Abstract
Background
Ankylosing spondylitis (AS) is characterized by inflammation and osteoblastic changes in the sacroiliac joint. As a potential imaging method for the early assessment of AS, positron emission tomography (PET) can quantify systemic disease activity, which is conducive to monitoring the progression of disease activity and assisting in evaluating the efficacy of the treatment.
Objective
The study was to evaluate the diagnostic value of aluminium-[18F]fuoride(Al18F)-labelled fibroblast activation protein inhibitor (FAPI) PET/computed tomography (CT) in AS and to investigate its ability to assess disease activity during the development of AS.
Material and Methods
Twenty AS participants who met the Assessment of SpondyloArthritis international Society criteria and were in an active disease stage were included in this study from May 2022 to April 2023. Sixteen healthy controls were also inrolled. All participants underwent Al18F-NOTA-FAPI-04 PET/CT imaging after collecting clinical assessment and laboratory results. The correlation between positive joint count (PJC) and systemic joint standard uptake value ratio (SUVR, the mean SUVmax of the 5 highest joints/SUVmax of the uninvolved sacrum) on PET and clinical disease activity assessment and various laboratory tests were analyzed.
Results
A total of 2820 joints were observed in 20 participants (median age 34.5,[21-61]range, 15 men), with a PJC of 1300 (46.7%), and 39 positive uptakes were found in 40 sacroiliac joints (97.5%). PET/CT images revealed FAPI-04 uptake in both sacroiliac joints in 2 participants without radiographic sacroiliitis in the early stages of AS and varying degrees of uptake in the sacroiliac joints and spinal joints in the remaining participants. PJC and SUVR were positively correlated with most clinical assessments and laboratory findings (P < .05), and SUVR of the sacroiliac joint was positively correlated with C-reactive protein (CRP) (mg/L; r = 0.498, P = .026).
Conclusion
Al18F-NOTA-FAPI-04 PET/CT was highly sensitive to systemic arthritic changes in AS participants and correlated with clinical disease activity and laboratory tests.
Introduction
As a chronic inflammatory disease, ankylosing spondylitis (AS) affects mainly the sacroiliac joints, spine joints, and ligaments. It can be accompanied by peripheral arthropathy, which in turn leads to changes in posture, restricted movement, spinal deformity, ankylosis, and even disability. 1 AS is a disease that leads to serious consequences. Currently, the pathophysiological mechanism of AS is not well understood, but 2 core characteristics can be identified: inflammation and new bone formation. 2 Inflammation is a key reversible factor determining dysfunction in the early stages of the disease, and irreversible structural changes become more important in the later stages of the disease.
For patients with AS, sacroiliitis is a relatively early symptom. The first symptom presenting is usually hip pain on one or both sides. Due to the complexity of the sacroiliac joint, sacroiliitis is insidious and progresses slowly in the early stages. Sacroiliitis is identified relatively late on plain radiography, usually after several years of persistent or recurrent inflammation. At this time, the lesion has progressed to a relatively late stage, and the best time for treatment has been missed. 3 As previously reported, TNF inhibitors effectively suppress inflammation but cannot prevent new bone formation. 4 The early detection and evaluation of inflammation are of great significance in diagnosing and treating AS. Besides, changes in structure on X-rays are insufficient for evaluating disease activity and assessing efficacy of the treatment. 5
As a sensitive molecular imaging technique, positron emission computed tomography (PET) is conducive to deepening the understanding of the pathophysiological process of the disease. 6 The specific binding of tracers to targets may be suitable for the early detection of inflammatory processes. Fibroblast activation protein (FAP) is minimally expressed in normal tissues and organs of healthy individuals. However, it is highly expressed under many pathophysiological conditions, such as inflammation, fibrosis, and in tumors, and is currently a widely studied popular molecular imaging target.7,8 FAP is overexpressed at tissue remodeling sites associated with arthritis and fibrosis (eg, fibroblast-like synoviocytes cells and chondrocytes), 9 and is normally expression at low levels in bone marrow. 10 Al18F-NOTA-FAPI-04 (Al18F-NOTA-FAPI-04, aluminum-[18F]-labeled 14,7-triazacyclononaneN,N′,N″-triacetic acid-conjugated fibroblast activation protein inhibitor 04) allows the visualization of systemic FAP expression for semiquantitative analysis and quantification of inflammatory processes. 11 This prospective study aimed to evaluate the potential of Al18F-NOTA-FAPI-04 PET/CT on FAP expression in AS joints, investigate the correlation between its uptake and inflammatory markers and disease activity, and thereby provide a new method for AS disease monitoring and efficacy evaluation.
