verify proper filtration efficiency prior to placing newly purchased units into service and for annual reverification. Results from on-site testing with this method have demonstrated that some PAPR/HEPA filter systems fail to perform at the expected level of filtration efficiency, thus emphasizing the need for including this enhancement and regular testing in a comprehensive biosafety program that includes respiratory protection at biosafety level 3 or animal biosafety level 3 (ABSL-3).
The Regional Biocontainment Laboratory (RBL) at Duke University is utilized to safely perform research activities involving infectious microbes at biosafety level 3 (BSL-3). Such containment is applicable for minimizing exposure risk when working with infectious agents that may cause serious or potentially lethal disease through the inhalation route of exposure. 1 A strict set of standard operating procedures governs all work activities in the laboratory and animal areas of the RBL, including defined requirements for personal protective equipment. A risk assessment of the high-containment research (ie, BSL-3) indicated a need for respiratory protection of laboratory personnel. The advantages and disadvantages of both powered and nonpowered respirators were considered when determining the most appropriate type of device to use in a BSL-3 environment. 2 It was determined that a loose-fitting powered air-purifying respirator (PAPR) equipped with a high-efficiency particulate air (HEPA) filter and a double-shrouded hood would best meet the needs of the research staff in the RBL. This respirator configuration is currently a required personal protective equipment component that is worn while actively handling risk group 3 (RG3) infectious microbes assigned to BSL-3 and infected animals assigned to ABSL-3.
The expected protection provided by a respirator when used within an effective respiratory protection program, as well as within compliance with Occupational Health and Safety Administration 29 CFR 1910.134, 3 is expressed as its assigned protection factor (APF).4,5 An APF represents the ratio of the concentration of a contaminant outside a respirator to the concentration of a contaminant inside the respirator; thus, the APF increases as this ratio increases. A loose-fitting PAPR with HEPA filtration has an APF of 1000 and, when worn properly, provides a higher level of respiratory protection when compared with negative-pressure half-mask respirators, such as N95 respirators, which have an APF of 10.2,3,5 This added level of protection has led to the use of PAPRs in many work environments that pose a risk of occupational exposure to airborne biohazards, as in BSL-3 laboratories. Other benefits of wearing a loose-fitting PAPR include the following: it offers respiratory protection without the need for tight face seal or fit testing; it can be worn by individuals with facial hair; the head cover and facepiece provide added splash and contact protection; it provides improved comfort for the wearer by constant flow of air through the unit; and the battery-powered blower pulls air through the filter, which results in no additional breathing resistance for the wearer.
Various types of HEPA-filtered containment equipment are commonly found in BSL-3 facilities. Examples include biological safety cabinets, ventilated animal caging systems, and exhaust air filtration systems. Several guidelines suggest the routine field-testing and certification of such equipment.1,6,7 In addition, inspectors from the Federal Select Agent Program (Centers for Disease Control and Prevention) require registered facilities to provide written test reports for all biological safety cabinets, animal caging systems, and exhaust filters during routine site visits. While all of the aforementioned entities and others place importance on routine testing of certain protective equipment, none of them currently require or recommend in situ efficiency testing of PAPR units.
Although HEPA-filtered PAPRs are worn to protect workers from potentially infectious bioaerosols during laboratory work—as would be generated from spills outside of biological safety cabinets—we are not aware of any current requirements to verify proper filtration efficiency in the field. American National Standards Institute Z88.2, “Practice for Respiratory Protection,” simply indicates that the PAPR high-efficiency filter should be replaced whenever the minimum airflow cannot be maintained, whenever the filter is damaged, or whenever unacceptable breathing resistance is noticed. 5 The American National Standards Institute report does not make a specific recommendation for routine field-testing of the PAPR system as it is to be used in the workplace. In addition, the Occupational Health and Safety Administration requires that PAPRs worn in the occupational setting be National Institute for Occupational Safety and Health (NIOSH) certified and used in compliance with a site-specific respiratory protection program; however, this standard does not specifically require any type of regular in situ testing. Thus, the safety team of the RBL at Duke University decided to investigate the feasibility of conducting on-site performance testing of the PAPR units used.
Both manufacturers of the PAPR systems used in the RBL (3M and MAXAIR) were contacted for guidance on how best to ensure proper unit filtration efficiency prior to use in a potentially hazardous work environment. Each emphasized the fact that its PAPR system was a NIOSH-certified respirator with an APF of 1000 and offered this as an indication of predicted performance over time in the field. The following summaries provide additional responses from each PAPR vendor.
3M recommends a thorough inspection of the whole unit prior to each use. This includes removal of the back cover and filter and careful inspection of multiple internal components (Figure 1). 3M also emphasizes the importance of verifying proper airflow by performing a flow check prior to each use. 3M does not recommend nor provide instruction on field-testing.

3M Air-Mate motor assembly and high-efficiency particulate air (HEPA) filter.
MAXAIR recommends close monitoring of the 5 LED safety-status indicators located on the underside front of the unit’s helmet (in the user’s peripheral vision). These indicators are designed to notify the user of inadequate airflow or battery charge. MAXAIR technical support indicated that the filter should be replaced if it becomes soiled or clogged with particulate, if it tears or breaks, or if the seals between the filter and the helmet become compromised (Figure 2). MAXAIR does not recommend nor provide instruction on field-testing.

MAXAIR controlled air-purifying respirator motor assembly and high-efficiency particulate air (HEPA) filter.
As noted previously, respirators worn in an occupational environment are required to be certified by NIOSH for their intended use. 3 NIOSH issues certificates of approval for chemical, biological, radiologic, and nuclear PAPRs, as well as certificates of approval for specific filtration requirements (chemical, particulate, etc; 42 CFR 84). 8 According to NIOSH, the chemical, biological, radiologic, and nuclear standards and tests provide scientifically based, industry-accepted evaluation criteria that establish levels of protection for approved respirators. The NIOSH approval designations for PAPR systems usually fall under the “21C” or “23C” rating. The 21C designation is noted as being an approved “powered air-purifying respirator with particulate filter,” while the 23C designation indicates the addition of a “chemical cartridge [to the] powered air-purifying respirator with particulate filter.” 9
Primary components of these certifications are described in the NIOSH standard testing procedures RCT-APR-STP-0012 (“Determination of Air Flow for Powered Air-Purifying Respirators”) and TEB-APR-STP-0001 (“Determination of Particulate Filter Penetration Test, Powered Air-Purifying Respirator Filters”). Minimum certification standards for these standards are set in 42 CFR, part 84, subpart G, section 84.63(a)(c)(d), and subpart KK, section 84.1157(a)(c). 8 In brief, these standards require testing of the complete filtration system to verify the following: (1) a minimum of 170 L/min of airflow and the ability to effectively operate with a breathing resistance of 85 L/min; and (2) HEPA filtration efficiency of at least 99.97% when challenged with dioctyl phthalate at a concentration of 100 ± 10 mg/m3. Here, the complete system is defined in section 84.1131 as having a facepiece with a hood or helmet, a filter unit, a harness, an attached blower, and a breathing tube.
The initial approval by NIOSH requires that the PAPR manufacturer submit “data that demonstrate(s) the respirator under evaluation meets or exceeds the performance requirements set forth” in the standards. 9 NIOSH then performs the previously mentioned certification tests for the complete respirator system under laboratory conditions and issues an approval if the appropriate requirements are met (42 CFR 84.30). 8 All postdesignation aspects (eg, quality control and instructions for use) for the approved respirator are the responsibility of the manufacturer. NIOSH does not designate nor describe field use conditions in the approval process.