Materials and Methods
Study Design and Participants
This prospective study was approved by the Ethics Review Board of the [details omitted for double-anonymized peer review]. Twenty-nine patients were enrolled in the study between May 2022 and April 2023 after they had provided written informed consent. All patients underwent physical examination and were clinically diagnosed by 2 experienced rheumatologists (WH and LZG). A total of 20 patients who met the Assessment of SpondyloArthritis international Society (ASAS) criteria for AS and were in the active stage of disease were included in the clinical study. Patients who (a) suffered from other diseases involving the spine or sacroiliac joint (such as bone metastases, trauma, or other rheumatic diseases), (b) were female and pregnant or breastfeeding, and (c) those who could not complete the examination due to pain were excluded from the study. In addition, we included 16 healthy controls of similar age to the AS group.
Blood samples were collected from all patients for laboratory tests, which included human leukocyte antigen B27 (HLA-B27), erythrocyte sedimentation rate (ESR) (mm/h), and C-reactive protein (CRP) (mg/L), within 2 days of the patient undergoing Al18F-NOTA-FAPI-04 imaging. Disease activity assessments and calculations, including the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) score, Ankylosing Spondylitis Disease Activity Score-CRP (ASDAS-CRP), and Ankylosing Spondylitis Disease Activity Score-ESR (ASDAS-ESR), were made by rheumatologists.
Radiopharmaceutical and PET/CT Protocol
Al18F-NOTA-FAPI-04 drug was synthesized according to the published synthesis method. 11 Kits were supplied by Wuxi Jiangyuan Industrial Technology & Trade Co., Ltd, with the product number JY2102. 18F was extracted after being produced with a medical cyclotron (HM12, Sumitomo Heavy Industries, Ltd, Japan).
Each patient was injected with about 185 MBq of Al18F-NOTA-FAPI-04 and subjected to PET/CT (Philips, GEMINI GXL) imaging with a field of view of 600 mm, tube current of 120 mA, tube voltage of 120 Kv, and layer thickness of 5 mm. Imaging from the middle of the femur to the top of the skull occurred 30 min after an intravenous injection of the drug. Patients with peripheral joint involvement were scanned to the plantar after an interview. The single-bed PET scan was performed for 120 s, and the PET image was reconstructed on a 144 × 144 matrix using a standard iterative algorithm method. The line of response reconstruction algorithm was used to reconstruct the image without postreconstruction filtering after automatic random and scattering correction.
Image Analysis
Al18F-NOTA-FAPI-04 PET/CT scan images were interpreted by 2 senior nuclear medicine physicians (FP and ZCJ), and the fused PET/CT images were accessed on the workstation in a randomized order. Both physicians were aware of the patient's medical history and routine imaging findings. AS-like lesions were distinguished from osteoarthritis (OA) lesions. They were considered to be degenerative if there were no signs of AS near the lesion. According to the standardized scale of AS spine and sacroiliac joints reported previously, 12 the regions of interest were drawn one by one and semiquantitatively analyzed. The maximum standard uptake value (SUVmax) of the following sites was recorded: bilateral sacroiliac joints, anterior and posterior vertebrae, spinous processes, and facet joints (including vertebral facet joints and costovertebral joints). The SUVmax value in the uninvolved portion of the sacrum was defined as baseline SUVj. Lesions exceeding SUVj were defined as PET-positive lesions, and the number of PET-positive lesions was indicated by the positive joint count (PJC). The mean SUVmax values of the 5 lesions with the highest SUVmax values were calculated for each patient, and their ratio to SUVj was defined as the standard uptake value ratio (SUVR). The ratio of SUVmax to SUVj of the sacroiliac joint was defined as SUVRsj.