There are documented examples of performance evaluations conducted in the “workplace” by respirator manufacturers and certain industrial sectors. These studies used workplace protection factor (WPF) and simulated WPF (SWPF) studies to better understand the protection level offered by PAPRs under occupational or in situ condition.10 –13 A WPF study is conducted at the job site under normal working conditions and operation, while a SWPF is done in a laboratory where conditions can be more closely controlled and repeated from test to test. The results of these studies sometimes demonstrated significant differences in the APF and the actual measured workplace performance. DaRoza et al 11 conducted a SWPF study on 3 brands of PAPRs. In this study, aerosol penetration into the respirator facepiece was measured as subjects exercised on a treadmill. One of the tested PAPRs provided a protection factor of 10, when the usual APF for a PAPR is 1000. Myers and Peach conducted a WPF study of PAPRs used in a silica bagging operation and reported that the laboratory performance of the PAPR unit (APF of 1000) was better than the field performance of the PAPR. 13 The WPF measured on the unit ranged from 16 to 215. Although informative, none of these studies outlined a test method for the routine verification of the filtration efficiency of the PAPR systems currently used in the RBL at Duke University.
Therefore, the purpose of this study was to develop and document a test method for routine on-site total leak testing of PAPR systems currently used in the RBL by researchers in BSL-3 laboratories. Francisella tularensis, Mycobacterium tuberculosis, and Yersinia pestis are examples of RG3 microbes that can be transmitted via inhalation and are involved in laboratory and aerobiology animal studies in the RBL. Data collected during this study were generated by testing the actual PAPR systems currently used by RBL investigators. During the past 6 years (2010-2015), the RBL safety team has coordinated annual in situ testing of all PAPR units (3M Air-Mate) in service in the BSL-3 laboratories. On average, 23 units have been tested annually since 2010, per the Institute of Environmental Sciences and Technology’s (IEST’s) recommended practice 034.3 for testing HEPA filters. More recently, a new brand of PAPR (MAXAIR controlled air-purifying respirator disposable lens cuff [CAPR DLC]) has been purchased and evaluated for use in the BSL-3 laboratories of the RBL. The 3M unit test procedure was adapted to the MAXAIR configuration and used to assess performance. The evaluation process therefore included performance testing of these newly purchased MAXAIR units with the 2 HEPA filter options available for the CAPR system.
Methods and Results
PAPR Systems Evaluated
Two PAPR systems were evaluated as part of this study: the 3M Air-Mate Belt-Mounted High Efficiency PAPR (3M Occupational Health and Environmental Safety Division, St Paul, Minnesota) and the MAXAIR CAPR DLC (Bio-Medical Devices International, Inc, Irvine, California). All 3M PAPR units were leak tested with a 3M NIOSH-approved HEPA filter installed (p/n 451-02-01) and with a double-shrouded hood (p/n BE-10) attached to the assembly. MAXAIR provides a choice of 2 NIOSH-certified HEPA filters (p/n 2163-10XP and p/n 2164-10) for its PAPR unit. The performance of the MAXAIR PAPR unit was evaluated by testing it in 2 configurations: with the 2163-10XP filter installed and with the 2164-10 installed. Both MAXAIR test configurations were tested with a double-shrouded head cover (p/n 01031396) attached to the assembly.
Leak Testing
The PAPR blower unit and filter for the 3M and MAXAIR products are shipped separately to the customer (Figures 1 and 2). Instructions on proper filter installation are provided with each shipment. Test data for the 3M Air-Mate were collected after initial filter installation and all subsequent annual reverifications. Each test data point for the MAXAIR CAPR DLC was collected after initial filter installation only. Once a performance test was completed for the MAXAIR, the filter was replaced with another filter, and the “new” system was then tested.
Total leak testing of the PAPR systems was designed to simulate standard HEPA filter performance testing and was performed in accordance with the IEST’s recommended practice 034.3 (IEST-RP-CC034.3: “HEPA and ULPA Filter Leak Tests”). When installed, the filter on the 3M and MAXAIR units is covered and protected by hard plastic shielding (Figures 1 and 2). This method includes details on proper testing of installations where filters are inaccessible, therefore making scan testing of the filter impossible. 14
The test procedure (Figures 3A and 4A) was as follows:

Test setup for the 3M Air-Mate powered air-purifying respirator: (A) Whole test apparatus. (B) Upstream and downstream sample collection sites.

Test setup for the MAXAIR controlled air-purifying respirator disposable lens cuff: (A) Whole test apparatus. (B) Upstream and downstream sample collection sites.
The filter system was tested at the normal operating airflow for the PAPR unit.
A Laskin nozzle was used to generate a heterogeneous aerosol from poly-alpha olefin. An aerosol generated by a Laskin nozzle had a light scattering mean diameter of approximately 0.3 μm, with a geometric standard deviation of about 1.7.
Use of an aerosol photometer (ATI Model 2i; Air Techniques International, Owings Mills, Maryland) that is capable of measuring aerosol concentrations of up to 100 mg/m3 had a threshold sensitivity capable of measuring 1 µg/m3 and a nominal sampling flow rate of 28.3 L/min.
A challenge aerosol was introduced upstream of the HEPA filter in a manner that promotes proper mixing and uniform smoke concentration in the airstream as it enters the air intakes of the PAPR unit.
Use of a 4-ft flexible hose promoted turbulence in the aerosol challenge stream.
Prior to testing, we collected multiple samples at various locations around the air intakes and confirmed a constant and uniform aerosol challenge concentration.
The upstream concentration of challenge aerosol was adjusted to 10 to 90 µg/L of air, with subsequent adjustment of the photometer to be capable of reading 100% of upstream concentration.
The downstream air sample near the breathing zone of the wearer was collected (Figures 3B and 4B).
IEST indicates that the acceptance criteria for total leakage of the filter system should be agreed on between the customer and the testing vendor. The acceptable total leakage allowance for PAPRs in the RBL at Duke University is no more than 0.01%, or 99.99% removal efficiency (RBL pass). These criteria were instituted for the PAPR systems in the RBL to account for the possibility that a total leak test on the PAPR units is less sensitive than a filter scan test. This possibility is evident in a commonly referenced test standard for Class II biological safety cabinets and requires a filter efficiency of 99.995% for filters that cannot be scanned. 6 It is important to note that a HEPA filter has traditionally been defined as having a minimum particle collection efficiency of 99.97% (traditional pass).1,14
Leak Test Setup
Each PAPR system was performance tested “as used” while running with a double-shrouded headpiece attached. A quarter Laskin nozzle cold smoke generator (PAT model MM2500; Precision Air Technology, Inc, Morrisville, North Carolina) was used to produce aerosol challenge smoke (poly-alpha olefin, brand name Emery 3004) with an expected size range of 0.1 to 1.0 µm. To ensure a homogenous challenge of the whole face of the filter, the challenge aerosol was introduced into one side of a 4-ft-long section of flexible hose that was connected to the PAPR unit intake. An aerosol photometer was used to collect upstream and downstream samples. The upstream sample tube was positioned in close proximity to the upstream side of the filter, and the downstream sample tube was positioned inside the PAPR hood near the breathing zone of the worker. Figure 3 shows the setup for the 3M Air-Mate PAPR. Figure 4 shows the setup for the MAXAIR CAPR DLC. A total of 139 leak tests were performed on the 3M Air-Mate PAPR system equipped with HEPA filter 451-02-01, over the past 6 years. Seventeen independent leak tests were performed on the MAXAIR CAPR DLC system with filter 2163-10XP installed and on the MAXAIR CAPR DLC system with filter 2164-10 installed, as part of an initial evaluation of this new PAPR system for deployment in the RBL at Duke University.
Leak Test Results
Leak test results for the 3 HEPA filter types were tested for normality via Shapiro-Wilks tests (GraphPad Prism 6.0; GraphPad Software, La Jolla, California). All data were right skewed and natural log-transformed to satisfy normality assumptions. Differences among the 3 filter types were determined through 1-way analysis of variance with Greenhouse-Geisser corrections and Tukey post hoc tests for multiple comparisons. Multiple comparisons used an α of 0.05 and multiplicity-adjusted P values. 15 Table 1 summarizes the leak test results for all 3 PAPR/HEPA filter configurations.