Radiological findings were graded according to the New York criteria for consensus. There are 5 grades: 0 points indicated normal, 1 point indicated changes in sacroiliac joint arthritis; 2 points indicated minor abnormalities with small local areas of erosion, sclerosis, widening, or narrowing of the joint space; 3 points indicated definite abnormalities with erosion, sclerosis, widening, or narrowing of the joint space, or partial ankylosis; and 4 points indicated complete ankylosis.13,14
Statistical Analysis
SPSS 25.0 statistical analysis software was adopted for statistical analysis, and GraphPad Prism v.8.0 software was used for plotting. All normally distributed variables are presented as the mean ± standard deviation, and all non-normally distributed variables are presented as the median and interquartile ranges (IQRs; 25%−75%). The differences between the 2 groups were examined using independent samples t-tests and the Mann-Whitney U test for normally and non-normally distributed parameters, respectively. One-way analysis of variance was applied to compare PJC and SUVR among groups with different disease activities. A Pearson's correlation coefficient analysis was performed for normally distributed data, and a Spearman's rank correlation coefficient analysis was performed for skewed data. P < .05 indicates that the difference was statistically significant.
Results
Participant Characteristics
A total of 20 patients with AS and 16 healthy controls were included in this prospective study (Figure 1), and the clinical characteristics of these patients with AS are summarized in Table 1. Twenty patients (15 male and 5 female, with a median age of 34.5 years) had a median disease duration of 29 months (range: 1-395). Ten of the 20 patients were initially diagnosed without treatment, and 10 had a history of AS that had now relapsed. There were 16 (80%) HLA-B27-positive patients, 17 (85%) ESR-positive patients, and 15 (75%) CRP-positive patients. According to the ASDAS-CRP disease activity scoring system, 3 patients had moderate disease activity (1.3-2.1), 11 patients had high disease activity (2.1-3.5), and 4 patients had very high disease activity (> 3.5). All laboratory tests and disease activity scores were higher in the AS group than in the control group (P < .05).

Flow chart of the patient recruitment criteria and study procedure. Abbreviations: Al18F-NOTA-FAPI-04, aluminum-[18F]-labeled 14,7-triazacyclononaneN,N′,N″-triacetic acid-conjugated fibroblast activation protein inhibitor 04; AS, ankylosing spondylitis; ASDAS, Ankylosing Spondylitis Disease Activity Score; BASDAI, The Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HLA-B27, human leukocyte antigen B27; SAPHO, synovitis, acne, pustulosis, hyperostosis, and osteitis.
Clinical Characteristics and Laboratory Findings of the Participants (n = 20).

Radiographs grading: 0: indicated normal; 1: indicated changes in sacroiliac joint arthritis; 2: indicated minor abnormalities with small local areas of erosion, sclerosis, widening, or narrowing of the joint space; 3: indicated definite abnormalities with erosion, sclerosis, widening, or narrowing of the joint space, or partial ankylosis; 4: indicated complete ankylosis.
ASDAS-CRP: 0.12 × back pain + 0.06 × duration of morning stiffness + 0.11 × patient global + 0.07 × peripheral pain/swelling + 0.58×Ln(CRP + 1); ASDAS-ESR: 0.08 × back pain +0.07×duration of morning stiffness +0.11×patient global + 0.09×peripheral pain/swelling + 0.29 ×√(ESR).