Leak Testing Summary Statistics.
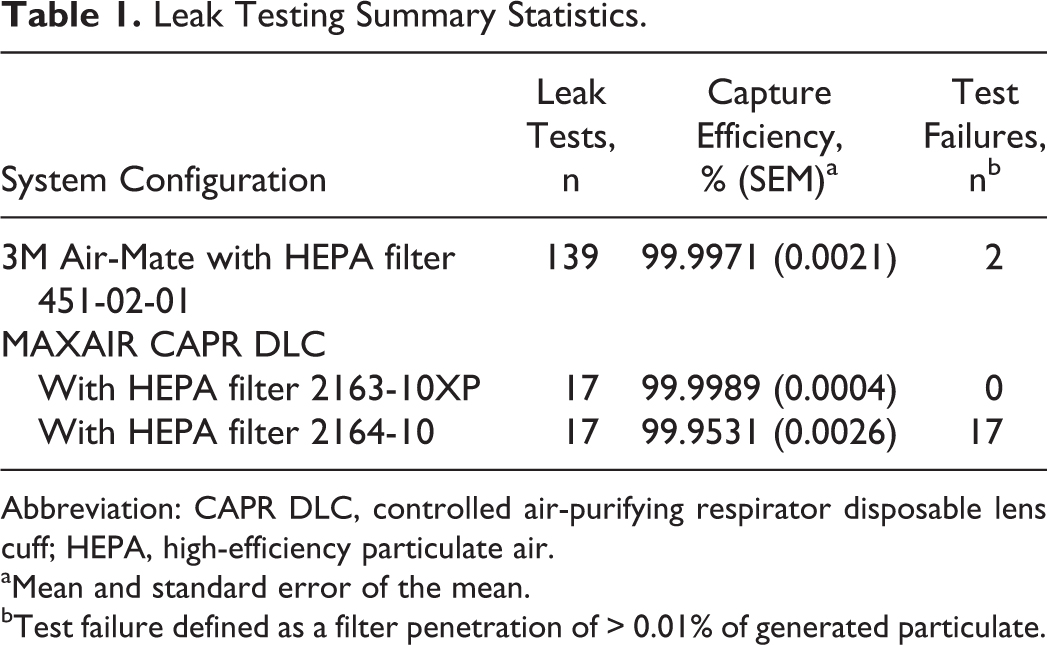
Abbreviation: CAPR DLC, controlled air-purifying respirator disposable lens cuff; HEPA, high-efficiency particulate air.
Mean and standard error of the mean.
Test failure defined as a filter penetration of > 0.01% of generated particulate.
The MAXAIR CAPR DLC with filter 2163-10XP had the best capture efficiency of the 3 filtration systems, followed by the 3M Air-Mate with filter 451-02-01 and the MAXAIR CAPR DLC with filter 2164-10. The MAXAIR configuration with filter 2163-10XP also had the smallest amount of variation in capture efficiency across the 17 leak tests, while the MAXAIR system with filter 2164-10 had the largest amount of variation in capture efficiency.
Figure 5 illustrates the significant decrease in capture efficiency exhibited by the MAXAIR CAPR DLC system with filter 2164-10 installed, as compared with the other filter configuration or the 3M Air-Mate setup. The average capture efficiencies from the leak tests for the 3M Air-Mate and MAXAIR CAPR DLC system with filter 2163-10XP were both well above the “RBL pass” criteria of 99.99%. However, the MAXAIR system with filter 2164-10 had a mean capture efficiency that was below the RBL cutoff as well as the traditional definition of HEPA filtration (99.97%). In fact, none of the 17 leak tests performed on this system with filter 2164-10 exceeded the 99.97% capture efficiency mark.