Abbreviations: AS, ankylosing spondylitis; ASDAS, The Assessment of SpondyloArthritis international Society; BASDAI, The Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HLA-B27, Human leukocyte antigen B27; PET, positron emission tomography; SUV, standard uptake value.
Positive Lesions and SUVmax Measurements of Al18F-NOTA-FAPI-04 PET/CT
Unlike the 16 healthy controls (Figure 2b), 20 patients with AS enrolled exhibited varying degrees of uptake of FAPI-04 in the sacroiliac joints and spine (Figure 2a and c). A total of 2820 joints (600 in the cervical vertebrae, 1680 in the thoracic vertebrae, 500 in the lumbar vertebrae, and 40 in the sacroiliac joints) were observed in the sacroiliac joints and spine of all patients, with a PJC of 1300 and a detection rate of 46.70%. Of these joints, 228 (38%, 228 of 600) were positive cervical vertebrae, 828 (49.7%, 828 of 1680) were positive thoracic vertebrae, 205 (41%, 205 of 500) were positive lumbar vertebrae, and 39 (97.5%, 39 of 40) were positive sacroiliac joints. Of the other joints in these 20 patients, there were 15 positive sternoclavicular joints (37.5%, 15 of 40), 1 positive ankle (2.5%, 1 of 40), and 4 positive knees (10%, 4 of 40). Twenty patients had a mean PJC of 65.75 ± 18. The mean SUVmax (± SD) of the 5 joints with the highest SUVmax was 3.29 ± 0.93. The mean SUVR (± SD) was 2.26 ± 0.67, the median sacroiliac SUVmax (IQR) was 2.41 (1.36-6.03), and the median SUVRsj (IQR) was 1.60 (1.15-3.51). Supplemental Table 1 demonstrates the demographics and SUV values of patients with AS with different radiological classifications, PET/CT of the sacroiliac joints in the 5 patients with different radiological grades is shown in Figure 3.

(a) A 28-year-old male patient with sacroiliac and low back pain of gradual worsening for 1 year, HLA-B27 positive, C-reactive protein 32.68, erythrocyte sedimentation rate 17, SUVR 3.51.ASDAS-CRP 4.2, whole-body maximum projection MIP image (A) shows bilateral sacroiliac joint and multiple vertebral joint uptake (arrows), and cross-sectional images (B-D) show bilateral sacroiliac joint radiotracer uptake with a SUVmax of 6.03, suggesting bilateral sacroiliac arthritis. (b) A 27-year-old healthy patient with Al18F-NOTA-FAPI-04 positron emission tomography images and a negative serological human leukocyte antigen B27 test did not meet the Ankylosing Spondylitis International Association criteria for Assessing Spondylitis. Maximum intensity projection image (A) and transverse axial image of the sacroiliac joint (B-D) are indicated as normal scans. (c) A 21-year-old male patient with malaise and sacroiliac joint pain for more than 1 month, HLA-B27 positive, CRP 37, erythrocyte sedimentation rate 39, SUVR 2.41, ASDAS-CRP 3.26. CT and radiographs showed no abnormalities in the sacroiliac joints with a radiological grade of 0 (A and C), and PET showed radiological aggregates in the sacroiliac joints bilaterally with a SUVmax of 2.79 (B), suggestive of bilateral sacroiliac arthritis, which was consistent with that shown by MRI (D). Abbreviations: Al18F-NOTA-FAPI-04, aluminum-[18F]-labeled 14,7-triazacyclononaneN,N′,N″-triacetic acid-conjugated fibroblast activation protein inhibitor 04; ASDAS, Ankylosing Spondylitis Disease Activity Score; CRP, C-reactive protein; CT, computed tomography; HLA-B27, human leukocyte antigen B27; MRI, magnetic resonance imaging; PET, positron emission tomography; SUVR, standard uptake value ratio.

CT and PET/CT images of 5 patients with different radiological grades of sacroiliac joints. Abbreviations: CT, computed tomography; PET, positron emission tomography.
Correlation Analysis of Disease Activity, Laboratory Examination, and PET/CT
The correlation of PJC and SUVR with various laboratory findings and clinical physical examinations in all patients with AS with positive Al18F-NOTA-FAPI-04 PET is illustrated in Table 2. PJC was positively correlated with CRP (R = 0.517, P = .020), the BASDAI score (R = 0.554, P = .011), and the ASDAS-ESR score (R = 0.511, P = .021), and was strongly positively correlated with the ASDAS-CRP score (R = 0.684, P = .001). SUVR was positively correlated with CRP (R = 0.510, P = .021), the ASDAS-CRP score, and the ASDAS-ESR score (R = 0.554, P = .011, R = 0.459, P = .042, respectively). The Spearman's analysis revealed that SUVRsj was positively correlated with CRP (R = 0.468, P = .038).
Correlation Analysis of PET/CT with Laboratory Tests and Clinical Disease Activity.

*P < .05, **P < .01.
Abbreviations: ASDAS, Ankylosing Spondylitis Disease Activity Score; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein; CT, computed tomography; ESR, erythrocyte sedimentation rate; PET, positron emission tomography; PJC, positive joint count; SUVR, standard uptake value ratio.
Patients with AS with different disease activity grades were compared by the mean ± standard deviation. As shown in Table 3 and Figure 4, PJC and SUVR gradually increased as the disease activity increased, and PJC was significantly higher in patients with very high disease activity (74.00 ± 11.90) than in those with moderate disease activity (43.67 ± 27.57). There was no statistical difference in the others.

Bar chart comparing differences between groups of PJC and SUVR between different levels of disease activity. Two sets of bar graphs show clear trends, using one-way ANOVA, P < .05 was statistically significant. Abbreviations: ANOVA, analysis of variance; PJC, positive joint count; SUVR, standard uptake value ratio.
Comparison of 3 Between-Group Indicators of Disease Activity.

Abbreviations: PJC, positive joint count; SUVR, standard uptake value ratio.
Al18F-NOTA-FAPI-04 Uptake After Anti-TNF Treatment
The scan results of 3 patients with AS who were diagnosed for the first time and who had not undergone any drug treatment before the PET/CT scan were defined as the baseline. Three patients were treated with adalimumab (adalimumab solution for injection) at the end of imaging. The Al18F-NOTA-FAPI-04 PET/CT was repeated 24 weeks later. Laboratory tests and physical examinations were performed before the PET/CT (Table 4 and Figure 5). Efficacy assessment based on ASDAS disease activity status and treatment improvement cutoff values 15 revealed significant clinical improvement in patient #3 (ASDAS-CRP score decreased from 4.12 to 1.58,very high disease activity to moderate disease activity) and significant clinical improvement in patient #8 (ASDAS-CRP score decreased from 3.44 to 1.94, high disease activity to moderate disease activity). In addition, there was a decrease in all laboratory and physical examinations in both patients and in SUVR, SUVRsj, and PJC. Patient #10 had high disease activity before and after treatment (score of 3.29 before treatment and score of 2.33 after treatment). Moreover, after treatment, the repeated whole-body PET revealed diffuse increased uptake of FAPI-04 with increased uptake in the liver and spleen (SUVmax 1.48 vs4.84 of liver before and after treatment and SUVmax 1.27 vs 4.27 of spleen before and after treatment). There was also decreased SUVR in some joints and increased uptake in major joints such as bilateral sacroiliac joints.