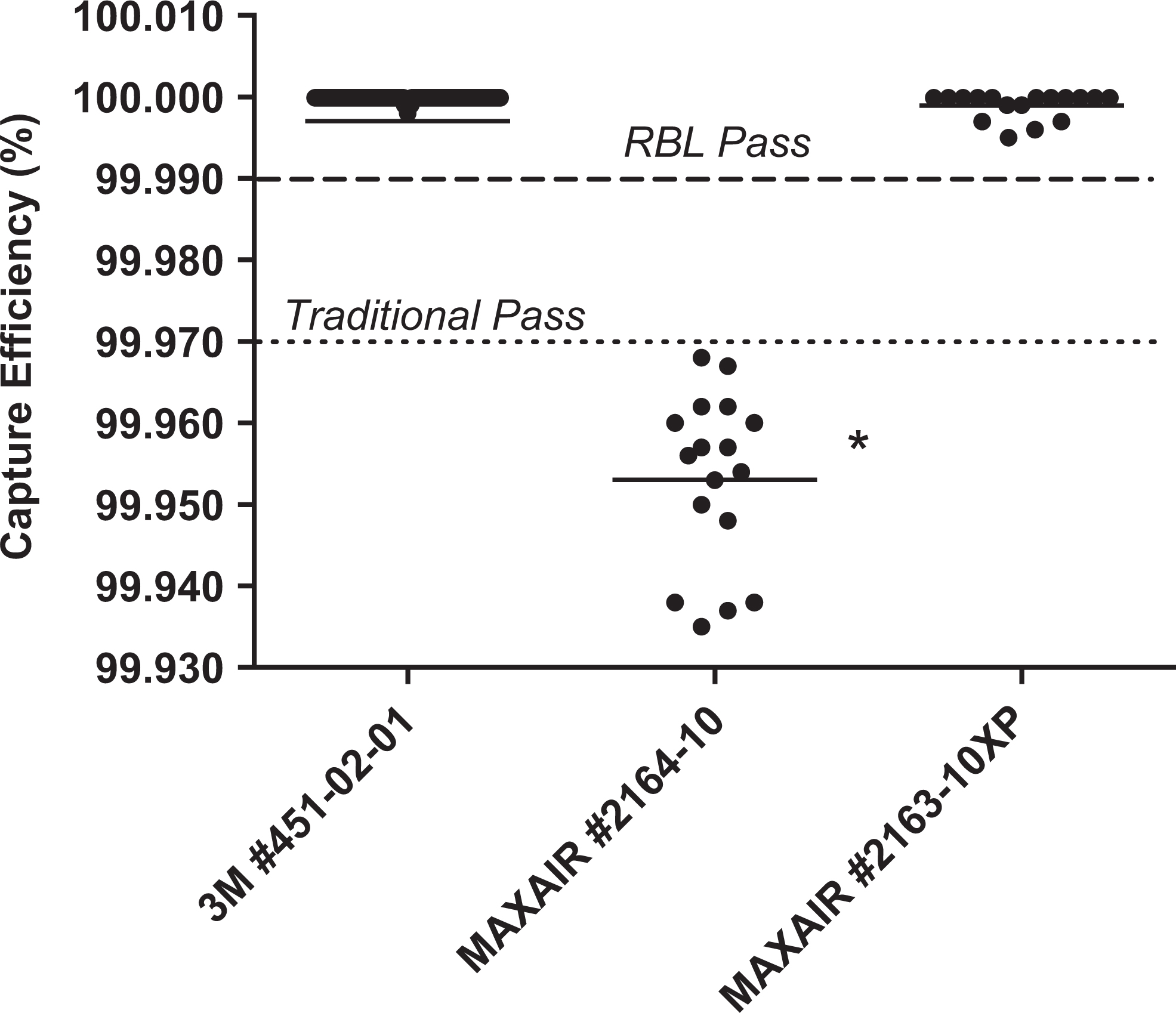
Leak test capture efficiency results for 3 PAPR/HEPA filter configurations. Mean percentage efficiency ± standard error of the mean is shown. *Significant difference (P < .0001) between the MAXAIR controlled air-purifying respirator disposable lens cuff and the 2 other tested filters. Note that 2 data points for the 3M Air-Mate (99.750 and 99.860) are not displayed owing to the scale of this chart. HEPA, high-efficiency particulate air; PAPR, powered air-purifying respirator.