Baseline and post-treatment Al18F-NOTA-FAPI-04 PET/CT images of 3 patients, patient #3 is a 35-year-old male with an initial diagnosis of ankylosing spondylitis who was treated with adalimumab and reviewed 7 months later, patient #8 is a 36-year-old man who had been diagnosed with ankylosing spondylitis 1 month earlier and was followed up 6 months after treatment with adalimumab. Patient #10 is a 28-year-old woman with an initial diagnosis of ankylosing spondylitis. She was treated with adalimumab and followed up 6 months later. Abbreviations: Al18F-NOTA-FAPI-04, aluminum-[18F]-labeled 14,7-triazacyclononaneN,N′,N″-triacetic acid-conjugated fibroblast activation protein inhibitor 04; CT, computed tomography; PET, positron emission tomography.
Comparison of Pre- and Post-Treatment Data in 3 Review Patients.
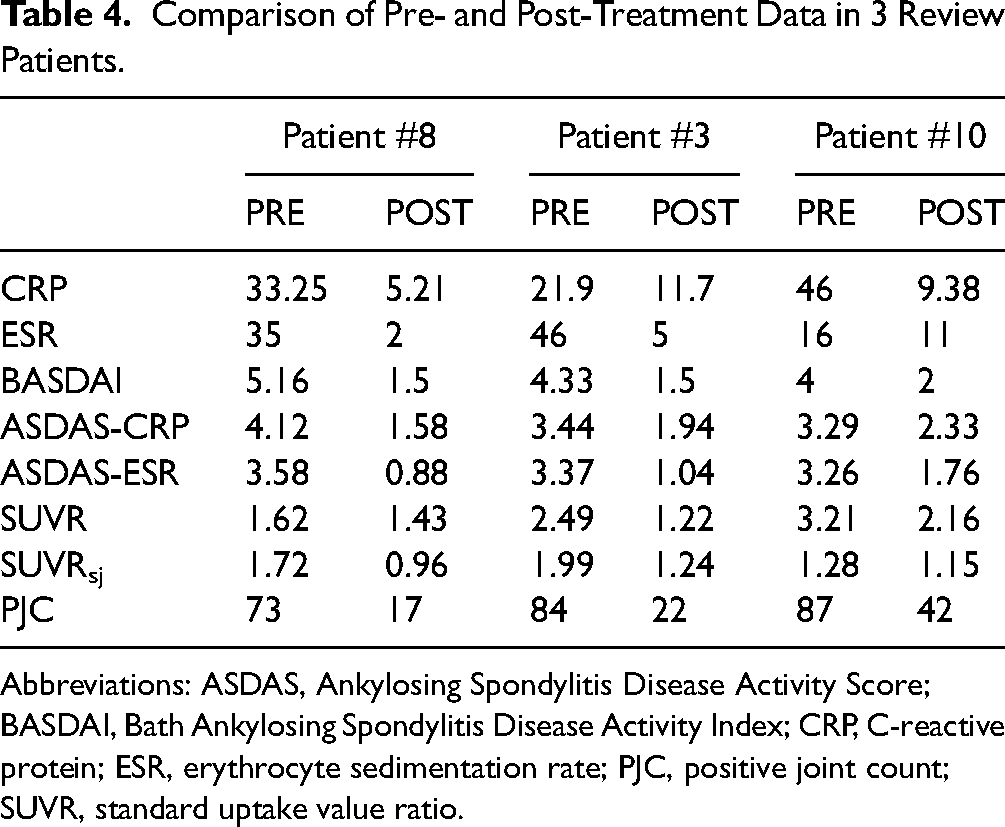
Abbreviations: ASDAS, Ankylosing Spondylitis Disease Activity Score; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; PJC, positive joint count; SUVR, standard uptake value ratio.
Discussion
To our knowledge, this is the first prospective exploratory study using FAPI-04 (a small molecule inhibitor of FAP) after labeling it with radionuclide 18F in the diagnosis and treatment of AS. The aims were to assess inflammatory changes in systemic joints, disease progression, and treatment outcomes in patients with AS at the active stage of the disease. Al18F-NOTA-FAPI-04 PET/CT examination can be used as a new imaging evaluation method for AS and is sensitive to inflammatory lesions.