Discussion
A primary assumption when using a NIOSH-certified PAPR is that the filtration system will perform over time in the field as it was certified in the test laboratory. This confidence originates from the fact that the full system was previously tested in the laboratory by both the manufacturer and NIOSH and confirmed to be operating as designed. The reality is that PAPR filtration systems are shipped to the customer in multiple pieces and require the end user to assemble the PAPR system and install the HEPA filter (Figures 1 and 2). As is recommended for other HEPA filtration devices (ie, biological safety cabinets, ventilated animal caging systems), it is our opinion that field-testing of PAPR systems used in a BSL-3 environment should be performed to ensure proper filtration efficiency. The in situ testing approach described in this report not only confirms the integrity of the installed HEPA filter but also identifies any leaks in the housing frame or around the filter gasket.
Both PAPR systems used in the RBL at Duke University are NIOSH certified and have been performance tested on site with the method described. The 3M Air-Mate Belt-Mounted High Efficiency PAPR with filter 451-02-01 has been used for the past 6 years and has consistently performed as marketed (Figure 5). Only twice (2 of 139 tests) has the 3M PAPR system failed to achieve the minimum filtration efficiency of 99.99% set by the RBL safety team (Table 1). In both cases, it was determined that the leak occurred around an improperly seated filter and that the integrity of the filter itself was intact. This was proven by removal of the filter, replacement of the filter gasket, reinstallation of the same filter, retesting of the PAPR system, and confirmation of filtration efficiency >99.99%. The MAXAIR CAPR DLC system was recently purchased for use in the BSL-3 laboratories of the RBL, as an additional option for respiratory protection. Given our experience with the 3M PAPR system, we decided to validate the performance of the MAXAIR PAPR before approving it for use in laboratories where active research with RG3 agents is conducted. The evaluation process included testing of the PAPR in 2 configurations—one with the 2163-10XP filter and the other with the 2164-10 filter. According to MAXAIR sales and technical support representatives, both filter installations are appropriate and safe for BSL-3 laboratory work, but the XP filter would provide a higher level of filtration efficiency if desired. Data from our performance evaluation of the MAXAIR CAPR DLC system demonstrated, as expected, that the highest level of respiratory protection was provided when the PAPR is equipped with the 2163-10XP filter. It is this later configuration of the MAXAIR system that was subsequently approved for use in the RBL at Duke University.
This report demonstrates the value of implementing an on-site performance evaluation process for HEPA-filtered PAPR units used for respiratory protection in a BSL-3 laboratory environment. Use of IEST recommended practice 34.3 as a guide to perform total leak testing of a HEPA-filtered PAPR system is a viable option for enhancement of a comprehensive biosafety program. Our data suggest that in situ testing can help to ensure the desired filtration efficiency of the PAPR units before they are put into service and, through regular testing, their continued performance. The data also demonstrate that supplementing an Occupational Health and Safety Administration–compliant site-specific respiratory protection program with on-site performance testing can aid in the identification of not only improperly installed filters but also overall filtration performance that differs from NIOSH and manufacturer test data.
Footnotes
Acknowledgments
We acknowledge the biosafety expertise of James Burch, who coordinated much of the powered air-purifying respirator testing over the past 6 years. The Regional Biocontainment Laboratory at Duke University was built with a construction grant from the National Institute of Allergy and Infectious Diseases, National Institutes of Health (UC6-AI58607). This work was supported in part by generous support from the Duke School of Medicine and the Duke Human Vaccine Institute, Duke University, Durham, North Carolina.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