PET/CT images could visualize the development of AS lesions in the bones and joints of a patient's body. Most joint uptake occurred in the thoracic vertebrae and sacroiliac joints, consistent with a previous report. 16 In the peripheral joints, uptake in both knees was found in 2 patients, and increased radioactivity concentration was observed in the ankles in one patient, probably due to peripheral joint accumulation caused by AS, which highlighted the advantages of PET/CT systemic examination.
In general, increased FAPI uptake can be observed in both OA and AS lesions. We observed spinal OA lesions in a few older control groups. Typical cases are shown in Supplemental Figure 1. There are few points to differentiate the AS and OA lesions. First, the age of onset of OA is higher. Second, in contrast to OA, laboratory tests including HLA-B27, CRP, and ESR are mostly positive in patients with AS. Third, the imaging studies usually reveal sacroiliac joint changes in patients with AS while the lesions of OA mainly accumulate in the spine. The main value of FAPI PET/CT in the diagnosis of AS lies in the detection of early inflammatory lesions.
Sacroiliac joints with radiographic grade 0, which cannot yet be diagnosed by anatomical changes on conventional radiological examinations such as X-ray and CT, were defined as preradiological AS and were also included in the AS diagnostic criteria. 17 In the PET/CT images of these patients, a positive distribution of Al18F-NOTA-FAPI-04 was comparable to that on magnetic resonance imaging (MRI) (Figure 2b), demonstrating its ability to diagnose early inflammatory lesions in AS. Al18F-NOTA-FAPI-04 PET/CT is expected to be a new option for detecting early inflammatory changes in the sacroiliac joint, and early detection of sacroiliac inflammation and timely treatment can greatly slow disease progression, 3 which is of great clinical significance.
Also, it was noted that SUVmax (6.03) and SUVR (3.51) were highest in grade 2 sacroiliac joints and lowest (2.07 and 1.46) in grade 4 sacroiliac joints, probably because the grade 4 patients were already in an advanced stage of the disease. Bilateral sacroiliac joints were completely closed on CT, and the pathological process of osteogenesis had ended with the formation of complete ankylosis and a substantial reduction in the extent and degree of the inflammatory response. Thus, the uptake of Al18F-NOTA-FAPI-04 was subsequently reduced. Therefore, Al18F-NOTA-FAPI-04 did not show a significant advantage in diagnosing patients with grade 4 AS. However, the imaging results demonstrated that Al18F-NOTA-FAPI-04 was absorbed in a small amount in the bone joints of patients with advanced AS, indicating that the inflammatory pathological development process may not be terminated entirely. Grade 4 patients were able to be diagnosed by X-ray, CT, and other imaging methods, which can complement PET and CT to a certain extent. Hybrid PET/CT imaging may possess a higher sensitivity for detecting sacroiliitis.
PJC and SUVR have not been demonstrated to be effective in assessing AS disease progression. The PJC on 18F-NaF PET/CT images12,18 has been counted in some studies. However, its correlation with disease progression or various laboratory parameters has not yet been analyzed. In PET fusion images, patients with AS tended to have more PJC around joint sites where they complained about pain. In addition, the SUVmax was higher in the more painful joints, suggesting that inflammatory changes in the joints may be the main cause of pain in patients 19 and PJC may correlate with disease activity scores. It was found that PJC was significantly correlated with ASDAS-CRP and correlated with CRP, the BASDAI score, and the ASDAS-ESR score. Given the heterogeneity of different patients, it may be more accurate to apply SUVR rather than SUVmax for analytical diagnosis in this study. Besides, considering that laboratory tests and disease activity scores are based on the whole-body assessment, it is also more reasonable to adopt SUVR of the whole-body joints for correlation analysis between various tests. It was revealed that SUVR correlated with CRP and the ASDAS-CRP and ASDAS-ESR scores. The SUVR in the sacroiliac joints correlated with CRP only. For the 3 groups of patients with different degrees of disease activity, the difference in PJC among the 3 groups was statistically significant. This was not the case with SUVR, probably because of the different pathological findings at each stage of AS development. 20 Some joints were in the osteogenic stage or even closed, the inflammatory response is not as strong at this point. Al18F-NOTA-FAPI-04 visualizes mainly the presence and degree of the inflammatory response, which resulted in no significant differences among the different groups.
PJC and SUVR were also used to compare patients before and after monoclonal antibody treatment. Notably, Al18F-NOTA-FAPI-04 PET images revealed that the inflammatory uptake in the sacroiliac joints and spinal joints was significantly reduced. However, there was slight structural progression on the CT images, which was consistent with previous follow-up data after the use of TNF inhibitors. Most patients with AS had significantly reduced inflammation after drug treatment. However, no effect on bone formation was observed, and treatment only slowed the disease progression. 21 Further, the ASDAS-CRP scores of 2 patients were also reduced to varying degrees. Another patient had decreased PJC and SUVR compared with pretreatment. However, additional positive uptake in joints at the same time and diffusely increased systemic Al18F-NOTA-FAPI-04 uptake, especially in the liver and the spleen, and the therapeutic effect was not satisfactory. Due to the small number of patients who received post-treatment examinations, this study did not investigate correlations between SUVR and PJC or the clinical scores and laboratory tests in patients before and after treatment. However, the possibility of applying Al18F-NOTA-FAPI-04 to monitor inflammatory changes in patients with AS before and after treatment was explored to assist in clinical efficacy evaluation. The correlation between them needs further investigation.
The study had some limitations. First, the sample size of the enrolled patients was small, and the enrolled patients were in an active disease stage. No imaging was performed for patients with inactive AS, and there might be an imbalance between the different groups. Furthermore, relevant pathological findings to further define the nature of the lesion could not be obtained. Finally, only a correlation between PJC and SUVR was analyzed in this study, and there was no more definitive scoring system to grade the disease activity. In the future, these 2 findings may be converted into Spondyloarthritis Research Consortium of Canada (SPARCC) MRI scores,22,23 or a new PET formula will be established for better disease activity grading to assess disease progression.
Conclusions
Al18F-NOTA-FAPI-04 PET/CT is highly sensitive to inflammatory changes in patients with AS. It has good blood clearance efficiency, a low background, and good inter-reader agreement. In addition, it can visualize systemic inflammatory lesions in one imaging. Therefore, it has great potential as an imaging tool for diagnosing AS and in the early assessment of inflammation. As a molecular imaging method, PET can reflect changes in molecular expression in the pathophysiological process of AS, which may be favorable for studying AS pathogenesis in clinical and scientific research. The 2 main findings, PJC and SUVR, which correlate well with clinical scores and laboratory tests, can be used as complementary data for the clinical assessment of disease activity and progression. As a repeatable imaging method, PET/CT images are significant for evaluating efficacy with good inter-reader agreement, which is conducive to clinical efficacy evaluation and making treatment plans.
Supplemental Material
sj-doc-1-mix-10.1177_15353508241270405 - Supplemental material for Preliminary Exploration of Al18F-NOTA-FAPI-04 PET/CT in the Management of Ankylosing Spondylitis: A Prospective Clinical Study
Supplemental material, sj-doc-1-mix-10.1177_15353508241270405 for Preliminary Exploration of Al18F-NOTA-FAPI-04 PET/CT in the Management of Ankylosing Spondylitis: A Prospective Clinical Study by Shibo Guo, MD, Zhehao Lyu, PhD, Chunyu Duan, MD, Hui Wang, PhD, Peng Xu, MD, Wei Han, BD and Peng Fu, PhD in Molecular Imaging
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Institutional Review Board approval was obtained.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by The First Affiliated Hospital of Harbin Medical University (grant number 2023M06 and 2021M16).
Informed Consent
Written informed consent was obtained from all subjects (patients) in this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
